Review Article - (2023) Volume 6, Issue 1
Urine Drug Testing in Pain Medicine
Received Date: Jul 27, 2023 / Accepted Date: Aug 01, 2023 / Published Date: Sep 23, 2023
Copyright: ©Â©2023 Rehan Haider. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Haider, R. (2023). Urine Drug Testing in Pain Medicine. Toxi App Pharma Insights Research, 6(1), 21-30.
Abstract
The recent Institute of Medicine report declared that greater than 100 million Americans have chronic pain with an associated cost of up to $635 billion each year due to medical treatment and lost productivity.[1,2] The explosion in opioid prescribing since the 1990s has resulted in opioids being the most frequently prescribed medication in the United States.[3–5] Manchikanti et al.'s study revealed a 149% increase in retail opioid sales and a 402% increase in average sales of opioids per person in the United States from 1997 to 2007 [6].
Looking at 1999–2012, the National Center for Health Statistics found that among patients aged 20 or older prescription opioid use increased from 5% to 6.9% until 2006 and then stayed at 6.9% until 2012.7 However, the percentage of patients using an opioid stronger than morphine dramatically increased from 17% to 37% over the same period.[7] Among the top 25 dispensed prescriptions in the United States, hydrocodone, tramadol, and oxycodone accounted for 1, 21, and 22 dispensed prescriptions, respectively. 5 Opioids represent one of many therapeutic options for treating chronic nonmalignant pain (CNMP), but their widespread use has resulted in a concomitant increase in misuse, abuse, addiction, overdose, and diversion. While there is evidence of the short-term benefit of opioids for treating pain based on randomized trials lasting less than 3 months, there are few studies reporting outcomes at 12 months or longer [8,9].
Keywords
Urine Drug Testing, Chronic Pain, Addiction Forensic Testing, Compliance Testing
Distribution of urine drug testing reports for Quest Diagnostics (January 2014 to December 2014). (From Blatt A, Chen Z, McClure L, Niles J, Kaufman M: Prescription drug misuse in America: diagnostic insights in the continuing drug epidemic battle. Quest Diagnostics Health Trends Prescription Drug Monitoring Report 2015, 16.
Distribution of urine drug checking reports for Quest Diagnostics (January 2014 to December 2014). (From Blatt A, Chen Z, McClure L, Niles J, Kaufman M: Prescription drug misuse in America: diagnostic insights in the continuing drug epidemic conflict. Quest Diagnostics health trends Prescription Drug monitoring document 2015, 16)
Introduction
Research has confirmed the lengthy-time period benefits of persistent opioid therapy (COT), and mounting evidence has confirmed the capacity-damaging dangers (opioid use ailment, overdose, and motor car harm) associated with COT.2 [10].
Loss of life due to opioid overdoses fell one hundred 65,000 individuals from 1999 to 2014,[11] and most victims who are on COT will no longer expand an opioid-use ailment, the chance is greater than that inside the well-known population. One has a look at established that an opioid-use disease happened in 3.8% of those on COT as opposed to zero.9% of the overall populace, and different studies recommend abuse prices can be as excessive as 18% to 41%.[12,13] among patients with continual ache who were either taking or now not taking a controlled substance, illicit substance abuse become stated in 14% to sixteen% and 34%, respectively.2 In 2014, approximately 27.0 million people elderly 12 or older had been contemporary (past month) illicit drug users.14 At the same time as marijuana turned into through ways the maximum commonly used illicit drug (22.2 million Americans), the usage of ache relievers (opioid and non-opioid) for nonmedical functions become second (4.3 million Americans) [14].
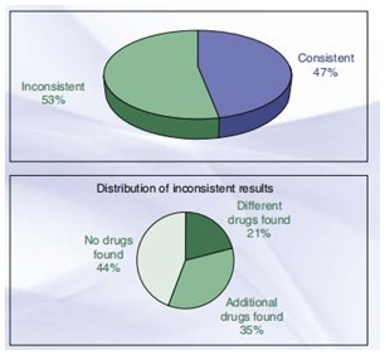
Of the humans aged 12 or older who had a prescription pain reliever use disorder (1.9 million humans, or 0.7% of the people aged 12 or older), the proportion became largely similar to what it became from 2005 to 2013.14 based totally on an analysis of two.1 million affected person urine drug checking out reviews, the most current annual Quest Diagnostics fitness developments Prescription Drug tracking file 2015 discovered consistent results in 47% and inconsistent results in fifty three%; and of the inconsistent effects, 44% had no drug, 35% had additional drugs, and 21% had exclusive capsules (Fig. forty six.1) 15.
While the inconsistent consequences are excessive, it represents a decline from sixty three% in 2011, probably because of a multitude of things: implementation and use of the prescription drug tracking application in the prescribers state, federal, and kingdom law on opioid prescribing guidelines, practitioner training, public focus campaigns, and usage of regulation enforcement [15].
For those patients for whom opioids might be beneficial, Many clinicians are reluctant to prescribe them for diverse reasons, including lack of schooling and education in opioid prescribing, concerns about misuse, abuse, dependency, tolerance, aspect consequences, worry about regulatory research, and lack of proof that COT is valuable in dealing with CNMP.2 [16].
Such concerns present boundaries for the use of COT for CNMP. Several screening instruments have been developed to risk-stratify sufferers on COT for substance misuse and addiction; however, it is far as much as the clinician can decide which one(s) to apply, considering that no personal instrument is advanced to all others. Use of hazard evaluation gear (e.g., Screener and Opioid evaluation for sufferers with pain [SOAPP] and Opioid chance tool [ORT], and Drug Abuse Screening check [DAST]) and obtaining cautious records and bodily examination can help the clinician in determining the level of threat related to prescribing opioids for an affected person and the associated level of monitoring wanted [17].
In a retrospective analysis of urine drug testing outcomes in 470 patients, one observer determined that noncompliance with COT passed off in 45% [18]. No longer taking medicine as a reason for noncompliance can be due to the subsequent: affected person persona and beliefs, sociodemographic and environmental issues, affected person-clinician conversation, severity and chronicity of health problems, comorbid ailments, the complexity of the remedy plan, facet effect profile, and drug interactions [19,20].
Taking medicine now not as prescribed as a cause of lack of compliance can be due to trouble seeing a clinician, self-escalation, abuse, addiction, or diversion. All of these issues have good-sized scientific and/or societal results [2]. For patients on COT, urine drug checking out represents one of many methods to objectively monitor for compliance with a designated remedy plan, pick out substance misuse or abuse, and guide medical selections to retain or stop COT 21. Several studies have proven that affected person self-reports are notoriously erroneous, with patients underreporting or denying noncompliance or illicit drug abuse 12, 13, 17, [22].
Patients may additionally fail to file or underreport a beyond or present-day history of dependency or drug misuse for worry of not having their ache dealt with.23 Furthermore, profiling primarily based on an affected person's race, socioeconomic fame, and intercourse is bad at determining who could have a bizarre urine drug testing result 12, 17, 18, 22. The reason for acting urine drug trying out, which is to help the affected person care, has to be explained to the patient. Consent for urine drug checking must be included in an opioid remedy agreement earlier than the beginning of COT, thereby reducing a patient's confusion or surprise when a specimen is requested. Urine drug checking ought to be covered as part of a complete monitoring program that consists of getting access to prescription drug tracking software (PDMP), conducting tracking (self-escalation, reviews of misplaced or stolen prescriptions, frequent smartphone calls to the clinic, requesting an identical-day appointment for a top off, allegations of more than one drug intolerance or hypersensitive reactions), and documenting alleviation, functional improvement, and side effects. With suitable patient training, a thoughtfully dependent urine drug checking out coverage can beautify patient care via [23–25]. Optimizing medicine remedies presenting objectivity to the treatment plan Reinforcing therapeutic compliance with the affected person identifying substances that contribute to unfavorable events or drug-drug interactions figuring out the presence of an undisclosed substance(s) and/or absence of prescribed medicine(s), which recommend abuse, misuse, diversion, and dependency, to inspire appropriate behavioral adjustments helping the need for referral to an ache and/or addiction expert Complying with medico-legal policies, which demonstrate patient evaluation and monitoring
HISTORY OF URINE DRUG TESTING
To assure a drug-free workplace, the Mandatory Guidelines for Federal Workplace Drug Testing Programs came about as a result of the Federal Drug-Free Workplace Act established in the 1980s [26]. The five drugs tested in the federal urine drug test (UDT) included opiates, marijuana, cocaine, phenyl cyclohexyl piperidine (PCP), and amphetamine/methamphetamine. Urine drug testing as a means of deterring and detecting illicit drug use has expanded to non-federal workplaces, sports organizations, schools, forensics, and the healthcare arena 17. Unfortunately, the type of urine drug testing performed and the method of testing employed by federal testing is suboptimal for the healthcare setting. With federal testing, thresholds for the detection of opioids and illicit substances are higher than determined by statutes, the testing panel is limited with a fewer number of opioids tested, specimen collection may be witnessed, there is a strict chain of custody, and there is no therapeutic relationship between the individual and testing entity[27].
BODILY SPECIMENS THAT CAN BE TESTED
Why is urine tested as opposed to other bodily specimens, such as blood, hair, sweat, or saliva? Unless a patient has a neurogenic bladder requiring intermittent catheterization, urine is an easily obtainable noninvasive specimen, is cheap to analyze, can be analyzed with in-office point-of-care (POC) tests, is detectable for days, has higher concentrations of the parent drug and/or metabolites than serum, and has the most extensively published information and evidence for adherence testing.2,28 The disadvantages of urine specimens include ease of adulteration or substitution, a short to the intermediate window of detection, and difficulty for the same patients to spontaneously provide a specimen 28. Other bodily specimens possess apparent advantages, but limitations currently override widespread use as part of an adherence-monitoring program. Measuring the serum concentration of a drug is useful in the setting of an anuric patient or for detecting recent substance use, such as in an intoxicated patient seen in an Emergency Department setting. There are established testing methods for serum 28. The disadvantages of serum specimens are the cost, the narrow window of detectability (hours), and invasiveness 17, [28,29]. Because drawing blood requires a trained professional, the use of this specimen in the workplace setting is not practical or economically feasible 17.
Saliva concentrations reflect serum concentrations since the salivary gland is highly perfused [30]. In addition to being noninvasive, testing saliva offers more potential advantages compared to testing urine: ease of specimen collection, ability to witness collection in a less embarrassing manner than a urine specimen, low likelihood of adulteration, ability to target the parent drug instead of the metabolite, and availability as a POC test 28[31,32].
Saliva testing, however, has limitations: salivary concentration is influenced by salivary pH, with basic drugs being found in a higher concentration; low concentrations of a drug may be difficult to detect if the salivary volume is low; elution solvent must adequately remove the drugs adsorbed to the collection device; drug-interference patterns, cross-reactivity, and impact of potential adulterants are not as well studied; and because it reflects serum concentrations, the window of detectability is only hours.17,28,29,[33]. Hair checking out presents several benefits: minimally invasive; the specimen is easy to keep and transport; series can be discovered with minimal embarrassment or risk of substitution; and, relying on the period of the hair pattern, the window of detectability expands from days to months.28 scientific utility of hair trying out is challenged using several factors: doubtful dating to timing and dosing of the drug as opposed to hair length; problem detecting low-stage use and occasional drug concentrations; issue acquiring a sufficient hair pattern from those who shave or who otherwise have minimal frame hair; hair can be adulterated by way of the usage of dyes or different stylistic treatments; a drug is extra simply recognized in obviously happening darkish-colored hair as opposed to lighter colored or white hair, thereby inherently biasing check outcomes; and expensive and time-eating sample training 28,29,33. In the end, hair evaluation is without a doubt beneficial from a forensics angle because it's by a long way most dependable for detecting persistent drug use in choice to current and rare drug use 28, 29.
Sweat sorting out gives numerous blessings as it's far miles minimally invasive, tough to adulterate, and has a window of detectability that permits detection of a drug that has been used within 24 hours (the use of a sweat wipe) or as plenty as weeks (using a sweat patch worn for 1 to two weeks), which is right for tracking in a chemical dependency or probation software program.28,29 Sweat patch sorting out requires visits for software and removal of the patch.28 Drug detection is based upon the drug's diffusion from the vasculature to the sweat gland, molecular mass, pKa, protein binding, lipophilicity, in which the patch is carried out, patch adherence to the pores and pores and skin, and impact of capability adulterants.17 development of greater successful technical and clinical pointers is preferred for the one's alternate specimens earlier than they will be used for a urine specimen.
URINE DRUG TESTING
Complete urine drug checking out is a two-step system (desk, forty-six. 1). the first step is an enzyme-mediated immunoassay display (IAS), that's a qualitative take a look at used to decide the presence or absence of drug commands.21, 23 IASs were covered within the federal administrative center, taking a take a look at five lessons mandated by way of the Substance Abuse and intellectual health services management (SAMHSA): amphetamine, cocaine, marijuana, PCP, and opiates (restrained to codeine, morphine, and 6-mono acetyl-morphine to check for heroin use). maximum IASs completed in the clinical placing check for the identical "SAMOSA-five," but maintaining this kind of confined panel isn't endorsed{34} other capsules or schooling which includes benzodiazepines, barbiturates, methamphetamine, semi-synthetic opioids, methadone, and buprenorphine need to also be protected. As it's miles neither rate-inexperienced nor practical to check for all capsules and illicit substances, clinicians may also want to personalize their IAS and UDT panels to pick the most generally used tablets and illicit materials. Moreover, because it's miles very difficult to choose all drugs found in a specimen, let alone all pills within the identical elegance, astute clinicians have to apprehend that IAS isn't always definitive. Therefore, IAS may be very beneficial in making initial remedy selections. IASs usually work by measuring antigen-antibody reactions. Antigens—the loose drug and/or free drug metabolite and the identical classified drug and/or categorized drug metabolite—compete for antibodies to elicit an enzymatic reaction 20,34.
While the free drug and metabolite are absent, the classified drug and metabolite bind the antibody to save you enzymatic interest 20, 34. However, while the unfastened drug or metabolite is a gift, it displaces the labeled drug or metabolite to create an enzymatic response this is measurable and proportional to the attention of the loose drug or metabolite 20, 34. Regarding an IASs antigen- antibody specificity, this will account for why an immunoassay can be specific for particular drugs (e.g., it mainly detects the cocaine metabolite, benzodiazepines, which has a longer half of-life than cocaine itself and, if used as a valid topical anesthetic, does now not pass-react with another nearby anesthetic); it could be so particular for a particular drug that it excludes similar pills (e.g., it detects morphine and codeine, but no longer methadone); or it may be so nonspecific that it has trouble differentiating capsules in the equal elegance (e.g., it can not distinguish morphine from codeine, and it has low sensitivity for detecting semi-artificial opioids); or it cross reacts with unrelated drugs which are structurally similar enough to trigger a false positive for a particular drug class (Table 46.2) 23.
In addition to identifying illicit drugs, the advantages of an IAS are that it is cheap, yields result within minutes and is convenient (either performed with an in-office POC test or sent to a laboratory for testing) 17. Originally developed to monitor workers for illicit substance use, a POC IAS offers the advantage of a faster turnaround time (minutes to hours) while the patient could still be in the clinic than a laboratory-based IAS.12,28,[35] However, the performance of a POC IAS depends on having clinic personnel who are trained in precisely following the manufacturer's specific instructions (e.g., amount of sample to use, sample application, and timing of reaction) and understanding how to interpret the result given the test 'inherent limitations.12,17,21,28 Since a POC IAS is a non-instrumented test, in most cases, the person handling the "device" (dipstick, cup, card, or cassette) must visually interpret its appearance and manually enter the data into the electronic medical record 17. Because the interpretation of some devices depends on a color change after the application of a specimen, any delay in when the change is interpreted, even as little as 10 minutes, can result in an inaccurate analysis since the color can continue to change over time 26.
Some POC IASs, however, can be read by a meter, which then transfers the result electronically to the electronic health record 17. A POC IAS should also include a "control" to be certain that the device is working as expected 26. Because manufacturers of POC IASs may differ in terms of the threshold of detectability, specificity for identifying the target drugs of interest, reproducibility, and complexity of performing the test, the IAS results can differ between brand manufacturers. But not only can there be variations between brand manufacturers, but there can also be variations between manufactured lots/batches that can lead to differing outcomes 26,34. Studies have also demonstrated inconsistencies in product performance versus the manufacturer's claims and deviations in threshold reporting (identifying drugs or metabolites below the threshold or not identifying drugs or metabolites above the threshold) [36]. Crouch et al. compared the utility and accuracy of five POC IAS brand manufacturers (800 specimens) and found that each brand had a false-negative rate of less than 1% for all drug classes and false-positive rates of less than 0.25% (marijuana, cocaine, and opiates), less than 1.5% (PCP), and less than 1.75% (amphetamine) [37]. While studies comparing the accuracy of POC IASs and laboratory-based IASs suggest both provide similar results, limitations of the studies were because laboratory-trained personnel performed the POC IASs, and challenges in interpreting the results were greatest when at the lower end of the threshold of detectability 17.
For clinicians who opt to use a POC IAS, it must be certified by the Clinical Laboratory Improvement Amendments of 1988 (CLIA) depending on the Food and Drug Administration's (FDA) categorization of test complexity (e.g., waived, moderate, or high).34 Medicare reimbursement also requires this certification. A CLIA Certificate of Waiver can be obtained for those POC IASs that the FDA approves for use in non laboratory settings (e.nonlaboratoryg., home, and office) provided the POC IAS is easy to use and interpret and the clinic follows the manufacturer's explicit testing instructions.36 To minimize testing errors and maximize quality assurance, the Centers for Disease Control and Prevention's Division of Laboratory Systems created "Good Laboratory Practices for Waived Testing Sites "{38}. In addition, the manufacturers of POC IASs include specific instructions on specimen collection, handling, storage and testing, reference values, and recording of test results. It is up to the clinician to review the device manufacturer's instructions on whether confirmatory testing is required. Violating the manufacturer's instructions may otherwise invalidate the device's CLIA waived status 26. It is up to the clinician to determine which POC IAS is covered by any given insurance company and to compare the cost and expediency of performing a POC versus laboratory-based IAS. If an immediate result is required to assist in making a clinical decision while the patient is still in the clinic, POC IAS would be most prudent.
If an immediate result is not required, the clinician may consider sending the specimen to a laboratory. From an insurance standpoint, Medicare will not be reimbursed for either a POC IAS or laboratory-based IAS at the same clinical encounter 25. In a laboratory setting, the IAS is performed by licensed personnel using automated analyzers. The advantage of using an automated analyzer is that the results can be automatically exported to a patient's electronic health record. Similar to a POC IAS, a laboratory IAS is also subject to variability among testing products. Because of its complex regulatory status (ensuring both quality control of the test product and analyzer and proficiency of the personnel doing the testing) and overhead (equipment and trained personnel), the cost and reimbursement are higher than with a POC IAS.26.
The second step in comprehensive urine drug testing is a confirmatory UDT, which requires laboratory-based testing using gas chromatography-mass spectrometry (GC/MS) or liquid chromatography-mass spectrometry (LC/MS). A UDT is a quantitative test that adds a much higher level of sensitivity and specificity than an IAS and identifies, where applicable, the specific drug within a class as well as the drug metabolite and drug concentration. Unlike IAS, most UDTs identify codeine, fentanyl, hydrocodone, hydromorphone, methadone, morphine, oxycodone, and oxymorphone. This is especially important for patients with chronic pain who take more than one type of opioid. With some opioids, the metabolism of the parent opioid can lead to a metabolite of an alternate opioid (e.g., codeine is metabolized to morphine, and oxycodone is metabolized to oxymorphone). If both a prescribed drug and its metabolite or only the metabolite are identified on an IAS, it is unclear if the patient is compliant with abusing non-prescribed opioids and highlights why one might see conflicting results with an IAS versus UDT.17 Because an IAS is only a qualitative screening test, quantitative confirmatory testing with a UDT should also be performed as part of the adherence-monitoring program. From a simple technical standpoint, chromatography is a separation method that involves both a stationary phase (contained with the analytical column) and a mobile phase (gas or liquid) 26. A drug that does not interact with the stationary phase exits at the identical float rate because of the cell section, whereas a drug that interacts with the stationary section is retained in the column and exits at a slower fee. There are two types of chromatography techniques: gas chromatography (GC) and liquid chromatography (LC).
There are two varieties of LC: extremely-overall performance liquid chromatography (UPLC) and high-performance liquid chromatography (HPLC). Whilst GC is the most popular technique, UPLC is more and more a way of preference because of its many blessings over GC: greater sensitivity, the requirement for a smaller sample length, faster analysis time, and much less onerous sample guidance time 20. Even as chromatography separates the analyte in a specimen, mass spectroscopy (MS) can be blended with both GC and LC to discover the unique molecular shape of man or woman tablets and/or metabolites primarily based on their mass- to-price ratio alongside their concentrations 17. Due to the fact GC/MS and LC/MS allow for qualitative and quantitative analysis of the specimen, ambiguous immunoassay effects may be resolved by using reading the attention of both the parent compound and its metabolite.
This improvement in sensitivity and specificity allows for the discount of fake positives and negatives. The secondary gain to testing for drug metabolites is the extra guarantee that the drug has been eaten up and processed using the body, while the presence of only the discerned drug does not rule out the opportunity that the urine specimen was adulterated by immediately including the discern drug 26.
THRESHOLD OF DETECTABILITY
The accuracy of each IAS and UDT in figuring out a drug or illicit substance relies upon several factors: the IAS's antigen- antibody specificity, the drug concern traction in the pattern, and the edge of detectability 17. wherein a laboratory chooses to set its threshold of detectability, might also depend upon whether or not it is being run for nonclinical (e.g., administrative center or forensic) purposes as opposed to clinical purposes. Unlike nonclinical testing requirements, the threshold concentrations in an ache control setting aren't standardized, 26.
The threshold for detectability can be intentionally set higher in nonclinical testing to yield the fewest fake negatives and false positives. As an example, morphine's thresh antique detection is ready at 2000 ng/mL for a workplace IAS as compared with 300 ng/mL for a scientific IAS or 50 ng/mL for a clinical UDT.2 When the drug is absent or the concentration falls beneath the brink of detectability, the result is reported as poor, and where the concentration rises above the threshold of detectability, it's far stated as positive.39 For medical purposes, one must ask the lab to set the lowest threshold of detectability to yield the truest positives and fewest false negatives. But, the byproduct of placing a low threshold is that a higher sensitivity may additionally detect impurities or contaminants associated with the producing process; for example, two exceptional opioids manufactured using the same device may additionally bring about low ranges of a non-prescribed opioid being detected on an IAS despite the usage of simplest one prescribed opioid. Whilst a laboratory should conceivably take away its threshold, there is a certain awareness underneath which a laboratory can not reliably file the result with an excessive degree of self-belief, thereby yielding inaccurate effects 17.
WHY perform AN IMMUNOASSAY screen AND URINE DRUG take look at even though a diabetic-affected person may additionally self-file precise glycemic management, clinicians nonetheless periodically acquire laboratory tests to objectively affirm the retrospective degree of diabetes management without accusing the affected person of mendacity 27. This notion system must.
Be of no kind regarding the appearance of an IAS and UDT in the affected person on COT. This currently consists of not the most effective tracking for prescribed opioid(s), but additionally for non- prescribed opioid(s) or illicit substances that discover a treatable substance use disease. For some sufferers, understanding they may be being monitored may additionally support a healthful way of life changes; and for those who engage in or relapse into complex drug use, a sudden take look at the result may additionally provide a secure harbor for the affected person and/or clinician to explore treatment options for a substance use disorder 27.
A checking-out policy also indicates to patients, law enforcement government, and regulatory authorities that one's vigilance demonstrates a commitment to the affected person and network safety.27 Used in this way, checking out is one device that can be used in supporting the clinician's selection to hold as opposed to discontinue COT. caution should be exercised whilst interpreting take a look at results because of barriers related to thresholds of detection, variables that affect drug awareness (Pharmacokinetics, pharmacodynamics, and pharmaco genetics), and variables associated with the specimen itself (series, managing, and evaluation).{forty} those obstacles and variables also, in component, explain why there is no scientifically confirmed dating among the urine drug concentration and the quantity of drug taken, the timing of whilst the drug became closing taken, or the supply of the drug 24. WHO should GET AN IMMUNOASSAY display AND URINE DRUG.
Take a look at although every affected person is entitled to pain management, COT Will not be the safest option for each affected person. While a chance-advantage analysis shows blessings outweigh dangers, checking out should be done in patients In whom a trial of COT is being taken into consideration, sufferers already on COT Who’s moving care, sufferers in whom an opioid rotation is being taken into consideration, patients being stated an ache specialist, as part of a random procedure to reveal for adherence, or whilst behavioral symptoms advocate problem (lost or stolen medications, requests for early refills, unannounced sanatorium visits, Doctor shopping, frequent Emergency department visits, request for positive Opioid, or allergic reactions to a couple of opioids).23, 24, [41] people who are Applicants for COT and consent to their use have to also accept responsibility for the use of the medicine as prescribed and under the situations stipulated through the opioid agreement the affected person signs with the clinician 27. One of the Conditions of prescribing COT have to consist of the affected person consenting to Random testing. Whilst the patient is within his or her right to the item to random try out, the clinician isn't obligated to initiate or retain COT because opioid analgesics are not required. For the patient who states he/ she cannot post a specimen (e.g., because he/she already urinated, is overdue for paintings, or has any other appointment to attend), alternatives consist of maintaining the prescription until the the patient can urinate later that day vs. giving a constrained delivery (e.g., one-day supply) and making further refills contingent upon filing a specimen inside a special length (e.g., 24 hours) 27. A uniform exercise coverage allows keeping away from bias in figuring out who receives tested and reducing the stigma related to filing an IAS and UDT 23.
Whilst TO GET AN IMMUNOASSAY screen AND URINE DRUG take a look at Katz et al. showed that urine drug checking out simplest for those who have a history of addiction or show off aberrant conduct missed a vast variety of patients who had unexpected test consequences 22. To confirm the veracity of a patient's responses to questions about his/her controlled substance use or illicit substance use, the clinician should consider obtaining an IAS and UDT before the initiation of COT, upon inheriting a patient who is already on COT that was initiated by another provider, before dose escalation or opioid rotation, in those instances when a patient exhibits behaviors or makes statements that may arouse suspicion, and randomly during maintenance therapy as part of an ongoing monitoring program 23. Combining urine drug testing with other monitoring techniques makes the most sense.21 Using this formulaic approach helps the clinician avoid profiling and stigmatizing patients based on race, ethnicity, socioeconomic status, or gender.
HOW OFTEN TO GET AN IMMUNOASSAY SCREEN AND URINE DRUG TEST
Because there is no evidence-based guideline that pinpoints which patients with chronic pain should be tested, definitive criteria for the frequency of testing do not exist. The frequency of testing beyond baseline urine drug testing is left to the clinician's discretion based on individual patient needs and documented medical necessity. 25 Table 46.3 shows one suggestion for monitoring frequency 25[42,43]. However, some state guidelines may suggest or require certain frequencies.
Fig Monitoring Recommendation Table 46.2
ADULTERATION
Some patients intentionally attempt to subvert the tests using simple or sophisticated adulteration techniques, products, or devices. Based on an analysis of 2.1 million patient urine drug testing reports, the most recent annual Quest Diagnostics Health Trends Prescription Drug Monitoring Report 2015 revealed that 33,396 (1.5%) urine specimens were adulterated by various means (Fig. 46.3).15 Information On adulteration techniques, products, and devices can be readily found online. Products purchased on the internet can be broadly categorized as "cleansing" agents (e.g., Extra Clean; Green Clean), additives, synthetic urine substitutes, and prosthetic devices.39 [57–59] There are commercially available products that claim to dilute the urine, or patients will ingest copious amounts of water for the same purpose, thereby decreasing the concentration of substances that could be detected 17. Because ingestion of large volumes of water can result in pale- colored urine, some patients will concurrently consume niacin and/or vitamin B to give the urine a yellow color. Urine specimens can also be directly adulterated by adding water from a sink or toilet bowl, household products (bleach, vinegar, and detergent), over-the-counter medications (eye drops and sodium chloride), and commercially purchased chemicals (glutaraldehyde, sodium or potassium nitrite, pyridinium chlorochromate, or peroxide/ peroxidase) 34,[60].
CONCLUSION
An estimated 100 million Americans experience chronic pain and 1.9 million aged 12 or older have a pain reliever use disorder 1, 14. The expansion of opioid prescribing for CNMP has led to a concurrent increase in diversion and deaths from overdose 10, [61, 62]. To preserve legitimate access to these potent but potentially dangerous pain relievers, clinicians must employ tools to guard against diversion, misuse, and abuse. Patient selection is critical for identifying individuals who are most likely to benefit from COT. Risk stratification based on detailed history and physical examination can help determine the level of surveillance needed to monitor compliance or avoid prescribing drugs in the first place. Tools such as PDMP and urine drug testing objectively evaluate adherence to a COT plan and can help support clinical decisions. Given the dire need to reduce risk related to opioid-related adverse events, various medical societies and regulatory agencies have erred.
Explain the absence of opioids), diversion, self-remedy of aches, mental health problems, drug abuse, or addiction. 44 Consequently, the examinations must be used alongside other tools to assist clinicians in developing, continuing, or editing treatment plans. For those who have a dual analysis of dependency and chronic pain, neither disease can be addressed in isolation and the simplest remedy for each illness will efficiently treat the alternative 23. Urine drugs trying out must be protected as a part of risk assessment and mitigation plans. Similar to other hazard management protocols (e.g., checking blood pressure to display for high blood pressure, acting pap smears to prevent cervical cancer, and performing colonoscopies to prevent colon cancer), urine drug testing may be coupled with scientific findings to enhance patient care.27 in the end, Having a urine drug checking out a policy may additionally make one's exercise less appealing to individuals focused on physicians for opioids and may endorse regulation enforcement and regulators that one has a risk management protocol in the area.
The recent Institute of Medicine report declared that greater than 100 million Americans have chronic pain with an associated cost of up to $635 billion each year due to medical treatment and lost productivity. [1,2] The explosion in opioid prescribing since the 1990s has resulted in opioids being the most frequently prescribed medication in the United States.[3–5] Manchikanti et al.'s study revealed a 149% increase in retail opioid sales and a 402% increase in average sales of opioids per person in the United States from 1997 to 2007.[6] Looking at 1999–2012, the National Center for Health Statistics found that among patients aged 20 or older prescription opioid use increased from 5% to 6.9% until 2006 and then stayed at 6.9% until 2012.7 However, the percentage of patients using an opioid stronger than morphine dramatically increased from 17% to 37% over the same period. [7] Among the top 25 dispensed prescriptions in the United States, hydrocodone, tramadol, and oxycodone accounted for 1, 21, and 22 dispensed prescriptions, respectively.5 Opioids represent one of many therapeutic options to treat chronic nonmalignant pain (CNMP), but their widespread use has resulted in a concomitant increase in misuse, abuse, addiction, overdose, and diversion. While there is evidence of the short-term benefit of opioids for treating pain based on randomized trials lasting less than 3 months, there are few studies reporting outcomes at 12months or longer. [8,9]
Acknowledgement
The completion of this research project would not have been possible without the contributions and support of many individuals and organizations. We are deeply grateful to all those who played a role in the success of this project. I would also like to thank My Mentor [Dr. Naweed Imam Syed Prof. Department of Cell Biology at the University of Calgary and Dr. Sadaf Ahmed Psychophysiology Lab University of Karachi for their invaluable input and support throughout the research process. Their insights and expertise were instrumental in shaping the direction of this project
Declaration of Interest
I at this moment declare that I have no pecuniary or other personal interest, direct or indirect, in any matter that raises or may raise a conflict with my duties as a manager of my office Management
Conflicts of Interest
The authors declare that they have no conflict of interest. Financial support and sponsorship No Funding
References
- Institute of Drugs (IOM) of the countrywide Academy of Sciences (NAS): Relieving pain in the USA: A Blueprint for Transforming Prevention, Care, schooling, and Studies. Washington, DC, 2011, Countrywide Academies Press, p. 364.
- Christo, P. J., Manchikanti, L., Ruan, X., Bottros, M., Hansen, H., Solanki, D. R., ... & Colson, J. (2011). Urine drug testing in chronic pain. Pain physician, 14(2), 123.
- Olsen, Y., Daumit, G. L., & Ford, D. E. (2006). Opioid prescriptions by US primary care physicians from 1992 to 2001. The Journal of Pain, 7(4), 225-235.
- Zerzan, J. T., Morden, N. E., Soumerai, S., Ross-Degnan, D., Roughead, E., Zhang, F., ... & Sullivan, S. D. (2006). Trends and geographic variation of opiate medication use in state Medicaid fee-for-service programs, 1996 to 2002. Medical care, 44(11), 1005-1010.
- IMS health. National Prescription Audit. 2013.
- Manchikanti, L., Fellows, B., Ailinani, H., & Pampati, V. (2010). Therapeutic use, abuse, and nonmedical use of opioids: a ten-year perspective. Pain physician, 13(5), 401.
- Frenk, S. M., Porter, K. S., & Paulozzi, L. (2015). Prescription opioid analgesic use among adults: United States, 1999-2012 (No. 2015). US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Health Statistics.
- Dowell, D., Haegerich, S. M., Chi, R., & Data, M. CDC Guidelines available Ordaining Opioids for Chronic Pain— United States, 2016.
- Chou, R., Turner, J. A., Devine, E. B., Hansen, R. N., Sullivan,S. D., Blazina, I., ... & Deyo, R. A. (2015). The effectiveness and risks of long-term opioid therapy for chronic pain: a systematic review for a National Institutes of Health Pathways to Prevention Workshop. Annals of internal medicine, 162(4), 276-286.
- Dowell, D., Haegerich, T. M., & Chou, R. (2016). CDC guideline for prescribing opioids for chronic pain—United States, 2016. Jama, 315(15), 1624-1645.
- a couple of purposes for death facts. 2014. (Accessed June 11,2016, at http://wonder. cdc.gov/mcd.html.)
- McCarberg, B. H. (2011). A critical assessment of opioidtreatment adherence using urine drug testing in chronic pain management. Postgraduate medicine, 123(6), 124-131.
- Fleming, M. F., Balousek, S. L., Klessig, C. L., Mundt, M. P., & Brown, D. D. (2007). Substance use disorders in a primary care sample receiving daily opioid therapy. The journal of pain, 8(7), 573-582.
- Center for Behavioral Fitness information, and exceptional. Behavioral fitness tendencies within the united states: effects from the 2014 Countrywide Survey on Drug Use and Health (HHS e Book No. SMA 15–4927, NSDUH collection H-50). 2015.
- Blatt, A., Chen, Z., McClure, L., Niles, J., & Kaufman, M. (2015). Prescription drug misuse in America: diagnostic insights in the continuing drug epidemic battle. Quest Diagnostics Health Trends Prescription Drug Monitoring Report, Quest Diagnostics.
- Bhamb, B., Brown, D., Hariharan, J., Anderson, J., Balousek, S., & Fleming, M. F. (2006). Survey of select practice behaviors by primary care physicians on the use of opioids for chronic pain. Current medical research and opinion, 22(9), 1859-1865.
- Hammett-Stabler, C. A., and L. Webster. "Urine drug testing: augmenting pain management & enhancing patient care." PharmaCom Group (2008).
- Michna, E., Jamison, R. N., Pham, L. D., Ross, E. L., Janfaza,D., Nedeljkovic, S. S., ... & Wasan, A. D. (2007). Urine toxicology screening among chronic pain patients on opioid therapy: frequency and predictability of abnormal findings. The Clinical journal of pain, 23(2), 173-179.
- Robiner, W. N. (2005). Enhancing adherence in clinical research. Contemporary clinical trials, 26(1), 59-77.
- Carlozzi, A. F., Fornari, F. A., Siwicki, D. M., Barkin, R. L.,Nafziger, A. N., Woodcock, M., ... & Empting, L. D. (2009). Urine drug monitoring: opioids.
- Webster LR: The role of Urine Drug testing in continual pain control: 2013 update. Big Apple: New York, 2013; McMahon Publishing, p.. 6.
- Katz, N. P., Sherburne, S., Beach, M., Rose, R. J., Vielguth, J., Bradley, J., & Fanciullo, G. J. (2003). Behavioral monitoring and urine toxicology testing in patients receiving long-term opioid therapy. Anesthesia & Analgesia, 97(4), 1097-1102.
- Heit, H. A., & Gourlay, D. L. (2004). Urine drug testing in pain medicine. Journal of pain and symptom management, 27(3), 260-267.
- Gourlay D, Heit H Urine drug trying out in scientific exercise. Dispelling the Myths and Designing the Techniques. 2006 ed., Stamford, CT, 2006, PharmaCom Group, Inc., p 28.
- centers for Medicare & Medicaid Services: nearby insurance dedication (LCD): controlled substance tracking and drug abuse testing (L36668). Rockville, MD, 2016, U.S. Department of Health and Human Services 25.
- McMillin, G. A., Slawson, M. H., Marin, S. J., & Johnson- Davis, K. L. (2013). Demystifying analytical approaches for urine drug testing to evaluate medication adherence in chronic pain management. Journal of Pain & Palliative CarePharmacotherapy, 27(4), 322-339.
- Heit, H. A., & Gourlay, D. L. (2015). Using urine drug testing to support healthy boundaries in clinical care. Journal of Opioid Management, 11(1), 7-12.
- Substance Abuse and intellectual health offerings management: medical Drug Checking out in number one care, Rockville, MD, 2012, U.S. Department and Human offerings, p 96.
- Caplan, Y. H., & Goldberger, B. A. (2001). Alternative specimens for workplace drug testing. Journal of Analytical Toxicology, 25(5), 396-399.
- Nichols, J. H., Christenson, R. H., Clarke, W., Gronowski, A., Hammett-Stabler, C. A., Jacobs, E., ... & Zucker, M.L. (2007). Executive summary. The National Academy of Clinical Biochemistry Laboratory Medicine Practice Guideline: evidence-based practice for point-of-care testing. Clinica chimica acta, 379(1-2), 14-28.
- Cone, E. J., & Huestis, M. A. (2007). Interpretation of oral fluid tests for drugs of abuse. Annals of the New York Academy of Sciences, 1098(1), 51-103.
- Cone, E. J. (2006). Oral fluid testing: new technology enables drug testing without embarrassment. Journal of the California Dental Association, 34(4), 311-315.
- Yacoubian, G. S., Wish, E. D., & Perez, D. M. (2001). A comparison of saliva testing to urinalysis in an arrestee population. Journal of psychoactive drugs, 33(3), 289-294.
- DePriest, A. Z., Black, D. L., & Robert, T. A. (2015). Immunoassay in healthcare testing applications. Journal of opioid management, 11(1), 13-25.
- Laxmaiah Manchikanti, M. D., Sairam Atluri, M. D., & Andrea Trescot, M. D. (2008). Monitoring opioid adherence in chronic pain patients: Tools, techniques and utility. Pain Physician, 11, 1.
- Melanson SE. Drug-of-abuse assessment out of the factor of care. Clin Lab Med 29:503–509, 2009.
- Crouch, D. J., Hersch, R. K., Cook, R. F., Frank, J. F., & Walsh, J. M. (2002). A field evaluation of five on-site drug- testing devices. Journal of analytical toxicology, 26(7), 493- 499.
- Gudin J: realistic considerations in Urine Drug Screening: Compliance and Rules for workplace-based testing. Big Apple: New York, 2014; McMahon Publishing, p.. 6.
- Reisfield, G. M., Salazar, E., & Bertholf, R. L. (2007). Rational use and interpretation of urine drug testing in chronic opioid therapy. Annals of Clinical & Laboratory Science, 37(4), 301- 314.
- Nafziger, A. N., & Bertino Jr, J. S. (2009). Utility and application of urine drug testing in chronic pain management with opioids. The Clinical journal of pain, 25(1), 73-79.
- Gourlay D, Heit H, Caplan Y: Urine Drug testing in clinical practice: The artwork and science of patient Care. 2015 ed., Stamford, CT, 2015, PharmaCom group, Inc, p 32.
- Washington nation corporation scientific directors' Institution: Interagency guiding principle on Prescribing Opioids for pain control ed 3. Olympia, WA, 2015, the Nation of Washington branch of health, and 105.
- Webster, L. R., & Webster, R. M. (2005). Predicting aberrant behaviors in opioid-treated patients: preliminary validation of the Opioid Risk Tool. Pain medicine, 6(6), 432-442.
- Chou, R., Fanciullo, G. J., Fine, P. G., Adler, J. A., Ballantyne,J. C., Davies, P., ... & Miaskowski, C. (2009). Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. The journal of pain, 10(2), 113-130.
- Fishman S is responsible for opioid prescription: a clinician’s guide. Revised and elevated. ed 2. Washington DC (2014) Waterford lifestyle sciences.
- Turner, J. A., Saunders, K., Shortreed, S. M., Rapp, S. E., Thielke, S., LeResche, L., ... & Von Korff, M. (2014). Chronic opioid therapy risk reduction initiative: impact on urine drug testing rates and results. Journal of general internal medicine, 29, 305-311.
- Berland, D., & Rodgers, P. (2012). Rational use of opioids for management of chronic nonterminal pain. American Family Physician, 86(3), 252-258.
- Popish SJ, Wells DL, Kimura H, Yee M, Christopher MLD: ache control Opioid protection education manual. Washington, DC, 2014, US branch of Veterans Affairs, p 24.
- Hooten WM, Timming R, Belgrade M, et al: assessment and control of chronic aches. ed 6. Bloomington, MN, 2013, Institute for medical systems improvement, p 106.
- Utah Branch of Health: Utah medical pointers on Prescribing Opioids for remedy of ache. Salt Lake City, UT 2009; Utah Department of Fitness, p. 92.
- American University of Occupational and Environmental Medicinal Drug: ACOEM recommendations for continued Use of Opioids. Elk Grove Village, IL, 2011, ACOEM, p 25.
- Manchikanti L, Abdi S, Atluri S, et al: American Society of Interventional pain medical doctor's (ASIPP) tips for accountable opioid prescribing in continual non-most cancers pain: part I— proof assessment. ache Phys 15:S1–S65, 2012.
- Furlan, A. D., Reardon, R., & Weppler, C. (2010). National Opioid Use Guideline Group. Opioids for chronic noncancer pain: a new Canadian practice guideline. CMAJ, 182(9), 923- 30.
- Nuckols, T. K., Anderson, L., Popescu, I., Diamant, A. L., Doyle, B., Di Capua, P., & Chou, R. (2014). Opioid prescribing: a systematic review and critical appraisal of guidelines for chronic pain. Annals of internal medicine, 160(1), 38-47.
- clinical Board of California: suggestions for Prescribing controlled materials for pain. Sacramento, CA, 2014, Scientific Board of California, p. 96.
- Starrels, J. L., Becker, W. C., Alford, D. P., Kapoor, A., Williams, A. R., & Turner, B. J. (2010). Systematic review: treatment agreements and urine drug testing to reduce opioid misuse in patients with chronic pain. Annals of internal medicine, 152(11), 712-720.
- Pesce, A., West, C., Egan City, K., & Strickland, J. (2012). Interpretation of urine drug testing in pain patients. Pain Medicine, 13(7), 868-885.
- always test smoothly. (Accessed May also 16, 2016, at http:// alwaystestclean.com/detoxify-extra-clean-instructions/.)
- Detoxify LLC. (Accessed May additionally sixteen, 2016, at http://www.detoxify commercial enterprise.com/ cellular/ client-Questions-answers.html.)
- Jaffee, W. B., Trucco, E., Levy, S., & Weiss, R. D. (2007). Is this urine really negative? A systematic review of tampering methods in urine drug screening and testing. Journal of Substance Abuse Treatment, 33(1), 33-42.
- Gilson, A. M., Ryan, K. M., Joranson, D. E., & Dahl, J. L. (2004). A reassessment of trends in the medical use and abuse of opioid analgesics and implications for diversion control: 1997–2002. Journal of pain and symptom management, 28(2), 176-188.
- Rudd, R. A., Aleshire, N., Zibbell, J. E., & Gladden, R. M. (2016). Increases in drug and opioid overdose deaths—United States, 2000–2014. American Journal of Transplantation, 16(4), 1323-1327.
- Chen, W. J., Fang, C. C., Shyu, R. S., & Lin, K. C. (2006).
- Underreporting of illicit drug use by patients at emergency departments as revealed by two-tiered urinalysis. Addictive behaviors, 31(12), 2304-2308.


