Research Article - (2026) Volume 2, Issue 1
To Compare the Manual Liquid-Based Cytology (MLBC) Technique on Fine-Needle Aspiration Cytology (FNAC) Material with Conventional Technique
2Senior Resident,Department of Pathology, MM collage of medical Sciences & research, Ambala, Haryana, India
3Professor, Department of Pathology, MM collage of medical Sciences & research, Ambala, Haryana, India
Received Date: Mar 02, 2026 / Accepted Date: Apr 10, 2026 / Published Date: Apr 15, 2026
Copyright: ©2026 Dr. Dayal Singh Bisht, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Bisht, D. S., Sanjyokta., Gulia, S. P. (2026). To Compare the Manual Liquid-Based Cytology (MLBC) Technique on Fine-Needle Aspiration Cytology (FNAC) Material with Conventional Technique. J Path Lab Med, 2(1), 01-07.
Abstract
Liquid Based Cytology (LBC) is a monolayer slide preparation technology that has outperformed conventional pap smears due to improved fixation, reduced masking factors and standardised cell transfer. In LBC, samples are collected by total immersion of the sampling apparatus in a vial containing a preservative solution, with cells being preserved and fixed at the same time, as opposed to conventional smears where the sample is sprayed onto a glass slide and fixed separately. Two main liquid preparation methods are currently known - ThinPrep and SurePath. In our study we used MLBC in fine-needle aspiration cytology (FNAC). It aimed to observe morphological characteristics, compare the outcomes of direct smear examinations with those of manual liquid-based smears, and correlate these findings with histopathological results in cases where a biopsy was also performed.
AIM: The aim was to establish manual liquid-based cytology (MLBC) technique on fine-needle aspiration cytology (FNAC) material and compare its results with conventional technique.
Material and Methods: In this progressive study, a total of 100 fine needle aspiration (FNA) samples from various anatomical sites were examined using MLBC and CS preparations. Direct smears and MLBC smears were assessed for cellularity, background, cellular preservation, and nuclear preservation. The slides were reviewed independently by two cytologists, each with more than 5 years of experience. The ANOVA test was utilized for statistical analysis. A p-value of less than 0.05 is considered statistically significant.
Result: The cellularity observed in MLBC was marginally lower than that found in conventional smears; nevertheless, the cellularity, in conjunction with the quality of nuclear assessment, background, and necrosis, is less evident when compared to traditional methods. Overlapping of nuclei is reduced, and occurrences of hemorrhage and necrosis are lessened, facilitating a better examination of cell morphology through the MLBC technique. The P value obtained was <0.05, which is considered statistically significant.
Conclusion: The process of preparing MLBC in FNAC is a method that is secure, uncomplicated, and time-efficient, potentially providing considerable diagnostic benefits when evaluating FNA samples from various anatomical sites. However, it is recommended to employ both MLBC and CS preparations to maximize the diagnostic yield.
Keywords
Conventional Smears (CS), Fine-Needle Aspiration (FNA) Cytology, Liquid-Based Cytology (LBC), Manual Liquid Based Cytology (MLBC)
Introduction
Liquid Based Cytology (LBC) is a monolayer slide preparation technology that has outperformed conventional pap smears due to improved fixation, reduced masking factors and standardised cell transfer [1]. In LBC, samples are collected by total immersion of the sampling apparatus in a vial containing a preservative solution, with cells being preserved and fixed at the same time, as opposed to conventional smears where the sample is sprayed onto a glass slide and fixed separately. Two main liquid preparation methods are currently known - ThinPrep and SurePath [2]. The two methods differ in the principles of cell collection, but produce similar products. SurePath operates on the principle of gradient sedimentation. In this procedure, the sample is vortexed and strained to break up the mucus and the large cell groups, and then treated with a gradient centrifugation process to remove the blood and debris. The cell pellet is reconstituted and allowed to settle on the glass slide. The advantages of liquid cytology include increased sensitivity and specificity as the fixation is better and the nuclear details are preserved. Abnormal cells are not covered or diluted by other epithelial cells or by inflammatory cells. Therefore, there is a lower incidence of non-compliant cervical cytology samples. Residual cell suspension may be used for the preparation of other cytological products or for other tests such as the detection of DNA of human papillomavirus (3-4). In our study we used MLBC in fine-needle aspiration cytology (FNAC). It aimed to observe morphological characteristics, compare the outcomes of direct smear examinations with those of manual liquid-based smears, and correlate these findings with histopathological results in cases where a biopsy was also performed.
Material & Methods
The prospective study received approval from the Institutional Ethical Committee at MM College of Medical Sciences & Research in Sadopur, Ambala, conducted by the Department of Pathology from August 2024 to August 2025. Informed consent was secured from all patients prior to the commencement of the study. A total of 100 Fine Needle Aspirations (FNAs) from various locations, including lymph nodes, thyroid, breast, salivary glands, and soft tissue, were incorporated into the study. For each site, FNA was executed using a 23-gauge needle attached to a 05 mL syringe. Two passes were performed for each case; the first pass was designated for conventional smear (CS), while the second was for material for MLBC preparations. All cases that allowed for a definitive diagnosis using conventional techniques were included, whereas cases with insufficient material for diagnosis by either method were excluded. For CS, the sample was directly placed on a slide to create smears. One smear was wet-fixed in 95% ethyl alcohol and stained using the Pap method. An air-dried smear was prepared for MGG staining. The MLBC samples were collected in vials containing an ethanol-based preservative, which simultaneously preserves and fixes the sample. The material underwent centrifugation at 1,500 rpm for five minutes and then process the sample in smi automated BD SurePath™ which operates on the principle of density gradient sedimentation. In this process, the cell/sample is vortexed and strained to disrupt mucus and large cell aggregates, followed by treatment through a density gradient centrifugation process to eliminate blood and debris. The cell pellet is then resuspended and allowed to sediment onto a glass slide. Wet-fixed smears were stained using the Pap method, while air-dried smears underwent MGG staining. The CS and MLBC preparation parameters were evaluated using a scoring system based on various criteria. These criteria were categorized into cellularity, background, cellular preservation, and nuclear preservation, as utilized by Arul P et al.16 in their study.
Results
Among the 100 FNA samples, 47 cases received surgical intervention, and the corresponding final histopathological diagnoses were obtained. Histopathological correlation could be done only for 47 cases (47%) out of 100. This study involved a comparison of fine-needle aspiration cytology (FNAC) diagnoses of CS and MLBC preparations with their respective histopathological diagnoses. A total of one hundred cases were analyzed in our research. All cases exhibited consistent diagnoses when assessed using both conventional and MLBC methods. The cellularity observed in MLBC was slightly less as compared to conventional smears, and the nuclei exhibited less overlap, facilitating a more detailed examination of cell morphology through MLBC. For the background analysis, we employed standard error of proportion statistics for the derived data. In MLBC, instances of hemorrhage and necrosis were diminished in comparison to conventional methods, resulting in a P value. The P value obtained is <0.05, indicating a highly significant result.
Table 1: ANOVA Test for Backgrounds Surrounding in Conventional And MLBC
|
Surrounding |
Conventional N-100 |
MLBC N-100 |
F value |
P value |
|
Necrosis |
30 |
10 |
16.32 |
0.0003 |
|
Fibromixoid |
14 |
04 |
7.49 |
0.0081 |
|
Mucin |
15 |
05 |
8.67 |
0.0052 |
|
Blood |
36 |
15 |
22.58 |
0.0001 |
|
Colloids |
05 |
01 |
4.21 |
0.0456 |
Table 2: ANOVA Test for Scoring System in Conventional and MLBC
|
Scoring system |
Conventional(n100) |
MLBC(n100) |
f value |
P value |
|
Cellularity |
96 |
85 |
7.33 |
0.007 |
|
Cell architecture |
86 |
94 |
4.57 |
0.034 |
|
Monolayered sheets |
82 |
92 |
5.88 |
0.016 |
|
Nuclear detail |
84 |
96 |
7.20 |
0.008 |
|
Cytoplasmic detailed |
85 |
96 |
6.49 |
0.012 |
Table 3: Scoring System used in Various FNAC Smears
|
CYTOLOGY FEATURE |
SCORE 1 |
SCORE 2 |
SCORE 3 |
SCORE 4 |
|
Cellularity |
Nil |
Scanty |
Adequate |
Abundant |
|
Background blood, cell debris |
Nil |
Occasional |
Good amount |
Abundant |
|
Cell architecture |
Not recognized |
Partially recognized |
Well recognized |
Cell architecture |
|
Nuclear detail |
Poor |
Fair |
Good |
Very good |
|
Cytoplasmic detail |
Poor |
Fair |
Good |
Very good |
Figure 1: Comparison of Necrotic Background in Case of Squamous cell Carcinoma in Conventional and Manual Liquid-Based Cytology Technique
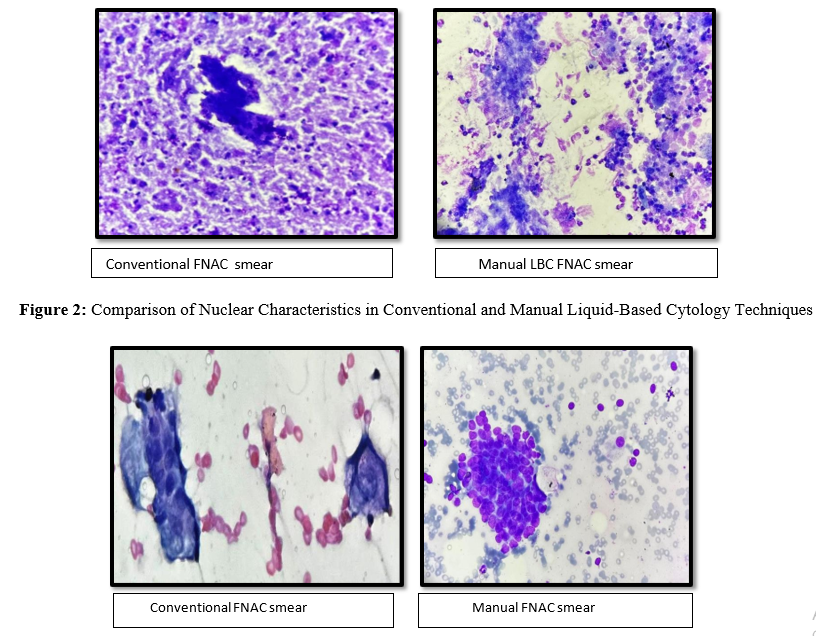
Figure 3: Comparison of Cellularity Background in Case of Diffuse Large B –Cell Lymphoma in Conventional and Manual Liquid-Based Cytology Technique
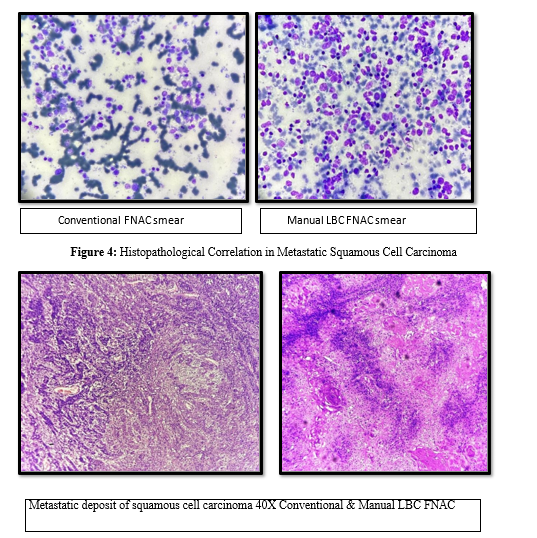
Table 4: Cases Dignosed on Conventional FNAC, Manual LBC and Correlation in Histopathology
|
Organ |
Total No of cases |
Convention FNAC |
Manual LB FNAC |
H&E |
|
Breast |
Fibroadenoma |
12 |
Same |
12 |
|
|
Benign breast disease(fibrocystic disease of breast) |
05 |
|
|
|
|
Carcinoma of breast |
04 |
|
4 |
|
Lymph node |
Reactive lymphadenitis |
08 |
Same |
08 |
|
|
Granulomatous lesion |
07 |
|
05 |
|
|
Lymphoma/Metastatic deposit |
12 |
|
10 |
|
Salivary glands |
Pleomorphic adenoma |
04 |
Same |
03 |
|
|
Warthin tumor |
01 |
|
|
|
|
Mucoepidermoid carcinoma |
03 |
|
01 |
|
Soft tissue swelling |
Lipoma |
06 |
Same |
|
|
|
Epidermal inclusion cyst |
08 |
|
|
|
Thyroid |
Colloid goitre |
20 |
Same |
|
|
|
|
|
|
|
|
|
Atypia of undetermined significant |
01 |
|
|
|
|
Follicular neoplasm |
02 |
|
|
|
|
Papillary carcinoma |
03 |
|
02 |
|
|
Lymphocytic thyroidtis |
04 |
|
02 |
|
Total |
|
100 |
|
47 |
Discussion
This study was conducted at MM College of Medical Sciences & Research located in Ambala. Liquid-based cytology is increasingly favored in non-gynecological fine needle aspiration cytology (FNAC) due to its benefits, which include a clearer background, enhanced cellular morphology, and the capability to gather samples for additional techniques [5]. In our clinical environment, patients present with caseating lymph nodes alongside extensive necrotic metastasizing malignancies. Nevertheless, malignancy may still be insufficiently diagnosed, even in necrotic inflammatory or hemorrhagic smears [6]. In our investigation, we opted for the MLBC technique we employed the MLBC technique, which is significantly easier, faster, and safer, requiring less expertise instead of the cell block method to evaluate cases such as breast, thyroid, and lymph nodes for better results. A comparable study was conducted by Saleh et al., which indicated that ThinPrep (liquid-based cytology [LBC]) preparation is more effective than the cell block technique in detecting atypical or neoplastic thyroid lesions [7].
In our research, from the pathologist’s viewpoint, the advantages of employing the LBC technique encompass minimal to no confounding factors (such as blood, debris, and necrotic materials), exceptional cell preservation, fewer fixation artifacts (including air-drying artifacts), consistent distribution, reduced cell overlap, and a lower number of slides requiring examination, which were found to be more significant with a P value <0.05 when compared to conventional FNAC. This method seeks to reduce hemorrhage and necrosis while enhancing nuclear cell preservation for better outcomes. A comparable study was carried out by Pawar et al., which also corroborates the findings presented by Dey et al. [8,16]. They objectively compared various cytological characteristics of TP and CS in FNAC materials and discovered that TPs outperformed CS in terms of clear background, monolayer cells, and cell preservation.
We have made efforts to implement this MLBC technique. In environments with limited resources, the semi-automated LBC technique serves as a viable and recognized alternative to automated LBC techniques, offering a cost-effective solution compared to automated methods [8]. In the evaluation of salivary gland lesions, this study revealed that the chondromyxoid matrix was less pronounced in MLBC preparations than in CS when diagnosing PA. Two studies support this observation, showing significant morphological differences in the quantity and characteristics of the stroma [9,12]. The diagnostic efficacy appears to be greater in CS than in LBC preparations for identifying PA, which is the most commonly diagnosed condition among salivary gland lesions [13]. In our investigation, the effectiveness of lymph node FNAC is equivalent for both CS and LBC. However, in instances of necrosis and extensive hemorrhage, the LBC preparation in this study exhibited superiority over CS in certain aspects. Similar findings were reported by Pawar et al., while the study by Garbar et al. indicates that there is no difference in outcomes between CS and MLBC [8,11]. In this research, the number of nuclear grooves and pseudoinclusions is somewhat higher in MLBC preparations compared to conventional preparations. This observation is consistent with the study conducted by Lee et al., which noted that the background material was slightly more pronounced in LBC preparations than in CS preparations. In this study, the concentration of colloid in MLBC preparations was reduced [12]. This finding is consistent with the research conducted by Mygdakos et al., which indicates that the amount of colloid present in the background of MLBC is less than that found in CS, a distinction that is significant concerning follicular thyroid lesions [10].
In the current investigation, all instances of breast lumps were identified through MLBC preparation. The diagnosis of fibroadenoma was validated by recognizing ductal cell aggregates and bipolar cells, despite our results indicating changes or reductions in stromal fragments. Nevertheless, diagnosing fibroadenoma appears to be particularly challenging with LBC preparations, as some studies indicate a lower diagnostic rate in comparison to CS [14]. However, due to the presence of abundant cellularity, distinct nuclear characteristics, and a clear background, carcinoma was diagnosed in this study, which is consistent with the findings reported by Dey et al [15]. In the current investigation, statistically significant differences were noted between MLBC and CS preparations regarding the absence of blood and debris, the presence of monolayers, and the preservation of cytoplasmic and nuclear details (P<0.05), which is significant. These findings are in agreement with the research conducted by Tripathy et al., and Mygdakos et al., [10,17].
Conclusion
The process of preparing MLBC in FNAC is a method that is secure, uncomplicated, and time-efficient, potentially providing considerable diagnostic benefits when evaluating FNA samples from various anatomical sites. However, it is recommended to employ both MLBC and CS preparations to maximize the diagnostic yield.
Financial Support and Sponsorship: Nil
Conflicts of Interest: There are no conflicts of interest.
References
- Mutuku, O. M. (2020). The utility of a Manual Liquid Based Cytology in Screening for Pre-cancerous Lesion and Cervical Ca (Doctoral dissertation, JKUAT COHES).
- Kavatkar, A. N., Nagwanshi, C. A., & Dabak, S. M. (2008). Study of a manual method of liquid-based cervical cytology. Indian Journal of Pathology and Microbiology, 51(2), 190-194.
- Zuna, R. E., Moore, W., & Dunn, S. T. (2001). HPV DNAtesting of the residual sample of liquid-based Pap test: utility as a quality assurance monitor. Modern Pathology, 14(3), 147-151.
- Cuschieri, K. S., Seagar, A. L., Moore, C., Gilkison, G., Kornegay, J., & Cubie, H. A. (2003). Development of an automated extraction procedure for detection of human papillomavirus DNA in liquid based cytology samples. Journal of virological methods, 107(1), 107-113.
- Hoda, R. S., VandenBussche, C., & Hoda, S. A. (2017). Diagnostic liquid-based cytology (pp. 45-73). New York: Springer.
- Nga, M. E. (2024). Pitfalls in lymph node fine needle aspirationcytology. Acta Cytologica, 68(3), 260-280.
- Saleh, H. A., Hammoud, J., Zakaria, R., & Khan, A. Z. (2008). Comparison of Thin-Prep and cell block preparation for the evaluation of Thyroid epithelial lesions on fine needle aspiration biopsy. Cytojournal, 5, 3
- Pawar, P. S., Gadkari, R. U., Swami, S. Y., & Joshi, A. R. (2014). Comparative study of manual liquid-based cytology (MLBC) technique and direct smear technique (conventional) on fine-needle cytology/fine-needle aspiration cytology samples. Journal of cytology, 31(2), 83-86.
- Parfitt, J. R., McLachlin, C. M., & Weir, M. M. (2007). Comparison of ThinPrep and conventional smears in salivary gland fineâ?needle aspiration biopsies. Cancer Cytopathology: Interdisciplinary International Journal of the American Cancer Society, 111(2), 123-129.
- Mygdakos, N., Nikolaidou, S., Tzilivaki, A., & Tamiolakis,D. J. R. J. M. E. (2009). Liquid Based Preparation (LBP) cytology versus Conventional Cytology (CS) in FNA samples from breast, thyroid, salivary glands and soft tissues. Our experience in Crete (Greece). Rom J Morphol Embryol, 50(2), 245-250.
- Garbar, C., Remmelink, M., & Mascaux, C. (2008). Fine needle aspiration cytology of lymph node: experience of 2 university hospitals with conventional smears and liquid-based cytology. Acta cytologica, 52(4), 418-423.
- Lee, K. R., Papillo, J. L., St John, T., & Eyerer, G. J. (1996). Evaluation of the ThinPrep processor for fine needle aspiration specimens. Acta cytologica, 40(5), 895-899.
- Perez-Reyes, N., Mulford, D. K., Rutkowski, M. A., Logan-Young, W., & Dawson, A. E. (1994). Breast fine-needle aspiration: a comparison of thin-layer and conventional preparation. American journal of clinical pathology, 102(3), 349-353.
- Perez-Reyes, N., Mulford, D. K., Rutkowski, M. A., Logan-Young, W., & Dawson, A. E. (1994). Breast fine-needle aspiration: a comparison of thin-layer and conventional preparation. American journal of clinical pathology, 102(3), 349-353.
- Dey, P., Luthra, U. K., George, J., Zuhairy, F., George, S. S., & Haji, B. I. (2000). Comparison of ThinPrep and conventional preparations on fine needle aspiration cytology material. Acta cytologica, 44(1), 46-50.
- Arul, P. (2016). Utility of manual liquid-based cytology and conventional smears in the evaluation of various fine-needle aspiration samples. Journal of Cytology, 33(4), 177-181.
- Tripathy, K., Misra, A., & Ghosh, J. K. (2015). Efficacy of liquid-based cytology versus conventional smears in FNA samples. Journal of Cytology, 32(1), 17-20.




