Research Article - (2023) Volume 7, Issue 1
Splenic Artery Aneurysm Management with Detachable-Coil Embolization Moderate Packing
2Aortic Center Hirslanden, Zurich, Switzerland
3Department of Surgical, Oncological and Oral Sciences, University of Palermo, Italy
4Department of Public Health, Vascular Surgery Unit, University Federico II of Naples, Italy
Received Date: May 04, 2023 / Accepted Date: Jun 06, 2023 / Published Date: Jun 05, 2023
Copyright: ©Â©2023 Ettore Dinoto, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Dinoto, E., Mirabella, D., Lachat, M., Dolce, A., Turchino, D., et al. (2023). Splenic Artery Aneurysm Management with Detachable-Coil Embolization Moderate Packing. Stem Cell Res Int, 7(1), 29-37.
Abstract
Splenic artery aneurysm (SAA) management is various but endovascular solutions are preferred for a lower incidence of mor- tality and morbidity. The aim is to report SAA management using moderate packing of detachable coil embolization without routine splenectomy. From January 2015 to September 2022, we retrospectively analyzed patients presenting SAA and treated by moderate packing detachable coil embolization. SAA treatment indications were transverse diameter >2 cm, any size pseu- doaneurysm, any size women in childbearing age and abdominal symptoms. The measured outcomes were technical success; mortality, morbidity, abdominal symptoms onset, and splenic infarction. Median follow-up was 34.77±9 months. Seven patients (median transverse diameter of 23±20 mm) were included. Technical success was achieved in all cases. No early mortality, abdominal pain onset or recurrence, reintervention, or splenic infarction was registered. Detachable coil moderate packing embolization is safe and effective for SAA management. The moderate coil packing maintained a limited splenic vasculariza- tion in the early period and avoided splenic infarction. During the follow-up, the coil embolization allowed SAA exclusion with no SAA growth. Larger patient samples with longer follow-up are required to validate this technique.
Keywords
Splenic, Aartery, Aneurysm, Coil Embolization, Endovascular, Percutaneous
Introduction
Splenic artery aneurysm (SAA) is the most common visceral artery aneurysm with a reported incidence between 0.02% and 10.4% in the general population [1]. Most SAAs are asymptomatic and in- cidentally found in imaging studies [2]. The initial SAAs clinical presentation is a rupture In 2% to 10% of patients with a signif- icative hemorrhage, often associated to fatal complications [3]. Treatment options depend primarily on aneurysm characteristics, position of lesion, operative risk and comorbidities [4]. Tradition- ally, open surgical treatment have been used to resolve SAAs, but more recently endovascular approach has been widely accepted as an alternative for the management of these aneurysms [5]. Herein, we report our experience of SAA endovascular treatment using de- tachable coil embolization with moderate density packing without routine splenectomy.
Materials and Methods
From January 2015 to September 2022, patients presenting with SAA and treated by detachable coil embolization with moder- ate density packing were retrospectively analyzed and inserted into standardized piloted forms. This retrospective study aimed to evaluate the efficacy of the coil embolization technique in the treatment of true SAAs on an intention-to-treat basis using a coil moderate density packing. Splenectomy was not performed as a routine intervention associated with SAA coil embolization. In- dications to SAA repair were a transverse diameter > 2 cm, pseu- doaneurysm at any size, and/or women in childbearing age at any size or abdominal symptoms. Ruptured SAAs were excluded from the study. All patients were considered not anatomically candidate to SAA exclusion using stent-graft. The study was performed in agreement with the Declaration of Helsinki and STROBE guide- lines for reporting observational studies were followed [6]. All pa- tients gave informed consent for the procedure itself, anonymous data collection, and analysis. The study was approved by the lo- cal institutional review board, according to the National Policy in the matter of Privacy Act on retrospective analysis of anonymized data. The retrospective and anonymized nature of the study did not require medical ethical committee approval. Demographics, comorbidities, medical treatment, clinical presentation, and proce- dural data were collected. The preoperative assessment consisted of computed tomography angiography (CTA) and splenic duplex ultrasound (DUS) in all cases (Fig 1).
Figure 1: Computed Tomography 3d Reconstruction Showing A Saccular Splenic Artery Aneurysm Located In The Median Splenic Artery.
Baseline laboratory investigations including complete white blood count, liver function tests, renal function test, and LDH were col- lected in all cases. SAA categorization was as proximal, median, or parailar according to its location. Embolization procedure. SAA embolization procedures were planned on preoperative CTA. The more appropriate route to the target lesion was selected according to the condition of remote access and the vessel employed to reach the SAA. The angle of the origin of the celiac trunk (CT) and the aorta was another determinant for the route to the target lesion. The procedures were carried in a dedicated angiosuite equipped with a fixed radioscopy system (GE Innova 4100 Cath Angio Sys- tem; General Electric Medical Systems, EU) in a standard fash- ion. The patient was positioned on to the angiographic table and draped. Vascular access was gained under local anaesthesia and DUS guidance through the common femoral artery (CFA) or the brachial access (left or right) according to the planned procedure.
An introducer sheath was inserted via the vascular access and ad- ministered 2500 IU of heparin. The origin of the CT and subse- quently the splenic artery were engaged using a 5F C2 or Simmons catheter. Subsequently a 5F Flexor sheath (Cook Medical, Bloom- ington, IN, US) introducer was advanced until the CT origin; a 45 cm length or a 90 cm introducer length was employed for the femoral or the brachial access respectively. Multiplanar pre-proce- dural arteriograms were performed to determine the SAA charac- teristics and confirm CTA findings. The SAA was cannulated using a .018 wire (Command; Abbott Vascular, Santa Clara, CA, US) or a .014 wire (Balance Middle Weight -BMW, Abbott Vascular), and a Rebar 18 microcatheter (EV3, Irvine, CA, US) was positioned into the SAA. At this stage, the Concerto Helix Detachable Coil System PGLA-fibered (Medtronic, Inc., Minneapolis, MN, US) was deployed inside the SAA until the aneurysm sac completely felling (Fig 2).
Figure 2: Intraoperative Imaging Showing (A-B) Splenic Artery Aneurysm Detachable Coil Embolizazion Filling And (C) Final Ar- teriography After Coils Deployment.
Coils number and dimension depended on the size of the aneurysm; as general principle larger and longer coils were employed at the filling beginning; smaller coils were employed to achieve a moderate SAA packing. A reduced contrast media caption into the SAA was evident in association to a 15-20% moderate volume coil packing (Fig. 3).
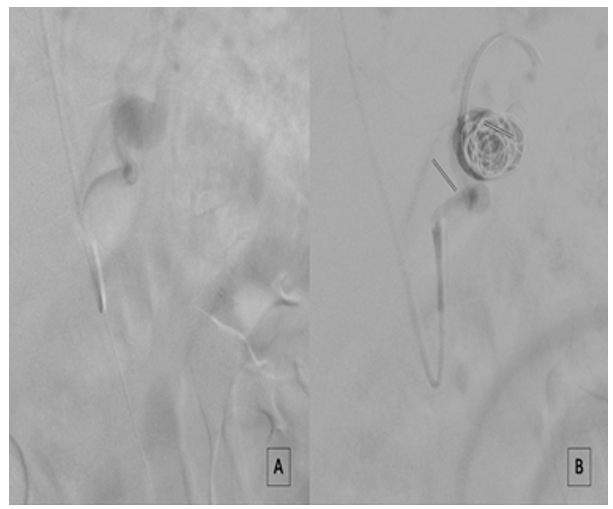
Figure 3: Intraoperative Imaging Showing (A) Splenic Artery Aneurysm Arteriography Before Coil Embolization Filling And (B) After Moderate (18%) Coil Packing Embolization With A Reduced Contrast Media Caption.
SAA luminal volumes was calculated using the images from the preoperative CTA imported into the Horos software (LGPL 3.0; GNU Lesser General Public License). The SAA was manually segmented using at least 3 ROI measurements for luminal volume calculation. The luminal volume was defined as the volume free of thrombus within the aneurysm sac. Coil volume was calculat- ed according to the length and the diameter of each coil with the following formula ‘Coil volume = π (radius/2) × length’. The ratio between the volume of the SAA and the inserted coils was defined as the packing density and was calculated using the equation ‘Coil volume/Aneurysm volume x100%’. As an example, for a 35 mm SAA maximal transverse diameter, presenting a luminal aneurysm volume of 22.438 mm3, to achieve a 15-20% of coil packing den- sity, a 14mmx30cm Concerto coil (Coil volume of 6.594 mm3) is employed. If the flow in efferent artery was significative and there was no stagnation of contrast after the first release, a second coil was placed inside the sac. This was often necessary because the aneurysm is not a perfect sphere. After the procedure, the ad- ministered heparin was not reversed and a single prophylactic low molecular weight heparin (LMWH) shot was administered after 6 hours from the index procedure to reduce the risk of deep venous thrombosis.
Therapy with single or double antiplatelet agent and/or LMWH was not discontinued during the SAA coil embolization. Antico- agulants were switched to LMWH before intervention. Outcomes. Technical success, early (within 30 days), and late (after 30 days) results were the measured outcomes. Technical success was de- fined as intraoperative SAA exclusion. Early and late outcomes included mortality, morbidity, abdominal symptoms onset, and splenic infarction on DUS. Follow-up consisted of clinical ex- amination, laboratory tests, and splenic DUS on 1st postoperative day; laboratory test at 14th POD; DUS and laboratory test at 1 month; laboratory test and basal CT at 2 months (Fig 4); laboratory test and DUS at 6 months. Median follow-up was 34.77 ± 9 (mean: 25.49; IQR: 9-38) months. Statistical analysis. Parametric data are presented as the mean and interquartile range (IQR) or median and min-max range; absolute values and percentages for non-paramet- ric data. Differences in preoperative and postoperative outcomes were assessed using the Student t test. Statistical significance was considered at P<.05. Statistical analysis was performed using SPSS 16.0 (SPSS Inc., Chicago, IL, USA).
Figure 4: Computed Tomography At 2 Months Follow-Up Showing Correct Position Of The Detachable Coils Into The Splenic Artery Aneurysm In (A) Coronal And (B) Transverse View.
Results
A total of 27 (12; 44% female) patients with a mean age of 59.34 ± 6 years were included in the study. Preoperative comorbidities and clinical findings are reported in table 1.
|
|
No. (%) |
|
Patients, N |
27 |
|
Hypertension, N (%) |
15 (55.5) |
|
Diabetes, N (%) |
8 (29.6) |
|
Cardiac Disease, N (%) |
8 (29.6) |
|
Cancer, N (%) |
11 (40.7) |
|
Lipid disorders, N (%) |
12 (44.4) |
|
COPD, N (%) |
8 (29.6) |
|
ASA |
|
|
II |
4 (14.8) |
|
III |
16 (59.2) |
|
IV |
8 (29.6) |
|
Medical therapy |
|
|
Antiplatelet |
8 (29.6) |
|
Lipid-lowering |
16 (59.2) |
|
Antihypertensive |
11 (40.7) |
|
Contraceptive |
4 (14.8) |
Table 1: Comorbidities and Preoperative Findings.
N: number; COPD: Chronic obstructive pulmonary disease; GFR: Glomerular filtration rate; SD: standard deviation; PAD: peripher- al arterial disease; TASC: Trans-Atlantic consensus society; ASA: American Society of Anesthesiologists.
No patients presented signs of splenic infarction at preoperative CT or DUS. Median SAA maximal transverse diameter was 34 ± 8 (mean: 34.7; IQR: 22-71) mm. SAA localization was in the me- dian splenic artery in 15 (56%) and the distal in 12 (44%) patients. In 1 (4%) patient another visceral aneurysm was associated with the SAA. Preoperative laboratory findings were in the physiologic range in all patients. SAA indication to treatment were symptoms in 4 (15%) patients and primary prevention in 23 (85%). All symp- tomatic patients presented abdominal pain; in 2 symptoms onset was referred within 24 hours; in 2 within 48 hours. The pain was reported to increase during breathing phases; in 3 patients was as- sociated with nausea and in 1 vomiting. No weight loss or bowel disorders were reported. In the 23 asymptomatic patients, the in- dication was a transverse diameter > 2 cm in 23 patients; pseudo- aneurysm in 2; and women in childbearing age in 2. A total of 54 Concerto Helix Detachable Coil System (Medtronic, Inc.) were employed to address the 27 treated SAA; median 2 ± 1 (mean: 2; IQR: 1-3) for each patient. The Concerto Helix Detachable Coil System (Medtronic, Inc.) median length was 40.00 ± 8 (mean: 39.7; IQR 30-50) cm; and median diameter was 16.00 ± 4 (mean 16.04; IQR: 12-20) mm. In 1 symptomatic patient with lesion near to origin of splenic artery, a covered stent was released in celiac trunk sealing the root of artery and minimizing the bleeding risk. All the procedures were performed percutaneously and under local anesthesia, through the CFA in 20 (74%) patients and the left bra- chial artery in 7 (26%) (table 2).
|
Pa#ent |
|
Size SAA |
n. Coils |
Diameter Coils |
Symptomatic (1) Asymptomatic (0) |
Time of procedure |
|
1 |
|
25 |
2 |
12x30 (2) |
0 |
60’ |
|
2 |
|
32 |
1 |
20x50 |
0 |
60’ |
|
3 |
|
71 |
2 |
20x50 + 16x40 |
0 |
60’ |
|
4 |
|
22 |
3 |
12x30 (3) |
1 |
120’ |
|
5 |
|
21 |
2 |
14x30 + 12x30 |
0 |
60’ |
|
6 |
|
28 |
2 |
20x50 + 18x40 |
0 |
60’ |
|
7 |
|
32 |
2 |
20x50 + 18x40 |
0 |
45’ |
|
|
8 |
23 |
3 |
16x40 + 14x30 (2) |
1 |
90’ |
|
|
9 |
24 |
1 |
12x40 |
0 |
50’ |
|
|
10 |
43 |
3 |
20x50 + 18x40 + 16x40 |
0 |
50’ |
|
|
11 |
55 |
2 |
20x50 + 18x40 |
0 |
60’ |
|
|
12 |
26 |
2 |
20x50 (2) |
0 |
60’ |
|
|
13 |
34 |
3 |
12x30 (2) + 14x30 |
1 |
120’ |
|
|
14 |
44 |
1 |
16x40 |
0 |
60’ |
|
|
15 |
37 |
3 |
12x30 (2) + 14x30 |
0 |
60’ |
|
|
16 |
27 |
2 |
12x30 (2) |
0 |
45’ |
|
|
17 |
39 |
2 |
12x30 + 14x30 |
0 |
45’ |
|
|
18 |
38 |
1 |
16x40 |
0 |
55’ |
|
|
19 |
57 |
2 |
20x50 + 18x40 |
0 |
50’ |
|
|
|
|
|
|
|
|
|
|
20 |
58 |
2 |
20x50 +16x40 |
0 |
60’ |
|
|
21 |
22 |
2 |
12x30 + 16x40 |
0 |
60’ |
|
|
22 |
24 |
2 |
16x40 (2) |
1 |
60’ |
|
|
23 |
39 |
2 |
14x30 (2) |
0 |
60’ |
|
|
24 |
35 |
2 |
16x40 + 14x30 |
0 |
60’ |
|
|
25 |
27 |
1 |
20x50 |
0 |
60’ |
|
|
26 |
28 |
2 |
16x40 + 14x30 |
0 |
45’ |
|
|
27 |
36 |
2 |
20x50 +12x30 |
0 |
60’ |
Table 2: Intraoperative Data
No blood transfusions were required; in 1 case presenting cardiac disease, cancer, and diabetes, 24-hours monitoring in the intensive care unit was required. Technical success was achieved in all cas- es. In the early postoperative period, no mortality, abdominal pain onset or recurrence, reintervention, or splenic infarction were reg- istered; 1 access site brachial hematoma not requiring reinterven- tion or prolonged hospitalization occurred. During the follow-up, no SAA-related death occurred but 1 patient died after a colectomy for cancer. After coil SAA intervention, laboratory findings, DUS, and basal CT remained stable without evidence of splenic isch- emic lesions or infarction (table 3).
|
|
Preoperative |
1st POD |
Follow-up |
P |
|
Variables |
mean ± SD |
mean ± SD |
mean ± SD |
|
|
White blood cells |
|
|
|
|
|
Neutrophils; (10^3/μL) |
3.74 ± 1.4 |
4.37 ± 1.9 |
3.49 ± 0.3 |
.529 |
|
Macrophages; (10^3/μL) |
0.5 ± 0.2 |
0.59 ± 0.3 |
0.37 ± 0.08 |
.159 |
|
Eosinophils; (10^3/μL) |
0.19 ± 0.1 |
0.19 ± 0.1 |
0.21 ± 0.1 |
.798 |
|
Basophils; (10^3/μL) |
0.04 ± 0.1 |
0.04 ± 0.1 |
0.02 ± 0.1 |
.416 |
|
Lymphocytes; (10^3/μL) |
2.07 ± 0.9 |
2.17 ± 1.1 |
1.76 ± 0.4 |
.513 |
|
LDH; (U/L) |
157.33 ± 17.6 |
168 ± 38.1 |
149 ± 28.1 |
.344 |
|
GGT; (U/L) |
13.5 ±3.5 |
22.67 ± 12.8 |
19.03 ± 8.6 |
.528 |
|
AST; (U/L) |
41.5 ± 10.6 |
15.17 ± 7.4 |
21 ± 7.3 |
.139 |
|
ALT; (U/L) |
11.75 ±11.7 |
14.33 ± 9.3 |
21.25 ± 11.9 |
.325 |
|
eGFR |
86.44 ± 18.0 |
83.66 ± 14.2 |
87.63 ±12.1 |
.913 |
Table 3. Laboratory Tests Findings.
POD: Postoperative day; SD: standard deviation; LDH: lactate de- hydrogenase; GGT: Gamma-glutamil transferase; AST: Aspartate transaminase; ALT: Alanine transaminase; eGFR: Estimated glo- merular filtration rate.
Discussion
SAAs are rare but their diagnosis is continuously increasing due to diagnostic exams for other diseases [7]. The most commonly incriminated causes for the development of a true splenic artery aneurysm include arterial or portal hypertension, cirrhosis, liver transplantation, or pregnancy [8]. Recent SVS practical guidelines identified thin slice CT as the main tool for SAA for diagnosis and treatment planning [4]. SAA is a potentially lethal condition if a rupture occurs with reported mortality up to 25%, thus an aggres- sive approach towards SAA treatment is suggested especially in young pregnant women in the third trimester of pregnancy who have a higher risk of SAA rupture [9, 10] Pseudoaneurysms have been associated with a higher rupture risk when compared to true SAA and thus in these circumstances treatment has been indicated independently from size [11]. Actual indications to SAA treatment include rupture; a diameter >2cm; pseudoaneurysms; and women of childbearing age. Treatment options include conventional open surgery and endovascular SAA exclusion. For both surgical and endovascular SAA repair, maintaining splenic artery patency is not generally mandatory due to the large collateral network [4].
The possibilities of treatment for SAA are different. The first kind of approach was given traditionally by open surgery where we can have an aneurysmectomy and reconstruction of vessel with a vein bypass if the lesion is in proximal o middle part of splenic artery; the alternative, when SAA is near to spleen, is the splenectomy after ligation of artery. In this last option is possible to resort to an laparoscopic approach [4, 12-14]. In all series reported, the open surgical approach is efficient but requires more invasively, more hospital time and involves more morbidities overall especially in fragile patients [15-17]. Endovascular treatment is already consid- ered the first-line approach in visceral and SAA presenting ade- quate anatomy due to a high technical success rate; reduced mor- tality; and reduced morbidity when compared to conventional open surgery [18, 19]. Furthermore, the endovascular technique is less invasive and associated with a shorter length of stay with a mean hospital stay [20, 21]. In absence of a splenectomy rate statistical significant between the two different approaches [15]. Different endovascular options can be employed in SAA repair including splenic artery coil embolization; SAA sac embolization; SAA stent grafting; endoluminal ablation; and thrombin injection [4, 9, 22, 23]. Independently from the endovascular technique employed, a significant concern of these techniques has been related to splenic infarction, especially in the short period [24]. Splenic artery stent grafting represents an exception to splenic infarction because of SAA exclusion maintaining splenic artery patency, but its use is generally limited to SAA located in the mid splenic artery. Advan- tages of coil embolization are related to low-profile microcatheter employed to position coils, especially in tortuous splenic arteries. A coil embolization drawback has been related to a considerably risk of recanalization and the related risk of aneurysm rupture. An- eurysm diameter and coil packing density have been identified as risk factor for future aneurysm recanalization. Larger aneurysms are more difficult to be coiled due to a larger amount of volume to be packed. Low packing density can lead to a reduced efficacy in disrupting incoming pulsatile flow and the amount of scaffold of thrombus [25].
Despite the identification of at least 20% of coil dense packing to reduce the risk of aneurysm recanalization [26]. In case of tight packing density with covered of long tracts of efferent and afferent artery to SAA, the acute exclusion of splenic circulation can po- tentially lead to splenic infarction [27-29]. The employed Concer- to Helix Detachable Coil System (Medtronic, Inc.) are construct- ed of a platinum embolization coil linked to a pushable delivery system enclosed to the spiral. A dedicated device is employed to detach the coil from the pushable delivery system. In this study, the employed Concerto Helix Detachable Coil System (Medtronic, Inc.) were enlaced with PGLA fibers, that been shown an enhance- ment of the thrombogenic potential when compared to standard platinum coils [30, 31].
Based on these data, a lower coil packing density was used to re- duce the risk of potential splenic infarction; on the other hand, it was argued that the employed platinum coils with PGLA fibers with the related higher thrombogenity would determine a progres- sive SAA thrombosis through a moderate slowdown and decrease of flow in splenic artery after the lesion treated. Therefore, the pe- culiarities of Concerto coil system (Medtronic, Inc.), permitted us to obtain a complete exclusion of SAA in all cases treated without to release coils in splenic artery before and after the aneurysm, with decrease of time procedure and number of devices.
In this series, no morbidity related to splenic infarction or SAA recanalization during the follow-up occurred. It can be argumented that the coil packing with a residual SAA perfusion at comple- tion arteriography could have a role in terms of progressive SAA thrombosis, allowing splenic collateral adaptation and avoiding splenic infarction. Moreover, the repositionability of the employed Concerto Helix Detachable Coil System (Medtronic, Inc.) allows positioning, and eventually repositioning, before precise release until desired. Also, other new generation coil systems with dif- ferent technologies, as the Azure hydrocoil (Terumo, Corporation, Tokyo, Japan), with a reported higher filling volume and packing density when compared to platinum coils have been shown to be safe and effective. The study reports an uncommon patient workup including DUS, CT and laboratory findings at baseline and during the follow-up. The baseline imaging and laboratory were used also assess the splenic vascularization and parenchyma before the SAA coil embolization procedure. At different intervals, splenic func- tion was examined to assess potential splenic infarction.
Conclusion
Endovascular coil embolization with moderate density packing was safe and effective for SAA management. The moderate coil packing maintained a limited splenic vascularization in the early period and avoided splenic infarction. During the follow-up, the coil embolization allowed SAA exclusion with no SAA growth. The relatively small patient sample in the study is a limitation but it can be justified with the uncommon SAA findings. The lack of a control group and the limited follow-up period are additional lim- itations. Larger patient samples with longer follow-up are required to validate this technique.
Funding: This research received no external funding. The APC was funded by Department of Surgical, Oncological and Oral Sci- ences—University of Palermo.
Informed Consent Statement: Informed consent was obtained from all subjects involved in the study.
Conflicts of Interest: The authors declare no conflict of interest.
References
- Wang, T., Wang, J., Zhao, J., Yuan, D., & Huang, B. (2021). Endovascular Treatment of Aberrant Splenic Artery Aneu- rysm Presenting With Painless Progressive Jaundice: A Case Report and Literature Review. Vascular and Endovascular Surgery, 55(7), 756-760.
- Correia de Sá, T., Soares, C., Queirós, J., Rocha, T. M., & Ol- iveira, M. (2020). Laparoscopic resection of a splenic artery aneurism with spleen preservation. Case Reports in Surgery, 2020.
- Ouchi, T., Kato, N., Nakajima, K., Higashigawa, T., Hashimo- to, T., Chino, S., & Sakuma, H. (2018). Splenic artery aneu- rysm treated with endovascular stent grafting: a case reportand review of literature. Vascular and Endovascular Surgery, 52(8), 663-668.
- Chaer, R. A., Abularrage, C. J., Coleman, D. M., Eslami, M. H., Kashyap, V. S., Rockman, C., & Murad, M. H. (2020). The Society for Vascular Surgery clinical practice guidelines on the management of visceral aneurysms. Journal of vascular surgery, 72(1), 3S-39S.
- Mesbahi, M., Zouaghi, A., Zaafouri, H., Hadded, D., Benzarti, Y., Riahi, W., ... & Maamer, A. B. (2021). Surgical manage- ment of splenic artery aneurysm. Annals of Medicine and Sur- gery, 69, 102712.
- von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) State- ment: guidelines for reporting observational studies. Int J Surg Lond Engl. 2014;12:1495–9.
- Saba, L., Anzidei, M., Lucatelli, P., & Mallarini, G. (2011). The multidetector computed tomography angiography (MDC- TA) in the diagnosis of splenic artery aneurysm and pseudo- aneurysm. Acta radiologica, 52(5), 488-498.
- Bacalbasa, N., Balescu, I., Tanase, A., Pautov, M., Brezean, I., Vilcu, M., & Brasoveanu, V. (2018). Spleno-pancreatec- tomy en bloc with parcelar gastrectomy for splenic artery aneurysm–a case report and literature review. In Vivo, 32(4), 915-919.
- Stanley, J. C., Wakefield, T. W., Graham, L. M., Whitehouse,W. M., Zelenock, G. B., & Lindenauer, S. M. (1986). Clinical importance and management of splanchnic artery aneurysms. Journal of Vascular Surgery, 3(5), 836-840.
- Dave, S. P., Reis, E. D., Hossain, A., Taub, P. J., Kerstein, M. D., & Hollier, L. H. (2000). Splenic artery aneurysm in the 1990s. Annals of vascular surgery, 14(3), 223-229.
- Pitton, M. B., Dappa, E., Jungmann, F., Kloeckner, R., Schot- ten, S., Wirth, G. M., ... & Dueber, C. (2015). Visceral artery aneurysms: incidence, management, and outcome analysis in a tertiary care center over one decade. European radiology, 25, 2004-2014.
- Madoff, D. C., Denys, A., Wallace, M. J., Murthy, R., Gupta, S., Pillsbury, E. P., ... & Hicks, M. E. (2005). Splenic arterial interventions: anatomy, indications, technical considerations, and potential complications. Radiographics, 25(suppl_1), S191-S211.
- Ellebrecht, D. B., Horn, M., Pross, M., Keck, T., & Kleemann,M. (2019). Laparoscopic treatment of splenic artery aneu- rysm. Zentralblatt fur Chirurgie, 144(5), 445-448.
- Berek, P., & Štefanic, P. (2017). Surgical treatment of splenic artery aneurysm. European Journal of Vascular and Endovas- cular Surgery, 54(1), 11.
- Zhu, C., Zhao, J., Yuan, D., Huang, B., Yang, Y., Ma, Y., & Xiong, F. (2019). Endovascular and surgical management of intact splenic artery aneurysm. Annals of Vascular Surgery, 57, 75-82.
- Shabunin, A. V., Bedin, V. V., Tavobilov, M. M., Karpov, A. A., Tsurkan, V. A., Alieva, F. F., & Pilyus, F. G. (2022). Se- lecting the most appropriate surgical treatment of true splenic artery aneurysm. Khirurgiia, (10), 21-27.
- Wang, T. K. M., & Desai, M. Y. (2020). Optimal surveillance and treatment of renal and splenic artery aneurysms. Cleve- land Clinic Journal of Medicine, 87(12), 755-758.
- Tulsyan, N., Kashyap, V. S., Greenberg, R. K., Sarac, T. P., Clair, D. G., Pierce, G., & Ouriel, K. (2007). The endovascu- lar management of visceral artery aneurysms and pseudoan- eurysms. Journal of vascular surgery, 45(2), 276-283.
- Bracale UM, Narese D, Ficarelli I, Laurentis MD, Spalla F, Dinoto E, et al. Stent-assisted detachable coil embolization of wide-necked renal artery aneurysms. Diagn Interv Radiol Ank Turk. 2017;23:77–80.
- Hemp, J. H., & Sabri, S. S. (2015). Endovascular management of visceral arterial aneurysms. Techniques in vascular and in- terventional radiology, 18(1), 14-23.
- Batagini, N. C., El-Arousy, H., Clair, D. G., & Kirksey, L. (2016). Open versus endovascular treatment of visceral artery aneurysms and pseudoaneurysms. Annals of Vascular Sur- gery, 35, 1-8.
- Hogendoorn, W., Lavida, A., Hunink, M. M., Moll, F. L., Geroulakos, G., Muhs, B. E., & Sumpio, B. E. (2014). Open repair, endovascular repair, and conservative management of true splenic artery aneurysms. Journal of Vascular Surgery, 60(6), 1667-1676.
- Hongo, N., Kiyosue, H., Ota, S., Nitta, N., Koganemaru, M., Inoue, M., ... & Asayama, Y. (2021). Vessel Occlusion using Hydrogel-Coated versus Nonhydrogel Embolization Coils in Peripheral Arterial Applications: A Prospective, Multicenter, Randomized Trial. Journal of Vascular and Interventional Ra- diology, 32(4), 602-609.
- Saltzberg, S. S., Maldonado, T. S., Lamparello, P. J., Cayne,N. S., Nalbandian, M. M., Rosen, R. J., ... & Rockman, C. B. (2005). Is endovascular therapy the preferred treatment for all visceral artery aneurysms?. Annals of Vascular Surgery, 19, 507-515.
- Damiano, R. J., Tutino, V. M., Paliwal, N., Patel, T. R., Waqas,M., Levy, E. I., ... & Meng, H. (2020). Aneurysm character- istics, coil packing, and post-coiling hemodynamics affect long-term treatment outcome. Journal of neurointerventional surgery, 12(7), 706-713.
- Leng, B., Zheng, Y., Ren, J., Xu, Q., Tian, Y., & Xu, F. (2014).Endovascular treatment of intracranial aneurysms with de- tachable coils: correlation between aneurysm volume, pack- ing, and angiographic recurrence. Journal of neurointerven- tional surgery, 6(8), 595-599.
- Wang, W., Chang, H., Liu, B., Wang, W., Yu, Z., Chen, C., & Wang, Y. (2020). Long-term outcomes of elective transcath- eter dense coil embolization for splenic artery aneurysms: a two-center experience. Journal of International Medical Re- search, 48(1), 0300060519873256.
- Pecoraro, F., Dinoto, E., Bracale, U. M., Badalamenti, G., Farina, A., & Bajardi, G. (2017). Symptomatic deep femoral artery pseudoaneurysm endovascular exclusion. Case report and literature review. Annals of Vascular Surgery, 42, 303-e5.
- Koganemaru, M., Abe, T., Nonoshita, M., Iwamoto, R., Kuhara, A., & Kugiyama, T. (2014). Follow-up of true visceral artery aneurysm after coil embolization by three-dimensional contrast-enhanced MR angiography. Diagnostic and Interven- tional Radiology, 20(2), 129.
- Dinoto, E., Ferlito, F., Mirabella, D., Tortomasi, G., Bajar- di, G., & Pecoraro, F. (2021). Type 1A endoleak detachable coil embolization after endovascular aneurysm sealing: case report. International Journal of Surgery Case Reports, 83, 106024.
- Girdhar, G., Read, M., Sohn, J., Shah, C., & Shrivastava, S. (2014). In-vitro thrombogenicity assessment of polymer fila- ment modified and native platinum embolic coils. Journal of the Neurological Sciences, 339(1-2), 97-101.


