Research Article - (2023) Volume 8, Issue 1
Mechanical Digestion of Broccoli through Chewing and its Impact on Myrosinase Activity
2Dental School, University of Detroit Mercy, 2700 Martin Luther King Jr. Blvd, Detroit, MI 48208, USA
Received Date: Aug 10, 2023 / Accepted Date: Sep 10, 0202 / Published Date: Sep 22, 2023
Copyright: ©Â©2023 Anne-Marie Kosi-Kupe, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kosi-Kupe, A-M., Cobau, A., Beard, S., Carswell. A., Ahmad, M., et al. (2023). Mechanical Digestion of Broccoli through Chewing and its Impact on Myrosinase Activity. Adv Nutr Food Sci, 8(1), 40-49.
Abstract
Myrosinase activity on broccoli glucosinolates has been widely and extensively discussed.
Purpose: Studies on cruciferous vegetables, especially broccoli, have gained significance in the fight against cancer. Glucosinolates in broccoli transformation into sulforaphane occur after its exposure through chewing. However, the relationship between individual chewing patterns and denture morphology has not been studied extensively. Research on human digestion has demonstrated how the mechanical breakdown of larger food particles into smaller ones is a crucial precursor to chemical food breakdown. This study is a comparative analysis of how chewing on broccoli tissues by dif- ferent individuals enables the enzyme myrosinase to break down broccoli glucosinolates chemically. We investigate the individual chewing patterns linked to the surface anatomy of the pre-molars and molars and the myrosinase activity on broccoli glucosinolates.
Methods: Three individuals chewed a 4-millimeter broccoli floret four times sequentially (2 grams), and we measured the floret length at each bite until the sample was ground. Then, we combined the chewed broccoli with distilled water, filtered it, and myrosinase activity was measured using photo spectrometric measurements and an agar diffusion test. Individual pre-molars and molars samples were measured in millimetres.
Results: Data from the three human chewing mechanisms compared to the mechanical breakdown performed by an au- tomatic mixer shows different and individually specific values.
Conclusion: Individual chewing patterns link to the unique surface anatomy of the pre-molars and molars, subsequently impacting the myrosinase activity on broccoli glucosinolates’ breakdown individually
Keywords
Mechanical Digestion, Broccoli, Chewing, Glucosinolates, Myrosinase
Introduction
In humans, two significant events occur following food intake: mechanical and chemical digestion before absorption. The me-chanical part of the digestion event in the mouth, including chew-ing and grinding, has yet to be widely discussed compared to the chemical one, which includes the enzymatic breakdown of essen-tial nutrients. Glucosinolate is found in broccoli florets, and its transformation into sulforaphane is critical in cancer prevention [5,7]. Halkier et al. elucidated the biology and biochemistry of glu-cosinolates [33]. Glucosinolates' hydrolysis has been discussed [5-9,17,33,36]. The myrosinase structure, function, and occurrence are widely studied, and its identification, purification, and charac¬terization [7,10-27,29,31,32,34,35,37-43].
This study compares broccoli's mechanical breakdown pattern in three individuals and then investigates any correlation between chewing pattern and myrosinase activity. Therefore, we compared the length, enamel, and other parts of the pre-molars and molars as they play a crucial function in chewing and grinding foods (Ta¬ble 1, Figure 1). Molar mass effects on viscosity and diffusion in liquid foods and mechanical and other physical properties of sol¬id and semi-solid foods and films have been studied [20-23]. In these functions, the molar mass is one of the critical factors, and therefore, the effects of molar mass on various health problems related to noncommunicable diseases or symptoms such as cancer, hyperlipidemia, hyperglycemia, constipation, high blood pressure, knee pain, osteoporosis, cystic fibrosis, and dysphagia. Methods and tools for measuring chewing (mastication) force are available and have been discussed [26,27].
Postoperative dietary intake reduction was more common after an exclusively mandibular extraction than after an extraction includ¬ing the maxilla [21]. Age and sex also may play critical factors in mechanical food breakdown and intake [4,22,25,26]. The role of the chewing pattern and the bioaccessibility of food nutrients are discussed [6,23,24,28].
Objective
This study was undertaken to see if it is possible to replicate my-rosinase enzymatic activity in a lab setting and further understand how our natural physiology (the size and shape of our teeth) plays a role in the mechanical digestion of broccoli myrosinase. Further¬more, we collected comparative data between human chewing and an automatic blender.
Hypothesis
The experiment hypothesizes that there may not be any chewing pattern difference between the three tested individuals or any en-zymatic difference in myrosinase activity when comparing human chewing to an artificial blender.
Material & Methods
• Fresh, organic Broccoli from Neimans’ market
• Human pre-Molars and Molars (UDM Dental school models)\
• Blender (Rolling) & Morser
• Agar (Difco) for agar-diffusion-test
• Thioglucosinolase (Myrosinase), Sigma, Aldrich, masks
• Aqua dest (deionized)
• Graduated Beakers, Erlenmeyer flasks, cylinders, stirrer
• Lab scale, measuring ruler
• Spectrophotometer-Monochromator Unit-Genesys20 at 600 nm
Experiment
Glucosinolates –Extraction from Broccoli Water Extraction Versus Ethanol Extraction- More effective, less hazardous dilution
Day One - Created two broccoli solutions with crushed broccoli as the solute: 1) 100ml distilled water + 2.7 grams of broccoli, 2) 20ml ethanol + 2.7 grams of broccoli.
Our goal was to crush the broccoli to resemble the texture of chewed broccoli. The broccoli was mashed mechanically using a scientific/lab blender and then ground by hand with a mortar and pestle to achieve the desired texture—the ethanol-broccoli solu-tion was placed in the refrigerator. We strained the water-broccoli solution with filter paper and a strainer to keep all solid particles of the broccoli separated from the supernatant solution. We chose Water extraction over ethanol extraction because of convenience, effectiveness, and less hazard [1].
Broccoli-Agar-Formulation
45ml of this strained broccoli water was set aside to be added to the agar solution and poured into the Petri dishes. Agar Petri dishes were made using two grams of agar and 400 ml of distilled water. The flask containing this solution was stirred mechanically using a magnetic stirrer to keep it liquid until we placed it in the autoclave. After placing the solution in the autoclave, we stirred the 45ml of strained broccoli solution into the liquid agar solution (~50oC ). The agar-strained broccoli solution was filled in 13 Petri dishes and was left overnight to harden.
Day Two: Upon interacting with the hardened Petri dishes, we realized that the resulting agar solution was too firm to introduce the enzyme properly. Any attempt to create holes in the agar dishes resulted in cracks running through the plate, making it unsuitable for our purposes, so we prepared another agar solution. This solu¬tion consisted of 500ml of distilled water (100ml more than our previous solution) and two grams of agar. Again, we placed this solution in the autoclave for sterilization. We collected another 45 ml of strained broccoli water from the original water-broccoli ex¬traction, added it to the autoclaved agar solution, and stirred and filled 15 Petri dishes. (Final Solution: 2g agar + 500 ml distilled water + 45 ml broccoli extract). To prepare the enzyme solution for later amylase testing (as reference), we combined 0.1 grams of amylase with 100 ml of distilled water. This solution was placed in the freezer to preserve it when the new agar petri dish solution solidified.
Preparation of Chewed Broccoli Samples
Three individuals chewed four times sequentially into a four-mil-limeter-long broccoli floret (2 grams), and we measured the floret length at each bite until the sample was ground. Then, we com¬bined the chewed broccoli with distilled water and filtered it for photospectrometric measurements and an agar diffusion test.
To replicate human chewing, We placed 2 grams of broccoli florets in a lab blender and then crushed them into a paste by hand with a mortar and pestle. We combined the resulting paste with 100 ml of distilled water and filtered it for photospectrometric measurements and an agar diffusion test.
Agar-Diffusion-Test
The Myrosinase activity was measured spectrometrically using an agar diffusion test—0.1 ml inoculum in each hole.
The broccoli agar Petri dishes were incubated at 37 and 30 degrees Celcius for one day. The myrosinase Inhibition zone could be seen and measured in millimeters.
Human Pre-Molars-Molars measurements
The teeth were collected from extracted teeth at the Detroit Mercy Dental School. A periodontal probe was used to measure the width and height of each tooth in millimeters.
Chewed Broccoli Measurements
Three Individuals were selected to chew 2 g (4mm length) broccoli in four bites in approximately 2 minutes. -Chewed broccoli was spread on filter paper, dry, and spread. The size of the vegetables was measured and recorded.
Results
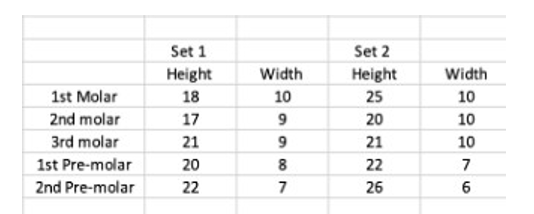
Table 1: Human Pre-Molars-Molars measurements (mm)
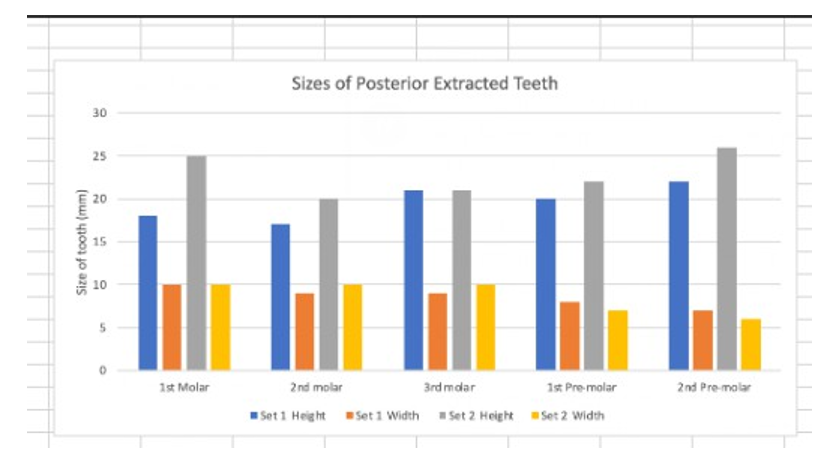
Figure 1: Human Pre-Molars-Molars measurements
|
Time ( Minutes) |
Photometric Measurements (A) |
|
0 |
0.970 |
|
15 |
1.032 |
|
30 |
1.038 |
|
45 |
1.046 |
|
60 |
1.102 |
|
90 |
1.123 |
Table 2: Individual#1-Spectrophotometric Measurement of Myrosinase Activity /Time (minutes)
Figure 2: Individual#1’s Spectrometric Measurements with data from Table 2
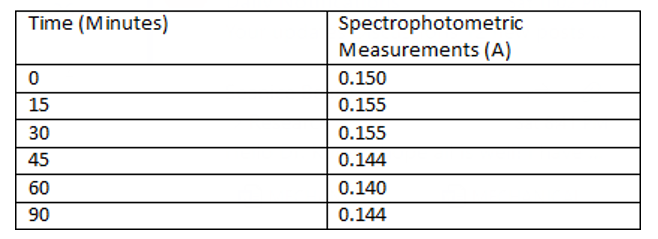
Table 3: Individual#2’s Spectrometric Measurements of Myrosinase Activity /Time (minutes)
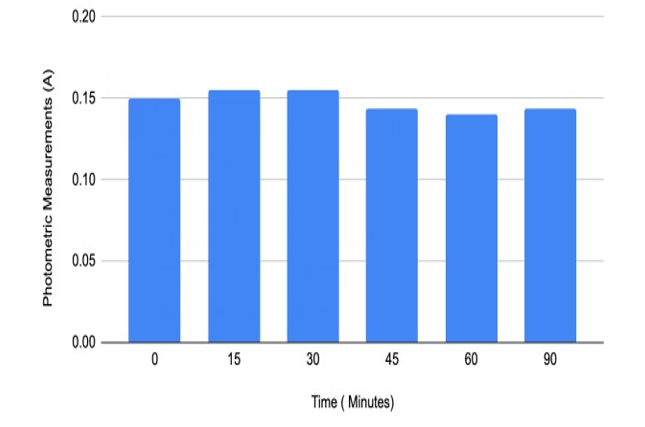
Figure#3: Individual#2’s Spectrometric Measurements with data from Table 3
Table 4: Individual#3’s Spectrometric Measurements of Myrosinase Activity /Time (minutes)
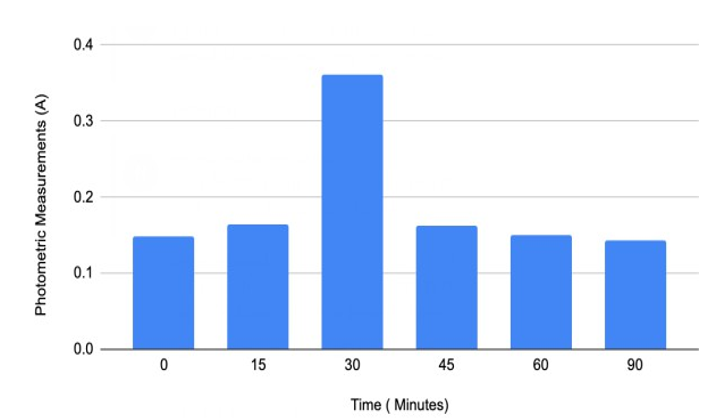
Figure#4: Individual#3’s Spectrometric Measurements with data from Table 4
|
Time (Minutes) |
Spectrophotometric Measurements (A) |
|
0 |
0.893 |
|
15 |
1.027 |
|
30 |
1.051 |
|
45 |
1.055 |
|
60 |
1.225 |
|
90 |
1.332 |
Table 5: Spectrometric Measurements of broccoli from Blender/minutes
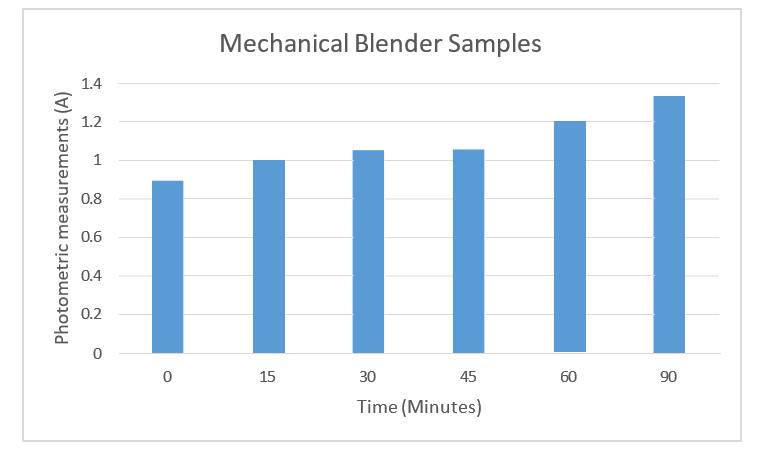
Figure 5: Spectrophotometric measurements of blended broccoli, with data from table 5
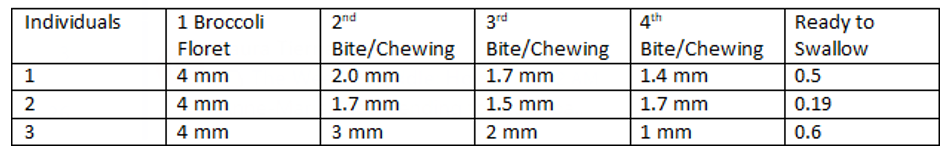
Table 6: Chewing Pattern Expressed in Broccoli Particles Measurements (mm)/bite
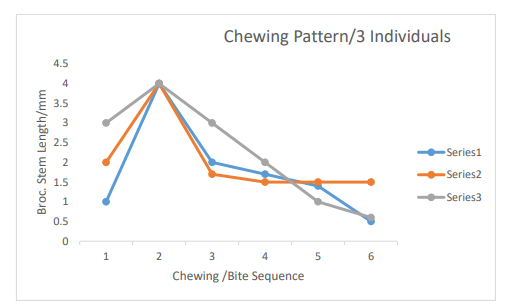
Figure 6: Legend-Series1-Individual#1 Series2-Individual#2 Series3-Individual#3

Discussion
This study was undertaken to see if it is possible to replicate my-rosinase enzymatic activity in a lab setting and further understand how our individual teeth anatomy (the size and shape of our teeth) plays a role in the mechanical digestion of broccoli myrosinase.
In the table above, data was collected by two third-year dental students. The teeth were collected from extracted teeth at Detroit Mercy Dental. Students measured sets #1 and #2 of teeth. A peri-odontal probe was used to measure the width and height of each tooth in millimeters. The x-axis shows the tooth that was mea-sured, and the y-axis demonstrates the measurements gathered in millimeters. The results from Table #1 and Figure #1 give us a clear idea of the variations in teeth anatomy that might impact chewing patterns and, subsequently, the myrosinase activity.
Molars 1, 2, and 3, as well as premolars 1 and 2 of set#1, vary in height and width compared to set#2 teeth. The results from this study show that myrosinase activity's effectiveness in transform-ing glucosinolate into isocyanates in older adults and patients with teeth issues may be negatively impacted. A mechanical blender could resolve this issue. According to orthognathic surgeons, hard food chewing occurs mainly on 1st molar and 2nd premolar areas [2]. Molars are perfect for grinding and crushing, and premolars are suitable for tearing and biting. Based on their anatomy, molars, have four cusps and more ridges, than premolars, which only have two cusps and fewer ridges.
Mastication efficacy has been studied and showed that it depends on the orofacial anatomical features of the subject, the coordina¬tion of these anatomical features, and the consistency of the food used during testing [6,23]. Studies confirm the impact of chewing and the bioaccessibility of nutrients after a meal [23-24].
Furthermore, the myrosinase activity on chewed broccoli samples varies among the three individuals (Figures 1, 2 and 3). Individu¬al 1 reached the highest myrosinase activity of 1.102 Absorbance after ninety minutes of incubation at 20oC (Table 2 and Figure 2), Individual 2 had the highest myrosinase activity at fifteen and thir¬ty minutes (Table 3 and Figure 3), and Individual 3 got the most increased myrosinase activity at thirty minutes (Table 4 and Figure 4). The results of the chewing pattern among the tested individuals in Table 5 and Figure 5 confirm our hypothesis [3]. In contrast, (Figure 6) demonstrates the correlation between teeth anatomy,chewing patterns, and impact on myrosinase activity.
Finally, the blended broccoli samples showed the highest myrosi-nase activity of 1.332 Absorbance after ninety minutes compared to human-chewed broccoli samples. And the myrosinase activity was confirmed and detected on glucosinate-containing agar. The inhibition zone varied between 1.1 to 1.7 mm in diameter (Figure 7). The size of the vegetables was measured and recorded in Figure 8.
The above-cited studies described the instability and properties complexity of myrosinase. Myrosinase, as an enzyme, is very sensitive to changes in temperature and the environment. Further¬more, glucosinolates must be converted to their bioactive form, known as isothiocyanates, better to exert its physiologic activ¬ity by the heat-labile enzyme myrosinase. However, Nandini et al., 2020 stated that this enzyme is destroyed on cooking or even steaming or blanching for more than a minute, as boiling the broc¬coli or thawing it below −85°C will spoil its anticancer properties. Shikita et al., 1999, Burmeister et al., 1997, and Cottaz et al. 1997 brought some awareness to myrosinase activation, inhibitor, and substrate recognition-active site mechanisms.
Three individuals chewed broccoli and then recorded their results to see how the enzymatic activity would compare to the crushed broccoli. Comparing Tables 2-6, they display the level of myrosi-nase activity through readings on the mass spectrometer; it is clear that crushed broccoli yields a level of enzymatic activity similar to chewed broccoli. While it will need to be investigated further, Ta-ble 9 reveals that the duration of enzymatic activity in the crushed broccoli appears longer than the chewed broccoli. This result could be due to other enzymes in the sample of chewed broccoli, such as amylase, further catalyzing the enzymatic activity. Com¬paring the chewed models, the selection of chewed broccoli from Individual #1 in Table 2 and Figure 2 yielded the most enzymatic activity. Further investigation into the physiology of their mouth might be warranted in future studies to determine how their enzy¬matic activity was so robust in comparison. As we only used one brand of blender, this experiment would need to be replicated with more samples of crushed/blended broccoli from different blenders to confirm this study's results.
These observations yield promise that there may be other ways to unlock the anticancer abilities of broccoli and myrosinase than chewing only. Unfortunately, the requirement that broccoli is fresh, uncooked, and naturally chewed to extract the benefits means these anticancer properties are unobtainable to specific populations. Many people do not necessarily have access to fresh fruits and vegetables, and specific populations, such as older peo¬ple, only sometimes possess the physiological requirements neces¬sary to mechanically digest broccoli by chewing. Knowing how to process fresh broccoli in a way that is accessible to these popula¬tions where these anticarcinogenic properties are preserved would be scientifically significant. Suppose we see what physiological properties of teeth, such as the measurements in our table, lead to the highest enzymatic activity. In that case, we could replicate the mechanism of artificially breaking down broccoli. Understand¬ing how to preserve the enzymatic activity of myrosinase properly also opens the possibility of more accessible and further study of this enzyme. Based on these reasons and others, it becomes clear that knowing how myrosinase acts and reacts within different tem¬peratures and environments is critical to unlocking its potential health benefits.
This experiment's results also raise the question of what methods can be used to create the optimal enzymatic environment to facili¬tate myrosinase activity. While we used a blender, would the same result be yielded by chopping the broccoli? What kind of pressure is optimal for breaking down the broccoli? Would more or less pressure result in the same enzymatic activity or somehow change it? Would exposing it to oxygen in the air affect enzymatic activ¬ity? Would this enzymatic reaction still happen in more relaxed environments like refrigerators? Could adding other substances to the broccoli, as Okunade et al. (2018) did with mustard seeds, foster the production of even more anticancerous isothiocyanates? Given the incredible health benefits, these and other questions re¬garding myrosinase production are worth further investigation.
Declarations
Ethical Approval: The University of Detroit Mercy Internal Re¬view Board has approved this study IRB#22-23-39 and guidelines have been followed. Chair: Dr. Elizabeth Hill: Elizabeth M. Hill,
Ph.D., Interim Chair
Professor Emerita of Psychology
Reno Hall 205
University of Detroit Mercy
4001 W. McNichols Road
Detroit, MI 48221
(313) 578-0405
Authors Contributions
Anne-Marie Kosi-Kupe's contributions to the paper: As Principal Investigator: Design of the project, chewing of broccoli samples, data collection, writing of the entire first draft, and main manu¬script
Alexis Cobau contributed to chewing broccoli samples, (partial¬ly) to reference collection and data collection, and (somewhat) to writing a portion of the first draft. Seanice Beard contributed to the chewing of the broccoli samples and data collection.
Amanda Carswell contributed to chewing broccoli samples and data collection.
Maha Ahmad, Maria Sanchez, and Hanaa Saleh contributed to the pre-molars and molars measurements and partial dental data anal¬ysis.
All authors have consented to participate and publish this study's results.
All authors reviewed and agreed upon the final manuscript. Study Data Declaration–Available All data related to this study are available upon request. Please get in touch with the corresponding author.
References
- Doheny-Adams, T., Redeker, K., Kittipol, V., Bancroft, I., & Hartley, S. E. (2017). Development of an efficient glucosinolate extraction method. Plant Methods, 13(1), 1-14.
- Posnick, J. C. (2013). Orthognathic surgery: principles andpractice, 227-263.
- Scheid, R. C. (2012). Woelfel's dental anatomy. LippincottWilliams & Wilkins, 130-155.
- Yamanaka, R., Akther, R., Furuta, M., Koyama, R., Tomofuji, T., Ekuni, D., ... & Kishimoto, E. (2009). Relation of dietary preference to bite force and occlusal contact area in Japanese children. Journal of oral rehabilitation, 36(8), 584-591.
- Nandini, D. B., Rao, R. S., Deepak, B. S., & Reddy, P. B. (2020). Sulforaphane in broccoli: The green chemopreven-tion!! Role in cancer prevention and therapy. Journal of oral and maxillofacial pathology: JOMFP, 24(2), 405.
- Sarvan, I., Kramer, E., Bouwmeester, H., Dekker, M., & Verk-erk, R. (2017). Sulforaphane formation and bioaccessibility are more affected by steaming time than meal composition during in vitro digestion of broccoli. Food chemistry, 214, 580-586.
- Soundararajan, P., & Kim, J. S. (2018). Anti-carcinogenic glucosinolates in cruciferous vegetables and their antagonistic effects on prevention of cancers. Molecules, 23(11), 2983.
- Agerbirk, N., Olsen, C. E., & Sørensen, H. (1998). Initial and final products, nitriles, and ascorbigens produced in myrosi-nase-catalyzed hydrolysis of indole glucosinolates. Journal of Agricultural and Food Chemistry, 46(4), 1563-1571.
- Angelino, D., Dosz, E. B., Sun, J., Hoeflinger, J. L., Van Tassell, M. L., Chen, P., ... & Jeffery, E. H. (2015). Myrosinase-dependent and–independent formation and control of isothiocyanate products of glucosinolate hydrolysis. Frontiers in plant science, 6, 831.
- Martinez-Ballesta, M. D. C., & Carvajal, M. (2015). Myrosinase in Brassicaceae: the most important issue for glucosinolate turnover and food quality. Phytochemistry reviews, 14, 1045-1051.
- Botti, M.G., Taylor, M.G. (1995). Studies on the Mechanism of Myrosinase:Investigation of the Effect of Glycosyl Accep-tors on Enzyme Activity. Journal of Biological Chemistry270(35):20530-20535.
- Masaru, O., & Tadao, H. (1972). Molecular properties of multiple forms of plant myrosinase. Agricultural and biological chemistry, 36(13), 2495-2503.
- Lönnerdal, B., & Janson, J. C. (1973). Studies on myrosinas-es. II. Purification and characterization of a myrosinase from rapeseed (Brassica napus L.). Biochimica et Biophysica Acta (BBA)-Enzymology, 315(2), 421-429.
- Nakamura, Y., Yoshimoto, M., Murata, Y., Shimoishi, Y., Asai, Y., Park, E. Y., ... & Nakamura, Y. (2007). Papaya seed represents a rich source of biologically active isothiocyanate. Journal of agricultural and food chemistry, 55(11), 4407-4413.
- Husebye, H., Arzt, S., Burmeister, W. P., Härtel, F. V., Brandt, A., Rossiter, J. T., & Bones, A. M. (2005). Crystal structure at 1.1 Å resolution of an insect myrosinase from Brevicoryne brassicae shows its close relationship to β-glucosidases. Insect biochemistry and molecular biology, 35(12), 1311-1320.
- Bridges, M., Jones, A. M., Bones, A. M., Hodgson, C., Cole, R., Bartlet, E., ... & Rossiter, J. T. (2002). Spatial organization of the glucosinolate–myrosinase system in brassica specialist aphids is similar to that of the host plant. Proceedings of the Royal Society of London. Series B: Biological Sciences, 269(1487), 187-191.
- Brabban, A. D., & Edwards, C. (1994). Isolation of glucosinolate degrading microorganisms and their potential for reducing the glucosinolate content of rapemeal. FEMS microbiology letters, 119(1-2), 83-88.
- Bones, A. M., & Rossiter, J. T. (1996). The myrosinaseâ?gluco-sinolate system, its organisation and biochemistry. Physiolo-gia plantarum, 97(1), 194-208.
- Koroleva, O. A., Davies, A., Deeken, R., Thorpe, M. R., To-mos, A. D., & Hedrich, R. (2000). Identification of a new glucosinolate-rich cell type in Arabidopsis flower stalk. Plant physiology, 124(2), 599-608.
- Nishinari, K., & Fang, Y. (2021). Molar mass effect in foodand health. Food Hydrocolloids, 112, 106110.
- Taniguchi, S., Abe, A., Ito, Y., Ishihama, T., Hayashi, H., Momokita, M., ... & Shibata, K. (2022). Factors associated with a reduced food intake after third molar extraction among adults: a cross-sectional study. BMC Oral Health, 22(1), 120.
- Le Révérend, B. J., Edelson, L. R., & Loret, C. (2014). Anatomical, functional, physiological and behavioural aspects of the development of mastication in early childhood. British Journal of Nutrition, 111(3), 403-414.
- Lemmens, L., Van Buggenhout, S., Van Loey, A. M., & Hen-drickx, M. E. (2010). Particle size reduction leading to cell wall rupture is more important for the β-carotene bioaccessibility of raw compared to thermally processed carrots. Journal of agricultural and food chemistry, 58(24), 12769-12776.
- Read, N. W., Welch, I. M., Austen, C. J., Barnish, C., Bartlett, C. E., Baxter, A. J., ... & Worlding, J. (1986). Swallowing food without chewing; a simple way to reduce postprandial glycaemia. British journal of nutrition, 55(1), 43-47.
- Rémond, D., Machebeuf, M., Yven, C., Buffière, C., Mioche,L., Mosoni, L., & Mirand, P. P. (2007). Postprandial whole-body protein metabolism after a meat meal is influenced by chewing efficiency in elderly subjects. The American journal of clinical nutrition, 85(5), 1286-1292.
- OrtuÄ?, G. (2002). A new device for measuring mastication force (Gnathodynamometer). Annals of Anatomy-Anato-mischer Anzeiger, 184(4), 393-396.
- Lucas, P. W., & Luke, D. A. (1983). Methods for analysing the breakdown of food in human mastication. Archives of Oral Biology, 28(9), 813-819.
- Miyawaki, S., Ohkochi, N., Kawakami, T., & Sugimura, M. (2000). Effect of food size on the movement of the mandibular first molars and condyles during deliberate unilateral mastication in humans. Journal of Dental Research, 79(7), 1525-1531.
- Ohtsuru, M., & Kawatani, H. (1979). Studies on the myrosinase from Wasabia japonica: Purification and some properties of wasabi myrosinase. Agricultural and Biological Chemistry, 43(11), 2249-2255.
- Iversen, T. H., & Baggerud, C. (1980). Myrosinase activity in differentiated and undifferentiated plants of Brassicaceae. Zeitschrift für Pflanzenphysiologie, 97(5), 399-407.
- El-Sayed, S. T., Jwanny, E. W., Rashad, M. M., Mahmoud, A. E., & Abdallah, N. M. (1995). Glycosidases in plant tissues of some brassicaceae screening of different cruciferous plants for glycosidases production. Applied Biochemistry and Biotechnology, 55, 219-230.
- Burmeister, W. P., Cottaz, S., Driguez, H., Iori, R., Palmieri, S., & Henrissat, B. (1997). The crystal structures of Sinapis alba myrosinase and a covalent glycosyl–enzyme intermediate provide insights into the substrate recognition and active-site machinery of an S-glycosidase. Structure, 5(5), 663-676.
- Halkier, B. A., & Gershenzon, J. (2006). Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol., 57, 303-333.
- Bones, A. M., & Rossiter, J. T. (2006). The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry, 67(11), 1053-1067.
- Shikita, M., Fahey, J. W., Golden, T. R., David Holtzclaw, W.,& Talalay, P. (1999). An unusual case of ‘uncompetitive acti-vation’by ascorbic acid: purification and kinetic properties of a myrosinase from Raphanus sativus seedlings. Biochemical Journal, 341(3), 725-732.
- Lambrix, V., Reichelt, M., Mitchell-Olds, T., Kliebenstein, D. J., & Gershenzon, J. (2001). The Arabidopsis epithiospecifier protein promotes the hydrolysis of glucosinolates to nitriles and influences Trichoplusia ni herbivory. The Plant Cell, 13(12), 2793-2807.
- Burmeister, W. P., Cottaz, S., Driguez, H., Iori, R., Palmieri, S., & Henrissat, B. (1997). The crystal structures of Sinapis alba myrosinase and a covalent glycosyl–enzyme intermediate provide insights into the substrate recognition and active-site machinery of an S-glycosidase. Structure, 5(5), 663-676.
- Cottaz, S., Rollin, P., & Driguez, H. (1997). Synthesis of 2-deoxy-2-fluoro-glucotropaeolin, a thioglucosidase inhibitor. Carbohydrate research, 298(1-2), 127-130.
- Björkman, R., & Janson, J. C. (1972). Studies on myrosinas-es: I. Purification and characterization of a myrosinase from white mustard seed (Sinapis alba, L.). Biochimica et Biophysica Acta (BBA)-Enzymology, 276(2), 508-518.
- Pessina, A., Thomas, R. M., Palmieri, S., & Luisi, P. L. (1990). An improved method for the purification of myrosinase and its physicochemical characterization. Archives of biochemistry and biophysics, 280(2), 383-389.
- Lüthy, B., & Matile, P. (1984). The mustard oil bomb: rectified analysis of the subcellular organisation of the myrosinase system. Biochemie und Physiologie der Pflanzen, 179(1-2), 5-12.
- Andréasson, E., Jørgensen, L. B., Höglund, A. S., Rask, L., & Meijer, J. (2001). Different myrosinase and idioblast distribution in Arabidopsis and Brassica napus. Plant physiology, 127(4), 1750-1763.
- Okunade, O., Niranjan, K., Ghawi, S. K., Kuhnle, G., & Meth-ven, L. (2018). Supplementation of the diet by exogenous myrosinase via mustard seeds to increase the bioavailability of sulforaphane in healthy human subjects after the consumption of cooked broccoli. Molecular nutrition & food research, 62(18), 1700980.



