Research Article - (2025) Volume 10, Issue 2
Effect of Methanol Extract of Justicia Carnea Leaf on Some Hematological Parameters of Phenylhydrazine-Induced Anaemic Rats
2Department of Home Economics, Federal College of Education Eha-Amufu, Nigeria
3Nutrition Department, Federal Ministry of Health and Social Welfare, Abuja, Nigeria
Received Date: Aug 01, 2025 / Accepted Date: Aug 26, 2025 / Published Date: Sep 03, 2025
Copyright: ©Â©2025 Ijioma Okorie, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Okorie, I., Chukwuemeka, E. N., Oguine, J., Chinyere, O. C. (2025). Effect of Methanol Extract of Justicia Carnea Leaf on Some Hematological Parameters of Phenylhydrazine-Induced Anaemic Rats. Adv Nutr Food Sci, 10(2), 01-07.
Abstract
Introduction/Objectiv: Anaemia is a major public health concern that can be prevented, and plant-based materials offer a cost-effective approach to prevention. This study investigated the potential benefits of methanol extract of Justicia carnea leaf in preventing anaemia by examining its effects on hematological parameters in rats with phenylhydrazine-induced anaemia.
Methods: Experimental study design was used to randomly assign 20 rats with similar weight into 4 groups of 5 rats each. J. carnea extract was performed using standard method. The groups are; Group 1 (300 mg/kg J. carnea extract), Group 2 (500 mg/kg J. carnea), Group 3 (100 mcg/mL fersolate), and Group 4 (no treatment). Rats received various doses of extract and fersolate, concurrently with 50 mg/mL of phenylhydrazine solution. Phytochemical, and micronutrient compositions, hemoglobin concentration, packed cell volume, red blood cell and white blood cell count were determined using standard method. Data were analysed using ANOVA and paired sample t-test, while significant level was set at p < 0.05.
Results: Justicia carnea contained alkaloids (1.11 mg/100g), flavonoid (1.53 mg/100g), phenol (2.26 mg/100g), iron (0.85 mg/100g), vitamin C (20.63 mg/100g), and vitamin B12 (3.68 mg/100g). Significant difference (p < 0.05) was observed between the baseline and end-line hemoglobin concentration, packed cell volume, red blood cell, and white blood cell count of phenylhydrazine-induced anaemic rats after administration with 300 mg/kg and 500 mg/kg of J. carnea leaf extract.
Conclusion: The study revealed dose-dependent effects of J. carnea leaf extract for improving anaemia-related health outcomes.
Keywords
Preventive, Anaemic Rats, Justicia Carnea Leaf, Hematology, Methanol Extract
Introduction
Worldwide, anaemia is a disease of public health importance, affecting approximately 1.9 billion people with the highest prevalence in developing countries [1]. Anaemia is marked by a reduction in red blood cells or hemoglobin levels, resulting in insufficient oxygen supply to the body’s tissues and organs [2]. Various factors including nutritional deficiencies, chronic disease, and genetic disorders contribute to anaemia [3]. Anaemia affects individuals across the lifespan, with higher prevalence in vulnerable groups like pregnant women and young children. Iron deficiency anaemia (IDA) is a significant contributor to the global disease burden [1]. The prevalence of anaemia is a growing concern due to its potential to cause severe health complications, including death, if left untreated or neglected. The findings from the National Food Consumption and Micronutrient Survey (NFCMS) in Nigeria reveal that iron deficiency affects 4.2% of adolescents, while iron- deficiency anaemia prevalence is 1.5%. Iron deficiency prevalence is 10.0% among women of reproductive age, with 4.6% aged 15–49 years experiencing iron deficiency anaemia in Nigeria, while the Nigeria Demographic Health Survey [NDHS] reported anaemia prevalence among under-five children (68%) and pregnant women (55%) [4,5]. This high rate of anaemia is cause for concern. Preventive medicine, such as vaccination/supplementation, plays a vital role in public health [6,7]. Historically, the focus on curative medicine has overshadowed preventive care, resulting in high rates of preventable diseases and mortality. Prioritizing preventive care can reduce healthcare costs, hospitalizations, and mortality rates, particularly in low and middle-income countries where preventable diseases claim millions of lives annually [8,7]. Oral iron supplements, despite being inexpensive, have limitations such as side effects and poor absorption, which can hinder their effectiveness in combating anaemia. This is particularly concerning in Nigeria, where government efforts to control anaemia through iron/folate supplementation, malaria control and nutrition education have yet to yield desired results, as evidenced by the persistently high prevalence of anaemia [9,5]. These challenges underscore the need for alternative solutions, particularly those of plants origin, which can provide a more accessible and potentially safer option for anaemia prevention and treatment, especially in developing countries. The use of plants as a cost-effective strategy in preventing iron-deficiency has led to their global acceptance in disease management [10,11].
Furthermore, study on Justicia carnea offers a promising alternative therapy for preventing anaemia. The plant have been traditionally used in folk medicine, and their anti-anaemic potential suggest further investigation [12]. The plant belongs to the Acanthaceae family, rich in bioactive compounds, and have been shown to exhibit antioxidant, anti-inflammatory, and blood tonic properties [12]. The study explored the potential preventive effects of methanol J. carnea leaf extract on haematological parameters in anaemic-induced rats, investigating its effect on anaemia.
Methods
Study Design:
Twenty rats were randomly assigned to four groups of five rats each using an experimental design. Rats with similar body weights were grouped together after individual weights were recorded.
Experimental Materials:
Justicia carnea leaves were harvested from the National Root Crops Research Institute, and authenticated at the Department of Plant Science and Biotechnology, Michael Okpara University of Agriculture Umudike (MOUAU). Twenty (20) rats were procured from the Department of Biochemistry MOUAU, while other experimental materials include methanol, fersolate tab, rat chow, and phenyl hydrazine.
Sample Preparation:
The method of leaf extraction described by Sukhder et al. was adopted for the study [13]. The leaves were sorted, washed, drained and dried at room temperature (250C) for 20 days. The leaves were chopped into small particles, milled and sieved to fine powder. A total of 100 g of the powdered leaves were submerged in 1800 mL of methanol and agitated on a mechanical shaker for about 10 minutes. The mixture were allowed to stand for 4 hours, thereafter, drained using a muslin cloth into a 4 L stainless steel bowl. The concentrate was subjected to dryness in a Hot air oven (Model No: DHG-9030) at 600C until a gummy extract was obtained. The gummy extract was transferred into sample bottles and stored in refrigerator until use. A stock solution of 100 mg/ mL was prepared by dissolving 1 g of J. carnea extract in 10 mL of distilled water. Analysis of the J. carnea extract was conducted to determine the amount of bioactive compounds (alkaloids, flavonoids and phenols). All analyses were carried out in triplicate in order to validate the findings using Ngounou et al. method as reported by Nnam et al. [11,14]. Twenty (20) healthy adult male albino rats weighing 120–145 g were purchased from the College of Natural Sciences MOUAU. The experiment was guided by the National Institutes of Health guidelines for the care and use of laboratory animals. The experimental protocol was designed to adhere to the moral guidelines for the use and care of animals. College of Veterinary Medicine Ethics Committee MOUAU approved the ethical clearance of experiments. The rats were kept in a cage at 25 ± 20C with a relative humidity (45% ± 5%) and 12- hour light and 12-hours dark condition in Department of Human Nutrition and Dietetics, MOUAU, Nigeria. The rats were given normal rat chow and water ad libitum. The rats were exposed to one-week acclimatization before experiment.
Administration of J. Carnea Leaf Extract and Anaemia Induction
The rats received the extract based on their individual body weight, and anaemia was induced using a 50 mg/mL phenyl hydrazine solution. The rats were administered 300 mg/kg or 500 mg/kg body weight of the J. carnea extract, concurrently with a 50 mg/ mL phenyl hydrazine solution. Phenyl hydrazine was administered for 3 days to induce anaemia, while the J. carnea extract and fersolate were given for 7 days. The study lasted 7 days, with groups receiving different doses via oral gavage: Group 1 (300 mg/kg J. carnea extract), Group 2 (500 mg/kg J. carnea), Group 3 (100 mcg/mL fersolate), and Group 4 (no treatment).
Phytochemical and Micronutrient Determination
Alkaloids, flavonoids, and phenols were determined using standard method described by AOAC [15].
Alkaloids Determination
The alkaloid content was determined by extracting the sample with acetic acid in ethanol, concentrating the extract, precipitating the alkaloids with ammonium hydroxide, and then weighing the dried residue.
Flavonoid Determination
The total flavonoid content was determined using the Dowd method with aluminum trichloride in methanol. The absorbance was read at 415 nm, and the results were expressed as milligrams of quercetin equivalents per 100 g of sample.
Polyphenol Content
The total polyphenol content was estimated using the Folin- Ciocalteu reagent. The absorbance was measured at 725 nm, and the results were expressed as milligrams of Gallic Acid Equivalent per gram of dry matter.
Iron: Sample blended, pH adjusted, reduced with hydroquinone, reacted with 1,10-dipridyl, and absorbance read at 520 nm using spectrophotometry [15].
Vitamin B12: Determined by High Performance Liquid Chromatography (HPLC, model C030 of AOAC after sample digestion and injection [15].
Ascorbic Acid: Sample dissolved in distilled water, TCA added, colour developed with 2,6-dichloroindophenol, read with spectrophotometer. Calculation using a specific formula [15].
Blood Sample Collection: A 2 mL syringe was used to collect baseline blood sample into EDTA test tube bottles for analysis of hemoglobin, red blood cell, packed cell volume, and white blood cell. After clotting for 30 minutes, the samples were centrifuged at 3000 rpm for 10 minutes. The serum supernatant was then aspirated and stored for analysis. Samples were collected on days 0 and 7 (end-line) for haematological evaluation.
Determination of Hemoglobin (Hb) Concentration
• Principle: Drabkin's reagent, containing potassium cyanide and potassium ferricyanide, was mixed with whole blood to form cyanmethemoglobin. The intensity of the resulting color was measured spectrophotometrically at 540 nm, with the optical density directly proportional to the hemoglobin concentration. Whole blood was added to Drabkin’s reagent, a solution containing potassium cyanide (KCn) and potassium ferri cyanide (K3Fe(Cn6)). KCn converts ferrous (Hb-Fe2+) to ferric (Hb-Fe3+) state to form methemoglobin which then combines with KCN to form a stable pigment, cyanmethemoglobin complex. The colour intensity of this mixture was measured in a spectrophotometer at a wavelength of 540 nm (using a yellow-green filter). The optical density of the solution was proportional to the hemoglobin concentration.
• Method: To measure hemoglobin levels, 5.0 mL of Drabkin's reagent was mixed with 0.02 mL of EDTA blood in test tubes. After 10 minutes at room temperature (25°C), the mixtures were read in a spectrophotometer at 540 nm. The Drabkin's reagent was used as a blank (setting the percentage transmittance at 100%), and hemoglobin values (g/dL) were determined using a pre-calibrated chart. A 5.0 mL of Drabkin’s reagent was pipetted into two test tubes 1 and 2, and a well-mixed sample of EDTA blood (0.02 mL) was pipetted into the tubes, rinsing the pipette five times with the reagent, until all the blood have been removed from the pipette. The solutions was mixed and allowed to stand at 2500C for 10 minutes in order to allow the formation of cyanmethemoglobin. The mixtures were transferred into cuvettes and read in a spectrophotometer at a wavelength of 540 nm. The Drabkin reagent in tube 1 was used as a blank, (setting the percentage transmittance at 100%). The readings was recorded and transferred into a pre-calibrated chart and the actual Hb values in g/dL was determined.
• Calculation: Hb (g/dL) = absorbance of test x concentration of standard (mg/dL)/absorbance of standard.
Determination of Packed Cell Volume (PCV)
• Principle: When whole blood sample is subjected to a centrifugal force for maximum RBC packing, the space occupied by the RBC is measured and expressed as percentage of the whole blood volume.
• Method: Using microhaematocrit method, a well-mixed anti- coagulated whole blood was allowed to enter capillary haematocrit tubes until they are approximately 2/3 full. Blood filling was done for each tube. One end of each tube was sealed with plastic and placed in the medial grooves of the centrifuge, head exactly opposite each other, with the sealed end away from the center of the centrifuge. All tubes were spun for five minutes at 1000 rpm. The tubes was removed as soon as the centrifuge stopped spinning.
• Calculation: PCV was obtained for each tube using microhaematocrit-reader by measuring the height of the RBC column and expressed as a ration of the height of the total blood column. PCV (%) = height of cell volume x 10/height of total blood column.
Determination of Red Blood Cell (RBC) Count
• Principle: Method by Dacie and Lewis [16]. Whole blood is diluted approximately (1/20) using an isotonic diluting fluid (formal citrate solution), sodium citrate formaldehyde 1.0 mL and distilled water 99.0 mL, this maintains disc-like appearance of red blood cell.
• Method: A 1:200 dilution was made by mixing 0.03 mL of blood with 4 mL of diluting fluid. The diluted sample was then loaded into a Neubauer chamber, and red blood cells were counted in the central squares using a ×40 microscope. The RBC count per microliter was calculated by multiplying the total count by 10,000.
Determination of White Blood Cell (WBC) Counts
The ocular method of counting white blood cells was used, as stated by Dacie and Lewis [16].
• Principle: Turk's solution was used to dilute whole blood; the glacial acetic acid in the solution lyses red blood cells, while gentian violet dyes the remaining white blood cells so that they may be counted visually under a microscope.
• Method: A 75 × 10 mm glass tube containing 0.38 ml of 2% acetic acid (diluting fluid) colored pale violet and 20µl (0.02 ml) of blood was used to create a 1 in 20 dilution. The tube was tightly covered, and then the suspension was mixed for one minute by rotating it in a cell-suspension mixer.
• A xxxxxs used to fill the Neubauer counting chamber. Using a ×6 eyepiece and a ×40 objects, the preparation was observed. In as many 1mm2 areas as required, at least 10 cells were counted
• The following method of calculating the total WBC count was as follows,

• Thus, if N cells are counted in 0.1µl, then the leukocyte count per liter is
• N × 20 × 106 = N × 200× 106
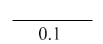
• E.g. if 115 cells are counted, the WBC is 115 × 200 × 106/L, which is equal to 23 × 109/L
• Statistical Analysis: Descriptive statistics (mean and standard deviation) were used to analyze bioactive compounds (alkaloids, flavoids and phenols), while a paired sample t-test was employed to assess the preventive effect of methanol J. carnea leaf extract on haematological parameters in anaemic rats. The significance level was set at p < 0.05 and percentage differences were also calculated to further quantify the changes observed in the study.
Results
The phytochemical composition of the methanol extract of J. carnea leaves is summarized in Table 1, highlighting the levels of alkaloids, flavonoids, phenols, iron, vitamin C, and vitamin B12 present in the extract.
|
Parameters (mg/100g) |
J. carnea leaves |
|
Alkaloids |
1.11 ± 0.02 |
|
Flavonoids |
1.53 ± 0.03 |
|
Phenols |
2.26 ± 0.05 |
|
Iron |
0.85 ± 0.01 |
|
Vitamin C |
20.63 ± 0.02 |
|
Vitamin B12 |
3.68 ± 0.02 |
Table 1: Phytochemical and micronutrient composition of methanol extract of J. carnea leaves
Table 2 shows mean hemoglobin (Hb) level of the rats within the groups. Preventive effect of Justicia carnea leaf extract at 300 mg/ kg body weight significantly increased Hb levels in anemic rats from 12.30 g/dL to 16.20 g/dL (31.71% increase, p < 0.05). At 500 mg/kg body weght, Hb levels increased from 12.32 g/dL to 17.20 g/ dL (39.61% increase, p < 0.05), indicating a dose-dependent effect. Preventive effect of fersolate at 100 mcg/kg significantly increased Hb levels from 12.38 g/dL to 18.39 g/dL (48.55% increase, p < 0.05). Similarly, untreated rats showed a significant increase in Hb levels from 12.26 g/dL to 14.42 g/d (17.62% increase, p < 0.05).
|
Groups |
Baseline |
End-line |
MD |
p-value |
%D |
|
JC300mg/kg |
12.30 ± 0.33 |
16.20 ± 0.16 |
3.90 ± 0.28 |
0.000 |
31.71 |
|
JC500mg/kg |
12.32 ± 0.31 |
17.20 ± 0.70 |
4.88 ± 0.72 |
0.000 |
39.61 |
|
STD100mcg/kg |
12.38 ± 0.33 |
18.39 ± 0.80 |
6.01 ± 0.82 |
0.000 |
48.55 |
|
NTG |
12.26 ± 0.32 |
14.42 ± 0.25 |
2.16 ± 0.36 |
0.000 |
17.62 |
|
MD = mean difference; %D = percentage difference; STD = standard drug (fersolate); NTG = no treatment group; JC = J. carnea at 300 mg/kg and 500 mg/kg body weight. |
|||||
Table 2: The preventive effect of methanol extract of J. carnea leaves on hemoglobin levels in anaemic rats, comparing mean baseline and end-line Hb levels
Table 3 shows the mean packed cell volume (PCV) level of the rats. Preventive effect of J. carnea leaf extract at 300 mg/kg body weight significantly increased PCV levels from 24.80% to 43.00 % (73.39% increase, p < 0.05). At 500 mg/kg body weight, PCV levels increased from 24.60% to 45.80% (86.18%), indicating a dose-dependent effect. Preventive effect of fersolate at 100 mcg/ kg body weight significantly increased PCV levels from 24.60% to 47.20% (91.87% increase, p < 0.05). Untreated rats also showed a significant increase in PCV levels from 24.60% to 34.80% (41.46% increase, p < 0.05).
|
Groups |
Baseline |
End-line |
MD |
p-value |
%D |
|
JC300mg/kg |
24.80 ± 1.10 |
43.00 ± 1.00 |
18.20 ± 1.48 |
0.000 |
73.39 |
|
JC500mg/kg |
24.60 ± 0.89 |
45.80 ± 1.92 |
21.20 ± 1.92 |
0.000 |
86.18 |
|
STD100mcg/kg |
24.60 ± 0.55 |
47.20 ± 2.39 |
22.60 ± 2.70 |
0.000 |
91.87 |
|
NTG |
24.60 ± 0.89 |
34.80 ± 2.86 |
10.20 ± 2.39 |
0.001 |
41.46 |
|
MD = mean difference; %D = percentage difference; STD = standard drug (fersolate); NTG = no treatment group; JC = J. carnea at 300 mg/kg and 500 mg/kg body weight. |
|||||
Table 3: The preventive effect of methanol extract of J. carnea leaves on packed cell volume levels in anaemic rats, comparing mean baseline and end-line PCV levels
Table 4 shows the mean red blood cell (RBC) level of the rats. Preventive effect of J. carnea leaf extract at 300 mg/kg body weight significantly increased RBC levels from 4.56 × 10^6 cells/ μL to 7.05 × 10^6 cells/μL (54.61% increase, p < 0.05). At 500 mg/ kg body weight, RBC levels increased from 4.42 × 10^6 cells/μL to 7.51 × 10^6 cells/μL (69.91% increase, p < 0.05), indicating a dose-dependent effect. Preventive effect of fersolate at 100 mcg/ kg body weight significantly increased RBC levels from 4.48 × 10^6 cells/μL to 7.80 × 10^6 cells/μL (74.11% increase, p < 0.05). Untreated rats also showed a significant increase in RBC levels from 4.44 × 10^6 cells/μL to 5.58 × 10^6 cells/μL (25.68% increase, p < 0.05).
|
Groups |
Baseline |
End-line |
MD |
p-value |
%D |
|
JC300mg/kg |
4.56 ± 0.11 |
7.05 ± 0.02 |
2.49 ± 0.11 |
0.000 |
54.61 |
|
JC500mg/kg |
4.42 ± 0.19 |
7.51 ± 0.33 |
3.09 ± 0.51 |
0.000 |
69.91 |
|
STD100mcg/kg |
4.48 ± 0.13 |
7.80 ± 0.41 |
3.32 ± 0.46 |
0.000 |
74.11 |
|
NTG |
4.44 ± 0.12 |
5.58 ± 0.44 |
1.14 ± 0.44 |
0.005 |
25.68 |
|
MD = mean difference; %D = percentage difference; STD = standard drug (fersolate); NTG = no treatment group; JC = J. carnea at 300 mg/kg and 500 mg/kg body weight. |
|||||
Table 4: The preventive effect of methanol extract of J. carnea leaves on red blood cell levels in anaemic rats, comparing mean baseline and end-line RBC levels
Table 5 shows the mean total white blood cell (WBC) level of the rats. Treatment with J. carnea leaf extract at 300 mg/kg body weight significantly decreased TWBC levels from 14.16 × 10^9 cells/L to 10.42 × 10^9 cells/L (26.41% decrease, p < 0.05). At 500 mg/kg, TWBC levels decreased from 13.74 × 10^9 cells/L to 10.27 × 10^9 cells/L (25.25% decrease, p < 0.05). Preventive effect of fersolate at 100 mcg/kg body weight significantly decreased TWBC levels from 13.46 × 10^9 cells/L to 9.48 × 10^9 cells/L (29.57% decrease, p < 0.05). Untreated rats also showed a significant decrease in TWBC levels from 14.17 × 10^9 cells/L to 11.99 × 10^9 cells/L (15.38% decrease, p < 0.05).
|
Groups |
Baseline |
End-line |
MD |
p-value |
%D |
|
JC300mg/kg |
14.16 ± 0.40 |
10.42 ± 0.30 |
3.74 ± 0.59 |
0.000 |
26.41 |
|
JC500mg/kg |
13.74 ± 0.43 |
10.27 ± 0.33 |
3.47 ± 0.48 |
0.000 |
25.25 |
|
STD100mcg/kg |
13.46 ± 0.47 |
9.48 ± 0.16 |
3.98 ± 0.58 |
0.000 |
29.57 |
|
NTG |
14.17 ± 0.40 |
11.99 ± 0.43 |
2.18 ± 0.73 |
0.003 |
15.38 |
|
MD = mean difference; %D = percentage difference; STD = standard drug (fersolate); NTG = no treatment group; JC = J. carnea at 300 mg/kg and 500 mg/kg body weight. |
|||||
Table 5: The preventive effect of methanol extract of J. carnea leaves on total white blood cell red blood cell levels in anaemic rats, comparing mean baseline and end-line TWBC levels
Discussion
Phenyl hydrazine administration caused a moderate and significant drop in hemoglobin concentration, packed cell volume (PCV), and red blood cell (RBC) count, effectively inducing anaemia in the rats. The results obtained are similar to that of Fagbohoun et al. [9], who observed a decrease in these hematological parameters of 42.29%, with an administration of 20 mg/kg of phenyl hydrazine in rats. On the 7th day of treatment, the rats having received fersolate and methanol extract of J. carnea leaf, initiated recovery. This recoverability of the anaemia due to discontinuation of administration of phenyl-hydrazine was described by Ryu and Yook and Fagbohoun et al. [17,9]. Methanol extract of Justicia carnea at 500 mg/kg effectively increased hemoglobin, packed cell volume, and red blood cell count in anaemic rats, showing comparable results to the standard treatment, fersolate. The 300 mg/kg dose of J. carnea showed some benefits, but to a lesser extent. The benefits of fersolate can be attributed to its iron content, which supports hemoglobin synthesis, and B-complex vitamins, which enhance hematopoiesis. Similarly, the bioactive compounds in J. carnea leaf extract likely contributed to the observed increase in hematological parameters, reversing the effects of phenyl- hydrazine-induced anaemia in rats. These findings align with Fagbohoun et al., who reported similar results with J. secunda extract administered to pheyl-hydrazine-induced anaemic rats [9]. The phytochemical analysis carried out on the methanol extract of J. carnea leaf studied showed the presence of alkaloids, flavonoids, and phenols, which are known for their antioxidant properties and ability to mitigate oxidative stress [18,9]. These compound may also promote blood tissue regeneration, contributing to the observed increase in red blood cell production, particularly in rats treated with the 500 mg/kg dose of the extract. The iron-chelating properties of these compounds might further support red blood cell regeneration [19,9].
The micronutrient analysis of J. carnea leaf extract revealed a potent combination of iron, vitamin C and vitamin B12, which are crucial for hematopoiesis. Vitamin C's ability to enhance non- heme iron absorption by converting ferric iron to ferrous iron in the stomach , likely played a significant role in the extract's efficacy against anaemia. This synergistic effect may have contributed to the observed increase in hemoglobin concentration, packed cell volume, and red blood cell count [20]. The findings underscore the importance of these micronutrients in supporting blood health and highlight the potential benefis of consuming J. carnea leaf to maximize iron absorption and alleviate anaemia. The observed decrease in total white blood cell count in rats treated with both fersolate and J. carnea leaf extract suggests that the treatment effectively addressed the underlying inflammatory response, potentially contributing to its anti-anaemic efficacy. The presence of bioactive compounds such as flavonoids, alkaloids, and phenols in the extract may have exerted immunomodulatory effects, normalizing the immune response to oxidative stress and subsequently reducing white blood cell count. This finding further underscore the extract's therapeutic potential in mitigating anaemia, possibly by modulating the immune system and reducing inflammation. The comparable effects observed with fersolate, a standard anti-anaemic agent, support the notion that J. carnea leaf extract possesses promising anti-anaemic properties. This finding align with Fagbohoun et al. who reported similar results with J. secunda extract [9].
Conclusion
This study highlights the potential benefits of Justicia carnea leaf extract in addressing anaemia, particularly iron-deficiency anaemia. The presence of phytochemicals (alkaloids, flavonoids, and phenols) and essential micronutrients (iron, vitamin C, and vitamin B12) supports its therapeutic potential. The dose- dependent effects observed suggest that Justicia carnea leaves may offer a natural, potentially side-effect-free alternative for improving anaemia-related health outcomes. These findings have implications for the development of complementary or alternative treatments for anaemia, particularly in resource-limited settings.
Author Contributions
Ijioma Okorie: Conception/design of the work; interpretation of data for the work; and Final approval of the version to be published. Elizabeth Ngozi Chukwuemeka: Conception/design of the work; Acquisition, analysis and interpretation of data for the work; Drafting of the work; Final approval of the version to be published. Joy Oguine: Acquisition, analysis and interpretation of data for the work; Drafting of the work; Data collection; Final approval of the version to be published.
References
- World Health Organization. (2002). The world health report 2002: reducing risks, promoting healthy life. World Health Organization.
- Shubham, K., Anukiruthika, T., Dutta, S., Kashyap, A. V., Moses, J. A., & Anandharamakrishnan, C. (2020). Iron deficiency anemia: A comprehensive review on iron absorption, bioavailability and emerging food fortification approaches. Trends in Food Science & Technology, 99, 58-75.
- Kresina, T. F. (2020). National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, Bethesda, Maryland. Immune Modulating Agents, 221.
- FGN, I. (2022). National Food Consumption and Micronutrient Survey 2021. Preliminary Report. Federal Government of Nigeria (FGN) and the International Institute of Tropical Agriculture (IITA).
- Nigeria Population Commission. (2019). Nigeria demographic and health survey 2018. NPC, ICF.
- Elder, C. (2017). Mind-body training for at-risk populations: preventive medicine at its best. The Permanente journal, 21, 16-174.
- Babatunde, A. O., Shobanke, H. A., Akinade, A. A., Michael,A. J., Osadare, M., Akanbi, O. K., ... & Adebisi, Y. A. (2021). Enhancing preventive medicine over curative medicine: Role of telemedicine. Public Health in Practice, 2, 100130.
- Luijten, N. (2010). When do the advantages of preventive healthcare overcome the disadvantages. Universiteit van Tilburg.
- Fagbohoun, L., Nonvidé, G. C., Orou, A. S., Houngbèmè, A., Sakirigui, A., Gunin, F., & Gbénou, J. D. (2022). Anti- anaemic activity and potential toxicity of extracts of four tinctorial plants used in the treatment of anemia in Benin: Gossypium barbadense, Sorghum bicolor, Hibiscus sabdariffa and Justicia secunda. American Journal of Plant Sciences, 13(12), 1460-1477..
- Gao, D. (2012). Hypolipidemia and Hyperglycemia.Antioxidant Enzyme, 169.
- M Nnam, N., J Okorie, I., & C Okorie, A. (2024). Effect of Aqueous Extract of Cymbopogon Citratus on Blood Glucose Level, Lipid Profile and Histology of Diabetic Rats. International Journal of Nutrition Sciences, 9(2), 149-158.
- Ekweogu, C. N., Ude, V. C., Nwankpa, P., Emmanuel, O., & Ugbogu, E. A. (2020). Ameliorative effect of aqueous leaf extract of Solanum aethiopicum on phenylhydrazine-induced anaemia and toxicity in rats. Toxicological research, 36(3), 227-238.
- Sukhder, K. K., Singh, R., Jain, S. K. and Kumar, A.. Phytochemical analysis and extraction methods of Justicia species (2008).
- Ngounou, F. N., Manfouo, R. N., Tapondjou, L. A., Lontsi, D., Kuete, V., Penlap, V., ... & Sondengam, B. L. (2005). Antimicrobial diterpenoid alkaloids from Erythrophleum suaveolens (guill. & perr.) brenan. Bulletin of the Chemical Society of Ethiopia, 19(2), 221-226.
- AOAC (2012). Official Methods of Analysis. Association of Official and Analytical Chemists. 18th Edition. Washington DC.
- Dacie, J. V., & Lewis, S. M. (2006). Practical Haematology 10th Edition Churchill Livingstone. Edinburgh, London, 398- 440.
- Ryu, J. H., & Yook, C. S. (2001). The effects of Sa-Mul- Tang (Si-Wu-Tang), a Traditional Chinese Medicine, on Phenylhydrazine-induced Anemic Rats. Biomolecules &Therapeutics, 9(1), 1-6.
- Ozyilkan, O., Colak, D., Akcali, Z., & Basturk, B. (2005). Olive: Fruit of peace against cancer. Asian Pacific Journal of Cancer Prevention, 6(1), 77-82.
- Kone, W. M., Koffi, A. G., Bomisso, E. L., & Bi, F. T. (2012).Ethnomedical study and iron content of some medicinal herbsused in traditional medicine in Cote d’Ivoire for the treatment of anaemia. African Journal of Traditional, Complementary and Alternative Medicines, 9(1), 81-87.
- Hu, L., Zhang, J. and Li, D. (2020). Effect of vitamin C on non-heme iron absorption: A review. Journal of Clinical Nutrition, 59(4), 323-330.



