Research Article - (2025) Volume 10, Issue 1
Effect of Different Edible Oil Coating on Post-Harvest Quality and Storage Life of Banana Fruit
2Department of Botany, University of Science and Technology Bannu, Pakistan
3Department of Agriculture Extension and Communication Education, The University of Agriculture Peshawar, Khyber Pakhtunkhwa, Pakistan
Received Date: Jan 24, 2025 / Accepted Date: Feb 10, 2025 / Published Date: Feb 28, 2025
Copyright: ©©2025 Ahmad Farooq, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Asghar, M., Khattak, A. M., Farooq, A., Habib, W., Bibi, R. et al. (2025). Effect of Different Edible Oil Coating on Post-Harvest Quality and Storage Life of Banana Fruit. Adv Nutr Food Sci, 10(1), 01-10.
Abstract
An experiment was carried out to study the “Effect of different edible oil coatings on postharvest quality and storage life of banana fruits” at the Postharvest Laboratory, Department of Horticulture, The University of Agriculture, Peshawar, Pakistan, during 2021-2022. The banana fruit cultivar (cv) Cavendish was obtained from a local market in Peshawar at the physiologically mature stage. The experiment was laid out using a completely randomized design (CRD) with two factors replicated two times. The fruits were then kept in packaging materials with one apple (cv. Anna) per bunch. When the fruits were ripened, they were divided into two groups: one group was coated with edible oil (olive, coconut and butter oil), and the other was stored (0, 3, 6, 9, 12 days) and kept at room temperature (26°C) at 90%-95% RH for 12 days. The data regarding banana fruits coated with butter oil showed the maximum fruit firmness (3.29 kg/cm2), titratable acidity (0.28%), ascorbic acid content (4.86 mg/100 g), fruit color score (7.3), fruit taste score (7.8) and fruit decay (0%). The maximum TSS concentration was observed in banana fruits coated with olive oil (26.19 °Brix). For the other means for storage duration, the maximum fruit firmness (4 kg/cm2), minimum fruit decay (0%) and minimum TSS (24 °Brix) were recorded for freshly ripened banana fruits. The maximum taste score and color score were observed for fruits stored for 12 days. Most of the studied attributes were significantly affected by the T × SD interaction. The present results revealed that banana fruits harvested at physiological maturity ripened under natural ripening agents, i.e., apple, coated with butter oil retained most of the quality attributes for 12 days, are recommended for better shelf life and consumer preference. Because of the impact of edible oil coatings on banana plants, consumer performance can be assessed through various methods, sensory evaluation, shelf life, quality attribute assessment and cost benefit analysis.
Keywords
Edible Coating, Postharvest, Storage Life, Banana Fruit, Musa Paradidica
Introduction
Banana (Musa paradidica L.) belongs to the family Musaceae. Banana is an edible fruit produced by several kinds of large herbaceous flowering plants in the genus Musa [1]. Banana evolved in the humid tropical regions of Southeast Asia, with India as one of its centers of origin. Modern edible varieties have evolved from two species—Musa acuminata and Musa balbisiana—and their natural hybrids, which were originally found in the rainforests of Southeast Asia. At present, banana plants are cultivated throughout warm tropical regions of the world between 300°N and 300°S at the equator [2]. Bananas are predominantly produced in Asia, Latin America and Africa. The largest producers are India and China, which produce 29 and 11 million tons per year, respectively. Most of the production in both countries serves the domestic market. Other large producers are the Philippines, Ecuador and Brazil [3]. Approximately 113.21 million tons of bananas are produced worldwide per year [3]. Approximately 5.6 million hectares of land are dedicated to banana production globally. The rapid expansion of the banana industry is evident in the evolution of the harvested area over time, which amounted to 3.6 million hectares in 1993 and 4.6 million hectares in 2000 [4]. In 2017-2018, the total area of Pakistan cultivated on bananas was 30.1 thousand hectares, with an average production of 135.1 thousand tons. The area under banana cultivation in Punjab is 0.2, that in Sindh is 28.1, that in Khyber Pakhtunkhwa is 0.7, and that in Balochistan is 1.1 thousand hectares, with total productions of 0.9, 109.5, 13, and 11.7 thousand tons, respectively [5]. Moreover, edible coatings, which intend to reduce ripening processes and protect fruits from water loss and spoilage, may be a good way to increase the shelf life of products. More recently, the inclusion of additives, such as essential oils and their constituents with antimicrobial and antioxidant activities, into these edible coatings to increase their effectiveness has been reported and patented [6]. Edible oils such as coconut, olive, butter oil, custard, and paraffin are used for coating fruits and vegetables [7]. The importance of a natural ripening agent for banana (both commercially and nutritionally) and problems related to poor shelf life and quality should be considered.
The use of an edible coating can reduce the respiration rate and retain the quality of banana plants; therefore, the present research was conducted with the following objectives:
• To identify an alternative source of ripening agent to calcium carbide, i.e., a natural ripening agent for the ripening of bananas.
• The interactive effect of edible oil and storage life on the storage performance of banana plants was investigated.
Materials and Methods
Experimental Site and Plant Material
The experiment was carried out to study the effect of different edible oil coatings and storage durations on the postharvest quality of banana fruits ripened with the natural ripening agent apple (cv. Anna) at the Department of Horticulture, The University of Agriculture Peshawar, Pakistan, during 2021-2022. Banana fruit cv. cavendish was obtained from the Peshawar local market brought from Hyderabad during the month of April 2021 at the physiologically mature stage.
Experimental Design and Treatment Combinations
The experiment was laid out using a completely randomized design (CRD) with two factors replicated two times. Banana fruits were taken at the physiologically mature stage from a local market in the Peshawar. The plants were treated with edible oil (olive, coconut and butter oil) and kept in cotton crates for 12 days at a room temperature of 26 °C and a relative humidity (RH) of 90%- 95%. The data were collected after 3 days.
Treatment with the Ripening Agent Apple
The banana fruits were carefully transported to wooden boxes for storage. The injured and bruised fruits were discarded. The fruits were washed with tap water and then sun-dried. Qualitative attribute data were recorded before and after ripening, and approximately 168 banana fruits at physiological maturity from the whole fruits were taken and ripened under polyethylene with 130 g of apple without slices in each bunch [8]. After ripening, the apples were removed from the packaging materials [9].
Preparation of Edible Oil
Edible oil was taken from the local market, Board Bazar, Peshawar. Twenty-four milliliters of olive oil was coated on 12 bananas; each banana was coated completely with 2 mL of olive oil through cotton. A similar procedure was carried out for coating banana fruits with coconut and butter oil [10].
Studied Parameters
Fruit Firmness (kg/cm2): Fruit firmness was determined by a penetrometer (FT 327, McCORMICK, Italy) with a probe diameter of 8 mm.
Procedure: A thin peel was removed from the fruit, and the probe of the penetrometer was used to penetrate the fruit. The reading displayed on the screen was recorded. At least two readings were taken from each sample [11].
Total Soluble Solids (°Brix): TSS was analyzed using the standard method of the Association of Official Agricultural Chemists with an Abbe refractometer (0-32%, Kernco, Japan) [12].
Procedure: The juice from each sample was put into a separate beaker. A small drop of the juice was placed on the lens of the refractometer. The lid of the lens was closed slowly, and a fine reading was taken by adjusting the scale [13].
Titratable Acidity (%)
Procedure: TA was determined by using the titrimetric method [12]. A 5 mL sample was taken in a graduated cylinder. The solution was then diluted with distilled water to a final volume of 50 mL. Then, a 10 mL sample was taken from the 50 mL solution in a flat bottom flask. The solution was titrated against NaOH. The decrease in the amount of NaOH in the burette was noted, and the results were recorded. This procedure was repeated for each sample [14].
NaOH Solution Standardization
Oxalic acid (6.3 g) was dissolved in distilled water, and the volume was adjusted to 1 L [15]. Again and 4 g of NaOH were dissolved in distilled water, and the volume was adjusted to 1 L. The total volume of NaOH was removed from the burette, and 10 mL of oxalic acid was added to the conical flask. One to two drops of phenolphthalein were added to the conical flask as an indicator. A pink color appeared due to the titration of NaOH solution against oxalic acid. The normality of the NaOH concentration was noted after three consecutive readings using the following formula:
N1V1 = N2V2
where N1 = volume of oxalic acid, V1 = normality of oxalic acid, and N2 = normality of NaOH.
V2 = Volume of NaOH.
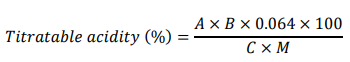
A = NaOH normality, B = NaOH (0.1 mL), M = diluted sample taken (mL), C = diluted sample taken for titration and 0.064 is a constant factor (mL).
Ascorbic Acid (Vitamin C)
Procedure: Five milliliters of juice was taken from each sample and diluted with 50 mL of oxalic acid. A 10 mL sample was taken from the 50 mL solution, and it was titrated against the dye. The reading on the burette was noted, and the result was calculated [16].
Standardization and Preparation of Dye Solution
To prepare the dye solution, 50 g of 2,6-dichlorophenol indophenol and 42 mg of NaHCO3 were added to distilled water, and the volume was adjusted to 250 mL. In a conical flask, 50 mL of typical ascorbic acid (vitamin C) was added [17]. The solution was diluted with 0.4% oxalic acid. Then, 2 mL of diluted ascorbic acid was titrated against the dye solution until a light pink color appeared, after which the mixture was incubated for 14 seconds. The dye factor was determined using the following equation:
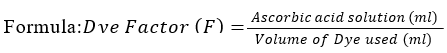
Sample Titration
To determine the content of vitamin C, a fruit sample was extracted. The solution was diluted with 0.4% oxalic acid, and the volume was adjusted to 100 mL. A 10 mL sample was taken from the 100 mL solution and titrated against the dye until a light pink color appeared [18]. Three consecutive readings were taken from all the samples. The ascorbic acid content was determined using the following formula:
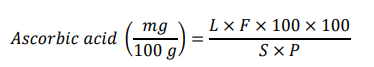
where F = Dye factor, L = mL of dye used, S = mL of dilute solution taken for titration, P = Sample volume for titration
Weight Loss (%)
Procedure: One fruit in each treatment was marked for weight loss analysis [19]. The percent weight loss was calculated using the following formula:

Decay Percentage (%)
At the end of the research, in each treatment, the rotten fruits were counted, and the decay percentage was calculated by using the following formula [12].
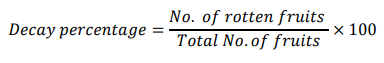
Taste and Color
Sensory evaluations of the banana samples were performed according to the methods outlined by using a twenty-member semi panelist consisting of students from the Department of Food Science and Technology, The University of Agriculture Peshawar, Khyber Pakhtunkhwa, Pakistan and some staff from the Food Processing Laboratory [20]. The panelists were either regular or occasional consumers of banana and were not allergic to any food. Banana samples subjected to different treatments were placed in coded white plastic containers. Water was provided to rinse the mouth between evaluations. The samples were presented randomly. The panelists were instructed to evaluate the coded samples for taste and color. Each sensory attribute was rated on a 9-point hedonic scale (9 for liked extremely and 1 for disliked extremely).
|
Score |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
|
Description |
Disliked extremely |
Disliked very much |
Disliked very much |
Disliked slightly |
Neither liked nor disliked |
Liked slightly |
Liked moderately |
Liked very much |
Liked extremely |
Table A: Table of Sensory Evaluations of Banana for Color
Statistical Analysis
The experimental data were analyzed using the software “Statistix 8.1 (Analytical software, Tallahassee, USA)”. When the data were significant, the least significant difference test (LSD) was used for mean comparison [21].
Results and Discussion
Firmness (kg/cm2)
The data relating to firmness are presented in Table 1 and Figure. 1. Statistical analyses revealed significant differences in fruit firmness among the edible coating and storage life conditions. The interaction between the edible coating and the storage life was significant. The mean data showed that banana fruits coated with butter oil had the highest firmness (3.29 kg/cm2) and the lowest firmness (2.80 kg/cm2) recorded for those coated with olive oil. The mean storage duration maximum firmness was found for fresh ripened fruit (4.00 kg/cm2), while the minimum firmness was found (1.86 kg/cm2) after 12 days. The treatment and storage duration interaction (T×SD) showed that the greatest firmness (3.85 kg/cm2) was observed for banana fruits coated with butter oil for 3 days, and the lowest firmness was recorded for banana fruits not treated with coating materials for 12 days.
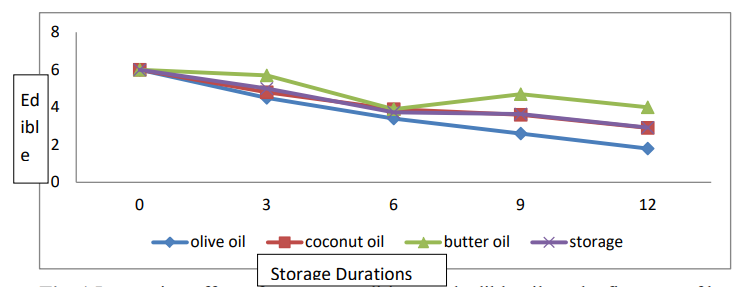
Figure 1: Interaction Effect of Storage Conditions and Edible Oil on the Firmness of Banana Fruits
|
Treatment Storage Time |
Firmness (kg/cm2) |
Attributes Total Soluble Solids (TSS °brix) |
Titratable Acidit (%) |
Ascorbic acid (mg/100 g) |
|
|
0 |
|
4 |
24 |
0.32 |
6 |
|
3 |
3.51 |
25.51 |
0.29 |
5.00 |
|
|
6 |
3.11 |
26.11 |
0.26 |
3.73 |
|
|
9 |
2.59 |
26.63 |
0.23 |
3.64 |
|
|
12 |
1.86 |
27.03 |
0.19 |
2.92 |
|
|
LSD (P≤ 5%) |
0.16* |
0.22* |
0.017* |
0.23* |
|
|
Coating Oil |
|||||
|
Olive Oil |
2.80 |
26.19 |
0.24 |
3.66 |
|
|
Coconut Oil |
2.95 |
25.89 |
0.25 |
4.25 |
|
|
Butter oil |
3.29 |
25.49 |
0.28 |
4.86 |
|
|
LSD (P≤ 5%) |
0.13* |
0.18* |
0.014* |
0.20* |
|
|
Interaction storage time× coating oil |
* |
NS |
NS |
* |
|
Table 1: Physicochemical Attributes of Banana Fruits Affected by Coating with Edible Oil and by Storage Life
The mean firmness gradually decreased as the storage interval increased in both the treated and untreated treatments. At the end, the control clearly showed the lowest firmness.
The retention of firmness can be explained by the decreased degradation insoluble protection afforded by the more soluble pectic acid and pectin. During fruit ripening, depolymerization or shortening of the chain length of pectin substances occurs via increased pectin sterase and polygalactronase activities [22]. Low oxygen and high carbon dioxide concentrations reduce the activities of these enzymes and allow retention of firmness during storage [23]. The application of a coating reduced the respiration rate of fruits after harvest, decreasing the amount of catabolic work and maintaining a decrease in firmness [24]. Our results strongly support the following conclusions. The edible coating significantly retained the firmness of Anna apple Malus domestica Borkh [25].
Total Soluble Solids (TSS °Brix)
The mean TSS data for the bananas are shown in Table 1. Statistical analyses revealed significant differences in the edible coating and storage life in terms of TSS. However, the interaction effect between the edible coating and storage life was not significant. The mean data showed that the maximum TSS concentration (26.19 °Brix) was detected in banana fruits coated with olive oil, followed by the maximum TSS concentration (25.89 °Brix) in banana fruits coated with coconut oil, while the lowest TSS concentration (25.49 °Brix) was detected in banana fruits coated with butter oil. For the mean storage duration, the highest TSS (27.03° brix) was recorded in banana fruits stored for 12 days, and the minimum TSS (24.00° brix) was observed in freshly harvested banana fruits after ripening.
It is evident from the mean table that the maximum TSS was recorded in the fruit stored for 12 days, while the minimum TSS was recorded in freshly harvested fruits. reported that an increase in the TSS of citrus fruits is mainly due to the breakdown of complex carbohydrates into sugar and the low moisture content of the fruit [26,27]. Additionally, an increase in the total soluble solids (TSS) level was found in all three treatments from day 1 to day 5. On the 5th day, the TSS concentration was significantly greater than that on the 1st day; thus, the genetic composition of the fruit plays a major role in increasing the total soluble solids (TSS) concentration day by day. A further more rapid increase in TSS due to a higher rate of cell metabolism results in a reduction in total acidity by converting different acids into sugars to soften the fruits, thus leading to a faster senescence process. Our result reflects the finding of that the TSS concentration is more slowly increased in mango fruits coated with butter oil than in fruits from other treatment groups [7].
Titratable Acidity (%)
The mean data regarding the titratable acidity of the bananas are shown in Table 1. Statistical analyses revealed significant differences in the edible coating and storage life in terms of titratable acidity. However, the interaction effect between the edible coating and storage life was not significant. The mean data showed that banana fruits coated with butter oil had the highest TA content (0.28%), which was significantly similar to the TA content (0.25%) in banana fruits coated with coconut oil, and the minimum TA content was recorded in banana fruits coated with olive oil. Concerning the mean data for storage duration, the maximum titratable acidity (0.32%) was recorded for freshly harvested ripe banana fruits, while the minimum titratable acidity (0.17%) was observed for Banana fruits stored for 12 days.
The results showed that the titratable acidity values gradually and significantly decreased with increasing storage duration. The untreated sample had the lowest level of titratable acidity at the end of the storage period, while the acidity in the other treatment groups was greater than that in the control group. Since organic acids such as malic or citric acid are primary substances for respiration, a reduction in acidity and hence an increase in pH are expected in highly respiring fruits. Coating may reduce respiration rates and may therefore delay the utilization of organic acids [22]. Therefore, our results are consistent with the findings of, who showed that coated apples exhibit a significant delay in titratable acidity [25].
Ascorbic Acid (mg/100 g)
The data relating to ascorbic acid are presented in Table 1 and Figure 2. Statistical analyses revealed significant differences in terms of ascorbic acid consumption and storage life among the edible coatings. The interaction between the edible coating and the storage life was significant. The mean data showed that the maximum ascorbic acid concentration (4.86 mg/100 g) was detected in banana fruits coated with butter oil, and the minimum ascorbic acid concentration (3.66 mg/100 g) was detected in banana fruits treated with olive oil. Regarding the mean storage duration, the highest ascorbic acid concentration (6.00 mg/100 g) was observed in freshly harvested ripe banana fruits, and the lowest ascorbic acid concentration (2.92 mg/100 g) was recorded in banana fruits stored for 12 days. The (T × SD) interaction showed that the highest ascorbic acid concentration (6.00 mg/100 g) was found in freshly harvested ripened banana fruits, and the lowest ascorbic acid concentration (0.60 mg/100 g) was found in banana fruits stored for 12 days.
Our results showed that the ascorbic acid content was greatest in fresh fruits after the last storage. Additionally, coating controlled the vitamin C content to some extent, and the best result was obtained for fruits coated with butter oil. The worst results were obtained for the uncoated and unpacked fruits. Ascorbic acid is water soluble and, for that reason, is depleted with moisture loss. The castor oil coating retained ascorbic acid content by reducing water loss and slowing the ripening process. Castor oil also contains antioxidants that inhibit oxidation of the fruit and, as a result, retain the ascorbic acid content of the fruit [28]. Therefore, our results are in good agreement with the results of, in which the maximum ascorbic acid concentration was found in the plum fruit- coated castor oil, followed by all the other treatments, including the control [29]. Influence of edible coating and storage duration on postharvest plum performance
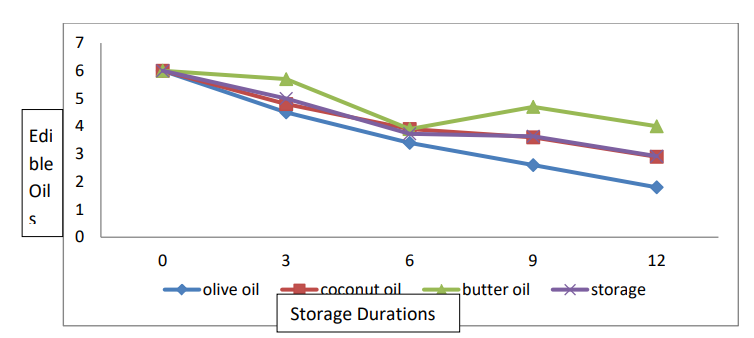
Figure 2: Effect of the Interaction between Storage Conditions and Edible Oil on the Ascorbic Acid Content of Banana Fruits
Weight Loss (%)
The data relating to weight loss are presented in Table 2 and Figure 3. Statistical analyses revealed significant differences in fruit weight loss among the edible coating and storage life conditions. The interaction between the edible coating and the storage life was significant. The mean weight loss (5.05%) was recorded for banana fruits coated with olive oil, followed by weight loss (4.56%) for those treated with coconut oil. The lowest weight loss (3.79%) was observed for the banana fruits coated with butter oil. For the mean data for storage duration, the greatest weight loss (9.3%) was recorded for the banana fruits stored for 12 days. The lowest weight loss (2.52%) was observed for the banana fruits stored for 3 days. With respect to the T×SD interaction, more weight loss (11.66%) was recorded for banana fruits not treated with coating materials for 12 days, while less weight loss (1.77%) was observed for banana fruits coated with butter oil stored for 3 days.
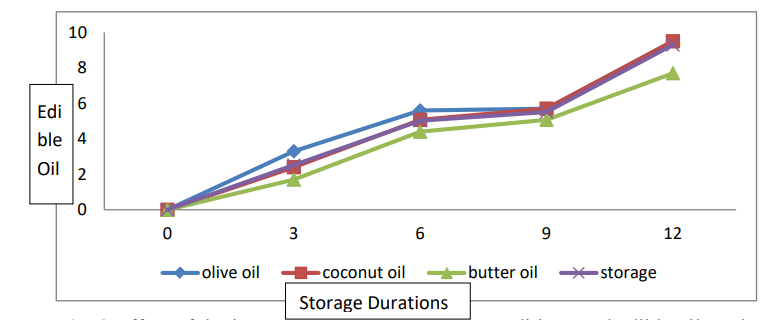
Figure 3: Effect of the Interaction between Storage Conditions and Edible Oil on the Weight Loss of Banana Fruits
|
Treatment Attributes Storage Time Weight loss (%) Decay percentage (%) Taste Color |
||||
|
0 |
0 |
0 |
6 |
4 |
|
3 |
2.52 |
0 |
6.33 |
6.16 |
|
6 |
5.02 |
0 |
7.16 |
7.16 |
|
9 |
5.51 |
3.47 |
7.66 |
7.5 |
|
12 |
9.3 |
11.10 |
7.67 |
7.66 |
|
LSD (P≤ 5%) |
0.08* |
0.9* |
0.18* |
0.18* |
|
Coating Oil |
||||
|
Olive Oil |
5.05 |
5.41 |
6.6 |
6.2 |
|
Coconut Oil |
4.56 |
3.33 |
6.5 |
6 |
|
Butter oil |
3.79 |
0 |
7.8 |
7.3 |
|
LSD (P≤ 5%) |
0.07* |
0.7* |
0.15* |
0.15* |
|
Interaction storage time× coating oil |
* |
* |
* |
|
Table 2: Weight Loss, Decay Percentage, Taste and Color of Banana Fruits as Affected by Edible Oil Coating and Storage Life
The primary mechanism of moisture loss from fresh fruits and vegetables is vapor-phase diffusion driven by a gradient of water vapor pressure at different locations [22]. On the other hand, respiration causes a weight reduction because a carbon atom is lost from the fruit during each cycle [30]. Wax coating decreases the rate of respiration and transpiration, which results in reduced weight loss of up to 65% compared to that of the control sample in the apple cultivar Tsugaru [31]. Weight loss in the control group was greater than that in the wax-treated group due to the high rates of transpiration and respiration. This is because of the barrier that is provided by the wax coating between the inner and outer environment of the fruits, which allows the weight of the fruits to be maintained throughout storage [32]. Therefore, our result is in agreement with the finding of that a butter oil coating has antioxidant and hydrophobic properties and hence significantly retains weight loss in mango plants [7].
Decay Percentage (%)
The data relating to the decay percentage are presented in Table 2 and Figure 4. Statistical analyses revealed significant differences in fruit weight loss among the edible oil coating and storage life conditions. The interaction between the edible oil coating and the storage life was significant. The mean fruit decay (5.41%) was the most common decay in banana fruits coated with olive oil, followed by that in banana fruits coated with coconut oil (3.33%). The lowest fruit decay (0%) was observed for banana fruits coated with butter oil. For the mean storage duration, the highest fruit decay (11.10%) was recorded for the banana fruits stored for 12 days. The lowest fruit decay (0%) was observed for freshly harvested ripened banana plants. With respect to the T×SD interaction, more fruit decay (25%) was recorded for the Banana fruits not treated with the coating materials and stored for 12 days.
The results showed that no signs of decay were observed after 6 days of storage. Compared with the control sample, the coating significantly reduced the percent decay during the storage period. The decay percentage of the control fruits at the end of the storage period was the highest, followed by the decay percentage of fruits coated with butter oil. The decrease in decay percentage of the treated sample was probably due to the effect of these coatings and packaging on delaying senescence, which allowed the commodity to be more vulnerable to pathogenic infection as a result of loss of cellular or tissue integrity [33]. Our result is in agreement with, who observed that an edible oil coating prevented the fruit (Anna apple) from decaying [25].
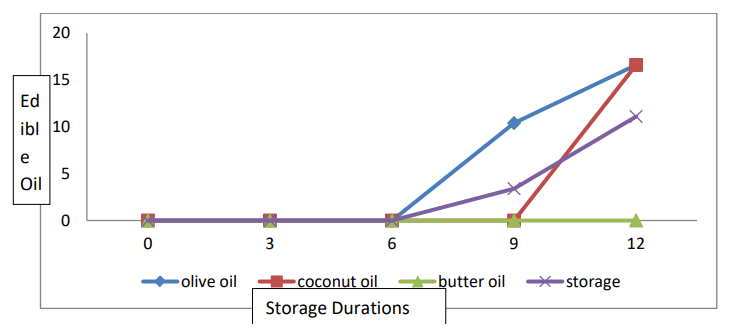
Figure 4: Interaction Effect of Storage Conditions and Edible Oil on the Percentage of Decayed Banana Fruit
Taste
The data relating to taste are presented in Table 2 and Figure 5. Statistical analyses revealed significant differences in taste among the edible oil coatings and in terms of storage life. The interaction between the edible oil coating and the storage life was significant. The mean taste score (7.8) was the highest for the banana fruits coated with butter oil. The lowest taste score (6.6) was observed for banana fruits in olive oil. According to the mean storage duration, the highest taste score (7.67) was recorded for banana fruits stored for 12 days. The lowest taste score (6.00) was observed for freshly harvested ripe banana fruits. Regarding the (T × SD) interaction, a greater taste score (9.00) was recorded for banana fruits treated with the butter oil coating, which were closely followed (9.00) and stored for 12 days.
During storage, the maximum taste was also recorded for fruits stored for 12 days, while the minimum was recorded for freshly harvested fruits. They are synthesized in the cytosol, localized in vacuoles and synthesized via the phenylpropanoid pathway
Two classes of genes are required for anthocyanin biosynthesis: structural genes, which encode enzymes that directly participate in the formation of anthocyanins and other flavonoids, and regulatory genes that control the transcription of structural genes. It has been reported that ethylene is involved in the regulation of genes related to anthocyanin biosynthesis [8]. Astringency, which arises due to tannins in fruits, tends to decrease during ripening. It has been reported that astringency depends on the molecular structure of tannins, which determines the cross-linking of proteins and glycoproteins [34]. Therefore, tannins have an astringent taste when they are dissolved in saliva. An increase in the molecular weight of tannins by polymerization, which occurs during ripening, causes a lack of astringency due to the insolubility of tannins [35]. Our results showed that the best taste was found in banana fruits coated with butter oil. Therefore, our results are in good agreement with the results of, in which the best taste score was found for the plum fruit-coated castor oil, followed by all the other treatments, including the control [29].
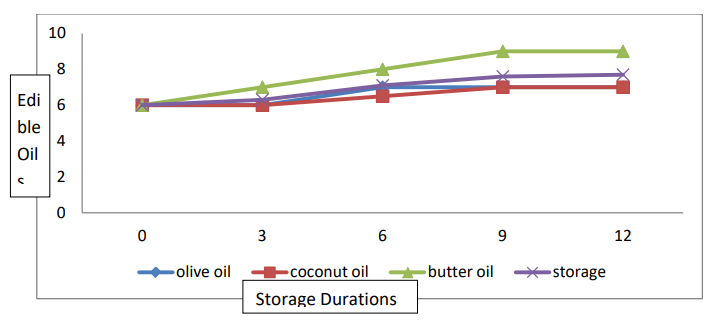
Figure 5: Interaction Effect of Storage Conditions and Edible Oil on the Taste of Banana Fruits
Color
The data relating to color are presented in Table 2 and Figure 6. Statistical analyses revealed significant differences in fruit color among the edible oil coating and storage life conditions. The interaction between the edible oil coating and the storage life was significant. The mean color score (7.3) was recorded for banana fruits coated with butter oil, and the minimum color score (6.00) was observed for banana fruits coated with coconut oil. For the mean storage duration, the highest color score (7.66) was recorded for the banana fruits stored for 12 days. The lowest color score (4.00) was observed for freshly harvested ripe banana fruits. Concerning the interaction and storage duration (T × SD), a greater color score (9.00) was recorded for banana fruits treated with butter oil than for those stored for 12 days (9.00), while the lowest color score (4.00) was observed for freshly harvested ripened banana fruits.
During storage, the maximum color was also recorded for fruits stored for 12 days, while the minimum was recorded for freshly harvested fruits. Color development is an important maturity index of many fruits and is associated with ripening. In many cases, the color change during fruit ripening is due to the unmasking of preexisting pigments by degradation of chlorophylls and synthesis of anthocyanin and carotenoids [36]. Carotenoid biosynthesis during ripening has been studied using tomato plants as a model. Carotenoids are derived from terpenoids and are synthesized in fruits at a high rate during the transition from chloroplasts to chromoplast [37]. Anthocyanins are responsible for orange, red, pink, blue and purple coloration in fruits and can be classified into two groups: flavonoids and phenolic compounds [38]. They are synthesized in the cytosol, localized in vacuoles and synthesized via the phenylpropanoid pathway. Two classes of genes are required for anthocyanin biosynthesis: structural genes, which encode enzymes that directly participate in the formation of anthocyanins and other flavonoids, and regulatory genes that control the transcription of structural genes. It has been reported that ethylene is involved in the regulation of genes related to anthocyanin biosynthesis [8]. Our results showed that the best color was found for banana fruits coated with butter oil. Therefore, our results are in good agreement with the results of, in which the best color score was found for the plum fruit-coated castor oil, followed by all the other treatments, including the control [29]. Influence of edible coating and storage duration on postharvest plum performance.
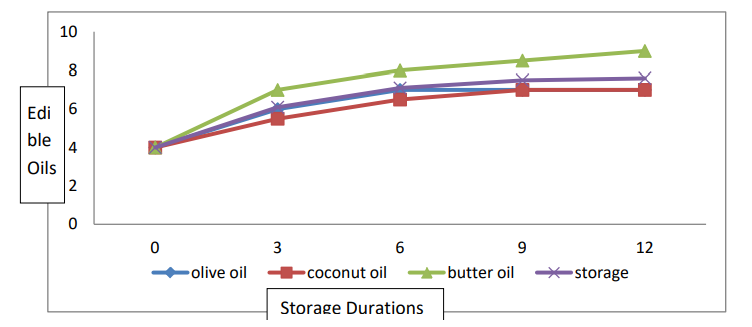
Figure 6: Interaction Effect of Storage Conditions and Edible Oil on the Color of Banana Fruits
Conclusion
It is concluded from the present experimental results that: Fruiting banana plants coated with butter oil significantly increased their firmness, titratable acidity (TA), and ascorbic acid (AA) content, as indicated by improved taste and color. Butter oil decreased weight loss and fruit decay compared to those of the other edible oils and the control treatment. The qualitative attributes of all the banana plants were significantly affected by storage duration. However, with increasing storage duration up to 12 days, the firmness, TA, and AA significantly decreased, while the TSS content, weight loss, and fruit decay increased. Furthermore, regarding the interaction between treatment and storage duration (TxSD), ascorbic acid (AA), firmness, color, taste, weight loss, and decay percentage were significantly affected in banana plants, but not in those with titratable acidity (TA) or total soluble solids (TSS).
Conflict of Interest Statement
The author(s) declare(s) that there are no conflicts of interest.
Declaration Section
Ethics Approval and Consent to Participate
The study was approved by Institutional Ethics Committee (Approval No. 61), and informed consent was obtained from all
Consent for Publication
Consent for publication was obtained from all participants included in the study.
Availability of Data and Materials
The data and materials are already mentioned in main manuscript.
Competing Interests
The author(s) declare(s) that there is no conflict of interest
Funding
We are not receiving any funds for this work, nor is there any funding available
Authors' Contributions - Provide Individual Author Contribution
Mr. Muhammad Asghar was the main author to conduct the research under the supervision of “Prof. Dr. Abdul Mateen Khattak,” Mr. Ahmad Farooq was helped me throughout the research, Mr. Waqas Habib and Ms. Razia Bibi are helped me in data analyses, Dr. Tariq Rahim guide me in graph preparation, Mr. Manzoor Ahmad help me in lab work.
Acknowledgement
Thanks to my parents, beloved brother, friends and Respectable teachers how support me throughout the research work
Authors Information
Mr. Muhammad Asghar (MS Horticulture) The University of Agriculture Peshawar
Prof. Dr. Abdul Mateen Khattak (Ph.D. in Horticulture) The University of Agriculture Peshawar
Mr. Ahmad Farooq (MS Horticulture) The University of Agriculture Peshawar
Mr. Waqas Habib (MS Horticulture) The University of Agriculture Peshawar
Ms. Razia Bibi (M. Phil in Botany) University of Science and Technology Bannu
Dr. Tariq Rahim (Ph.D. in Agriculture Extension) The University of Agriculture Peshawar
Mr. Manzoor Ahmad (MS Horticulture) The University of Agriculture Peshawar
Statements & Declarations
The authors declare no conflicts of interest. All data supporting the findings of this study are available upon request. This manuscript has not been previously published and is not under consideration elsewhere.
Acknowledgement
I would like to express my sincere gratitude to (Abdul Mateen Khattak, Ahmad Farooq, Waqas Habib, Razia Bibi, Tariq Rahim, Manzoor Ahmed and Shahbaz Ahmad) for their invaluable assistance, guidance, and support throughout my research. Their contributions were instrumental in the successful completion of this work.
References
- Memon, I. N., Wagan, H., Noonari, S., Lakhio, M. H., & Lanjar, B. A. (2016). Economic analysis of banana production under contract farming in Sindh Pakistan. Economic Analysis, 21, 14-21.
- Bantayehu, M. (2017). Fruit ripening and postharvest life of banana varieties at different temperatures and packaging. Journal of postharvest Technology, 5(1), 30-42.
- FAO: (2018) Food and agriculture Organization.
- FAOSTAT. (2017). Banana facts and figures.
- MNFSR. 2018. Ministry of National Food Security & Research. retrieved on 22, May 2021.
- D Antunes, M., M Gago, C., M Cavaco, A., & G Miguel, M. (2012). Edible coatings enriched with essential oils and their compounds for fresh and fresh-cut fruit. Recent patents on food, nutrition & agriculture, 4(2), 114-122.
- Baloch, M. K., Bibi, F., & Jilani, M. S. (2013). Effect of coatings over the quality and shelf life of mango (Mangifera indica L.) fruit. Journal of Food Processing and Preservation,37(1), 66-73.
- Elâ?Kereamy, A., Chervin, C., Roustan, J. P., Cheynier, V., Souquet, J. M., Moutounet, M., ... & Bouzayen, M. (2003). Exogenous ethylene stimulates the longâ?term expression of genes related to anthocyanin biosynthesis in grape berries. Physiologia plantarum, 119(2), 175-182.
- Singal, S., Kumud, M., & Thakral, S. (2012). Application of apple as ripening agent for banana.
- Xue, Z., Yu, Y., Yu, W., Gao, X., Zhang, Y., & Kou, X. (2020).Development prospect and preparation technology of edible oil from microalgae. Frontiers in Marine Science, 7, 402.
- Terasaki, S., Sakurai, N., Kuroki, S., Yamamoto, R., & Nevins, D. J. (2013). A new descriptive method for fruit firmness changes with various softening patterns of kiwifruit. Postharvest biology and technology, 86, 85-90.
- AOAC. (2012) Association of official and analytical chemist.
- Li, J. L., Sun, D. W., & Cheng, J. H. (2016). Recent advances in nondestructive analytical techniques for determining the total soluble solids in fruits: a review. Comprehensive Reviews in Food Science and Food Safety, 15(5), 897-911.
- Tyl, C., & Sadler, G. D. (2017). pH and titratable acidity. Food analysis, 389-406.
- Syrovaya, A., Tishakova, T., Savelieva, E., Petyunina, V., Makarov, V., Lukianova, L., ... & Kozub, S. (2017). Fundamentals of titrimetric analysis. Preparation and standardization of NaOH operating solution.
- Cunha-Santos, E. C. E., Viganó, J., Neves, D. A., Martínez, J., & Godoy, H. T. (2019). Vitamin C in camu-camu [Myrciaria dubia (HBK) McVaugh]: evaluation of extraction and analytical methods. Food Research International, 115, 160- 166.
- Resch-Genger, U., & Rurack, K. (2013). Determination of the photoluminescence quantum yield of dilute dye solutions (IUPAC Technical Report). Pure and Applied Chemistry, 85(10), 2005-2013.
- Beck, S., Méthot, M., & Bouchard, J. (2015). General procedure for determining cellulose nanocrystal sulfate half- ester content by conductometric titration. Cellulose, 22, 101- 116.
- Sohail, M., Ayub, M., Khalil, S. A., Zeb, A., Ullah, F., Afridi,S. R., & Ullah, R. (2015). Effect of calcium chloride treatment on post harvest quality of peach fruit during cold storage. International Food Research Journal, 22(6).
- Ukwo, S. P., Inyang, M. E., Udo, M. E., & Udofia, E. I. (2023). Quality parameters of banana fruit as influenced by traditional ripening techniques. Research Journal of Food Science and Quality Control (RJFSQC) E-ISSN, 2756, 5483.
- Myers, J. L., Well, A. D., & Lorch Jr, R. F. (2013). Research design and statistical analysis. Routledge.
- Bovi, G. G., Caleb, O. J., Linke, M., Rauh, C., & Mahajan, P.V. (2016). Transpiration and moisture evolution in packaged fresh horticultural produce and the role of integrated mathematical models: A review. Biosystems Engineering, 150, 24-39.
- Krupa, T., & Tomala, K. (2021). Effect of oxygen and carbon dioxide concentration on the quality of minikiwi fruits after storage. Agronomy, 11(11), 2251.
- Riva, S. C., Opara, U. O., & Fawole, O. A. (2020). Recent developments on postharvest application of edible coatings on stone fruit: A review. Scientia Horticulturae, 262, 109074.
- El-Anany, A. M., Hassan, G. F. A., & Ali, F. R. (2009). Effects of edible coatings on the shelf-life and quality of Anna apple (Malus domestica Borkh) during cold storage.
- Bindu, M. S., Jatinder, M., & Amandeep, K. (2017). Effect of fruit ripening agents on composition and storage quality of muskmelon. Int. J. Curr. Microbiol. App. Sci, 6(9), 2012- 2018.\
- Khan, M. N., Nawaz, M. A., Waqar Ahmad, W. A., Muhammad Afzal, M. A., Malik, A. U., & Saleem, B. A. (2010). Evaluation of some exotic cultivars of sweet orange in Punjab, Pakistan.
- Panigrahi, J., Patel, M., Patel, N., Gheewala, B., & Gantait,S. (2018). Changes in antioxidant and biochemical activities in castor oil-coated Capsicum annuum L. during postharvest storage. 3 Biotech, 8(6), 280.
- Shah, S. T., A. Basit, I. Ullah, M. Sajid, I. Ahmad, I. Ahmad,M.A. Khalid, S. Ullah, I. Ullah and B. Muhammad. (2021). Influence of edible coating and storage duration on postharvest performance of plum. Pure App. Biol. 10(1): 81-96.
- Ramaswamy, H. S. (2014). Post-harvest technologies of fruits& vegetables. DEStech Publications, Inc.
- Tzia, C., Tasios, L., Spiliotaki, T., Chranioti, C., & Giannou,V. (2015). Edible coatings and films to preserve quality of fresh fruits and vegetables.
- Kumar, P., & Sethi, S. (2018). Edible coating for fresh fruit: A review. International Journal of Current Microbiology and Applied Sciences, 7(05), 2619-2626.
- Banerjee, S., & Basu, P. S. (1992). Hormonal regulation of flowering and fruit development: Effect of gibberellic acid and ethrel on fruit setting and development of Momordica charantia L. Biologia plantarum, 34(1), 63-70.
- Soares, S., Brandão, E., Guerreiro, C., Soares, S., Mateus, N., & De Freitas, V. (2020). Tannins in food: Insights into the molecular perception of astringency and bitter taste. Molecules, 25(11), 2590.
- Jackson, R.S. (2008). Wine Science: Principles and Applications, Academic Press.
- Maduwanthi, S. D. T., & Marapana, R. A. U. J. (2019). Induced ripening agents and their effect on fruit quality of banana. International journal of food science, 2019(1), 2520179.
- Bouzayen, M., A. Latche, P. Nath and J.C. Pech. (2010). Mechanism of fruit ripening, in plant developmental biology- biotechnological perspectives, Pp. 319–339, Springer, Berlin, Germany.
- Rogez, H., Pompeu, D. R., Akwie, S. N. T., & Larondelle,Y. (2011). Sigmoidal kinetics of anthocyanin accumulation during fruit ripening: A comparison between açai fruits (Euterpe oleracea) and other anthocyanin-rich fruits. Journal of Food Composition and Analysis, 24(6), 796-800.



