Research Article - (2025) Volume 4, Issue 2
Combined Effect of Zingiber Officinale Rhizomes Extracts and Dried Fruits of Tetrapleura Tetraptera on Attenuated Monosodium-Iodoacetate-Induced Osteoarthritis Via its Analgesic, Anti-Inflammatory, Antioxidant and Anxiolytic Effects
2Laboratory of Biology and Physiology of Animal Organisms, Department of Biology of Animal Organisms, University of Douala, Cameroon
3Department of Biomedical Sciences, Faculty of Health Sciences, University of Buea, Cameroon
4Research Unit of Animal Physiology and Phytopharmacology, Department of Animal Biology, Faculty of Science, University of Dschang, Cameroon
Received Date: Sep 06, 2025 / Accepted Date: Oct 15, 2025 / Published Date: Oct 27, 2025
Copyright: ©�2025 Mbiantcha Marius, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Djami, M. B., Bilanda, D. C., Atsamo, A. D., Yousseu Nana, W., Bomba Tatsinkou, F. D., et al. (2025). Combined Effect of Zingiber Officinale Rhizomes Extracts and Dried Fruits of Tetrapleura Tetraptera on Attenuated Monosodium-Iodoacetate-Induced Osteoarthritis Via its Analgesic, Anti-Inflammatory, Antioxidant and Anxiolytic Effects. J Traditional Medicine & Applications, 4(2), 01-13.
Abstract
Ethnopharmacological Relevance: Zingiber officinale and Tetrapleura tetraptera are used by the population to treat many diseases.
Aim of the Study: This study aimed to evaluate the therapeutic effects of the mixture of dried fruits of Tetrapleura tetraptera extract and the rhizomes of Zingiber officinal extract on osteoarthritis.
Materials and Methods: Osteoarthritis was induced with monosodium-iodoacetate (50 μl, 100 mg/kg, i.a.). The extracts of the mixture of Zingiber officinale and Tetrapleura tetraptera (100, 200 mg/kg) was administered orally. Body mass, thermal hyperalgesia, paw volume, locomotion and anxiety states were assessed. The hematological, biochemical, anti- inflammatory, pro-inflammatory and oxidative stress parameters were measured. The microarchitecture of the knee joint was evaluated.
Results: Extracts (200 mg/kg) have reduced hypersensitivity (69.33%; 79.03%); and also, the paw volume (63.96%; 81.85%). The extracts improved the locomotion and reduced the total time of immobility. Extracts have increased the number of lines crossed and the mobility time episode and have improved the oxidative status of animals. It has inhibited TNF-α, IL-1β, MMP9, MPO, and stimulated IL-4, IL-10. Extracts have ameliorated histopathological alterations in the knee joint.
Conclusions: Results show antihyperalgesia and anti-inflammatory properties of the mixture of Zingiber officinale and Tetrapleura tetraptera, and is able to improve locomotion damage and anxiety status caused by osteoarthritis.
Keywords
Osteoarthritis, Zingiber Officinale, Tetrapleura Tetraptera, Analgesic, Anti-Inflammatory
Introduction
Osteoarthritis, a disabling joint pathology, which implies almost all parts of joint of a joint (the synovial membrane, cartilage, subchondral bone, adipose pads and meniscus). Nearly 250 million people in the world are affected (quality of their life), thus poses an important challenge of public and social health [1]. The structural destruction of the joint with partial and/or total loss of joint function accompanied by chronic pain is the consequence of the pathogenesis of osteoarthritis [2]. Pathophysiology of many human diseases (obesity, cancer, neurological diseases, rheumatoid arthritis and autoimmune diseases) implies chronic inflammation [3]. It is known that, people with osteoarthritis, presented a high level of pro-inflammatory chemokines and cytokines in their synovial fluids [4]. In the pathogenesis of osteoarthritis, synoviocytes, chondrocytes and macrophages release in quantities pro-inflammatory cytokines in the joint which will result an increase in the released of free radicals by degenerating the articular cartilage matrix [5,6]. In patients with osteoarthritis, the level of lipid peroxidation is very high in cartilage and biological fluids, while antioxidant enzymes such as peroxidase (GPX), superoxide dismutase (SOD) and catalase (CAT) are highly reduced [7,8].
The pathological effects of pro-inflammatory cytokines and ROS due to the activation of signaling pathways, resulting in a cascade of events leading to the activation of the NF-κB signaling pathway. This pathway intervenes in the positive feedback regulation of several inflammatory genes of cytokines, iNOS and an increased in the expression of proteases (MMP-1, MMP-9, MMP-13) [9]. Hematological disorders, including anemia, white blood cell, platelet abnormalities, coagulopathy and hematological malignancies can occur in many autoimmune rheumatic diseases [10]. Anxiety has been shown to be one of the psychological states of osteoarthritis. Castano et al. showed that 25 % anxiety cases in 1258 patients with osteoarthritis, and Axford et al. confirm 40.7 % anxiety cases in 54 patients with osteoarthritis [11,12]. Osteoarthritis is a chronic sickness that needs lifelong management with nutritional management and regular use of drugs such as analgesic therapies, steroidal anti-inflammatory drugs, non-steroidal anti-inflammatory drugs, immunosuppressants and biotherapies; these drugs have several limitations [13]. Despite great quantities of current medicines, public around the world are progressively turning to herbal medications for their primary health problems. In view of the key place that these plants occupy in pharmacopoeia, the WHO has suggested that studies be conducted on the effects of plants used in traditional medicine [14].
Tetrapleura tetraptera (T. tetraptera) (Fabaceae-Mimosoideae family) is a tree (20-25 meters) with dried fruit containing pulp [15]. These fruits are traditionally used in many African countries in the treatment of diabetes, hypertension, rheumatic pain and ar¬thritis [16]. In Cameroon, more precisely in the West region, these fruits are used for medicinal properties, nutritional and cultur¬al values [17]. Zingiber oficinale (Z. oficinale) (Zingiberaceae family) is an herbaceous plant with roots (ginger) known for its nutritional and medicinal values. Traditionally, ginger have gastro¬intestinal, cardiovascular, hepatoprotector, immunomodulator and antimicrobial properties [18]. Scientifically, the dried fruits of T. tetraptera have already proven their anti-inflammatory, antiarthri-tis, cardiovascular, neuromuscular and anti-ulcer properties, while Z. Oficinale has shown the anti-carcinogen, anti-inflammatory, anti-hyperglycemic, antioxidant and anti-apoptotics properties [19,20]. Phytochemical tests have shown the presence of flavo-noids, steroids, phenolic compounds, saponins, 7-hydroxy-6-Me-thoxy, Coumarin, Echinocystic Acid, Oleanolic Acid in T. tetrap-tera extract [5,21,22].
Also, the presence of Gingerol, Zingiberol, Beta-Phelllandrene, Cineole, Alpha-Faranesne, Beta-Bisabolene, Géraniol, Linalol and Zingerone was found in Z. oficinale [23]. Taken individually, T. tetraptera dried fruits and Z. oficinale rhizomes have shown pharmacological and nutritional properties. Although consumed for many generations in African societies, no scientific studies have been carried out to demonstrate the effect of these combined two nutrients in the management and/or prevention of a degenerative disease. The present study aimed to assess the therapeutic effects of supplementation of the mixture of dried fruits of T. tetraptera and the rhizomes of Z. oficinale on osteoarthritis animals induced by monosodium-iodoacetate (MIA).
Materials and Methods
Plant Identification and Extracts Preparation
Z. oficinale rhizomes and T. tetraptera dried fruits were bought at the Mendong market (Yaounde, Center Region, Cameroon, July 2023). Both plants were authenticated from the Cameroon National Herbarium compared to specimens deposited under the numbers: 4343SRF/CAM (Z. oficinale) and 2316SRF/CAM (T. tetraptera). Rhizomes and dried fruits were washed, peeled, cut into small pieces, dried in the shade at room temperature, and then crushed in powder. For aqueous extract, 500g of rhizomes powder and 500g of dried fruits powder were separately soaked in 3L of distilled water (agitated, 48 hours). Each mixture was filtered (Wattman n°4) and evaporated an oven (40°C). The extraction of rhizomes gave 7g (3.5%) while the extraction of the dried fruits gave 6g (3%). For the ethanolic extraction, rhizomes powder (250g) and dried fruit powder (250g) were soaked separately in 2L of ethanol (95°, 72h) and each mixture subsequently filtered (Wattman n°4) and concentrated (rotary evaporator, 70°C reduced). The ethanolic extraction rhizomes gave 9g (3.6%) while that of the dried fruits gave 8g (3.2%). The extracts prepared for the present study were made up with 50/50 of the extract of each plant for both extracts: meaning 50/50 aqueous and 50/50 ethanolic.
Chemicals and Reactive
MIA was bought from Markal-Za (France). Analytical kits for IL-1β, TNF-α, IL-10, IL-4, MPO and MMP-9 for Rat immuno-enzymatic tests were purchased from Sigma-Aldrich (St. Louis, Mo, USA). Obtained from Sigma-Aldrich, Ellman and Griess reagents were freshly prepared before use. Drug solutions were prepared fresh and administered per bone in a volume of 10 ml/kg of body weight.
Animals
Wistar rats (males and females, 3 months, 150–180 g) were raised in the pet store of the Animal Physiology Laboratory of the University of Yaoundé I (Cameroon) under the standard lighting (day/night cycle of 12h), animals have free access to a standard diet and the water of the Ad Libitum tap. They were housed in groups of 3 rats per cage (40 cm × 40 cm). All experiences followed the principles of the use and care of laboratory animals of "directives of the European Community" (ECE Directive 2010/63/ EEC) and were approved by the "Ethics Committee" of the Faculty of Sciences, University of Yaoundé I.
Osteoarthritis Induction
After submitting all the animals to the locomotion test (10 min) to assess the locomotor activity, the basic value of pain sensitivity and the initial paw volume of all animals were measured. Subsequently, the animals were anesthetized (diazepam (0.1 mg/ kg)/ketamine (0.2 mg/kg), i.p.), the left knee of each animal was disinfected (iodized alcohol) and MIA (50µl, 100 mg/kg) diluted in 0.9% NaCl were injected into the left knee of each animal.
Grouping and Treatment of Animals
Non-arthritis (six) and arthritis (36) rats were grouped as follows: group 1 (normal control, non-osteoarthritis rat, received NaCl 0.9%); group 2 (negative control, osteoarthritis rat, received NaCl 0.9%); group 3 (positive control, osteoarthritis rat, received diclofenac 5 mg/kg); groups 4, 5, 6 and 7 (tests groups, osteoarthritis rat, received aqueous (100 and 200 mg/kg) and ethanolic (100 and 200 mg/kg) extracts from the mixture of rhizomes and dried fruits. All the treatments were given orally. One hour (1h) after MIA injection, the treatments were administered and then by continued once a day for 21 consecutive days. During this period, the body mass, pain reaction threshold, paw volume and locomotor activity were measured.
Assessment of Antihyperalgesia and Anti-Inflammatory Effects
Each animal was placed on the heating surface (heating plate, Basile Ugo, Italy) (on day 0, 2, 4, 6, 8, 10, 12, 14, 16, 18 and 20), the weather was launched and as soon as the animal licked one of its legs or lifted the paw, the time was stopped. In order to avoid tissue lesions in animals, a maximum stimulation time of 15 seconds was used in the case event of non-reaction of the animal. The paw of each animal was plunged into a 10 ml graduated burette containing water (water plethysmometer) (on days 0, 3, 5, 7, 9, 11, 13, 15, 19 and 21). When the water reached the knee joint, the quantity of water (ml) moved to the burette was a function of the volume of the soaked leg.
Assessment Locomotion and Anxiety Effects
Open field tests (OFT) were carried out before and after the treatment period (between 8:00 p.m. and 11:00 p.m.) in an enclosed quite environment to observe animal record behavior and locomotion. This test was carried out at night due to an intense animal activity [24]. The test field was an open field built in wood (80 cm × 80 cm × 60 cm) with a white bother, divided into squares of 10 cm in length. In the test each rat was introduced into the device and was given 10 minutes to explore it. For the locomotion and anxiety disorder, several parameters were recorded (day 0, 8, 12, 16, 20) automatically recorded and analyzed by a 7.15 video system (Stoelting, Wood Dale, IL, USA) with a high-resolution Logitech camera [25].
Samples Collection and Preparation
On the 21st day, all the animals were anesthetized (thiopental 50 mg/kg, i.p.) then sacrificed. Blood was taken by catheterization of the abdominal aorta and collected into tube without anticoagulant and another tube containing EDTA (Ethylene diamine tetra acetic acid). The blood with anticoagulant was used for the blood formula, while the blood collected without anticoagulant was centrifuged (3000 rpm, 15 min) and the supernatant taken for biochemical analysis. The synovial fluid was taken by joint washing (100 µl, 0.9% NaCl) to determine the level of TNF-α, IL-1β, IL-10, IL-4, matrix metalloprotein 9 (MMP9) and myeloperoxidase (MPO). Liver, kidney, brain was weighed. Part of the joint, liver, kidney and brain were homogenized (crushed) in sorted buffer (10 mm, 4°C, pH7.4) cold at 10%, centrifuged (3000 rpm, 4°C, 25 min) and kept at -20°C for the evaluation of oxidative stress parameters (GSH, SOD, MDA, Catalase, NO). The other part of the knee joint was fixed in 10% formalin for histology.
Histological Analysis
The knee joint was used for routine coloring. This joint was dehydrated in alcohol and integrated into paraffin blocks, and tissues cuts of 5 µm thickness were colored with hematoxyline and eosin. Colorful and climbed slides were observed using a scientific STM-50 optical microscope (HSIDC Industrial Estate, Haryana, India) equipped with a Celestron 44421 digital camera connected to a computer (HP Pavilion, G series).
Statistical Analysis
The results are presented in the form of average ± standard error of the mean (SEM). Statistical analysis and data presentation were carried out using Graph Pad Prism. The data were analyzed using a unidirectional variance analysis (ANOVA) followed by the Tukey-Kramer test; then a multidirectional variance analysis (ANOVA) followed by Bonforoni test.
Results
Effects of on Pain, Inflammation, Body Mass, and Organs
Mass Oral treatments have reduced pain sensitivity whit 69.33% (20th day), 79.03% (10th day) and 23.75% (16th day) respectively for aqueous extract, ethanolic extract and diclofenac; and reduced paw volume with 63.96% (3rd day) for aqueous extract, 81.85% (21st day) for ethanolic extract and 32.43% (3rd day) for Diclofenac. Apart from Diclofenac, all treatments have significantly improved the body mass of animals. The mass of the liver was significantly reduced in the negative control group as compared to the normal control group. The extracts increase the mass of the liver in treated animals (Figure 1).
Figure 1: Effects of the Aqueous and Ethanolic Extracts of the Zingiber Officinale Rhizomes and Dried Fruits of Tetrapleura Tetraptera Mixture on Pain Sensitivity (A), Paw Edema (B), body weight (C), and Organ Weight (D) in arthritic rats. Data are shown as mean ± SEM, n = 6; αp<0.05; βp<0.01; yp<0.001 Significant Compared to Normal Control; ap<0.05; bp<0.01; cp<0.001 Significant Compared to NaCl (0,9%)
Effects on the Spontaneous Locomotion
Figure 2 shows that MIA injection has led to significantly affected the spontaneous locomotion in osteoarthritis animals not treated compared to animals of the normal control group. These results were evident in the illustration representing animal tracks during the test where very low activity was observed in osteoarthritis animals compared to the activity of normal animals (Figure 3). This diagram shows that osteoarthritis rats have a marked decrease in total mobility time, the total displacement distance and a total increase in immobility time as compared to normal control. The administration of the extracts has improved locomotion of treated animals.
Figure 2: Effects of the Aqueous and Ethanolic Extracts of the Zingiber Officinale Rhizomes and Dried Fruits of Tetrapleura Tetraptera Mixture on Spontaneous Locomotion. Data are Shown as Mean ± SEM, n = 6; αp<0.05; βp<0.01; yp<0.001 Significant Compared to Normal Control; ap<0.05; bp<0.01; cp<0.001 Significant Compared to NaCl (0,9%)
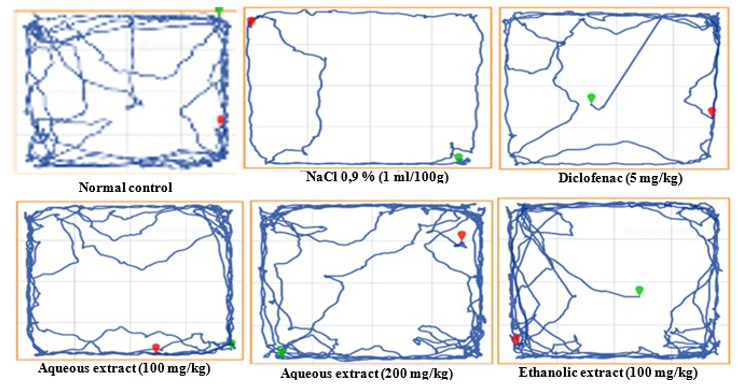
Figure 3: Effects of the Aqueous and Ethanolic Extracts of the Zingiber Oficinale Rhizomes and Dried Fruits of Tetrapleura Tetraptera Mixture on Spontaneous Locomotion.
Effects on Some Anxiety Parameters
Intraarticular injection of MIA caused an increase of stool, licking time and immobility time episode, with a significant decrease of the number of lines crossed and mobility time episode in non-treated animals as compared to the animals of the normal control group. The extracts administered, have reduced the stools, licking time, immobility time episode and increased the number of lines crossed and mobility time episode in osteoarthritis animals treated compared to animals of the negative control group (Figure 4)
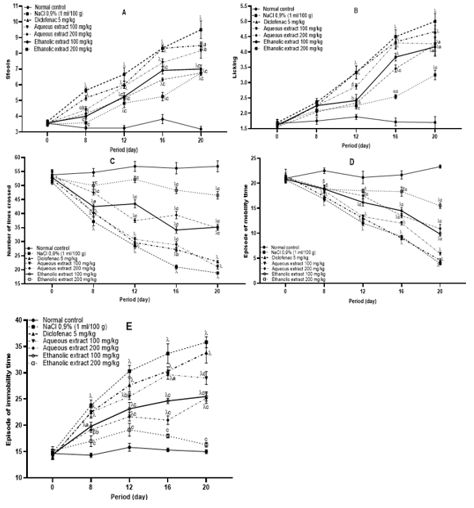
Figure 4: Effects of the Aqueous and Ethanolic Extracts of the Zingiber Officinale Rhizomes and Dried Fruits of Tetrapleura Tetraptera Mixture on Anxiety. Data are Shown as Mean ± SEM, n = 6; αp<0.05; βp<0.01; yp<0.001 Significant Compared to Normal Control; ap<0.05; bp<0.01; cp<0.001 Significant Compared to NaCl (0,9%).
Effects on Some Hematological Parameters and Serum Biochemical Parameters
The injection of MIA has disturbed hematological and serum biochemical parameters (Table). The various extracts administered orally restored the hematological disorder caused by the MIA. Extracts significantly reduced ALT, AST serum activities and serum creatinine level; it has also significantly increased the concentration of serum protein.
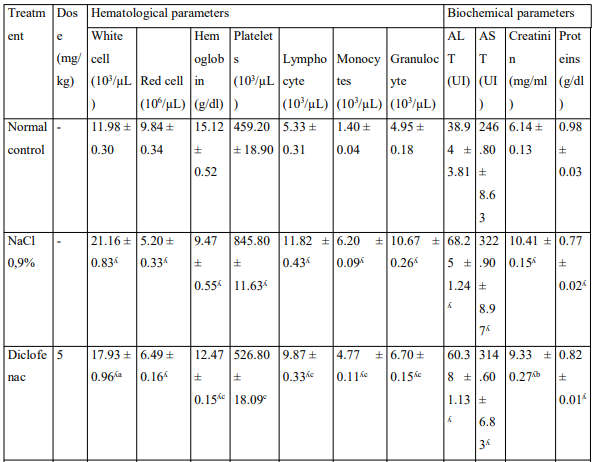
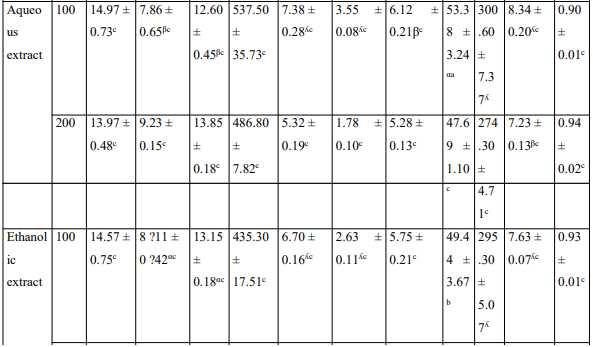
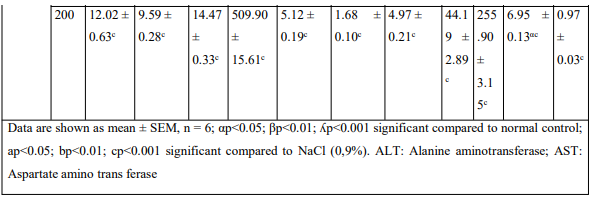
Table: Effects of the aqueous and ethanolic extracts of the Zingiber officinale rhizomes and dried fruits of Tetrapleura tetraptera mixture on hematological parameters and some biochemical parameters
Effects on Some Oxidative Parameters
It appears on Figure 5 that the injection of MIA caused a decrease SOD, CAT activity, GSH level and increase MDA level in animals of the negative control group as compared to animals of the normal control group. Aqueous and ethanolic extracts has showed a favorable effect by increasing SOD, CAT activities, GSH level and significantly reduced MDA level.
Figure 5: Effects of the Aqueous and Ethanolic Extracts of the Zingiber Officinale Rhizomes and Dried Fruits of Tetrapleura Tetraptera Mixture on Oxidative Parameters and NO levels. Data are Shown as Mean ± SEM, n = 6; αp<0.05; βp<0.01; yp<0.001 Significant Compared to Normal Control; ap<0.05; bp<0.01; cp<0.001 Significant Compared to NaCl (0,9%)
Effects on Inflammatory Parameters
Injection of MIA has led to a increase in the synovial concentrations of TNF-![]() , IL-1ß, MMP9 and MPO, and a decrease in concentrations of IL-4 and IL-10 in animals of the negative control group (figure 6). In the synovial liquid, extracts have reduced TNF-
, IL-1ß, MMP9 and MPO, and a decrease in concentrations of IL-4 and IL-10 in animals of the negative control group (figure 6). In the synovial liquid, extracts have reduced TNF-![]() IL-1ß, MMP9, MPO levels and increased IL-4, IL-10 levels. Furthermore, in animals treated with extracts, NO levels was significantly reduced
IL-1ß, MMP9, MPO levels and increased IL-4, IL-10 levels. Furthermore, in animals treated with extracts, NO levels was significantly reduced
Figure 6: Effects of the Aqueous and Ethanolic Extracts of the Zingiber Oficinale Rhizomes and Dried Fruits of Tetrapleura Tetraptera Mixture on Pro-Inflammatory (TNF-α, IL-1β, MMP9, MPO) and anti-inflammatory (IL-4, IL-10) parameters. Data are shown as mean ± SEM, n = 6; αp<0,05; βp<0,01; yp<0,001: Significant as Compared to Normal Control; ap<0.0, cp<0.001: significant as compared to NaCl 0,9%. TNF-α: Tumor Necrosing Factor Alpha, IL-1β: interleukine-1beta, IL-4: Interleukine 4, IL-10: Interleukine 10, MMP9: Matrix Metalloprotein 9, MPO: myeloperoxidase
Effects on the Microarchitecture of the Knee Joint
It emerges from Figure 7 those normal animals had a normal structure of the joint with a junction space, chondrocytes, epiphyses and very distinct articular cartilage. In negative control group, the presence of leukocyte infiltration granulomas and a synovial hyperplasia was noted. Aqueous and ethanolic extracts of the mixture of rhizomes and dry fruits have corrected these histopathological alterations.
Figure 7: Microphotography of the Knee Joint of the Rats After MIA-Induced Arthrosis (Hematoxyline & Eosine Coloration, X40) Ca: Cartilage; JE: Joint E Space; OT: Osteoclast; BM: Bone Marrow; SH: Synivial Hyperplasia; LI: Leucocyte Infiltration
Discussion
In the present study, the effects of aqueous and ethanolic extracts of the mixture of Z. oficinale rhizomes and dried fruits of T. tetraptera on hyperalgesia, inflammation, anxiety and locomotor dysfunction induced by the intraarticular injection of MIA in rat were evaluated. In the present study, the injection of MIA in animals caused hypersensitivity and joint inflammation from the 1st day of injection till the end of experimentation. The oral administration of the aqueous and ethanolic extracts of the mixture of rhizomes and dried fruits has significantly reduced hypersensitivity and inflammation in all animals treated. This effect was observed from the 2nd day to the end of treatments. It is known that the model of osteoarthritis induced by MIA is suitable to predict the analgesic and anti-inflammatory effects of the drugs which target synovial inflammation and the cartilage/subchondral bones [26].
One of the known complications of osteoarthritis pain is the reduction in mobility that may result in a significant reduction in body mass. This study indicated a significant decrease in body mass in non-treated osteoarthritis rats. During the development of osteoarthritis, hypersensitivity, chronic inflammation and oxidative stress that were developing are very important factors that can cause mitochondrial dysfunction, apoptosis, significant limitation of physical activities and locomotor disorders. The two extracts have improved body mass changes in osteoarthritis rats. These results showed that the extracts has improved the changes in body weight and muscle strength by lowering hypersensitivity, inflammations and oxidative status. The improvement of the locomotor activity is highlighted by a significant decrease in the number of lines crossed, total distance traveled, average speed, total mobility time and mobility time episode on non-treated osteoarthritis rats. The rats who received the extracts at different doses had traveled longer distances, were very mobile and very active as compared to non-treated rats. These results indicate that the association of rhizomes and dried fruits is capable of reducing hypersensitivity and joint inflammation, but can also to improve the locomotor function in osteoarthritis conditions.
It is known that hematological disorders, particularly anemia, anomalies of white blood cells, platelet anomalies, coagulopathy and hematological malignant tumors can develop in many autoimmune rheumatic diseases [10]. The results of this study have shown that non-treated animals had severe anemia (significant decrease in red blood cell level), a significant rise in blood platelets and white blood cells. Apart hematological disorders observed, osteoarthritis animals also showed alterations in liver and renal functions (increase in serum ALT, AST activity, creatinine level and significant reduction in protein concentration). The aqueous and ethanolic extracts of the mixture of rhizomes and dried fruits have corrected the hematological disorders caused by osteoarthritis and also restored the hepatic and renal functions. Biochemical analysis of the degenerative cartilage of patients with osteoarthritis has shown that there is a pathological relationship between oxidative stress and degeneration of cartilage in the progression of osteoarthritis [27]. The mechanical load promotes the production of free oxygen radical in the mitochondria of chondrocytes and a decrease in the expression activity of SOD in mitochondria, while the mitochondrial dysfunction induced by the superoxide in the mitochondria will still lead to degeneration of cartilage [28].
Furthermore, the NO in cartilage and synovial fluid is produced by iNOS, which mediates the expression of inflammatory factors, inhibits the synthesis of collagen and proteoglycan, and induces the apoptosis and pain of chondrocytes [29]. The production of NO in osteoarthritis cartilage is unusually high because osteoarthritis chondrocytes produce high levels of NO. NO regulates the expression of cytokines, resulting in oxidative damage and apoptosis of chondrocytes, thus promoting the pathogenesis of osteoarthritis [30]. In addition, the increase in NO leads to a lesion of cartilage by improving the activity of matrix metalloprotein (MMP) and by regulating the biosynthesis of proteoglycan and collagen [31]. By reacting with oxidants (superoxide anions), NO promotes cell lesions and makes chondrocytes vulnerable to apoptosis induced by cytokines [32]. It is known that under physiological conditions, a restless load for 48 hours can cause oxidation of glutathione in cartilage, resulting in a reduction in the protection of glutathione against additional oxidative stress [33]. The oral administration of the extracts showed an antioxidant activity by increasing SOD, CAT activities, GSH level, then reduce MDA and NO levels.
During the pathogenesis of osteoarthritis, inflammatory cytokines are very high thus regulating MMP-9, which amplifies the loss of articular cartilage [34]. In the human body, MMPs synthesized by chondrocytes and synoviocytes, release growth promoter protein factors, inactive proteinase inhibitors and influence cytokines and inflammatory chemokines [35]. The destruction of cartilage activate the chondrocytes to produce more MMP, reactive species of oxygen (ROS) responsible for the degradation of cartilage and the release of microcrystals and osteochondral fragments which stimulate the inflamed synovium cells (synoviocytes, macrophages, lymphocytes) to secrete cytokins, chemokines and lipid mediators, as well as more ROS and MMP, which can directly degrade the components of the cartilage matrix thus amplifying inflammation, creating a vicious cycle [36]. IL-1β and TNF-α secreted by fibroblasts, chondrocytes and immune cells are responsible for catabolism and metabolic imbalance by stimulating MMP synthesis in osteoarthritis joints [37]. The level of MMP-9 in the synovial liquid of patients with osteoarthritis is increased [38]. MPO is involved in the pathogenesis of several diseases generally associated with chronic or high inflammatory states [39]. IL-1β is capable of activating several signaling paths which when combined, lead to the progression of osteoarthritis [40].
Thanks to the signaling of the protein kinase activated by mitogen (MAPK), IL-1β induces catabolic events (degradation of cartilage) which lead to hypertrophy of chondrocytes, dedifferentiation and finally, apoptosis [41]. IL-1β can also activate NF-kB factor leading to the inhibition of type II collagen expression, increased production of MMP-9, TNF-α and NO [42]. TNF-α blocks the synthesis of type II collagen and also leads to a degradation of the extracellular matrix and considerably reduces the repair of the cartilage [43]. The extracts have reduced the MMP-9, MPO, IL-1β and TNF-α levels. Looking at these results, we can speculate that this mixture reduces hypersensitivity, inflammation and degradation of cartilage in osteoarthritis by inhibiting the production of MMP-9, TNF-α and IL-1β in synovial fluid and can therefore act as a natural anti-inflammatory agent in osteoarthritis. Furthermore, it is known that IL-4 has a notable chondroprotective effect, an inhibitor of the secretion of metalloproteinases, an inhibitor of the degradation of proteoglycans in articular cartilage, an inhibitor of the synthesis of inflammatory cytokines (IL-1β, TNF-α), and other inflammatory mediators (PGE-2, PLA2, COX-2, iNOS) [37,44,45]. IL-10 has chondroprotective, antiapoptotic, anti-inflammatory and inhibitory effects of MMP [46]. In combination, IL-4 and IL-10 protect against cartilage lesions and inhibit the apoptosis of chondrocytes [37,47]. Thus, a substance capable of stimulating the production of IL-4 and IL-10 would be beneficial in the control of osteoarthritis. The two extracts led to an increase in the production of IL-4 and IL-10 in all treated animals. These results are supported by the fact that extracts have corrected histopathological alterations in treated animals, which showed a normal structure of the joint with a junction space, chondrocytes, epiphyses, and well-defined articular cartilage.
Conclusion
In this study, the effectiveness of extracts from the mixture of Z. officinale rhizomes and the dry fruits of T. tetraptera on hypersensitivity, inflammation, anxiety and locomotor anomalies induced by MIA has been examined. The results obtained imply that these extracts have the capacity to reduce hypersensitivity, inflammation, anxiety and correct osteoarthritis damage due to their antioxidant and anti-inflammatory activities.
Authors Contribution Statement
Djami Bejoutance Marcelle, Mbiantcha Marius and Dzeufiet Djomeni Paul: involved in the study’s conceptualization and design, data collection and analytics supervision, and manuscript revision for essential intellectual content.
Djami Bejoutance Marcelle, Mbiantcha Marius, Dzeufiet Djomeni Paul, Bilanda Danielle Claude, Atsamo Albert Donatien, and Yousseu Nana William: involved in the conceptualization and design of the study, data gathering and analysis, and manuscript drafting.
Djami Bejoutance Marcelle, Mbiantcha Marius, Dzeufiet Djomeni Paul, Bomba Tatsinkou Francis Desire, Djuichou Nguemnang Stéphanie Flore and Tcheutchoua Yannick Carlos contributed to the data analysis and interpretation, as well as to the manuscript’s scientific quality.
Mbiantcha Marius, Bilanda Danielle Claude, Bakouatche Jovanie Destin, Kwenthe Ernest Allah Hoki, Kana Miariam Flore and Ateufack Gilbert helped with data collection and analysis as well as manuscript review. The final document for submission was read and approved by all authors.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data Availability
Data will be made available on request.
Acknowledgements
The authors would like to think the Laboratory of Animal Physiology, Department of Animal Biology and Physiology of the University of Yaoundé 1(Faculty of Science, Cameroon) and the Research Unit of Animal Physiology and Phytopharmacology, Department of Animal Biology of the University of Dschang (Faculty of Science, Cameroon) for assistance and helpful discussion.
References
- Chen, D. I., Shen, J., Zhao, W., Wang, T., Han, L., Hamilton, J. L., & Im, H. J. (2017). Osteoarthritis: toward a comprehensive understanding of pathological mechanism. Bone research, 5(1), 1-13.
- Lu, K., Ma, F., Yi, D., Yu, H., Tong, L., & Chen, D.(2022). Molecular signaling in temporomandibular joint osteoarthritis. Journal of orthopaedic translation, 32, 21-27.
- Straub, R. H., & Schradin, C. (2016). Chronic inflammatory systemic diseases: An evolutionary trade-off between acutely beneficial but chronically harmful programs. Evolution, medicine, and public health, 2016(1), 37-51.
- Griffin, T. M., & Scanzello, C. R. (2019). Innate inflammation and synovial macrophages in osteoarthritis pathophysiology. Clinical and experimental rheumatology, 37(Suppl 120), 57.
- Khan, N. M., Haseeb, A., Ansari, M. Y., Devarapalli, P., Haynie, S., & Haqqi, T. M. (2017). Wogonin, a plant derived small molecule, exerts potent anti-inflammatory and chondroprotective effects through the activation of ROS/ ERK/Nrf2 signaling pathways in human Osteoarthritis chondrocytes. Free Radical Biology and Medicine, 106, 288-301.
- Jiang, Y., Xiao, Q., Hu, Z., Pu, B., Shu, J., Yang, Q., ... & Hao,J. (2014). Tissue levels of leukemia inhibitory factor vary by osteoarthritis grade. Orthopedics, 37(5), e460-e464.
- Nemirovskiy, O. V., Radabaugh, M. R., Aggarwal, P., Funckes-Shippy, C. L., Mnich, S. J., Meyer, D. M., ... & Misko, T.P. (2009). Plasma 3-nitrotyrosine is a biomarker in animal models of arthritis: Pharmacological dissection of iNOS’role in disease. Nitric Oxide, 20(3), 150-156.
- Altindag, O., Erel, O., Aksoy, N., Selek, S., Celik, H., & Karaoglanoglu, M. (2007). Increased oxidative stress and its relation with collagen metabolism in knee osteoarthritis. Rheumatology international, 27(4), 339-344.
- Rigoglou, S., & Papavassiliou, A. G. (2013). The NF-κB signalling pathway in osteoarthritis. The international journal of biochemistry & cell biology, 45(11), 2580-2584.
- Yukawa, N., & Mimori, T. (2013). Hematologic, biochemical, and immunological tests in clinical practice of rheumatoid arthritis. Nihon rinsho. Japanese journal of clinical medicine, 71(7), 1178-1182.
- Carou, A. C., Fernández, S. P., Díaz, S. P., de Toro Santos,F. J., & group EVALÚA, S. (2015). Clinical profile, level of affection and therapeutic management of patients with osteoarthritis in primary care: the Spanish multicenter study EVALÚA. Reumatología Clínica (English Edition), 11(6), 353-360.
- Axford, J., Butt, A., Heron, C., Hammond, J., Morgan, J., Alavi, A., ... & Bland, M. (2010). Prevalence of anxiety and depression in osteoarthritis: use of the Hospital Anxiety and Depression Scale as a screening tool. Clinical rheumatology, 29(11), 1277-1283.
- Leung, T. F., Liu, A. P. Y., Lim, F. S., Thollot, F., Oh, H.M. L., Lee, B. W., ... & Struyf, F. (2015). Comparative immunogenicity and safety of human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine and HPV-6/11/16/18 vaccine administered according to 2-and 3-dose schedules in girls aged 9–14 years: Results to month 12 from a randomized trial. Human vaccines & immunotherapeutics, 11(7), 1689-1702.
- World Health Organization. (2000). Promotion du role de la m ˆ edecine traditionnelle dans ´ le systeme de sante. ´ Harare, 50, 4-5.
- Essien, C. A. (2021). Effects of Tetrapleura tetraptera on performance, carcass characteristics and organ indices of broiler chickens. Animal Research International, 18(2), 4134-4144.
- Nwaichi, E. O., & Anyanwu, P. (2013). Effect of heat treatment on the antioxidant properties of Tetrapleura tetraptera, Xylopia aethiopica and Piper guineense. International Journal of Biotechnology and Food Science, 1(1), 1-5.
- Bouba, A. A. (2009). Contribution à l'étude du développement d'un aliment fonctionnel à base d'épices du Cameroun: caractérisation physico-chimique et fonctionnelle (Doctoral dissertation, Institut National Polytechnique de Lorraine).
- Dieumou, F. E., Teguia, A., Kuiate, J. R., Tamokou, J. D., Fonge, N. B., & Dongmo, M. C. (2009). Effects of ginger (Zingiber officinale) and garlic (Allium sativum) essential oils on growth performance and gut microbial population of broiler chickens. Livestock research for rural development, 21(8), 23-32.
- Aladesanmi, A. J. (2007). Tetrapleura tetraptera: Molluscicidal activity and chemical constituents. African Journal of Traditional, Complementary and Alternative Medicines, 4(1), 23-36.
- Priya Rani, M., Padmakumari, K. P., Sankarikutty, B., Lijo Cherian, O., Nisha, V. M., & Raghu, K. G. (2011). Inhibitory potential of ginger extracts against enzymes linked to type 2 diabetes, inflammation and induced oxidative stress. International journal of food sciences and nutrition, 62(2), 106-110.
- Effiong, G. S., Udoh, I. E., Essien, G. E., Ajibola, D. O., & Archibong, K. O. (2014). Effect of aqueous extract of Tetrapleura tetraptera on excision wounds in albino rats. International Research Journal of Plant Science; 5 (4): 57, 60.
- Sharanya, A., Jayachandran, N. C. V., & Prashobh, G. R. (2023). A Review on Pharmacological Activities of Tetrapleura Tetraptera. Int J Res Publ Rev, 4(9),1181-1184.
- Incharoen, T., & Yamauchi, K. (2009). Production performance, egg quality and intestinal histology in laying hens fed dietary dried fermented ginger. International Journal of Poultry Science, 8(11), 1078-1085.
- Norton, S., Culver, B., & Mullenix, P. (1975). Development of nocturnal behavior in albino rats. Behavioral Biology, 15(3), 317-331.
- Zhou, X., Wang, S., Ding, X., Qin, L., Mao, Y., Chen, L., ... & Ying, C. (2017). Zeaxanthin improves diabetes-induced cognitive deficit in rats through activiting PI3K/AKT signaling pathway. Brain research bulletin, 132, 190-198.
- Sasaki, Y., Kijima, K., & Yoshioka, K. (2024). Validity evaluation of a rat model of monoiodoacetate-induced osteoarthritis with clinically effective drugs. BMC Musculoskeletal Disorders, 25(1), 975.
- Scott, J. L., Gabrielides, C., Davidson, R. K., Swingler, T.E., Clark, I. M., Wallis, G. A., ... & Young, D. A. (2010).Superoxide dismutase downregulation in osteoarthritis progression and end-stage disease. Annals of the rheumatic diseases, 69(8), 1502-1510.
- Koike, M., Nojiri, H., Ozawa, Y., Watanabe, K., Muramatsu, Y., Kaneko, H., ... & Shimizu, T. (2015). Mechanical overloading causes mitochondrial superoxide and SOD2 imbalance in chondrocytes resulting in cartilage degeneration. Scientific reports, 5(1), 11722.
- Suantawee, T., Tantavisut, S., Adisakwattana, S., Tanpowpong, T., Tanavalee, A., Yuktanandana, P., ... & Honsawek, S. (2015). Upregulation of inducible nitric oxide synthase and nitrotyrosine expression in primary knee osteoarthritis. J Med Assoc Thai, 98(1), S91-97.
- Scher, J. U., Pillinger, M. H., & Abramson, S. B. (2007). Nitric oxide synthases and osteoarthritis. Current rheumatology reports, 9(1), 9-15.
- Lepetsos, P., & Papavassiliou, A. G. (2016). ROS/oxidative stress signaling in osteoarthritis. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1862(4), 576-591.
- Amin, A. R., Dave, M., Attur, M., & Abramson, S. B. (2000). COX-2, NO, and cartilage damage and repair. Current rheumatology reports, 2(6), 447-453.
- Zhu, S., Makosa, D., Miller, B., & Griffin, T. M. (2020). Glutathione as a mediator of cartilage oxidative stress resistance and resilience during aging and osteoarthritis. Connective tissue research, 61(1), 34-47.
- Piao, S., Du, W., Wei, Y., Yang, Y., Feng, X., & Bai, L. (2020).Protectin DX attenuates IL-1β-induced inflammation via the AMPK/NF-κB pathway in chondrocytes and ameliorates osteoarthritis progression in a rat model. International immunopharmacology, 78, 106043.
- Nagase, H., Visse, R., & Murphy, G. (2006). Structure and function of matrix metalloproteinases and TIMPs. Cardiovascular research, 69(3), 562-573.
- Henrotin, Y., Pesesse, L., & Lambert, C. (2014). Targeting the synovial angiogenesis as a novel treatment approach to osteoarthritis. Therapeutic advances in musculoskeletal disease, 6(1), 20-34.
- Wojdasiewicz, P., Poniatowski, L. A., & Szukiewicz, D. (2014). The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediators of inflammation, 2014(1), 561459.
- Tchetverikov, I., Ronday, H. K., Van El, B., Kiers, G. H., Verzijl, N., TeKoppele, J. M., ... & Hanemaaijer, R. (2004). MMP profile in paired serum and synovial fluid samples of patients with rheumatoid arthritis. Annals of the rheumaticdiseases, 63(7), 881-883.
- Frangie, C., & Daher, J. (2022). Role of myeloperoxidase in inflammation and atherosclerosis. Biomedical reports, 16(6), 53.
- Molnar,V., Matišic,V., Kodvanj,I.,Bjelica,R.,Jelec,Z.,Hudetz, D., ... & Primorac, D. (2021). Cytokines and chemokines involved in osteoarthritis pathogenesis. International journal of molecular sciences, 22(17), 9208.
- Hwang, H. S., & Kim, H. A. (2015). Chondrocyte apoptosis in the pathogenesis of osteoarthritis. International journal of molecular sciences, 16(11), 26035-26054.
- Lepetsos, P., Papavassiliou, K. A., & Papavassiliou, A. G. (2019). Redox and NF-κB signaling in osteoarthritis. Free Radical Biology and Medicine, 132, 90-100.
- Xue, J., Wang, J., Liu, Q., & Luo, A. (2013). Tumor necrosis factor-α induces ADAMTS-4 expression in human osteoarthritis chondrocytes. Molecular medicine reports, 8(6), 1755-1760.
- Doi, H., Nishida, K., Yorimitsu, M., Komiyama, T., Kadota, Y., Tetsunaga, T., ... & Ozaki, T. (2008). Interleukin-4 downregulates the cyclic tensile stress-induced matrix metalloproteinases-13 and cathepsin B expression by rat normal chondrocytes. Acta Medica Okayama, 62(2), 119.
- Schuerwegh, A. J., Dombrecht, E. J., Stevens, W. J., Van Offel, J. F., Bridts, C. H., & De Clerck, L. S. (2003). Influence of pro-inflammatory (IL-1α, IL-6, TNF-α, IFN-γ) and anti-inflammatory (IL-4) cytokines on chondrocyte function. Osteoarthritis and cartilage, 11(9), 681-687.
- Schulze-Tanzil, G., Zreiqat, H., Sabat, R., Kohl, B., Halder, A., Muller, R. D., & John, T. (2009). Interleukin-10 and articular cartilage: experimental therapeutical approaches in cartilage disorders. Current gene therapy, 9(4), 306-315.
- van Meegeren, M. E., Roosendaal, G., Jansen, N. W., Wenting,M. J., van Wesel, A. C., van Roon, J. A., & Lafeber, F. P. (2012). IL-4 alone and in combination with IL-10 protects against blood-induced cartilage damage. Osteoarthritis and Cartilage, 20(7), 764-772.




