Research Article - (2022) Volume 7, Issue 3
Adsorption Isotherms and Thermodynamic Properties of Dried Tomato Slices
2Department of Nutrition and Dietetics, Federal University of Agriculture, Abeokuta, Nigeria
3Department of Hospitality and Tourism, Federal University of Agriculture, Abeokuta, Nigeria
4Department of Bio-resources Engineering, Federal University of Agriculture, Abeokuta, Nigeria
5Department of Agricultural Extension and Rural Development, Federal University of Agriculture, Abeok, Nigeria
6Postharvest Loss Alliance for Nutrition (PLAN), Global Alliance for Improved Nutrition, Abuja, Nigeria
Received Date: Jun 23, 2022 / Accepted Date: Jun 30, 2022 / Published Date: Jul 12, 2022
Copyright: ©Abdul-Rasaq Adebowale, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Adebowale AA, Akinniyi G, Shittu TA, Adegoke AF, Omohimi CI, Sobukola OP, Onabanjo OO, Adegunwa MO, Kajihausa OE, Dairo OU, Abdulsalam-Saghir P, Sanni LO, Siwoku BO, Okoruwa AE. (2022). Adsorption Isotherms and Thermodynamic Properties of Dried Tomato Slices. AdvNutr Food Sci. 7(3): 249-262.
Abstract
Moisture adsorption isotherms and thermodynamic properties of dried tomato slices obtained from four tomato varieties were investigated. Freshly harvested tomatoes were pretreated with potassium metabisulphite solution (0.2%, weight/volume) before drying in a cabinet dryer at 60± 2° C. Moisture adsorption behavior of the dried tomato slices was determined using the static gravimetric method at 25, 30 and 40o C. The data fitted to GAB, M.BET, DLP, Smith and Oswin sorption models. The isosteric heat of sorption was also determined. Moisture adsorption isotherm of dried tomato slices from UC82B, Roma, Eva-F1 and Kerewa varieties exhibited a sigmoid isotherm curve typical of type II BET classification. Increase in temperature resulted into a decrease in the equilibrium moisture content. The GAB, DLP and modified BET model adequately modeled the isotherms of dried tomato slices. The relationship between isosteric heat of sorption and the equilibrium moisture of dried tomato slices were accurately described by exponential model.
Keywords
Sorption Isotherms, Equilibrium Moisture Content, Sorption Models, Isosteric Heat of Sorption, Tomato
Introduction
Tomato (Solanum lycopersicum L.) is one of the highly cultivat-ed and substantially consumed vegetables in different parts of the world [1]. The most common color of tomato is red but, they also come in yellow, purple, green and black colors. Tomatoes possess various sizes and color and there are about 75,000 varieties world¬wide [2]. The cultivars that are predominantly cultivated and mar¬keted in Nigeria include: Ibadan local, Roma-VF and Ife-1 [3,4]. Nigeria is the second largest producer of tomato in Africa and ranked 14th in the world, with an annual production of about 1.6- 1.8 million tonnes at an average yield of 5.77 tonnes per hectare [5,6]. Thus, the country has comparative advantage and capacity to be the highest producer and exporter of tomato in Africa [7]. Unfortunately, Nigeria tomato sub-sector/value chain is battling with challenges such as critical inputs deficiency, lack of improved agronomic practices, low yield and productivity, high postharvest losses and lack of processing technology/method and inadequate market or transport infrastructure [8,9]. Hence, the consumers demand for tomato and its derivatives in Nigeria far exceeds the supply.
Tomato and tomato-based products supply a large array of nutri¬ents as well as many health-related benefits to humans. Tomatoes and tomato derived products are rich in nutrients and phytochem-icals such as carotenoids (predominantly, lycopene), flavonoids, ascorbic acid, potassium and vitamin E [10]. It contains all four major carotenoids: α- and β- carotene, lutein and lycopene which may have individual benefits, but also have synergistic effect as a group [11,12]. Tomato constituents such as lycopene, phenolics, ascorbic acid and flavonoids are important antioxidants in the diet [13]. The major pigment responsible for the red color of tomatoes - lycopene, contributes 87% of total carotenoids present in ripe tomatoes [14]. The antioxidant role of lycopene is associated with immune regulatory abilities, DNA damage reduction, lowering of biological oxidative damage of proteins, lipids and other cell com¬ponents as well as transformation of malignant tissues [15].
Despite the aforementioned properties of tomato, which justifies its nutritional and economic importance, tomato, especially in ripe form, is not suitable for long-term storage after harvest because it is extremely liable to rot. Thus, enormous losses and wastage takes place during and postharvest phase [16]. This situation leads to significant losses both nutritionally and economically. Therefore, a key intervention aimed at preventing losses and wastage of to-mato is of utmost priority in Nigeria and other developing tropical countries where there is marked difference between supply and demand at harvest and during off-season period [17]. Ugonna et al. reported that Nigeria has a large market for fresh and processed tomato products due to its large population of over 170 million and an estimated annual national population growth rate of 5.7% per annum coupled with an average economic growth rate of 3.5% per annum [18]. The demand for various processed products from tomato has been increasing progressively in the retail and food ingredients or additives markets and industries [18]. This under¬scores the need to develop appropriate techniques/technology for value addition to tomato to reduce postharvest losses, obtain value added products and in turn generate more revenue for the country while also serving as a source of nutrients and livelihood to farmer and processor. This will ensure the availability of tomato all year round and helps to lessen the need to till the land continuously, and thus reduces the adverse environmental effect.
Amid the commonly employed processing methods for tomato processing and preservation, drying remains one of the most suit-able because the final dried product moisture content is consider-ably reduced thereby preventing microbial deterioration [19]. In addition, the weight and volume of the final product reduced after dehydration, which may be responsible for vital savings in the cost of handling, transport and storage [20]. Dehydration processes present an alternative technique of delivering tomato to commerce and consumers. Hot air drying is the most popular form of tomato drying because it is simple to operate and inexpensive [17]. Water activity (aw) is an important factor that influence the biochemical reactions during storage and shelf stability of dried products [21]. Moisture sorption isotherms are practical thermodynamic param¬eters for estimating interactions of water and food substances, and elaborate information to evaluate processing operations, such as drying, packaging, mixing, and storage [22]. It can as well be em¬ployed to explore structural characteristics of a food product, such as pore volume, specific surface area and pore size distribution as well as crystallinity. Sorption isotherms data can be used for choosing appropriate storage conditions, and packaging methods and materials that optimize or maximize retention of aroma, color, texture, nutrients and biological stability [23-26].
This study investigated the sorption isotherms of dried tomato slices with a view to providing information suitable for solving the critical problem of post-harvest losses by transforming tomato fruits into dried slices, predict the hygroscopic properties of de¬hydrated tomato slices and also find an acceptable, available and affordable package for the slices to enhance its use and storage domestically.
Materials and Methods
Four tomato varieties; UC82B, Roma and Kerewa varieties were purchased from Olomore market, Abeokuta, Ogun State, Nigeria while Eva-F1, (the hybrid variety) was obtained from a greenhouse in Abeokuta, Ogun State, Nigeria. Packaging materials used (glass containers, high-density polyethylene bags, polypropylene con¬tainers and low-density polyethylene bags) were procured from Lafenwa market, Abeokuta, Ogun State, Nigeria.
Processing of Dried Tomato Slices
Wholesome tomatoes sorted from the bulk in terms of firmness, degree of ripeness (color) and freedom from defects. The tomatoes then cleaned rigorously by washing under running potable water. The washed tomatoes then sliced using a sharp stainless knife to 10 mm thickness and the slices were sulphited according to the Akanbi et al., method with few modifications by dipping it in 0.2% potassium metabisulfite solution for 10 min [17]. The slices were thereafter drained and blotted before the drying process. The to¬mato slices were arranged on several perforated rows in the cabi¬net dryer (APV & Pasilac, England) and dried at a temperature of 60° C to a final moisture content of below 10%. The dried tomato slices were cooled, wrapped with aluminum foil and packaged in zip-lock polythene bags and again in airtight plastic containers at 4° C prior to further analyses.
Determination of Adsorption Isotherms of Dried Tomato Slices
Adsorption isotherms of dried tomato slices were determined using the static gravimetric method of saturated salt solutions at different temperatures (25, 30 and 40o C) to depict the ambient temperature within different geopolitical zones of the country. Experiments was performed using 2-4 g of the dried tomato slices for all tem¬peratures in the water activity range from 0.11 to 0.96 as earlier described by Adebowale et al. [21]. The saturated salt solution was prepared by dissolving in a jar, an appropriate quantity of salt in distilled water. The jar was then allowed to stabilize in incubators at their respective pre-set temperatures. Duplicate samples were there after placed inside hermetically sealed jars, which were then arranged in temperature-controlled incubators. The samples were weighed at 2-day interval until equilibrium was attained. Equilibri¬um was assumed to have been attained when the change in weight did not exceed 0.001 g for three consecutive readings.
Mathematical Modeling of Adsorption Data of Dried To-mato Slices
The experimental sorption isotherms data matched into different models presented in Table 1.

Determination of Thermodynamic Properties
The isosteric heat of sorption which is a differential molar quantity derived from the temperature dependence of the sorption isotherm at a constant amount of adsorbed water moles (nm) is shown in equation 1 [25].

Figure 1: Adsorption Isotherms of Dried Tomato Slices from UC82B, Roma, Eva-F1 and Kerewa variety at 25, 32 and 45° C
By plotting ln(aw) vs 1/T for different constant moisture contents,the Qst nand S was determined from the slope and y-intercept, respectively [25].
Results and Discussions
Equilibrium Moisture Content of Dried Tomato Slices at Dif¬ferent Storage Temperatures and Water Activity
The equilibrium moisture content (EMC) of dried tomato slices from UC82B, Roma, Eva-F1 and Kerewa tomato varieties at 25° C, 35° C and 45° C at various water activities (0.11 to 0.97) are presented in Table 2. The hygroscopic equilibrium of dried tomato slices was attained at day 25. The equilibrium moisture content (EMC) values of the dried tomato slices ranged from 0.0272 to 0.1902, 0.0263 to 0.1647, 0.0259 to 0.1698 and 0.0269 to 0.1726 kg/kg at 25° C (aw = 0.11-0.97) for UC82B, Roma, Eva-f1 and Kerewa variety, respectively. EMC for UC82B, Roma, Eva-f1 and Kerewa at 32° C ranged from 0.0227 to 0.1511, 0.0197 to 0.1455, 0.0228 to 0.1493 and 0.0208 to 0.1344 kg/kg, respectively. While at 45° C, it ranged from 0.0195 to 0.0820, 0.0189 to 0.0996, 0.0204 to 0.0994 and 0.0186 to 0.0987 kg/kg for Uc82, Roma, Eva-f1 and Kerewa variety, respectively. The equilibrium moisture content of the dried tomato slices decreased with increasing temperature irre¬spective of variety.
Table 2: Equilibrium Moisture Content (kg water/kg d.b) Values of Dried Tomato Slices from Different Tomato Varieties underSelected Temperatures and Water Activity
|
Temp (°C ) |
Water activity (aw) |
Equilibrium moisture content (kg water/kg d.b) |
|||
|
UC82B |
Roma |
Eva-f1 |
Kerewa |
||
|
25 |
0.113 |
0.0272 |
0.0263 |
0.0259 |
0.0269 |
|
|
0.225 |
0.0251 |
0.0245 |
0.0306 |
0.0271 |
|
|
0.328 |
0.0270 |
0.0263 |
0.0322 |
0.0289 |
|
|
0.432 |
0.0362 |
0.0261 |
0.0354 |
0.0331 |
|
|
0.529 |
0.0328 |
0.0307 |
0.0408 |
0.0358 |
|
|
0.649 |
0.0393 |
0.0354 |
0.0481 |
0.0437 |
|
|
0.753 |
0.0574 |
0.0553 |
0.0714 |
0.0663 |
|
|
0.81 |
0.0868 |
0.0611 |
0.0838 |
0.0731 |
|
|
0.851 |
0.0958 |
0.0958 |
0.1157 |
0.0896 |
|
|
0.973 |
0.1902 |
0.1647 |
0.1698 |
0.1726 |
|
32 |
0.113 |
0.0227 |
0.0197 |
0.0228 |
0.0200 |
|
|
0.218 |
0.0236 |
0.0202 |
0.0241 |
0.0214 |
|
|
0.322 |
0.0247 |
0.0226 |
0.0255 |
0.0251 |
|
|
0.432 |
0.0313 |
0.0253 |
0.0297 |
0.0278 |
|
|
0.507 |
0.0310 |
0.0285 |
0.0392 |
0.0302 |
|
|
0.603 |
0.0366 |
0.0338 |
0.0382 |
0.0341 |
|
|
0.752 |
0.0559 |
0.0497 |
0.0648 |
0.0520 |
|
|
0.805 |
0.0776 |
0.0566 |
0.0705 |
0.0599 |
|
|
0.825 |
0.0897 |
0.0821 |
0.0973 |
0.0840 |
|
|
0.969 |
0.1511 |
0.1455 |
0.1493 |
0.1344 |
|
45 |
0.112 |
0.0195 |
0.0189 |
0.0204 |
0.0186 |
|
|
0.208 |
0.0198 |
0.0183 |
0.0209 |
0.0196 |
|
|
0.316 |
0.0202 |
0.0195 |
0.0220 |
0.0210 |
|
|
0.432 |
0.0246 |
0.0231 |
0.0233 |
0.0240 |
|
|
0.469 |
0.0271 |
0.0261 |
0.0350 |
0.0262 |
|
|
0.524 |
0.0304 |
0.0316 |
0.0365 |
0.0317 |
|
|
0.749 |
0.0494 |
0.0475 |
0.0573 |
0.0473 |
|
|
0.779 |
0.0563 |
0.0534 |
0.0642 |
0.0550 |
|
|
0.797 |
0.0819 |
0.0630 |
0.0842 |
0.0770 |
|
|
0.962 |
0.0820 |
0.0996 |
0.0994 |
0.0987 |
The observed increase of equilibrium moisture content with re-duced temperature could be due to fact that the kinetic energy connected with water molecules present in tomato increased with increase in temperature resulting in decreased attractive forces and release of water molecules [27]. In addition, at constant tempera-ture, the equilibrium moisture content (EMC) increased with in-creasing relative humidity. Previous authors have reported similar trends for plants food commodities [28-36]. Shatadal and Jayas have also reported similar findings for low sugar containing food commodities [37]. The increase of EMC with humidity can be fur-ther explained according to Ronald et al. who postulated change in excess enthalpy of water binding, dissociation of water, or increase in solubility of solute in water as temperature increases [38]. This implies that at any relative humidity, dried tomato slices become less hygroscopic with an increase in temperature. Consequently, in an atmosphere of constant relative humidity, it is expected to ad-sorb more moisture at lower temperatures than it would at a higher temperature. Temperature affects the mobility of water molecules and the dynamic equilibrium between the vapour and adsorbed phases [39]. According to Al-Muhtaseb, at higher temperatures, the molecules of water are activated at energy levels that allow them to be detached from their sorption sites, thereby decreasing the equilibrium moisture content [39]. The increase in equilibrium moisture content (adsorbed moisture) as water activity increased at constant temperature implies that at lower water activities of the environment, fewer water was accessible for surface assimilation by the dried tomato slices.
Moisture Adsorption Isotherm Behavior of Dried Toma- to Slices from Different Varieties
The sorption isotherm curves in Figure 1 were obtained from the plot of equilibrium moisture content values in kg water/kg dry sol-ids versus the water activity (aw) at the test temperatures (25o C, 32o C and 45o C) [40]. The moisture isotherm curve describes the water ingression capacity of the dried tomato slices under different temperature and relative humidity. The adsorption isotherm curves of different varieties of dried tomato slices for the three tempera¬tures was sigmoidal, characteristic of many dried biological mate¬rials. The adsorption isotherms possessed sigmoidal-shaped char¬acteristics in accordance with BET classification indicating that equilibrium moisture increased swiftly as water activity increases. The experimental adsorption isotherms showed the temperature dependencies of isotherms for all the four varieties of dried tomato slices.
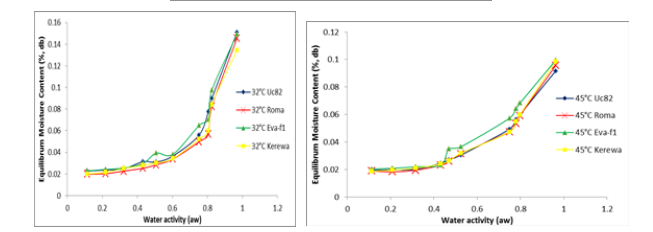
Figure 1: Adsorption Isotherms of Dried Tomato Slices from UC82B, Roma, Eva-F1 and Kerewa variety at 25, 32 and 45° C
All of the isotherm curves of the dried tomato slices were ob-served to follow a pattern of type II sigmoid shape, as described by Brunauer et al classification of isotherm curves [41]. The isotherms are characteristics of plant food commodities and pattern of amor¬phous materials that is rich in hydrophilic constituents [39,42-47]. An increase in temperature at constant moisture content lowers the isotherm curves, reduce water activity and lower the vulnerability of the product to microbial deterioration [24,25,47]. According to Giovanelli et al, this product requires higher water removal during drying to reduce water activity and is more sensitive to moisture variations during storage [48-50].
The relationship between water activity and moisture adsorption of dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa varieties is shown in Figures 2, 3 and 4, respectively. The adsorbed water corresponding to water activity below 0.75 was small for a relatively large increase in water activity. This shows that the critical equilibrium relative humidity (ERH) for dried tomato slic¬es from UC82B, Roma, Eva-f1 and Kerewa varieties lies above 60% for the temperature range of 25 to 45° C. Beyond this level, there will be a huge rise in the moisture adsorbed with relatively low increase in water activity which depicts the region of rapid spoilage [21-23]. This finding is in agreement with the report of Loong et al for dried foods [50]. From Table 2, at higher water activity (> 0.75 aw) and temperature of 25° C, dried tomato slices of UC82B variety adsorbed more water than that of Kerewa, Roma and Eva-f1 variety. Subsequent increase in the temperature to 32° C and 45° C, the adsorbed moisture reduced across all varieties but at different rates.
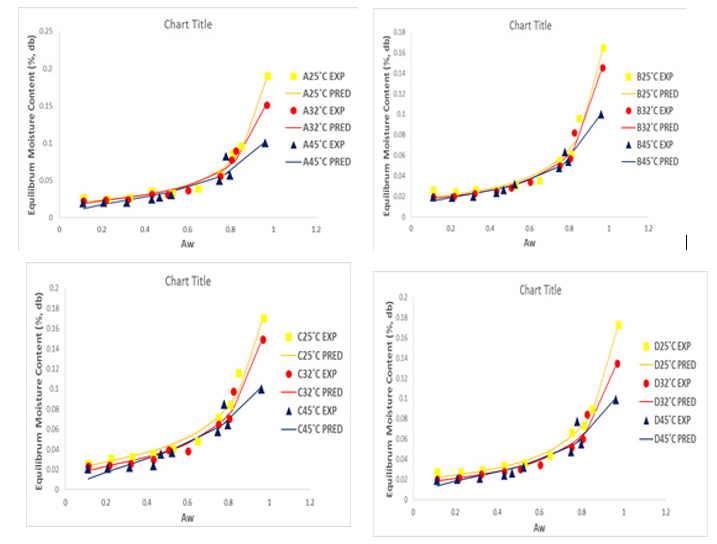
Figure 2: Adsorption Isotherms of Experimental and Predicted Values (GAB model) of Dried Tomato Slices from UC82B, Roma, Eva-F1 and Kerewa varieties at 25, 32 and 45° C.
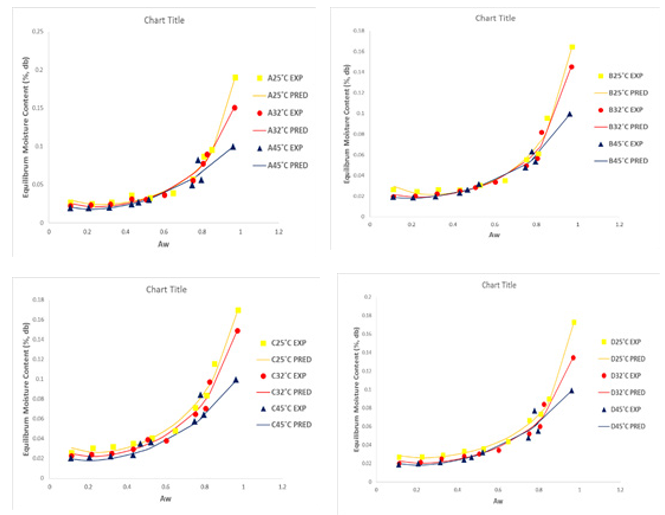
Figure 3: Adsorption Isotherms of Experimental and Predicted Values (DLP model) of Dried Tomato Slices from UC82B, Roma, Eva-F1 and Kerewa varieties at 25, 32 and 45° C.
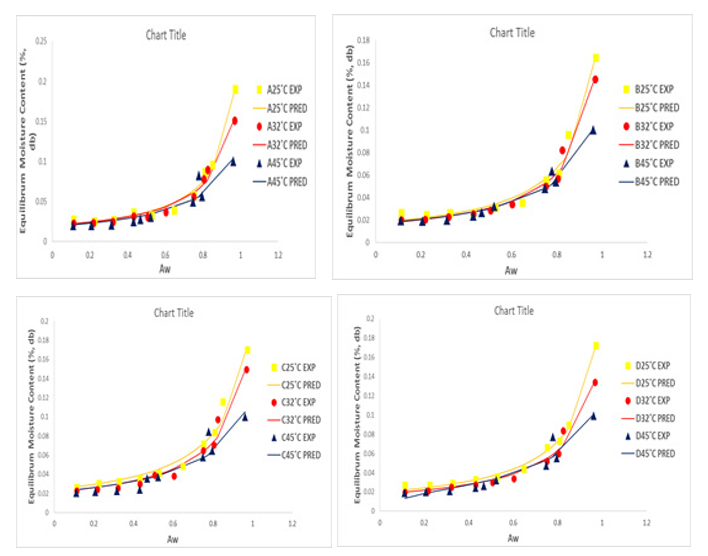
Figure 4: Adsorption Isotherms of Experimental and Predicted Values (modified BET model) of Dried Tomato Slices from UC82B,Roma, Eva-F1 and Kerewa varieties at 25, 32 and 45° C
The difference in sorption capacity between the different variet-ies of dried tomato slices at higher water activity (≥0.75 aw) may be due to variation in the structural cohesion of the dried tomato slices as well as their total soluble solids content. The analogy of water adsorption behavior at lower water activity established by the estimated values of the GAB monolayer moisture content in Table 3A which indicated that dried tomato slices from Roma vari-ety was the most hygroscopic and dried tomato slices from Kerewa variety was the least hygroscopic.
The experimental adsorption data for the dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa variety at 25º C, 32º C and 45º C were plotted with predicted results from Guggenheim–An-derson–de Boer (GAB), Double Log Polynomial (DLP), modified BET model equation as presented in Figures 2, 3 and 4, respective¬ly in line with the fitting criteria of lower percentage error (E%) and higher coefficient of determination (r2) to determine sorption parameters and plots. The GAB, DLP, modified BET model result¬ed into a Type II curve. It is evident from Figures 2, 3 and 4 that the isotherm curves are typically sigmoidal in shape.
Table 3A: Estimates of the Parameters of GAB, DLP and Modified BET Model Equations for the Moisture Adsorption of Dried Tomato Slices from Uc82 (A), Roma (B), Eva-f1 (C) and Kerewa (D) variety at 25, 32 and 45ÃÂ?? C
|
|
|
25°C |
32°C |
45°C |
|||||||||
|
A |
B |
C |
D |
A |
B |
C |
D |
A |
B |
C |
D |
||
|
GAB |
M |
0.229 |
0.231 |
0.226 |
0.220 |
0.198 |
0.219 |
0.190 |
0.184 |
0.145 |
0.153 |
0.138 |
0.130 |
|
|
C |
16.50 |
17.98 |
16.07 |
15.99 |
8.49 |
8.60 |
8.43 |
8.27 |
9.078 |
34.272 |
4.793 |
13.08 |
|
|
K |
0.834 |
0.860 |
0.815 |
0.809 |
0.849 |
0.875 |
0.824 |
0.813 |
20.89 |
22.11 |
20.00 |
19.90 |
|
|
r2 |
0.987 |
0.976 |
0.977 |
0.996 |
0.989 |
0.98 |
0.972 |
0.975 |
0.991 |
0.975 |
0.999 |
0.992 |
|
|
RSS |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0.001 |
0 |
0.001 |
0.001 |
|
|
E% |
6.284 |
4.728 |
4.541 |
3.6 |
3.571 |
4.207 |
3.57 |
3.473 |
7.167 |
6.534 |
2.641 |
2.907 |
|
|
RMSE |
0.013 |
0.11 |
0.10 |
0.049 |
0.099 |
0.089 |
0.094 |
0.099 |
0.113 |
0.06 |
0.013 |
0.107 |
|
M.BET |
A |
0.020 |
0.018 |
0.024 |
0.020 |
0.020 |
0.017 |
0.021 |
0.018 |
0.019 |
0.017 |
0.022 |
0.018 |
|
|
B |
0.922 |
0.919 |
0.885 |
0.909 |
0.898 |
0.916 |
0.887 |
0.896 |
0.848 |
0.865 |
0.824 |
0.851 |
|
|
r2 |
0.987 |
0.976 |
0.977 |
0.996 |
0.979 |
0.880 |
0.970 |
0.975 |
0.890 |
0.973 |
0.893 |
0.912 |
|
|
RSS |
0.024 |
0.018 |
0.019 |
0.018 |
0.015 |
0.014 |
0.015 |
0.012 |
0.007 |
0.006 |
0.007 |
0.007 |
|
DLP |
b0 |
0.003 |
0.003 |
0.004 |
0.002 |
0.003 |
0.002 |
0.003 |
0.002 |
0.004 |
0.002 |
0.004 |
0.003 |
|
|
b1 |
0.019 |
0.019 |
0.021 |
0.014 |
0.019 |
0.014 |
0.017 |
0.014 |
0.016 |
0.011 |
0.015 |
0.013 |
|
|
b2 |
-0.011 |
-0.008 |
-0.017 |
-0.013 |
-0.014 |
-0.012 |
-0.017 |
-0.013 |
-0.016 |
-0.014 |
-0.019 |
-0.016 |
|
|
b3 |
0.026 |
0.022 |
0.029 |
0.029 |
0.024 |
0.022 |
0.026 |
0.023 |
0.022 |
0.022 |
0.025 |
0.022 |
|
|
r2 |
0.99 |
0.983 |
0.984 |
0.997 |
0.988 |
0.982 |
0.98 |
0.978 |
0.927 |
0.984 |
0.948 |
0.938 |
|
|
RSS |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0.001 |
0 |
0 |
0 |
|
|
E% |
10.419 |
11.7957 |
8.0909 |
5.6229 |
10.0346 |
7.424 |
9.5312 |
0.0083 |
6.8831 |
9.1423 |
6.6958 |
8.0311 |
|
|
RMSE |
0.2278 |
0.1407 |
0.098 |
0.076 |
0.119 |
0.088 |
0.124 |
0.091 |
0.094 |
0.114 |
0.225 |
0.064 |
Modelling of Adsorption Isotherms of Dried Tomato Slices from Different Varieties
The parameters of GAB, DLP, modified BET, Oswin and Smith equations along with coefficient of determination (r2), mean rel-ative percentage error (E%), root mean square error (RMSE) and residual sum of squares (RSS) for each of the models are summa¬rized in Table 3A & 3B. Based on the experimental data used to model the adsorption isotherm, the monolayer moisture content (MMC) of the dried tomato slices obtained through a non-linear regression analysis ranged from 0.145 to 0.229, 0.153 to 0.231, 0.133 to 0.226 and 0.130 to 0.220 for UC82B, Roma, Eva-f1 and Kerewa variety, respectively. The least MMC recorded at 45º C for Kerewa variety while the highest MMC recorded by Roma variety at 25º C. The MMC was observed to decrease with increasing tem¬perature. Modeling of sorption data with GAB model permits the calculation of monolayer moisture content values that represents the moisture of a material when the whole surface covered with uni-molecular layer water vapour molecules [16,51].
Table 3B: Estimates of the Parameters of Smith and Oswin Model Equations for the Moisture Adsorption of Dried Tomato Slicesfrom UC82B (A), Roma (B), Eva-f1(C) and Kerewa (D) Variety at 25, 32 and 45ÃÂ?? C.
|
|
|
25°C |
32°C |
45°C |
|||||||||
|
A |
B |
C |
D |
A |
B |
C |
D |
A |
B |
C |
D |
||
|
Smith |
A |
0.005 |
0.006 |
0.014 |
0.01 |
0.009 |
0.005 |
0.011 |
0.009 |
0.013 |
0.012 |
0.017 |
0.013 |
|
|
B |
0.048 |
0.042 |
0.044 |
0.043 |
0.041 |
0.039 |
0.04 |
0.035 |
0.028 |
0.027 |
0.028 |
0.028 |
|
|
r2 |
0.957 |
0.943 |
0.968 |
0.947 |
0.971 |
0.96 |
0.968 |
0.965 |
0.89 |
0.957 |
0.885 |
0.913 |
|
|
RSS |
0.001 |
0.001 |
0.001 |
0.001 |
0.001 |
0.001 |
0.001 |
0.001 |
0.001 |
0 |
0.001 |
0.001 |
|
|
E% |
21.234 |
23.473 |
11.978 |
14.174 |
14.182 |
18.458 |
11.871 |
14.105 |
13.014 |
9.094 |
12.75 |
11.416 |
|
|
RMSE |
0.279 |
0.279 |
0.146 |
0.191 |
0.187 |
0.222 |
0.152 |
0.164 |
0.156 |
0.115 |
0.182 |
0.138 |
|
Oswin |
A |
-0.936 |
-0.943 |
-0.934 |
-0.932 |
-0.936 |
-0.943 |
-0.934 |
-0.943 |
-0.949 |
-0.952 |
-0.945 |
-0.95 |
|
|
B |
0.035 |
0.033 |
0.036 |
0.037 |
0.035 |
0.033 |
0.036 |
0.031 |
0.025 |
0.023 |
0.026 |
0.024 |
|
|
r2 |
0.708 |
0.665 |
0.735 |
0.667 |
0.708 |
0.665 |
0.735 |
0.704 |
0.744 |
0.764 |
0.799 |
0.755 |
|
|
RSS |
0.004 |
0.004 |
0.004 |
0.004 |
0.004 |
0.004 |
0.004 |
0.004 |
0.002 |
0.001 |
0.001 |
0.001 |
|
|
E% RMSE |
41.126 |
44.457 |
33.293 |
41.781 |
44.489 |
48.878 |
41.752 |
42.213 |
37.93 |
33.88 |
32.38 |
36.455 |
|
|
|
0.534 |
0.565 |
0.477 |
0.536 |
0.575 |
0.615 |
0.534 |
0.535 |
0.456 |
0.429 |
0.426 |
0.443 |
The increase of sorption temperature from 25 to 45 º C resulted in decreased monomolecular layer capacity in all varieties of dried tomato slices, which can be due to the exothermic nature of the sorption phenomenon. The values obtained in this study are in agreement with the monolayer moisture content reported by Kira¬noudis et al and Alakali et al for dried tomato [16,51]. A decrease in GAB monolayer moisture content with corresponding rise in temperature depicts that the absorbed water molecules gained ki¬netic energy making the attractive forces to be loosened and this permitted some water molecules to dissociate from their sorption sites thereby decreasing the equilibrium moisture values [26,49]. It could also be linked to the variation of excitation states, distance and attractive forces between molecules as temperature was altered and to a reduction in the number of active sites because of physi¬cal-chemical changes caused at elevated temperatures [42,52,53].
The decrease in monolayer moisture with temperature obtained in this study is in tandem with findings of Akanbi et al for dried toma¬to slices; Goula et al for dried tomato pulp; Mariem and Mabrouk for dried tomato slices; Ariahu et al for fresh water crayfish and for freeze dried Mexican red sauce by Escobedo-Avellaneda et al. [3,27,54-56]. According to these authors, this may be due to the reduction in the number of active sites for water binding because of the physical and chemical changes in the product induced by temperature. In addition, there is another possibility that as the temperature is raised, there is an increase in kinetic energy of wa¬ter vapour molecules and this makes them less stable, thereby, fa¬voring their departure from the binding sites of food material, thus causing the monolayer moisture content to decrease [57]. These observations imply that the optimum moisture for shelf stability of tomato slices will be determined in relation to the anticipated storage temperature. Therefore, the monolayer moisture content represents a critical parameter because it is the moisture content that guarantees minimal loss of product quality for a long period at a particular temperature, with minimal deteriorating reaction rate below this value, except for unsaturated fat oxidation [54].
The energy constant (C) obtained from the GAB model, ranged from 8.49 to 20.89, 8.60 to 22.11, 8.43 to 20.00 and 8.27 to 19.90 for UC82B, Roma, Eva-f1 and Kerewa dried slices, respectively with the Roma variety having the highest value at 45 ºC and Kere¬wa variety having the least value at 32 ºC. As reported by Lewicki, the value of the C parameter is an indication of how appropriate is the choice of GAB model to describe empirical sorption data [58]. It has been postulated that strong adsorbent-adsorbate interactions, which are exothermic, are favoured at lower temperature, resulting in an increase in parameter C and decrease in temperature [59]. However, the trend observed in the present study is in contrast with findings of Diosady et al as the value of C was found to be irregular for dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa variety since it neither decreased with temperature nor in¬creased with temperature. Goula et al, Akanbi et al, Ouafi et al, Rhim, and Hong also reported a similar trend for the C value of dried tomato pulp, dried tomato slices, Bay leaves and red pepper, respectively [17,54,59-61]. This phenomenon agrees with litera¬ture data such as the findings of Iglesias and Chirife which showed that for many food products one cannot talk about an increasing tendency of C constant depending on the decrease of temperature [62]. Iglesia and Chirife studied more than 30 different foods and reported that in about 74% of the samples, C parameter did not decrease with increase in temperature, which might be due to ir¬reversible changes associated with increasing temperature such as enzymatic reaction, or protein denaturation [62]. Pérez-Alonso et al indicate the possibility of the C constant losing its physical state because of compensation of parameters [63].
The constant correcting properties (K) ranged from 0.834 to 0.870, 0.860 to 0.881, 0.815 to 0.840 and 0.809 to 0.824 for UC82B, Roma, Eva-f1 and Kerewa variety, respectively. The highest K val¬ue recorded for Roma variety at 45º C while the least was found in Kerewa variety at 25 ºC. The K value increased as temperature increased. Furthermore, coefficient of determination (r2) ranged from 0.987 to 0.901 with UC82B variety having the highest value at 25 ºC and the least value at 45 ºC. The C values obtained are much higher than K value indicating that the heat of sorption of the first layer is higher than that of the multilayers. The value of the K parameter depicts the scope of application of the GAB model [58]. With increase in temperature from 25 to 45 ºC, the K parameter as-sumed higher values for dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa variety, which imply a change in the energy level of water molecules forming a multilayer system. Increase in K was an indication that the multilayer molecules became more entropic at higher temperature [59]. The calculated figures for the constant K was lower than unity (1.0) and this agrees with the GAB model’s assumption that the multilayer has character¬istics between those of the monolayer and bulk liquid. When k = 1, the multilayer has bulk liquid properties. This is in agreement with previous reports for similar products such as by Mariem and Mabrouk for dried tomato slices; Alakali et al for dried ginger slic¬es and Lomauro et al for dried onions [16,55,64].
The residual sum of square (RSS) for the GAB model ranged be¬tween 0 and 0.001 across the varieties, while the percentage error (E%) varied between 2.641 and 7.167% with UC82B dried slices having the highest at 45 ºC while the Eva-f1 had the least at 45º C. The root mean square error (RMSE) ranged from 0.06 to 0.113 with UC82B variety having the highest value at 45º C and Roma variety having the least value at 45º C.
From Table 3A, the empirical constants b0, b1, b2 and b3 (ob-tained from DLP model) ranged from 0.002 to 0.004, 0.011 to 0.019, -0.019 to -0.008 and 0.022 to 0.029, respectively. The coef-ficient of determination (r2) from the DLP model ranged between 0.927 and 0.997 with Kerewa variety having the highest value at 25 ºC while UC82B variety had the least value at 45 ºC. The resid-ual sum of square (RSS) ranged from 0 to 0.001 with Roma variety at 45 ºC having the highest value of 0.001 while the others at all temperatures had 0 value for RSS. From the DLP model, Kerewa variety had the least percentage error (E%) at 25 ºC while Eva-f1 variety had the highest (E%) at 25ºC, the values ranged from 4.316 to 9.48%. Similarly, for the root mean square error (RMSE), the values ranged from 0.076 to 0.2278 with Kerewa variety having the least at 25º C while UC82B variety had the highest at 25º C. Percentage error ranged between 4.32 and 9.48% with Kerewa va-riety having the least percentage error (E%) at 25 ºC while Roma variety had the highest at 45º C.
Generally, a model with a P value of less than 10% is considered suitable [64]. From the parameters presented in Table 3A & 3B, the mean relative percentage error (E%), root mean square error (RMSE) and residual sum of squares (RSS) for all four varieties of dried tomato slices were the least for GAB model followed by DLP and modified BET models. On the other hand, mean relative percentage error (E%), root mean square error (RMSE) and residu¬al sum of squares (RSS) for Oswin and Smith were relatively high. The coefficient of determination (r2) for all four varieties of dried tomato slices was highest for GAB equation followed by DLP and modified BET equations while that obtained for Oswin and Smith equations was the least. Therefore, GAB, DLP and modified BET sorption models could model adsorption behaviour of dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa varieties with high predictive accuracy.
This is in agreement with previous authors that GAB model has been proven to be the best fitting model in several food products such as potato, tomato, green pepper and carrot as reported by Kiranoudis et al, yellow dent corn by Samapundo et al, fruits and vegetables as well as meat products by Lomauro et al, and maize flour by Oyelade [51,64-66]. These findings is in agreement with that obtained by previous authors, who had studied the sorption isotherms of many fruits and vegetables. Kiranoudis et al inves- tigated the equilibrium moisture content of the potato, carrot, to¬mato, green pepper and onion in the range of 10 to 90% relative humidity and at three different temperatures (30° C, 45° C and 60° C) and concluded that GAB model is the best for predicting the ex¬perimental sorption data. Mc Laughlin and Magee determined the sorption isotherms of potatoes at temperatures from 30 to 60° C, and among the models tested, the GAB model gave the best adjust¬ment with the experimental data [51,52]. According to Noumi et al, the GAB model adequately represents the sorption isotherms of Canarium schweinfurthii fruit, while Akanbi et al concluded that the equilibrium moisture content of tomato slices follows closely the GAB equation [17,67]. According to Mariem and Mabrouk, the GAB model adequately represents the desorption isotherms of tomato pulp and the adsorption isotherms of tomato powders [55]. Goula et al studied the adsorption isotherms of tomato powder within a temperature range of 70 and 20 °C, and reported that the GAB model adequately describe the sorption isotherms at varying water activity [54].
Isosteric Heat of Sorption
The isosteric heat of sorption (HoS) was estimated from the slope of the plot between ln (aw) and 1/T (K) in Figure 5 at different moisture contents. The HoS values varied from 3.756 to 20.244 KJ/mol for UC82B dried tomato slices, 2.976 to 14.951 KJ/mol for Roma dried tomato slices, and 2.321 to 17.594 KJ/mol for Eva-f1 dried tomato slices and from 1.959 to 16.055 KJ/mol for Kerewa dried tomato slices. The HoS value at the lowest moisture level (0.02 kg/kg) was highest for dried tomato slices from UC82B vari¬ety (20.244 KJ/mol) while dried tomato slices from Roma variety (14.951 KJ/mol) had the least HoS value. Similarly, the HoS val¬ue at the highest moisture level (0.12 kg/kg) was lowest for dried tomato slices from Kerewa variety (1.959 KJ/kg) and highest for dried tomato slices from UC82B variety (3.756 KJ/kg). The result¬ing HoS were then plotted against equilibrium moisture content in Figure 6 to obtain the net isoteric heat of sorption. The net isoteric heat of sorption curve indicated that the net isosteric heat of ad¬sorption decreased with an increase in moisture content. As shown in the isotherm plots, a steep slope was observed. This implies that there is intermolecular attraction forces between sorptive sites and water vapour. However, at lower moisture content, the isosteric heat of sorption was high which thereafter decreased when mois¬ture contents became higher.
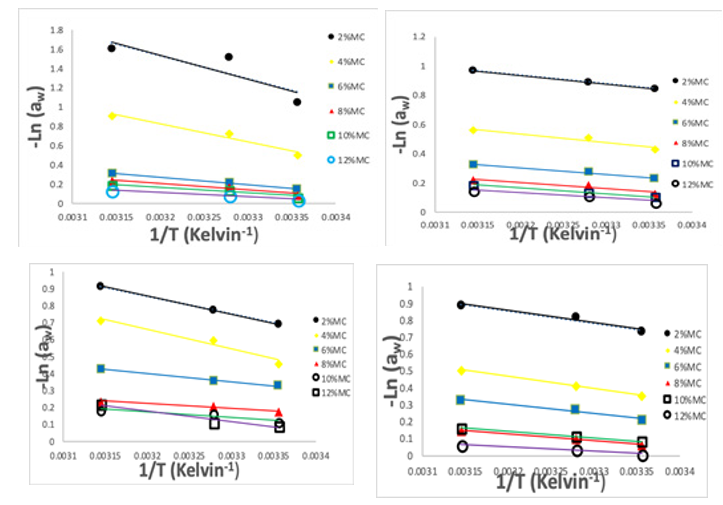
Figure 5: Net-isoteric plot of ln(aw) versus 1/T (K) at different moisture content levels of dried tomato slices from UC82B, Roma Eva-F1 and Kerewa variety
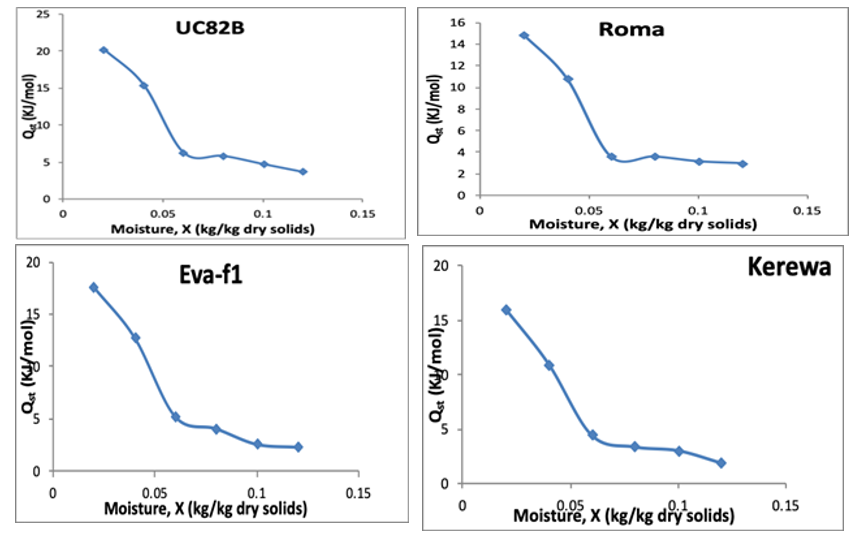
Figure 6: Plot of Isoteric Heat of Sorption Versus Equilibrium Moisture Content for Dried Tomato Slices from UC82B, Roma, Eva-F1 and Kerewa variety
The association between ln(aw) and the reciprocal of absolute temperature (1/T) for dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa varieties at constant moisture content showed that the isoteric were straight lines which agrees with findings of Ariahu et al and Labuza et al [49,56]. The HoS also termed as differential enthalpy is the minimum amount of heat required to remove or add a given amount of water [24,68]. Thus, the HoS is considered as indicative of the binding forces between the sorption sites (adsorbent) and the water vapour (adsorbate) [42]. The ob¬tained curves for dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa varieties showed that the sorption heat increases when the water content decrease. The higher heat of adsorption at lower moisture content can be justified because of the greater resistance to migration of water molecules from interior to the outer surface of the food samples [29].
In effect, when the moisture content increased, the sorption sites available decrease thus, resulting in a reduction in the sorption heat [69]. Tsami postulated that high value of the sorption heat at low moisture content, is due to the existence of the highly active polar sites on the surface of the product that are covered with water molecules forming a monolayer [70]. Iglesia and Chirife showed that sorption occurs firstly on available sites, which are highly ac¬tive resulting into high interactive energy [62]. Therefore, it takes place on less active sites bringing about a reduction in the isosteric heat and then the active sites become busy with the increase of the equilibrium moisture content. Salgado et al explained this occur¬rence by the fact that in a very restricted field of moisture, when the moisture content increases, some products swell and ease the opening of new adsorption sites of strong connections, which in¬creased the isosteric heat [71]. The isosteric heat of adsorption curve thus exhibit a regular fall with higher moisture content as earlier reported for dried tomato slices; Ziziphus leaf powder; mint leaf; and Red onion slices [17,47,55,72,73].
Conclusion
Moisture adsorption isotherm of dried tomato slices from UC82B, Roma, Eva-f1 and Kerewa varieties exhibited a sigmoid isotherm curve typical of the type II BET classification. Temperature shown to affect the sorption behavior because the equilibrium moisture content decreased with increasing temperature at constant water activity. Within the temperature range studied, the GAB, DLP and the modified BET models found to best describe the experimental data within the entire water activity range. An exponential rela¬tionship significantly describe the interdependence of the isoteric heat of sorption on the equilibrium moisture content [74,75].
References
- Grandillo, S., Zamir, D., & Tanksley, S. D. (1999). Genetic improvement of processing tomatoes: A 20 years perspective. Euphytica, 110(2), 85-97.
- Allen, A. (2008). A passion for tomatoes. Smithsonian magazine.
- Akanbi, C. T., & Oludemi, F. O. (2004). Effect of processingand packaging on the lycopene content of tomato products.International Journal of Food Properties, 7(1), 139-152.
- Adedeji, O., Taiwo, K. A., Akanbi, C. T., & Ajani, R. (2006). Physicochemical properties of four tomato cultivars grown in Nigeria. Journal of food processing and preservation, 30(1), 79-86.
- FAOSTAT (2014). Global tomato production in 2012. Rome, FAO.
- Ugonna, C. U., Jolaoso, M. A., & Onwualu, A. P. (2015). Tomato value chain in Nigeria: Issues, challenges and strategies.J. Sci. Res. Rep, 7(7), 501-515.
- FAO (2010). FAOSTAT Database (FAO). Avalaible from http://faostat.fao.org. Food and Agricultural Organization of the United Nations (FAO). 2010. FAOSTAT. Available: http:// faostat.fao.org/
- Gabriel, C. (2014). Poor infrastructure: The sad story of a nation. Vanguard, August, 24.
- Rufus, A., & Bufumoh, P. E. (2017). Critical infrastructure decay and development crises in Nigeria. Global Journal of Human-Social Science Research, 17, 59-66.
- Sousa, A. S. D., Borges, S. V., Magalhães, N. F., Ricardo, H. V., & Azevedo, A. D. (2008). Spray-dried tomato powder: re-constitution properties and colour. Brazilian Archives of Biology and Technology, 51, 607-614.
- Shi, J. (2000). Lycopene in Tomatoes: Chemical and Physical Properties Affected by Food Processing. Critical Reviews in Biotechnology ,
- 12. Bhowmik, D., Kumar, K. S., Paswan, S., & Srivastava,S. (2012). Tomato-a natural medicine and its health benefits.Journal of Pharmacognosy and Phytochemistry, 1(1), 33-43.
- Beutner, S., Bloedorn, B., Frixel, S., Hernández Blanco, I., Hoffmann, T., Martin, H. D., & Walsh, R. (2001). Quantitative assessment of antioxidant properties of natural colorants and phytochemicals: carotenoids, flavonoids, phenols and indi-goids. The role of βâ?ÂÃÂ?ÂÂcarotene in antioxidant functions. Journal of the Science of Food and Agriculture, 81(6), 559-568.
- Kaur, C., George, B., Deepa, N., Singh, B., & Kapoor, H. C. (2004). Antioxidant status of fresh and processed tomato-A review. Journal of Food Science and Technology-Mysore, 41(5), 479-486.
- Krajcovicova-Kudlackova, M., & Dusinska, M. (2004). Oxidative DNA damage in relation to nutrition. Neoplasma, 51(1), 30-33.
- Joseph, A., Agomuo, J. K., & Alaka, I. C. (2015). Storage stability of tomato paste as influenced by oil-citric acid and packaging materials. African Journal of Food Science, 9(3), 120-125.
- Akanbi, C. T., Adeyemi, R. S., & Ojo, A. (2006). Drying characteristics and sorption isotherm of tomato slices. Journal of food engineering, 73(2), 157-163.
- Verlent, I., Hendrickx, M., Rovere, P., Moldenaers, P., & Loey, A. V. (2006). Rheological properties of tomatoâ?ÂÃÂ?ÂÂbased products after thermal and highâ?ÂÃÂ?ÂÂpressure treatment. Journal of food science, 71(3), S243-S248.
- Fellows, P. J. (2009). Food processing technology: principles and practice. Elsevier.
- Doymaz, I. (2007). Air-drying characteristics of tomatoes. Journal of Food engineering, 78(4), 1291-1297.
- Adebowale, A. R., Sanni, L., Awonorin, S., Daniel, I., & Kuye, A. (2007). Effect of cassava varieties on the sorption isotherm of tapioca grits. International journal of food science & technology, 42(4), 448-452.
- Debnath, S., Hemavathy, J., & Bhat, K. K. (2002). Moisture sorption studies on onion powder. Food Chemistry, 78(4), 479-482.
- Rizvi, S. S. (2014). Thermodynamic properties of foods in dehydration. In Engineering properties of foods (pp. 261-348). CRC Press.
- Adebowale, A. A., Sanni, L. O., & Kuye, A. (2006). Effect of roasting methods on sorption isotherm of tapioca grits. Electronic J. Environ. Agric. Food Chem, 5(6), 1649-1653.
- Eim, V. S., Rosselló, C., Femenia, A., & Simal, S. (2011). Moisture sorption isotherms and thermodynamic properties of carrot. International Journal of Food Engineering, 7(3).
- Owo, H. O., Adebowale, A. A., Sobukola, O. P., Obadina,A. O., Kajihausa, O. E., Adegunwa, M. O., ... & Tomlins, K. (2016). Adsorption isotherms and thermodynamics properties of water yam flour. Quality Assurance and Safety of Crops and Foods, 9(2), 221-227.
- ESCOBEDO-AVELLANEDA, Z. A. M. A. N. T. H. A., PÉREZ-PÉREZ, M. C., BÁRCENAS-POZOS, M. E., &WELTI-CHANES, J. O. R. G. E. (2011). Moisture adsorption isotherms of freeze-dried and air-dried Mexican red sauce. Journal of Food Process Engineering, 34(6), 1931-1945.
- Adam, E., Mühlbaucr, W., Esper, A., Wolf, W., & Spieβ, W. (2000). Effect of temperature on water sorption equilibrium of onion (Allium cepa L). Drying Technology, 18(9), 2117-2129.
- Chen, C. (2000). A rapid method to determine the sorption isotherms of peanuts. Journal of Agricultural Engineering Research, 75(4), 401-408.
- Mohamed, A., Kouhila, M., Jamali, A., Lahsasni, S., & Mahrouz, M. (2005). Moisture sorption isotherms and heat of sorption of bitter orange leaves (Citrus aurantium). Journal of food Engineering, 67(4), 491-498.
- Jamali, A., Kouhila, M., Mohamed, L. A., Jaouhari, J. T., Idlimam, A., & Abdenouri, N. (2006). Sorption isotherms of Che-nopodium ambrosioides leaves at three temperatures. Journal of Food Engineering, 72(1), 77-84.
- Mehta, S., & Singh, A. (2006). Adsorption isotherms for red chilli (Capsicum annum L.). European Food Research and Technology, 223(6), 849-852.
- Janjai, S., Bala, B. K., Tohsing, K., Mahayothee, B., Haewsungcharern, M., Mühlbauer, W., & Müller, J. (2006). Equilibrium moisture content and heat of sorption of longan (Dimocarpus longan Lour.). Drying technology, 24(12), 1691-1696.
- Ghodake, H. M., Goswami, T. K., & Chakraverty, A. (2007). Moisture sorption isotherms, heat of sorption and vaporiza-tion of withered leaves, black and green tea. Journal of Food Engineering, 78(3), 827-835.
- Sinija, V. R., & Mishra, H. N. (2008). Moisture sorption isotherms and heat of sorption of instant (soluble) green tea powder and green tea granules. Journal of food engineering, 86(4), 494-500.
- Vullioud, M., Marquez, C. A., & De Michelis, A. (2006). Equilibrium sorption isotherms and isosteric heat of rose hip fruits (Rosa eglanteria). International Journal of Food Properties, 9(4), 823-833.
- Shatadal, P. and D. S. Jayas. (2000). Sorption isotherm of foods. In: Development of Drying. Volume 1: Food Dehydration. Eds. A.S. Mujumdar and Sirikalaya Suvachittanont. Kastsart University Press, Bangkok, Thailand. pp 43-58.
- Wrolstad, R. E., Acree, T. E., Decker, E. A., Penner, M. H.,Reid, D. S., Schwartz, S. J., ... & Sporns, P. (Eds.). (2005). Handbook of food analytical chemistry, volume 1: Water, proteins, enzymes, lipids, and carbohydrates. John Wiley & Sons.
- Al-Muhtaseb, A. H., McMinn, W. A. M., & Magee, T. R. A. (2002). Moisture sorption isotherm characteristics of food products: a review. Food and bioproducts processing, 80(2), 118-128.
- Monsurat, B., Matthew, O. O., & Victor, N. E. (2019). Modeling of the adsorption isotherm of Pleurotus ostreatus using Guggenheim-Anderson-de Boer (GAB) equation. Journal of Engineering and Technology Research, 11(4), 41-46.
- Brunauer, S., Emmett, P. H., & Teller, E. (1938). Adsorption of gases in multimolecular layers. Journal of the American chemical society, 60(2), 309-319.
- Arslan, N., & TogÃÃÂ???rul, H. (2006). The fitting of various models to water sorption isotherms of tea stored in a chamber under controlled temperature and humidity. Journal of Stored Products Research, 42(2), 112-135.
- Ait-Mohammed, L., Kouhila, M., Jamali, A., Lahsasni, S., & Mahrouz, M. (2004). Experimental study of adsorption-de-sorption isotherms of bitter orange leaves. In Proceeding of the 14th International Drying Symposium (pp. 1404-1410).
- Kouhila, M., Kechaou, N., Otmani, M., Fliyou, M., & Lahsasni, S. (2002). Experimental study of sorption isotherms and drying kinetics of Moroccan Eucalyptus globulus. Drying Technology, 20(10), 2027-2039.
- Sogi, D. S., Shivhare, U. S., Garg, S. K., & Bawa, A. S. (2003). Water sorption isotherm and drying characteristics of tomato seeds. Biosystems Engineering, 84(3), 297-301.
- Idlimam, A., Lamharrar, A., Abdenouri, N., Akkad, S., Kane,C. E., Jamali, A., & Kouhila, M. (2008). Thermodynamic properties and moisture sorption isotherms of Argania spinosa and Zygophyllum gaetulum. Journal of Agronomy.
- Saad, A., Touati, B., Draoui, B., Tabti, B., Abdenebi, A., & Benaceur, S. (2014). Mathematical modeling of moisture sorption isotherms and determination of isosteric heats of sorption of Ziziphus leaves. Modelling and Simulation in Engineering, 2014.
- Giovanelli, G., Zanoni, B., Lavelli, V., & Nani, R. (2002). Water sorption, drying and antioxidant properties of dried tomato products. Journal of food engineering, 52(2), 135-141.
- Labuza, T. P., Kaanane, A., & Chen, J. Y. (1985). Effect of temperature on the moisture sorption isotherms and water activity shift of two dehydrated foods. Journal of Food science, 50(2), 385-392.
- Lim, L. T., Tang, J., & He, J. (1995). Moisture sorption characteristics of freeze dried blueberries. Journal of food science, 60(4), 810-814.
- Kiranoudis, C. T., Maroulis, Z. B., Tsami, E., & Mari-nos-Kouris, D. (1993). Equilibrium moisture content and heat of desorption of some vegetables. Journal of Food engineering, 20(1), 55-74.
- McLaughlin, C. P., & Magee, T. R. A. (1998). The determination of sorption isotherm and the isosteric heats of sorption for potatoes. Journal of Food Engineering, 35(3), 267-280.
- Yogendrarajah, P., Samapundo, S., Devlieghere, F., De Sae-ger, S., & De Meulenaer, B. (2015). Moisture sorption isotherms and thermodynamic properties of whole black peppercorns (Piper nigrum L.). LWT-Food Science and Technology, 64(1), 177-188.
- Goula, A. M., Karapantsios, T. D., Achilias, D. S., & Ada-mopoulos, K. G. (2008). Water sorption isotherms and glass transition temperature of spray dried tomato pulp. Journal of Food Engineering, 85(1), 73-83.
- Mariem, S. B., & Mabrouk, S. B. (2015). Moisture sorption isotherms and isosteric heats of sorption of tomato slices. American Journal of Renewable and Sustainable Energy, 1(3), 140-155.
- Ariahu, C. C., Kaze, S. A., & Achem, C. D. (2006). Moisture sorption characteristics of tropical fresh water crayfish (Procambarus clarkii). Journal of Food Engineering, 75(3), 355-363.
- Tunç, S., & Duman, O. (2007). Thermodynamic properties and moisture adsorption isotherms of cottonseed protein isolate and different forms of cottonseed samples. Journal of Food Engineering, 81(1), 133-143.
- Lewicki, P. P. (1997). The applicability of the GAB model to food water sorption isotherms. International journal of food science & technology, 32(6), 553-557.
- Diosady, L. L., Rizvi, S. S. H., Cai, W., & Jagdeo, D. J. (1996). Moisture sorption isotherms of canola meals, and applications to packaging. Journal of Food Science, 61(1), 204-208.
- Ouafi, N., Moghrani, H., Benaouada, N., Yassaa, N., Maachi, R., & Younsi, R. (2015). Moisture sorption isotherms and heat of sorption of Algerian bay leaves (Laurus nobilis). Maderas. Ciencia y tecnología, 17(4), 759-772.
- Rhim, J. W., & Hong, S. I. (2011). Effect of water activity and temperature on the color change of red pepper (Capsicum annuum L.) powder. Food Science and Biotechnology, 20(1), 215-222.
- Iglesias, H. A., & Chirife, J. (1976). Local isotherm concept and modes of moisture binding in food products. Journal of Agricultural and Food Chemistry, 24(1), 77-79.
- Perez-Alonso, C., Beristain, C. I., Lobato-Calleros, C., Rodrí-guez-Huezo, M. E., & Vernon-Carter, E. J. (2006). Thermodynamic analysis of the sorption isotherms of pure and blended carbohydrate polymers. Journal of Food Engineering, 77(4), 753-760.
- Lomauro, C. J., Bakshi, A. S., & Labuza, T. P. (1985). Evaluation of food moisture sorption isotherm equations part II: Milk, coffee, tea, nuts, oilseeds, spices and starchy foods. LWT-Food Science and Technology, 18(2), 118-124.
- Samapundo, S., Devlieghere, F., De Meulenaer, B., Atuk-wase, A., Lamboni, Y., & Debevere, J. M. (2007). Sorption isotherms and isosteric heats of sorption of whole yellow dent corn. Journal of Food Engineering, 79(1), 168-175.
- Oyelade, O. J., Tunde-Akintunde, T. Y., Igbeka, J. C., Oke,M. O., & Raji, O. Y. (2008). Modelling moisture sorption isotherms for maize flour. Journal of Stored Products Research, 44(2), 179-185.
- Noumi, GB, Laurent, S., Ngameni, E., Kapseu, C., Jannot, Y., & Parmentier, M. (2004). Modeling of the dehydration of the fruit pulp of Canarium schweinfurthii engl. Tropicultura , 22 (2), 70-76.
- Aguerre, R. J., Suarez, C., & Viollaz, P. E. (1989). New BET type multilayer sorption isotherms. Part II: Modelling water sorption in foods. Lebensm. _Wiss. a. Technol, 22, 192-195.
- Pezzutti, A., & Crapiste, G. H. (1997). Sorptional equilibrium and drying characteristics of garlic. Journal of food Engineering, 31(1), 113-123.
- Tsami, E., Maroulis, Z. B., Marinos-Kouris, D., & Saravacos,G. D. (1990). Heat of sorption of water in dried fruits. International Journal of Food Science & Technology, 25(3), 350-359.
- Salgado, M. A., Lebert, A., Garcia, H. S., Muchnik, J., & Bim-benet, J. J. (1994). Development of the characteristic drying curve for cassava chips in monolayer. Drying technology, 12(3), 685-696.
- Dalglç, A. C., Pekmez, H., & Belibal, K. B. (2012). Effect of drying methods on the moisture sorption isotherms and thermodynamic properties of mint leaves. Journal of Food Science and Technology, 49(4), 439-449.
- Gazor, H. R., & Eyvani, A. (2011). Adsorption isotherms for red onion slices using empirical and neural network models. International Journal of Food Engineering, 7(6).
- Oswin, C. R. (1946). The kinetics of package life. III. The isotherm. Journal of the Society of Chemical Industry, 65(12), 419-421.
- Smith, S. E. (1947). The sorption of water vapor by high polymers. Journal of the American Chemical Society, 69(3), 646-651.



