Research Article - (2026) Volume 11, Issue 1
Validation of Analytical Method for Quantification of Six Pesticide Residues in Dried Chili Powder Using GC-MS/MS
Received Date: Feb 07, 2026 / Accepted Date: Feb 28, 2026 / Published Date: Mar 13, 2026
Copyright: ©2026 Neha Gupta. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Gupta, N., Singh, R., Bhat, B., Das, M. (2026). Validation of Analytical Method for Quantification of Six Pesticide Residues in Dried Chili Powder Using GC-MS/MS. Adv Nutr Food Sci, 11(1), 01-11.
Abstract
The study describes an analytical method for determination of six pesticide residues namely Quinalphos, Dimethoate, alpha- endosulfan, beta-endosulfan, Triazophos and Endosulfan sulfate, in dried chilli using GC-MS/MS (Gas chromatography triple quad mass spectrometer) instrument with modified QuEChERS (Quick, Easy, Cheap, Effective, Rugged and Safe) sample preparation technique. The developed method was validated with respect to various performance attributes as per SANTE 11312/2021 and was found to be linear within the range from 10 ng/g to 200 ng/g with the correlation of coefficient (r2) > 0.990 for all the 6 pesticides. The method was found to be precise with a method LOQ (Limit of Quantification) concentration level of 10 ng/g, with % RSD (Relative standard deviation) for 6 replicates as less than 15% for all the six compounds. Recovery studies in dried chilli shows that method is accurate at all the three recovery levels i.e. 10 ng/g, 20 ng/g and 50 ng/g with % RSD (n-6) < 15 for all the six pesticides. Mean recovery control charts were plotted for recovery data, as a performance based quality assessment tool. Validated method was also applied to 8 popular market brands and 6 loose unbranded samples collected from the local market of India to check pesticide load in them.
Keywords
QuEChERS SANTE, LOQ, % RSD, Control Chart
Introduction
Chili (Capsicum annum L.) belonging to the "Solanaceae" family, is one of the most important Indian spices, owing to its worldwide usage in one form or another. Dried chili powder is a widely consumed spice, valued for its pungency, color, and flavor. India is the world leader not only in production of chili, but also its largest consumer and world exporter [1].
Chilies specifically are prone to qualitative and quantitative loss in growth period due to attack of insects and non-insect pests including chili thrips, Scirtothrips dorsalis hood and yellow mite [2]. To compensate for this loss, wide ranges of pesticides are used to protect these fresh produce from the damage caused by insects, mites, rodents and other pests. Owing to the uncontrolled usage of pesticides to increase crop yield, residual contamination of pesticides is evident in dried chili production [3]. Dried chili powder is found to be susceptible to pesticide contamination due to its post-harvest processing and storage conditions. Processing of chili fruit to produce dried chili used as spice, leads to increase in the concentration of the pesticides owing to the added processing factor [4]. However, selection of these particular insecticides for a commodity must not only work upon its efficacy for pest control but its residues in the commodity must also be acceptable [5]. The potential presence of pesticide residues in dried chili powder raises concerns regarding food safety, regulatory compliance, and human health risks. Therefore, analysis of residual amounts of pesticides in raw agricultural crops and processed foods is a leading preventive measure of public health. Efficient method of analysis for determination of these residual pesticides in these types of commodities is of utmost importance.
Analysis of agrochemical residues in red chili powder is always considered difficult because of higher matrix interference due to capsaicinoids, essential oils, pigments and other co-extractives, posing analytical challenges due to matrix interferences [6]. Various sample preparation techniques, including QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) and dispersive solid-phase extraction (d-SPE), have been extensively studied for their effectiveness in pesticide residue extraction. The QuEChERS method, in particular, has been widely adopted due to its simplicity, high recovery rates, and is still evolving for pesticide analysis in diverse applications, indicating its multidisciplinary nature and ongoing significance in research [7]. Sample preparation in chili powder however involves using solvents like acetonitrile and adsorbents such as PSA and C18 to clean up the samples [8,9]. The QuEChERS methodology has undergone rigorous validation for a multitude of pesticide residues across various food matrices and has been formally recognized as the official method 2007.01 by the Association of Analytical Communities (AOAC) and by CEN in 2008 [10]. Modifications to the QuEChERS method, such as using different dispersive adsorbents, have been shown to improve the extraction efficiency and reduce matrix interference [6]. Gas chromatography-triple quadrupole mass spectrometry (GC-MS/MS) is widely utilized for pesticide residue analysis due to its superior selectivity, sensitivity, and robustness. Studies have demonstrated its effectiveness in detecting multi-class pesticide residues in spices, including chili powder, with high accuracy and reproducibility [6,11]. The use of multiple reaction monitoring (MRM) in GC-MS/MS enhances the detection capabilities by reducing background noise and improving quantification. GC- MS/MS is employed for its high separation power and selectivity, making it suitable for analyzing complex matrices like chili powder [12]. While extensive research has been conducted on pesticide residue analysis in various food matrices, including studies focused on dried chili powder using GC-MS/MS in MRM mode of acquisition [5,6,13-16]. None of the study covers all the 6 pesticides focused in this work. Therefore the present study aims optimization of an analytical method for estimation of six pesticides in dried chili including Quinalphos, Dimethoate, Alpha- Endosulfan, Beta-Endosulfan, Triazophos and Endosulfan sulfate, all of which have already set Maximum residue limits (MRLs) at national (FSSAI) as well as international level. Method detailed in paper is based on modified QuEChERS as sample preparation procedure along with GC-MS/MS as instrumental technique, with quantification at concentrations below MRLs (Maximum Residue Limits) specified in national and international regulations for the identified pesticides. This paper further elaborates validation methodology including various performance attributes and statistical representation of results in the form of mean recovery control charts for the developed method of analysis, ensuring the reliability and reproducibility of analytical method. Mean recovery control chart is an important performance based quality assessment tool for statistical control and acceptance of analytical results produced. Random branded and unbranded samples were picked up from the Indian market to judge applicability of developed & validated method of analysis for 6 pesticide residues studied.
Material and Method
Procurement of Standard and Other Chemicals
• Procurement of Standards
CRM (Certified Reference Material) for selected pesticides of study were procured from Chem Service for Quinalphos, Dr. Ehrenstorfer for Dimethoate, Endosulfan Sulfate & Triazophos and Restek for alpha & beta Endosulfan. All the CRM’s used in analysis had purity above 99 %.
• Procurement of Other Chemical and Reagents
Chemicals used in sample preparation procedures were majorly QuEChERS salts including Magnesium Sulphate Hepta Hydrate, Carbon 18 & Graphitized Carbon Black i.e. GCB from Agilent; Sodium Chloride from Merck; Sodium Citrate Dibasic Sesqui Hydrate & Sodium Citrate Tribasic Dihydrate from Sigma Aldrich. Other chemical & reagents used in analysis include, anhydrous Sodium Sulphate of 99% purity from Merck; Acetonitrile of 99.8 % purity from Qualigens; Ethyl acetate of 99.9 % purity from SDFCL and Ultrapure water produced by a Milli-Q system (Millipore Advantage A10).
Standard Preparation
• Standard Stock Preparation
Individual 100 µg/ml (approximately) stock standard for all the 6 pesticides (Quinalphos, Dimethoate, alpha-Endosulfan, beta-Endosulfan, Triazophos, Endosulfan Sulfate) were made by diluting 1 mg of individual reference standard with 10 ml of Acetone, in a 10 ml volumetric flask.
• Working Standard Preparation
Mixed standard of 10 µg/ml for all the 6 pesticides, namely Quinalphos, Dimethoate, Alpha-Endosulfan, Beta-Endosulfan, Triazophos, Endosulfan sulfate was prepared by diluting individual standard stock solutions tenfold using Ethyl acetate as solvent. Further, mixed standard of 1 µg/ml in Ethyl acetate as solvent was prepared by serial dilution.
• Solvent Calibration Standard Preparation
Calibration standards in solvent at concentration levels of 5, 10, 25, 50 and 100 ng/ml were prepared using a working standard of 10 µg/ml, for solvent linearity.
• Matrix-Matched Calibration Standard Preparation
The co-extraction of various natural compounds, including those found in chlorophyll-rich foods and other naturally occurring pigments (e.g., spices), lipids or fats (e.g., nuts), essential oils, and flavonoids (e.g., herbs), can result in detrimental effects such as the suppression or enhancement of signals, a phenomenon commonly identified as the matrix effect (ME) [17]. These matrix effects induce analytical complications that compromise the precision of the results and may manifest in the recovery rates of pesticides by either diminishing or elevating the acceptable limits for validation purposes from 70 to 120% [18].
To compensate for the matrix effect introduced by chilli matrix components, matrix match calibration standards were prepared using appropriate matrices (free from pesticides under study). Blank matrix of dried chilli was prepared using sample preparation technique, mentioned in section 2.3. Matrix match calibration standards were prepared at concentration levels of 5, 10, 25, 50 and 100 ng/ml by diluting 10 times higher solvent standards in the blank chilli matrix, keeping the matrix equal for all the 5 calibration standards.
Sample Preparation
Dried chilli samples were homogenized well before taking representative quantities for analysis. 5 g of sample was weighed in a 50 ml centrifuge tube, to which 20 ml of water was added to make a slurry. To this sample slurry, 10 ml of acetonitrile was added. Mixture was vortex for 1 minute and QuEChERS salts namely, 4 g Magnesium Sulphate, 1 g Sodium Chloride, 1 g Sodium Citrate Tribasic Dihydrate and 0.5 g Sodium Citrate Tribasic Sesqui Hydrate were added for extraction. The mixture was vortexed for 5 minutes and then centrifuged for 10 minutes at 5000 rpm for separation. After centrifugation, 6 ml of supernatant layer was carefully transferred to another 15 ml centrifuge tube. To this supernatant layer, 900 mg of Magnesium Sulphate, 150 mg of C-18 and 45 mg of Graphitized Carbon Black were added for further sample clean-up process. It was then vortexed for 5 minutes and again centrifuged at 10,000 rpm for about 5 minutes. 1 ml of supernatant was then dried under nitrogen, made up with 1 ml of Ethyl Acetate and filtered with 0.45 µm membrane filter before injecting onto the GC-MS/MS system.
Instrumental Conditions
Gas chromatography mass spectrometry triple quad instrument (GC-MS/MS) with MRM (Multiple reaction monitoring) was used for this particular study and combined method of analysis was optimized for 6 pesticides namely: Quinalphos, Dimethoate, alpha-Endosulfan, beta-Endosulfan, Triazophos and Endosulfan sulfate. GC-MS/MS analysis was performed using a Shimadzu TQ 8030 equipped with an auto-sampler and coupled to a triple quadruple mass analyzer. GC was connected with a DB-5 MS column i.e. (5% phenyl) methyl polysiloxane (60 m x 0.25 µm ID x 0.25 mm film thickness). Sample injection was carried out in splitless mode at a temperature of 250°C. Helium was used as carrier gas at a flow rate of 1.0 ml/min. The oven temperature was programmed as follows: initial oven temperature was held at 70°C for 1 min., increased to 150°C at 20°C/min. and further increased to 250°C at a rate of 5 °C/min., with a hold of 10 min. Final temperature of 300°C was attained at 10°C/min. and held for 15 min. The ion source and interface temperature were set at 230°C and 280°C respectively.
With above optimized method parameters, a method with total run-time of 55 minutes was developed for effective separation and quantification of 6 pesticide residues in dried chilli matrix (Figure 1).
Figure 1: Typical Tic Of 100 Ng/Ml Mm Standard With Compound Name At Peak Top Of Chromatogram With Following Elution Order: Quinalphos–16.1 Min., Dimethoate–17.4 Min., Α-Endosulfan–25.1 Min., β-endosulfan– 27.4 min., triazophos–28.3 min. and endosulfan sulfate–29.4 min
Results and Discussion
The method discussed in this paper for analysis of six pesticide compounds in dried chilli is optimized using GC-MS/MS for specific MRM (Multiple Reaction Monitoring) transitions with quantifier and qualifier ions (Table 1). Mass spectra obtained for all the 6 pesticide compounds is given in Figure 2.
|
S.No. |
Compound name |
MRM transition |
Collision energy (CE) |
|
1 |
Quinalphos |
157.1 > 129.0 |
14 |
|
157.1 > 102.0 |
24 |
||
|
2 |
Dimethoate |
125.0 > 79.0 |
8 |
|
|
|
125.0 > 47.0 |
14 |
|
125.0 > 62.0 |
10 |
||
|
3 |
alpha-Endosulfan |
240.9 > 205.9 |
15 |
|
240.9 > 170.0 |
30 |
||
|
240.9 > 136.0 |
40 |
||
|
4 |
beta-Endosulfan |
195.0 > 159.0 |
10 |
|
195.0 > 125.0 |
25 |
||
|
195.0 > 161.0 |
10 |
||
|
5 |
Triazophos |
257.0 > 162.0 |
8 |
|
257.0 > 134.0 |
22 |
||
|
257.0 > 119.0 |
26 |
||
|
6 |
Endosulfan sulphate |
271.9 > 236.9 |
20 |
Table 1: Optimized MRM transitions with respective collision energy (CE) in eV for six pesticide compounds used in study
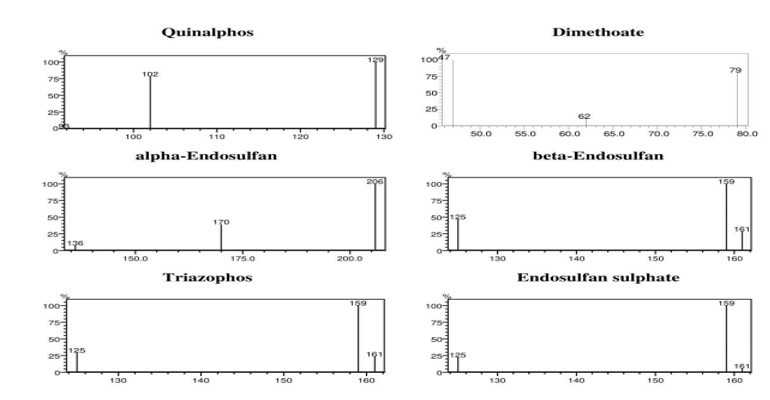
Figure 2: Mass Spectra of the Six Pesticide Compounds Showing Relative Intensities of Major Ions
Method Validation
The developed method of analysis was validated following guidelines of SANTE 11312/2021 for Analytical quality control and method validation procedures for pesticide residues analysis in food and feed, covering parameters like Specificity, LOD, LOQ, Linearity, Precision and Accuracy (Recovery) [18].
• Selectivity
The presence of co-extracted interferences in the chromatograms from blank samples was used to assess selectivity. By tracking the chromatograms run on MRM (Multiple Reaction Monitoring) mode of acquisition for every insecticide at the anticipated retention time window for each component, the existence of matrix interferences was investigated. The MRM detection mode reduces the signal of these inferences even though co-extracted substances from the chili matrix occur during the pesticide extraction process. This is because the most prevalent ions generated after each pesticide's fragmentation are used to quantify it [19]. The use of GC-MS/ MS with MRM mode of acquisition therefore adds selectivity to developed method of analysis.
• Specificity
The method was found to be specific for all the six pesticides by comparing matrix match mixed standard in chilli at LOQ level of 10 ng/g with blank chilli matrix (Figure 3).
Figure 3: Comparative chromatograms of Matrix match chili blank (A) and Matrix match pesticide standard of 10 ng/ml (B) concentration for specificity
• Sensitivity as Limit of Detection (LOD) and Limit of Quantification (LOQ)
Precision data as % RSD (Relative standard deviation) for LOD (Limit of Detection) at concentration level of 5 ng/g and LOQ (Limit of Quantification) at concentration level of 10 ng/g, for proposed method of analysis for 6 replicates is given in below table (Table 2). The results show that % RSD at LOQ is less than 15%, for all the six pesticides.
|
Compound Name |
LOD (Limit of Detection) – 2.5 ng/g |
LOQ (Limit of Quantification) – 5 ng/g |
||
|
Average Area |
% RSD (n-6) |
Average Area |
% RSD (n-6) |
|
|
Quinalphos |
2375 |
11.9 |
6126 |
9.7 |
|
Dimethoate |
1105 |
13.1 |
2509 |
12.3 |
|
alpha-Endosulfan |
897 |
9.4 |
2114 |
11.4 |
|
beta-Endosulfan |
884 |
9.8 |
2361 |
3.7 |
|
Triazophos |
2057 |
8.4 |
4752 |
4.6 |
|
Endosulfan sulfate |
1443 |
15.9 |
3769 |
7.8 |
Table 2: Precision Data With Average Area And % Rsd At Lod And Loq Concentration Levels For 6 Replicates, Given In Table For All The Six Pesticide Compounds Respectively.
• Linearity
Matrix match calibration standards were used for linearity so as to remove any interference due to the chilli matrix. The method is found to be linear over the range from 5 ng/g to 100 ng/g at Instrument, with correlation of coefficient greater than 0.990 for all the six compounds (Figure 4 & Table 3).
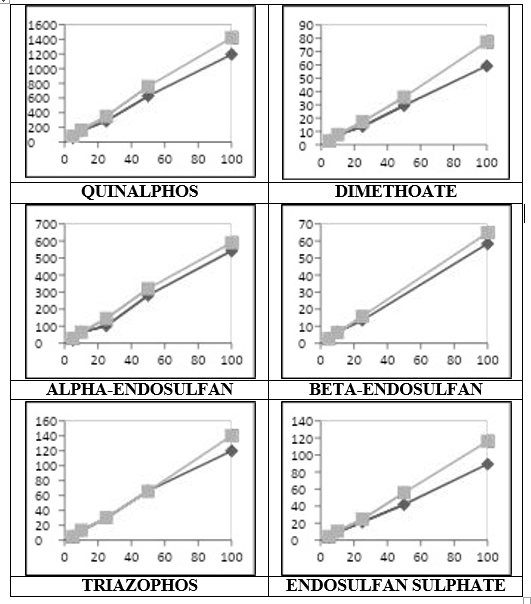
Figure 4: Comparative calibration curve of six pesticide compounds in solvent and chili matrix, with concentrations at 5, 10, 25, 50 and 100 ng/ml at horizontal axis and response in terms of area on vertical axis. Dark color line indicates Solvent linearity and light color line indicates Matrix match linearity for chili
|
Compound Name |
Solvent Linearity |
Matrix match – Chili Linearity |
Matrix factor |
||
|
|
Regression Equation |
r2 |
Regression Equation |
r2 |
|
|
Quinalphos |
y=1185.6x+1349.5 |
0.998 |
y=1418.6x+1528.9 |
0.999 |
19.7 |
|
Dimethoate |
y=584.79x+407.1 |
0.998 |
y=777.28x+1391.6 |
0.998 |
32.9 |
|
alpha-Endosulfan |
y=551.08x+787.96 |
0.993 |
y=593.29x+426.57 |
0.998 |
7.7 |
|
beta-Endosulfan |
y=581.92x+23.402 |
0.999 |
y=653.78x+457.71 |
1.000 |
12.3 |
|
Triazophos |
y=1210.7x+665.36 |
0.997 |
y=1418x+3101.8 |
0.998 |
17.1 |
|
Endosulfan sulphate |
y=895.08x-985.37 |
0.999 |
y=1180.2x-2531.8 |
0.999 |
31.9 |
Table 3: Regression equation and respective correlation of regression (r2) of Solvent and matrix match linearity run with associated matrix effect, for the six pesticide compounds studied
• Matrix Effect
According to earlier research, interactions between analyte functional groups like hydroxyl, amino, and phosphate and the active surfaces in the GC-MS (injector, column, detector) system result in a signal enhancer brought on by the matrix effect [20].
It is crucial to emphasize that at least one of these functional groups are present in the pesticides examined in this paper. This makes it an important aspect to study the matrix effect associated with chili matrix for concerned pesticides.
For this, solvent and matrix-matched chili linearities were run on GC-MS/MS instrument, so as to compare area response and finally slope of plotted calibration curves to calculate Matrix Factor, using below mentioned formulae:
% Matrix factor (MF) = [Matrix-matched slope –
Solvent slope] x 100
Solvent Linearity Slope
Also, the matrix effect was assessed based on a calculated matrix factor, classified as ‘Weak’, ‘medium’ and ‘Strong’ based on the value of the matrix factor [21]. Matrix match calibration curves are therefore required in order to prevent any associated area response and recovery enhancement owing to positive matrix factors observed, thus assuring accurate results [19].
• Accuracy
Recovery at three levels was carried out using blank dried chilli matrix spiked at concentrations of 20 ng/g, 50 ng/g and 100 ng/g. % Relative Standard Deviation (% RSD) for 6 replicates run at three recovery levels is found to be less than 15 % for all the 6 pesticide compounds in dried chilli matrix. (Table 4) Method accuracy in terms of calculated percent recovery is also evident for pesticides in chili powder ranging from 70% to 110%, indicating the method's reliability and in line with previous work by [6,9]. Mean recovery control charts were plotted for performance based quality assessment of recovery study carried out. Mean recovery control charts were plotted for all the 6 pesticides under study with data obtained for 18 replicates in total span at three different concentration levels covering the range of method from LOQ, twice of LOQ and five times that of LOQ. Control charts depicted that recovery control limits spanned from about 70 to 140% of spiked recovery concentration levels. (Figure 5)
Figure 5: Mean recovery control charts for 6 pesticides at 3 concentration levels with 18 replicates in total
|
Compound Name |
Spiked concentration (µg/g) |
% Mean Value of Recovery (MVa) |
Standard Deviation (SDa) |
% Relative Standard Deviation (RSDa) |
% Total Recovery |
|
Quinalphos |
0.01 |
90.1 |
4.7 |
5.2 |
86.2 |
|
0.025 |
81.8 |
3.2 |
3.9 |
||
|
0.5 |
86.8 |
4.6 |
5.4 |
||
|
Dimethoate |
0.01 |
103.6 |
4.4 |
4.3 |
94.3 |
|
0.025 |
87.2 |
3.2 |
3.7 |
||
|
0.5 |
91.9 |
4.4 |
4.8 |
||
|
alpha-Endosulfan |
0.01 |
104.5 |
6.3 |
6.0 |
100.3 |
|
0.025 |
95.1 |
7.4 |
7.7 |
||
|
0.5 |
101.2 |
5.0 |
4.9 |
||
|
beta-Endosulfan |
0.01 |
113.4 |
11.2 |
9.9 |
105.3 |
|
0.025 |
99.2 |
9.8 |
9.9 |
||
|
0.5 |
103.2 |
7.8 |
7.6 |
||
|
Triazophos |
0.01 |
117.4 |
5.1 |
4.3 |
103.1 |
|
0.025 |
95.3 |
4.9 |
5.1 |
||
|
0.5 |
96.5 |
5.8 |
6.0 |
||
|
Endosulfan sulfate |
0.01 |
117.6 |
10.9 |
9.3 |
107.1 |
|
0.025 |
100.2 |
3.7 |
3.7 |
||
|
0.5 |
103.6 |
8.2 |
7.9 |
Table 4: Recovery results of six pesticides in chili sample using developed method of analysis at three recovery levels i.e. 10 ng/ ml,25 ng/ml and 50 ng/ml
Method Application
Developed and validated method of analysis discussed in the paper was applied to 8 popular brands and 6 loose samples of chilli from market at random to assess pesticide load and applicability of developed method in real-world scenarios. Also, the method was found adaptable to matrices from different origins owing to analysis of both packed and loose samples of dried chilli powder. A similar study was done by, highlighting versatility of method in pesticide residue analysis. Out of 14 market chilli samples analysed, all the loose chilli samples were found to show presence of triazophos (Table 5) [12]. The presence of triazophos in dried chilli is supported by work done by . where triazophos was the most intense pesticide found in chilli fruits with concentrations higher than the EU MRL [23]. Triazophos content in one of the 6 unbranded dried chilli samples analysed was found exceeding the set MRL as per national (FSSAI) & international (EU) regulations. (Figure 6)
|
Compound Name |
Spiked concentration (µg/g) |
Repeatability |
Ruggedness |
||||||
|
MVa |
SDa |
% RSDa |
% Total Recovery |
MVa |
SDa |
% RSDa |
% Total Recovery |
||
|
Quinalphos |
0.01 |
96.4 |
6.0 |
6.2 |
91.0 |
94.9 |
8.3 |
8.7 |
92.0 |
|
0.025 |
86.4 |
5.4 |
6.2 |
|
86.6 |
7.5 |
8.6 |
|
|
|
0.5 |
90.4 |
3.1 |
3.5 |
|
94.5 |
6.7 |
7.1 |
|
|
|
Dimethoate |
0.01 |
105.9 |
5.7 |
5.4 |
91.7 |
105.0 |
8.1 |
7.7 |
96.9 |
|
0.025 |
84.0 |
5.6 |
6.6 |
|
89.9 |
7.5 |
8.4 |
|
|
|
0.5 |
85.2 |
5.0 |
5.8 |
|
95.7 |
5.8 |
6.0 |
|
|
|
alpha- Endosulfan |
0.01 |
100.8 |
5.5 |
5.5 |
94.4 |
91.1 |
9.7 |
10.6 |
93.2 |
|
0.025 |
90.3 |
4.6 |
5.1 |
|
88.8 |
9.6 |
10.8 |
|
|
|
0.5 |
92.3 |
3.2 |
3.4 |
|
99.7 |
4.6 |
4.6 |
|
|
|
beta-Endosulfan |
0.01 |
105.0 |
8.5 |
8.1 |
95.9 |
97.9 |
10.2 |
10.5 |
95.6 |
|
|
0.025 |
90.5 |
5.9 |
6.5 |
|
88.4 |
7.9 |
8.9 |
|
|
0.5 |
92.1 |
4.6 |
5.0 |
|
100.4 |
4.0 |
4.0 |
||
|
Triazophos |
0.01 |
110.3 |
4.0 |
3.6 |
96.6 |
101.0 |
9.4 |
9.3 |
94.2 |
|
0.025 |
90.2 |
5.1 |
5.7 |
|
87.3 |
7.4 |
8.4 |
||
|
0.5 |
89.4 |
2.9 |
3.2 |
|
94.2 |
4.3 |
4.6 |
||
|
Endosulfan sulfate |
0.01 |
110.1 |
5.6 |
5.1 |
97.9 |
96.3 |
9.0 |
9.3 |
93.6 |
|
0.025 |
90.6 |
6.5 |
7.2 |
|
86.7 |
8.5 |
9.8 |
||
|
0.5 |
93.1 |
4.2 |
4.5 |
|
97.8 |
6.1 |
6.2 |
||
|
a Number of replicates, n=6 Abbreviations: MV, Mean value; SD, Standard Deviation; RSD, Relative standard deviation |
|||||||||
Table 5: Average recovery of six pesticides in chili, for 6 replicates studied at 3 concentration levels with corresponding SD and% RSD for assessing method's reproducibility in terms of Repeatability and Ruggedness
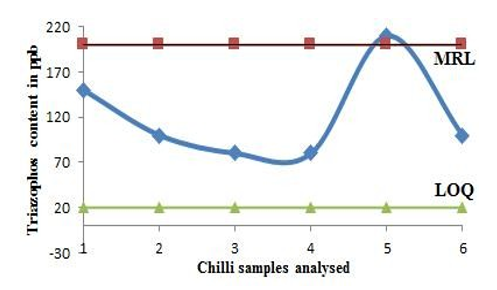
Figure 6: Triazophos pesticide found in random loose chilli samples from market
Conclusion
Pesticide residues in spices like dried chilli powder pose a direct threat to the noxious effect of these compounds to humans owing to their direct consumption. Also, processing factors involved in the production of dried chilli powder from fresh chilli harvested concentrates the level of pesticides in them. The proposed method of analysis in this paper is found to be simple, accurate, precise and multi-residue technique for simultaneous determination of six pesticides in dried chilli samples. LOQ of method being 20 ng/g for all the 6 compounds is appropriate in terms of achieving present national as well as international MRL’s. The method can be used for routine testing to assess safety of dried chilli samples with respect to pesticides. Application of developed method of analysis over multiple branded and non-branded loose chilli samples also confirms method’s robustness and authenticity. Also, presence of Triazophos seen in all the six loose chilli samples necessitates quality control requirements for the unorganized spice market [24,25].
|
Marketsamples |
Quinalphos |
Dimethoate |
alpha- Endosulfan |
beta- Endosulfan |
Triazophos |
Endosulfan sulphate |
|
Brand-A |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-B |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-C |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-D |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-E |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-F |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-G |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Brand-H |
ND* |
ND* |
ND* |
ND* |
ND* |
ND* |
|
Loose-1 |
ND* |
ND* |
ND* |
ND* |
0.15 |
ND* |
|
Loose-2 |
ND* |
ND* |
ND* |
ND* |
0.10 |
ND* |
|
Loose-3 |
ND* |
ND* |
ND* |
ND* |
0.08 |
ND* |
|
Loose-4 |
ND* |
ND* |
ND* |
ND* |
0.08 |
ND* |
|
Loose-5 |
ND* |
ND* |
ND* |
ND* |
0.21 |
ND* |
|
Loose-6 |
ND* |
ND* |
ND* |
ND* |
0.10 |
ND* |
|
ND - Not Detected * Method LOQ – 0.02 µg/g |
||||||
Table 6: Results for six pesticides under study in µg/g for 14 market chilli samples analysed as per developed method of analysis
Declarations
Funding: This research received no specific grant from any funding agency.
Conflicts of Interest: The authors declare no conflict of interest.
Author Contributions: Dr. Neha Gupta – writing-original draft preparation, methodology, data curation and validation; Dr. Rakhi Singh – investigation & visualization Dr. Binu Bhat - Conceptualization & supervisio and Dr. Mukul Das – resources and writing- review & editing.
Data Availability Statement: The datasets generated during and/ or analyzed during the current study are available from the corresponding author on reasonable request.
AI Usage Statement: Generative AI tools were not used at all.
Acknowledgement: The authors are grateful to the Analytical Bio-Science division of Shriram Institute for Industrial Research, Delhi for providing all the necessary facility, guidance and support. The manuscript is assigned SRI Manuscript number, SRI- MS#20230130-01.
References
- Geetha, R., & Selvarani, K. (2017). A study of chilli production and export from India. International Journal of Advance Research and Innovative Ideas in Education, 3(2), 205-210.
- Faisal Hussain, F. H., & Muhammad Abid, M. A. (2011). Pests and diseases of chilli crop in Pakistan: a review.
- Kulkarni, A., Jasti, P., Ravindran, S., & Shukla, S. (2017). Risk Assessment of Pesticide Residues in Selected Chilli Samples by Chromatography and Mass Spectrometry. Indian Journal of Public Health Research & Development, 8(4).
- Anugu, Anil. Reddy., Narendra, Reddy. C., Anitha, Kumari.D., Manohar, Rao. A., Narendar, Reddy. S., Srinivasa, Reddy.S. (2022). Processing factor for selective insecticides in chilli.The Pharma Innovation Journal 11(12), 4888-4890.
- Pooja, Sushil, Ahlawat., Ashu. (2023). Determination of Triazophos residues in/on Chilli fruits using Gas Chromatography-tandem Mass Spectrometry. Biological Forum - An International Journal; 15(3), 744-748.
- Kandaswamy, C., Presley, D., Ahammed, T. S., & Anadaram,S. (2022). Improved chromatographic performance in red chilli powder pesticide residue quantitation by retention gap introduction in gas chromatography tandem mass spectrometry. Journal of Food Science and Technology, 59(5), 1692-1703.
- Veiga-del-Bano, J. M., Andreo-Martinez, P., Pérez-Lucas, G., & Navarro, S. (2024). Overview of the evolution and trends of the QuEChERS sample preparation procedure. Reviews of Environmental Contamination and Toxicology, 262(1), 22.
- Srivastava, A., Singh, G. P., Chhabra, A., & Srivastava, P. C. (2022). Determination of pesticides residues and evaluation of decontamination treatments in chili. Indian Journal of Horticulture, 79(2), 249-255.
- Richhariya, N. (2017) Method validation for determination of 23 organophosphorus pesticide residues in chilli by Gas Chromatography. World Journal of Pharmaceutical Research, 1183-1197.
- Sharma, S. S., & Kapadnis, K. H. (1983). Development of Method for Determination of Pesticide Residues from Chilly and Ladyfinger by Using HPLC and GC-MS. Methodology.
- Naik, Harischandra. (2019). Standardization and validation of multi-residue analytical method for simultaneous determination of 34 chemical pesticides in red chilli powder using GC-MS/MS. Project: Establishment of Pesticide Residue and Food Quality Analysis Laboratory at UAS, Raichur.
- Kıvrak, Å?., & Harmandar, M. (2018). Development and validation of modified QuEChERS method coupled with GC-MS/MS for 123 pesticide residues in food. International Journal of Secondary Metabolite, 5(2), 130-139.
- C, Sivanandha. Reddy., S, Kulavardhana. Reddy., Gopi, Reddy., Venkata, Subba. Reddy., (2018). Multi-residue analysis of 13 pesticides in spice using QuEChERS based method and gas chromatography-triple quadrupole mass spectrometry. International Journal of Engineering Science Invention (IJESI), 7(4) Ver. IK.J.
- Balasubramani, K. J., Vijayan, L., & Sunitha, C. S. (2019). Pesticide residue contamination study in chilli in Karnataka by LC-MS/MS and GC-MS/MS analyses. International Advanced Research Journal in Science, Engineering and Technology, 6(11), 48-56.
- Song, L., Han, Y., Yang, J., Qin, Y., Zeng, W., Xu, S., & Pan,C. (2019). Rapid single-step cleanup method for analyzing 47 pesticide residues in pepper, chili peppers and its sauce product by high performance liquid and gas chromatography- tandem mass spectrometry. Food chemistry, 279, 237-245.
- Goon, A., Shinde, R., Ghosh, B., & Banerjee, K. (2020). Application of automated mini–solid-phase extraction cleanup for the analysis of pesticides in complex spice matrixes by GC-MS/MS. Journal of AOAC International, 103(1), 40-45.
- Rutkowska, E., Åozowicka, B., & KaczyÅ?ski, P. (2019). Three approaches to minimize matrix effects in residue analysis of multiclass pesticides in dried complex matrices using gas chromatography tandem mass spectrometry. Food chemistry, 279, 20-29.
- Damale, R. D., Dutta, A., Shaikh, N., Pardeshi, A., Shinde, R., Babu, K. D., ... & Banerjee, K. (2023). Multiresidue analysis of pesticides in four different pomegranate cultivars: Investigating matrix effect variability by GC-MS/MS and LC- MS/MS. Food Chemistry, 407, 135179.
- Pihlström, T., Fernández-Alba, A. R., Amate, C. F., Poulsen,M. E., Hardebusch, B., Anastassiades, M., ... & ORegan, F. (2021). Analytical quality control and method validation procedures for pesticide residues analysis in food and feed SANTE 11312/2021. Sante, 11312(v2).
- Pano-Farias, N. S., Ceballos-Magaña, S. G., Muniz-Valencia, R., & Gonzalez, J. (2017). Validation and assessment of matrix effect and uncertainty of a gas chromatography coupledto mass spectrometry method for pesticides in papaya and avocado samples. Journal of food and drug analysis, 25(3), 501-509.
- Kwon, H., Lehotay, S. J., & Geis-Asteggiante, L. (2012). Variability of matrix effects in liquid and gas chromatography–mass spectrometry analysis of pesticide residues after QuEChERS sample preparation of different food crops. Journal of chromatography A, 1270, 235-245.
- Pano-Farias, N. S., Ceballos-Magaña, S. G., Muniz-Valencia, R., & Gonzalez, J. (2017). Validation and assessment of matrix effect and uncertainty of a gas chromatography coupled to mass spectrometry method for pesticides in papaya and avocado samples. Journal of food and drug analysis, 25(3), 501-509.
- Pilla, Venkateswara. Rao., P, Uma. Mahesh., Avvs, Swamy., N, Srinivas. (2018). Relative Pesticide Residue in Chilli (Capsicum annum L.) Plant Parts at Different Growth Stages. International Journal of Science and Research (IJSR) ISSN: 2319-7064.
- Standard, B. (2008). Foods of plant origin-Determination of pesticide residues using GC-MS and. or LC-MS/MS following Acetonitrile Extraction/Partitioning and Clean up by Dispersive SPE-QuEChERS-method. BS EN 15662 (E).
- Lehotay, S. J. (2007). Determination of pesticide residues in foods by acetonitrile extraction and partitioning with magnesium sulfate: collaborative study. Journal of AOAC International, 90(2), 485-520.



