Research Article - (2018) Volume 3, Issue 1
Uterine Carcinosarcoma: A Single Centre Retrospective Clinical Cohort Analysis
2Clinical Oncology, University College London Hospital UCLH NHS Foundation Trust, London, UK
3Medical Oncology department, The Institute of Cancer Research (ICR), London, UK
4Clinical Oncology Royal Marsden Hospital, London, UK
5Cancer Research UK &UCL Cancer Trials Centre, UCL -University College London, London, UK
6Clinical Oncology, University College London Hospital UCLH NHS Foundation Trust, London, UK
7Medical Oncology,UCL cancer institute, UCL, UK
8Clinical Oncology, University College London Hospital UCLH NHS Foundation Trust, London, UK
Received Date: Nov 29, 2017 / Accepted Date: Dec 05, 2017 / Published Date: Jan 05, 2018
Copyright: ©C Perna, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Carcinosarcomas are rare, heterogeneous tumours with a poor prognosis and no well-defined treatment pathway. Through analysis of a cohort of University College London Hospital (UCLH) patients, we assessed potential known clinicopathological prognostic factors by looking at the association of baseline characteristics with progression free survival (PFS) and overall survival (OS).
Methods: Women with uterine carcinosarcoma treated at UCLH from 2003 to 2014 were retrospectively identified and analysed. Clinicopathological data and treatment history were collected from patient records.
Results: 73 patients were included. 69.9% were FIGO stage I/II, 62.2% had heterologous elements and 60.0% had lymphovascular space invasion (LVSI). Adjuvant chemotherapy and radiotherapy (RT) was received by 48% of patients, 27.4% had RT alone and 8.2%, chemotherapy alone. Median OS was 38.3 months and PFS 25.5 months. Two-year OS and PFS were greater in earlier stage disease (OS; 68.3% vs. 48.9%, PFS; 58.7% vs. 36.1%), homologous component (OS; 72.7% vs. 41.7%, PFS; 60.8% vs. 35.7%) and absence of LVSI (OS; 75.3% vs. 53.7%, PFS; 66.9% vs. 42.5%). Two years OS and PFS for patients who received adjuvant chemotherapy and RT was 64.9% and 58.2% respectively, which was similar to survival rate of those who received RT alone (OS 68.4% and PFS 57.4%). Patients who didn’t receive adjuvant treatment had worse survival (32.7% OS and 11.4% PFS).
Conclusions: Patients receiving RT alone had similar survival to those receiving RT and chemotherapy. Patients who didn’t receive adjuvant treatment had the poorest survival.
Keywords
“Uterine Neoplasm”, “Carcinosarcoma”, “Adjuvant Treatment”, “Outcome”, “Survival”.
Tweetable abstract
Adjuvant treatment for uterine carcinosarcoma showed better survival compare to surgery alone
Introduction
Carcinosarcoma of the uterus is an uncommon variant of uterine cancer, comprising less than 5% of all cases. The worldwide annual incidence is between 0.5-3.3 cases per 100.000 women [1]. It is a biphasic malignancy that contains both carcinomatous (epithelial) and sarcomatous (mesenchymal) elements. The mesenchymal component is described histologically as either homologous (in which the sarcomatous component is made of tissues found in the uterus) or heterologous type (comprised of tissues not found in the uterus, such as cartilage, skeletal muscle and/or bone) [2].
Overall, carcinosarcoma has a worse prognosis stage for stage compared with adenocarcinoma of the endometrium. The risk of haematogenous and lymphatic dissemination (with a propensity for the pelvic and para-aortic nodes) is 40-60% even in early stage (stage I-II) disease [3,4]. Five-year disease-free survival (DFS) is poor (30-50%) and median overall survival (OS) of all stages is about 21 months [5,6].
In contrast,endometrioid adenocarcinoma is confined to the uterus at diagnosis in 70% of the patients, and the five year OS is 75% for early stage disease, as observed in the ASTEC trial [7].
In addition to histological subtype, the most significant prognostic factor at diagnosis is stage [8]. Other prognostic factors include grade of the epithelial component, presence of lymphovascular space invasion (LVSI), and presence of postoperative residual tumour [6]. In 2011 a Canadian review attempted to evaluate molecular markers as potential therapeutic and prognostic targets in this rare tumour sub¬group. The identification of definitive biomarkers in this population is hampered by rarity of this neoplasm, small sample size of cases and tumour heterogeneity [9].
Immunohisto chemistry shows that Transforming Growth Factor-β (TGF-β), p16, p53, Estrogen Receptor β (ERβ) and Vascular Endothelial Growth factor (VEGF) are commonly over expressed whereas hormone receptors are infrequently expressed [8]. European society of medical oncology (ESMO) guidelines recommends treating uterine carcinosarcomas as high-grade carcinomas [8]. Despite the high risk of relapse, there is no clear consensus regarding adjuvant therapy.
We carried out a retrospective review of all patients with carcinosarcoma of the endometrium treated at University College London Hospital (UCLH) between 2003 and 2014. The review focussed on clinical outcomes, progression free survival (PFS) and OS, following treatment and the influence of factors such as the presence of heterologous elements and LVSI.
Materials and Methods
Two doctors independently retrospectively reviewed all patients with uterine carcinosarcoma treated at UCLH from January 2003 to August 2014 inclusive. To ensure capture of all patients, we created a patient list from the pathology department database. We then acquired clinical data from patient records and, if necessary, general practitioners. The only exclusion criterion for this analysis was metastatic disease at diagnosis.
The data set included age, tumour size, FIGO stage, presence of heterologous element and adjuvant treatment received as well as relapse patterns and subsequent treatment.
Kaplan-Meier Survival curves were generated using Stata version 14.1; the differences in survival were estimated using the long-rank test. A p-value <0.05 was used to determine statistical significance. OS and PFS rates at 2 years were estimated according to adjuvant treatment received and prognostic factors.
Survival times were measured from the date of surgery until relapse or death for PFS, or death for OS. Patients who did not experience any event were censored at the date last seen.
A literature search was conducted using Pubmed database selecting “uterine neoplasm” and “carcinosarcoma” as primary keyword plus “adjuvant treatment”, “outcome” and “survival”.
Results
Eighty-nine patients with uterine carcinosarcoma treated between 2003 and 2014 were identified on the pathology department database. Nine were excluded due to metastatic disease at presentation, 3 died before any treatment, 2 declined regular follow up, 1 declined treatment and 1 transferred care to an alternative hospital. The records of 73 patients were therefore thoroughly reviewed.
Mean age at diagnosis was 69 (range 50-90). The majority, 51 (69.8%) were early stage with 46 (63.0%) FIGO stage I and 5 (6.8%) FIGO stage II. Twenty-two (30.1%) were FIGO stage III. All patients underwent surgery as their primary treatment: total abdominal or laparoscopic hysterectomy with bilateral salpingo-oophorectomy. In addition 17 (23.3%) patients underwent a lymphadenectomy.
Pathological data about heterologous differentiation was available for 37 patients, 23 (62.2%) with heterologous element, and 14 (37.8%) without. The presence or absence of LVSI was documented for 68 patients of whom 41 (60.3%) had LVSI. Thirty-five (48.0%) patients received adjuvant chemotherapy and RT, 20 (27.4%) received adjuvant RT alone and 6 (8.2%) received adjuvant chemotherapy alone. Twelve (16.4%) declined adjuvant treatment (Fig 1).
Of the RT patients, 41 (74.5%) received external beam radiotherapy (EBRT) and vaginal vault brachytherapy (VVB), 6 (10.9%) had EBRT only and 8 (14.5%) had VVB alone. 39 of 41 (95.1%) patients who received chemotherapy were treated with carboplatin and paclitaxel; of the remaining 2 patients, one received carboplatin and caelyx and one received carboplatin as single agent. EBRT was delivered to a planned volume, using a 4-field box technique to treat the whole pelvis to a total dose of 45 Gray in 25 fractions over 5 weeks. The VVB boost was delivered using Iridium 192 high dose rate (HDR) brachytherapy to a total dose of 12Gray in 2 fractions (12Gy/2#) for 38 patients. Alternative VVB doses delivered were 16Gy/2# (1 patient), 13Gy/2# (1 patient) and 7Gy/1# (1 patient). 8 received only VVB to a total dose of 24Gy in 4#.
Figure 1
The median OS was 38.9 months (range: 1 day - 89.7 months) and the median was 25.5 months. As expected 2 year OS and PFS rates were lower in advanced stage; 68.3% and 58.7% respectively for stage I and II, versus 48.9% and 36.1% in stage III and IV (Fig.2).
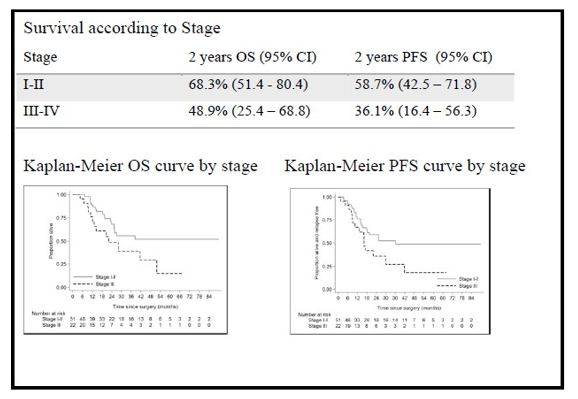
Figure 2
The presence of heterologous component was associated with a lower OS and PFS at 2 years (41.7% and 35.7% respectively) compared to those patients with none (72.7% and 60.8%). Although there is a suggestion that these patients had an inferior PFS and OS, this wasn’t statistically significant (p=0.44 for OS and p=0.23 for PFS), perhaps due to the small patient numbers (Fig.3).
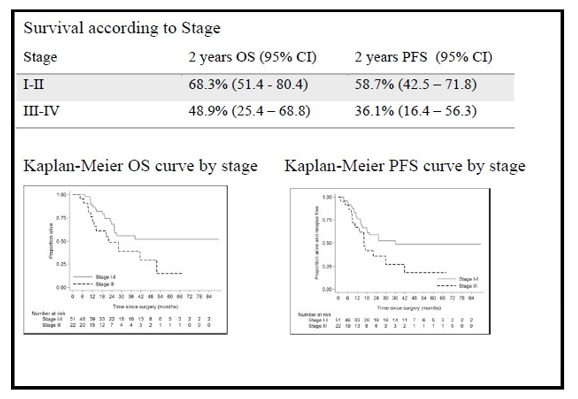
Two year OS and PFS in patients with LVSI were lower than patients without LVSI; OS 53.7% compared to 75.3% and PFS 42.5% compared to 66.9%. This was statistically significant (p=0.009 for PFS and p=0.005 for OS) and therefore can be confirmed as a prognostic factor (Fig. 4).
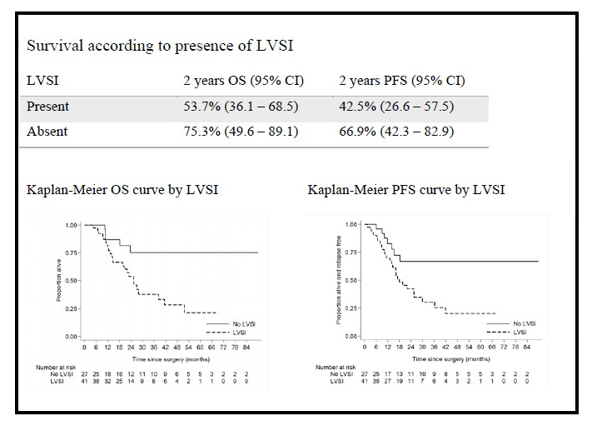
Figure 4
The 2 year OS and PFS for patients who received adjuvant chemotherapy and RT were 64.9% and 58.2% respectively. The 2 year OS and PFS rates were 32.7% and 11.4% respectively in those patients who declined adjuvant treatment.
Patients receiving chemotherapy alone demonstrated OS of 80.0% and PFS of 60%. The 21 patients, who received RT alone, had OS and PFS respectively of 68.4% and 57.4%, very similar to rates seen in patients who received both chemotherapy and RT (Fig 5).
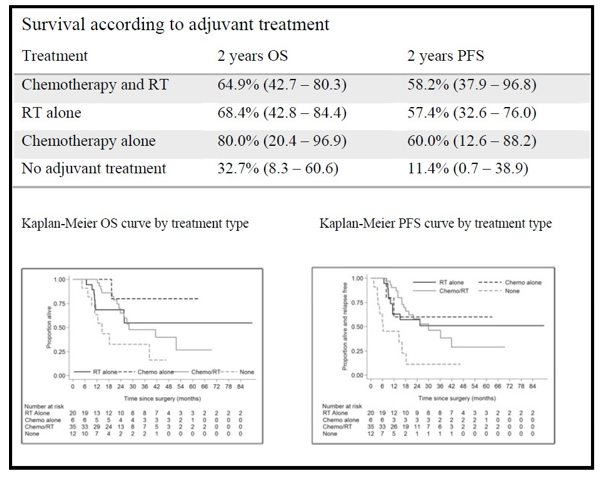
Figure 5
Lymphadenectomy in our study (not routinely performed) showed no survival difference (Fig. 6).
Figure 6
Discussion
Carcinosarcoma of the uterus is an aggressive high-risk carcinoma with a poor prognosis and a high risk of haematogenous and lymphatic dissemination [8]. The reported 5 year survival of patients with carcinosarcoma is 30-50% [10].
Our series shows a median PFS of 25.5 months and median OS of 38.9 months, in line with data previously reported. Two year OS for all the patients was 62.3% (48.6% - 73.3%) and PFS 51.3% (38.3% - 63.0%) which is also similar to that in literature [11].
A retrospective analysis conducted by De Jong in 2011, has shown that the behaviour of these tumours is determined by the epithelial component, and in the respective series of cases, the epithelial component was responsible for metastatic disease in 72% [12]. Epithelial elements invade the lymphovascular space and metastasize, whereas the mesenchymal component has limited metastatic potential [13,14]. In our series heterologous component was seen in 23 (62.2%) out the 37 patients with available data. Although its presence was associated with a poorer prognosis with 2 year OS of 41.7% and PFS of 35.7% compared to 72.7% and 60.8% in patients without it, this did not reach statistical significance maybe due to the small number of patients analysed. (Fig. 3-4)
All patients in our series underwent total hysterectomy and bilateral salpingo-oophorectomy as primary treatment with only 17 patients also undergoing lymphadenectomy. According to Bengt Sorbe lymphadenopathy has no impact on overall survival and this was supported by our data [15]. (Fig. 9-10)
LVSI was statistically significant as a poor prognostic factor; patient with positive LVSI had lower 2 year OS (53.7% vs. 75.3%) and PFS (42.5% vs. 66.9%). We also confirmed, as expected, that prognosis was better for those with early compared with advanced stage disease.
In endometrial cancer, randomised controlled trials have demonstrated a reduction in local recurrence rates with adjuvant post-operative radiotherapy (PORT) but without definite survival benefit [16,17].The role of adjuvant chemotherapy in high-risk early stage disease remains to be defined and the results of the PORTEC 3 trial are eagerly awaited. Data from our institution suggests that for patients with stage III endometrial cancer survival is improved in those treated with sequential chemotherapy and RT compared with either modality alone.
Patients with stage III disease undergoing chemotherapy and RT in our carcinosarcoma series had OS of 59.8% (28.5-81.0) and PFS of 46.8% (19.6-70.2), which is in line with our previous published data for patients with stage III endometrioid endometrial cancer [18].
Similarly, no studies have clearly defined the role of adjuvant chemotherapy for carcinosarcoma. Carboplatin and paclitaxel is usually administered as for other high-risk endometrial carcinomas but the optimal regimen is unclear [5]. For patients receiving chemotherapy in our analysis, 95% were treated with Carboplatin and Paclitaxel.
A Cochrane systematic review analysed three randomized trials comparing adjuvant chemotherapy to RT in women with FIGO stage III and IV endometrial cancer. Two trials (GOG 122 and Maggi 2006) of 620 patients, compared adjuvant chemotherapy with adjuvant RT; they reported a longer OS and PFS in the chemotherapy group [16,19]. A third trial (Susumo 2008) of 552 patients compared two chemotherapy regimens in patients who all underwent adjuvant RT. Despite no clear differences in PFS between the two chemotherapy regimens, a benefit of adjuvant sequential treatment (chemotherapy and RT) was observed [20].
In our data the 2 years PFS in chemotherapy only was 60% and 58.2% in chemotherapy and RT group. The OS was 80.0% in chemotherapy only (only 6 patients) and 64.9% in chemotherapy and RT group which is in keeping with published literature. Patients who did not receive adjuvant treatment, had a 2 year PFS of 11.4% and OS of 32.7%.
The 21 patients, who received RT alone, had OS and PFS of 68.4% and 57.4% respectively. This is almost identical to the chemotherapy and RT group survival rates. However, differences in the patient characteristics may have influenced the outcome as 85.0% in RT group had stage I/II disease compared with 57.1% in the chemotherapy and RT group. Similarly, LVSI was present in 68.6% of patients in the chemotherapy and RT group compared to 41.2% of patients in the RT alone group.
Accepting the limitations associated with small numbers and a retrospective review, we might speculate that those patients with LVSI and advanced stage benefit from both chemotherapy and RT. However RT alone may be sufficient for those with earlier stage disease and no LVSI. These findings merit further investigation in a randomised trial.
We can, however, identify a survival benefit of adjuvant treatment in endometrial carcinosarcoma that even within the limits of this retrospective study is of interest.
Conclusion
Our results are in line with current published literature and further support the need for randomised prospective studies to confirm the role of adjuvant treatments in this uncommon cancer. The most striking finding in this review was the difference in outcome between those who received any adjuvant therapy and those who had no adjuvant therapy. There was little difference in outcome according to type of adjuvant therapy although the numbers in the chemotherapy only group were small and baseline factors may have influenced the choice of therapy.
Acknowledgement
The authors would like to acknowledge support received from the UCL hospitals Biomedical Research Centre. Rebecca Kristeleit is supported by the UCH/UCL Biomedical Research Centre
References
- SE Brooks, M Zhan, T Cote, CR. Baquet (2004) Surveillance, Epidemiology, and End Results analysis of 2677 cases of uterine sarcoma. Gynecol. Oncol 93: 204-208.
- WG McCluggage (2002) Uterine carcinosarcomas (malignant mixed Mulleriantumors) are metaplastic carcinomas. Int. J. Gynecol. Cancer 12: 687-690.
- K Galaal, M Al Moundhri, A Bryant, AD Lopes, TA Lawrie (2014) Adjuvant chemotherapy for advanced endometrial cancer, Cochrane Database Syst. Rev 5.
- FJ Major, JA Blessing, SG Silverberg, CP Morrow, WT Creasman, et al. (1993) Prognostic factors in early-stage uterine sarcoma. A Gynecologic Oncology Group study. Cancer 71: 1702-1709.
- MA Powell, VL Filiaci, PG Rose, RS Mannel, P Hanjani, et al. (2010) Ueland, Phase II evaluation of paclitaxel and carboplatin in the treatment of carcinosarcoma of the uterus: a Gynecologic Oncology Group study. J. Clin. Oncol 28: 2727-2731.
- A Gadducci, E Sartori, F Landoni, P Zola, T Maggino, et al. (2002) The prognostic relevance of histological type in uterine sarcomas: A cooperation task force (CTF) multivariate analysis of 249 cases. Eur. J. Gynaecol. Oncol 23: 295-299.
- DPJ Barton, R Naik, J Herod (2009) Efficacy of systematic pelvic lymphadenectomy in endometrial cancer (MRC ASTEC Trial) A Randomized Study, Int. J. Gynecol. Cancer 8: 1465
- N Colombo, C Creutzberg, F Amant, T Bosse, A Gonzalez Martin, et al. (2015) ESMO-ESGO-ESTRO Consensus Conference on Endometrial Cancer: diagnosis, treatment and follow-up, Radiother. Oncol 117: 559-581.
- R Kanthan, JLSenger (2011) Uterine carcinosarcomas (malignant mixed müllerian tumours): a review with special emphasis on the controversies in management. Obstet. Gynecol. Int 2011: 13.
- M Callister, LM Ramondetta, A Jhingran, TW Burke, PJ Eifel (2004) Malignant mixed Mulleriantumors of the uterus: analysis of patterns of failure, prognostic factors, and treatment outcome. Int. J. Radiat. Oncol Biol. Phys 58: 786-796.
- A Otsuki, T Otsuki, H Tokunaga, H Niikura, S Nagase, et al. (2014) Evaluation of postoperative chemotherapy in patients with uterine carcinosarcoma: a retrospective survey of the Tohoku Gynecologic Cancer Unit. Int. J. Clin. Oncol 20: 574-578.
- RA de Jong, HW Nijman, TF Wijbrandi, AK Reyners, HM Boezen, et al. (2011) Molecular markers and clinical behavior of uterine carcinosarcomas: focus on the epithelial tumor component. Mod. Pathol 24: 1368-1379.
- P Bitterman, B Chun, RJ Kurman (1990) The significance of epithelial differentiation in mixed mesodermal tumors of the uterus. A clinic pathologic and immune histochemical study. Am. J. Surg. Pathol 14: 317-328.
- SG Silverberg, FJ Major, JA Blessing, B Fetter, FB Askin (1990) Carcinosarcoma (malignant mixed mesodermal tumor) of the uterus. A gynecologic oncology group pathologic study of 203 cases. Int. J. Gynecol. Pathol 9: 1-19.
- B Sorbe, G Paulsson, S Andersson, G Steineck (2013) A population-based series of uterine carcinosarcomas with longterm follow-up. ActaOncol. (Madr) 52: 1-8.
- M Randall, VL Filiaci, H Muss, NM Spirtos, RS Mannel (2006) GOG 122: Randomized phase III trial of whole-abdominal irradiation versus doxorubicin and cisplatin chemotherapy in advanced endometrial carcinoma: A gynecologic oncology group study. JCO 24: 36-44.
- CL Creutzberg, WL van Putten, PC Koper, ML Lybeert, JJ Jobsen, et al. (2000) Surgery and postoperative radiotherapy versus surgery alone for patients with stage-1 endometrial carcinoma: multicentre randomized trial. PORTEC Study Group. Post Operative Radiation Therapy in Endometrial Carcinoma. Lancet (London, England) 355: 1404 -1411.
- S Kuku, M Williams, M McCormack (2013) Adjuvant therapy in stage III endometrial cancer: treatment outcomes and survival. a single-institution retrospective study. Int. J. Gynecol. Cancer 23: 1056 -1064.
- R Maggi, A Lissoni, F Spina, M Melpignano, P Zola (2006) Adjuvant chemotherapy vs. radiotherapy in high-risk endometrial carcinoma: results of a randomized trial. Br. J. Cancer 95: 266-271.
- N Susumu, S Sagae, Y Udagawa, K Niwa, H Kuramoto (2008) Randomized phase III trial of pelvic radiotherapy versus cisplatin-based combined chemotherapy in patients with intermediate and high-risk endometrial cancer: A Japanese Gynecologic Oncology Group study, Gynecol. Oncol 108: 226-233.


