Research Article - (2019) Volume 4, Issue 3
Treatment of Acute Iliofemoral Deep Vein Thrombosis: Does Catheter Directed Thrombolytic Therapy Change Outcomes
2Department of General & Vascular Surgery, Benha University, Egypt
3Department of Vascular Surgery, Benha Insurance Hospitals, Egypt
4Department of Vascular Surgery, Zagazig University, Egypt
5Department of Vascular Surgery, AlBaha university, AlBaha, Saudi Arabia
6Department of General Surgery, Benha Teaching Hospitals, Egypt
Received Date: Jun 28, 2019 / Accepted Date: Jul 09, 2019 / Published Date: Jul 31, 2019
Copyright: ©Ayman M Fakhry, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Iliofemoral deep vein thrombosis (DVT) is associated with severe post-thrombotic morbidity when treated with anticoagulation alone. Catheter- directed thrombolysis (CDT) allows early removal of thrombus and reduces valvular reflux and Post-thrombotic Syndrome (PTS).
Patients and methods: This prospective randomized multi-center controlled two- arm blind study was conducted in 6 centers on 252 patients with iliofemoral DVT. Patients were randomly allocated by using simple random allocation cards method into two groups; Group (A): CDT followed by oral anticoagulation (N=126 (50%)), Group (B): Standard DVT therapy (N=126 (50%)). Follow-up was for 24 months.
Results: Patients of group (A) significantly complained less pain at 10 & 30 days (P-Value: 0.02 & 0.04 respectively). Also there was significant decrease in leg circumference in group (A) at 10 & 30 days (P-Value: 0.001 & 0.03 respectively). Patency of iliac vein segment was significantly higher in group (A) during the 24 months follow up (P-Value <0.001 (HS)). Patients in group (A) developed less PTS at six months, at one year and at two years (P-Value: 0.024, 0.017 and 0.035 respectively). Better Quality of life was observed in group (A) (P-Value: 0.003).
Conclusion: Addition of catheter-directed thrombolysis in the treatment of acute iliofemoral DVT; was safe and tolerated by most of the patients with better effect to reduce leg pain & circumference. It was considered a protecting weapon to prevent post-thrombotic syndrome and so improve quality of life and was related to achievement of higher iliac vein patency and less reflux.
Keywords
Iliofemoral venous thrombosis, Catheter-directed thrombolysis; Standard anticoagulation, Post-thrombotic Syndrome.
Introduction
Acute deep vein thrombosis (DVT) is ranging from asymptomatic calf vein thrombosis to painful, blue, swollen limb of phlegmasia cerulea dolens due to extensive multisegment thrombosis including iliofemoral venous segment [1].
DVT of the lower extremity is the cause of both pulmonary embolism and post-thrombotic syndrome. Therapeutic goals include, diminishing the severity and duration of lower extremity symptoms, preventing pulmonary embolism and preventing post-thrombotic syndrome (PTS) sequelae [2,3].
PTS is a debilitating condition that diminishes quality of life and often worsens over time. PTS is due to ambulatory venous hypertension, which is defined as elevated venous pressure during exercise that may lead to swelling, pigmentation, lipodermatosclerosis and microcirculatory changes leading to dermal breakdown [1-4].
Patients with PTS have a poorer quality of life than patients with chronic lung disease, diabetes, or arthritis. In addition, disease- specific quality of life worsened significantly with increasing severity of PTS, and those with severe form of PTS have quality of life comparable to that of patients with congestive heart failure or cancer [5].
Anatomic components causing ambulatory venous hypertension are venous valvular incompetence and luminal obstruction. The most severe postthrombotic morbidity is associated with the highest venous pressures, which occur in patients with both valvular incompetence and luminal venous obstruction [1-5]. The most severe post-thrombotic morbidity is found in patients who have had thrombosis of their iliofemoral venous segment. Additionally, the risk of recurrent venous thromboembolism for patients with iliofemoral DVT is significantly higher than patients with infrainguinal DVT [6-8].
Standard anticoagulant therapy aims at prevention of pulmonary embolism (PE) and recurrent DVT. However, over 50% of patients with proximal DVT may suffer post-thrombotic syndrome (PTS) within the first 2 years. Severe PTS occurs in 5–10% of all symptomatic DVTs, and may lead to venous ulceration [8].
There are many strategies for early thrombus removal, one of these strategies is operative venous thrombectomy, but it is not adopted by many vascular surgeons except for patients who are not candidates for CDT or CDT is not available [1-9].
Systemic thrombolysis has limitations of being associated with higher risks of bleeding and failure in patients with extensive occlusive venous thrombosis. CDT has emerged as an alternative endovenous treatment for DVT, and is now the preferred method of management of iliofemoral DVT if thrombus removal is desired [9,10].
CDT is the favored route of administration in recent years for preserving venous function, and patient selection is important to reduce the risk of complications [11-13].
The basic mechanism of thrombolysis is activation of fibrin- bound plasminogen and production of plasmin. Local delivery of plasminogen activator is more effective and potentially safer than systemic infusion. Additionally, intrathrombus delivery protects plasminogen activators from circulating plasminogen activator inhibitor and protects active enzyme plasmin from neutralization by circulating antiplasmin. This neutralization of circulating plasminogen is so effective that the half-life of Plasmin in the systemic circulation is only a fraction of a second [1].
Patients and Methods
After approval by local ethical committee of Benha and Zagazig universities, Military Academy of Alexandria , Benha Insurance Hospitals, Benha Teaching Hospitals, Egypt and AlBaha university, Saudi Arabia and obtaining written fully informed patients consent. Patients with iliofemoral deep vein thrombosis were enrolled at these 6 clinical centers from August 2015 till April 2019; the enrollment period was 19 months and the patient follow-up period was 24 months.
This prospective multi-center randomized controlled two-arm blind interventional study was conducted on 252 patients with iliofemoral DVT. Patients were randomly allocated by using simple random allocation method, where 252 cards (126 were signed as group (A) and other 126 were signed as group (B)) were prepared by the principal investigator and were put in closed envelops and mixed together. Each patient has chosen envelop after he had approved for participation. Double blind technique was applied where patients and care providers were blind about to which group the patients were allocated; the randomization sequence was generated by an independent statistician. MedCalc software version 16.1(© 1993- 2016 MedCalc Software) was used to calculate the required sample size. Level of significance (type I error) =0.05, Type II error (1-level of power) =0.1. Null hypothesis percentage= 50%; so, the least sample size =54 in each group.
Inclusion criteria
Patients with iliofemoral DVT with age 20-60 years old with the onset of symptoms within fourteen days with open distal part of the popliteal vein verified by duplex scan.
Exclusion criteria
1. Patients with bleeding diathesis; with hemoglobin <10 g/dL and/or thrombocytopenia (platelets <100000/mm3)
2. Renal impairment (estimated creatinine clearance <30 mL/min)
3. Acute DVT during pregnancy or within 7 days postpartum
4. Acute DVT within 14 days following major surgery or trauma
5. History of recent subarachnoid, intracerebral or GIT bleeding
6. Drug abuse or mental disease that could interfere with treatment and follow-up
7. Recurrent iliofemoral DVT
8. Current malignant disease.
Group (A) of CDT
In prone position; ten milliliters of 2% lignocaine was injected subcutaneously and under ultrasound guidance; a 19 gauge 10 cm needle was used to puncture the popliteal vein followed by advancing a 6-french standard sheath. Venography was then performed. A 0.035 inch 260 cm j-tip hydrophilic guide wire (standard type of ZIP wire (Boston Scientific, Chaska, MN, U.S.A.) was used to cross the thrombus to a healthy part of the IVC. When difficulty was encountered during crossing of the lesion, stiff type of ZIP wire 0.035 inch 270 cm (Boston Scientific, Chaska, MN, U.S.A.) was used.
After adequate flushing with heparinized saline, a 5 french 135cm length Uni Fuse catheter (Angiodynamics, Inc., Queensbury, NY, U.S.A.); with an infusion segment length of 40 or 50cm according the length affected, was advanced over the guidewire. The guidewire was exchanged for the occluding wire which was intended to occlude the end hole of the catheter, therefore, the infused thrombolytic agent exited the catheter only through its side holes (inside the thrombus). Correct placement of the catheter and occluding wire were checked radiographically.
Then; thrombolytic therapy i.e. Alteplase (Actilyse®; Boehringer- Ingelheim, Ingelheim am Rhein, Germany)); 50mg) (2 bottles, one powder and another solution 50ml: 10ml loading dose was injected followed by flush saline then patient was transferred to intermediate care unit where 0.01-0.05mg/kg/hour maintenance dose (1ml/hour till 40 hours) by the squirt pump with heparin ampoule IV every 4-6 hours or continuous infusion 500iu/hour using a syringe pump; then imaging and managing according findings.
A venogram was then performed to determine the need for venoplasty, and/or venous stents for culprit lesions; > 50% narrowing. At the end of the procedure, infusion catheter and sheath were removed and followed by manual compression for 20 min then crepe bandage was applied for further two hours. Low molecular weight heparin (enoxaparin 1 mg/kg/12 hours) was started 2 hours after removal of the sheath. Warfarin was started the 2nd day post-procedure. Anticoagulation was then continued as mentioned in group (B). Patients were discharged home whenever there was neither hematoma nor any suspicion of concealed hemorrhage and after initiation of warfarin therapy. Patients were advised to wear fit-sized above knee, 30-40mm Hg, graduated elastic compression stockings at the 10-day follow-up visit for 2 years (BELSANA medical) [14].
Thrombolysis grade was assessed by independent readers who scored venograms obtained before and after the procedure, using the proximal-vein components of the Marder score. The modified Marder score ranges from 0 to 24, with 0 representing no thrombus and 24 representing complete thrombosis [15].
Group (B) of Standard anticoagulation
Patients were prescribed anticoagulant therapy in accordance to local routines based on international guidelines using the low molecular weight heparin (LMWH) enoxaparin (clexane®; Sanofi, France) in a dose of 1 mg/kg/12 hours, for at least five days. Oral warfarin (Marevan®; GlaxoSmithKline, UK) was started the day of randomization, at a daily dose of 5 mg. The dose was modified according to the patient’s international normalized ratio (INR) with a target INR of 2.0 to 3.0. LMWH was stopped when the patient’s INR is 2.0 or above for at least 24 hours. Warfarin was continued for at least 3 months. Patients were advised to wear fit- sized above knee, 30-40mm Hg, graduated elastic compression stockings at the 10th day visit for 2 years (BELSANA medical) [14].
Follow-up
Bleeding complications were recorded based on World Health Organization bleeding scale that includes; four grades; grade (1) and (2) doesn’t necessitate blood transfusion i.e. minor bleeding but grade (3): Any bleeding requiring RBC transfusion over routine transfusion needs i.e. Any bleeding that led to drop of more than 2 gm/dl of hemoglobin and grade (4): Any bleeding associated with severe hemodynamic instability (hypotension; >50mm/Hg fall or >50% decrease in either systolic or diastolic blood pressure, with associated tachycardia (heart rate increase of >20% for 20 minutes) and requiring RBC transfusion over routine transfusion needs or Fatal bleeding from any source.
Patients returned for follow-up monthly for 24 months postrandomization. At each visit, the patient was evaluated for the signs and symptoms of DVT and/or PE and PTS. Because PTS varies in its clinical manifestations, its presence and severity was evaluated in complementary ways. Villalta score was recorded to assess for development of PTS. The occurrence of PTS was counted if the Villalta score at that visit was 5 or higher. This score consists of five venous symptoms (pain, paraesthesia, pruritis, cramps and heaviness) and six physical signs (pretibial edema, skin induration, redness, hyperpigmentation, venous ectasia and calf tenderness), which are rated on a four-point scale, whereas 0= none; 1= mild; 2= moderate; and 3= severe. Points are summed to produce score (0-33). Patients were classified as having mild PTS; when Villalta score was 5-9, moderate PTS; when Villalta score was 10-14 and severe PTS, while a score ≥15 or presence of venous ulcer [16].
All patients were subjected to a venous duplex examination of the treated venous segments at 1, 6, 12 and 24 months postprocedure and at any time a suspicion for PTS and DVT was raised. Recurrent DVT was considered if previously compressible vein segment became non-compressible or there was >5- mm increase in diameter of the thrombus on ultrasound evaluation. PE was suspected when symptoms of dyspnea, chest pain, cough, fever, hemoptysis, and/ or syncope has occurred and diagnosis was supported by multislice CT pulmonary angiography or/and echocardiography. Also venous duplex was used for detection of post-thrombotic iliofemoral wall thickening and residual thrombi and to evaluate iliofemoral venous flow and reflux. Venous reflux was defined as reversal of velocity curve lasting >0.5 second after distal compression while patient in standing position.
Patient-reported health-related quality of life at admission and 24 months was assessed with the use of the venous disease-specific Venous Insufficiency Epidemiological and Economic Study Quality of Life (VEINES-QOL) measure; this measure includes symptom- specific score that ranges from 0 to 100, with higher scores indicating better quality of life. A difference of 3 to 4 points is considered clinically meaningful [17]. Leg pain and leg swelling at 1st day, 10 days, and 30 days were assessed; Pain was evaluated using a visual analog score (VAS); Patients in both groups ranked the level of pain from 0 (no pain) to 10 (very severe pain). But leg swelling was evaluated by measuring calf circumference at 10 cm below the tibial tuberosity.
Statistical Analysis
Collected data were tabulated and analyzed using SPSS version 16 soft ware (SpssInc, Chicago, ILL Company) and Microstat W software (India, CNET Download.com). Categorical data were presented as number and percentages, using Chi square (X2) test or Z test of 2 proportions (ZPROP.) for their analysis. Continuous data were expressed as mean ± standard deviation, Data were tested for normality using Shapiro-Wilks test, assuming normality at P >0.05. Differences between groups were tested using student “t” for normally distributed variables or Mann Whitney U (ZMWU) test for non parametric ones. Accepted level of significance in this work was stated at 0.05 (P ≤0.05 was considered significant) [18]. Sample size was calculated with power of 90% with marginal error 5%.
The companies mentioned in this study played no role in the design or conduct of the trial or in the analysis or reporting of the data. All scores in this study were recorded by an independent investigators and statistician
<img src="https://www.opastpublishers.com/scholarly-images/1024-69ae45aeb3966-treatment-of-acute-iliofemoral-deep-vein-thrombosis-does-cat.png" width="500" height="300">
Figure 1: Duplex guided needle insertion
<img src="https://www.opastpublishers.com/scholarly-images/1024-69ae4615166d5-treatment-of-acute-iliofemoral-deep-vein-thrombosis-does-cat.png" width="500" height="600">
Results
This prospective randomized controlled two-arm blind interventional study was conducted on 252 patients (285 limbs) with acute lower limb proximal deep vein thrombosis; 219 patients with unilateral limb and 33 patients with bilateral limbs. During follow-up period of about 24 months; four patients died due to unrelated cause. Two patients of group (A) were with bilateral DVT and two patients of group (B) with unilateral DVT. Eight patients were lost during follow up; one of them with bilateral DVT. These 12 patients that died and lost during follow up were excluded from the per-protocol analysis. So at completion of 24 months follow-up data, clinical status was available for 240/252 (95.2%) patients (270/285 limbs (94.7%)); 117 patients (132 limbs) were assigned to catheter- directed thrombolysis; group (A) and 123 patients (138 limbs) were assigned to the control group; group (B). There was no statistical difference between both groups in demographic data. Tab. (1).
Table 1: Patients’ demographic and clinical data:
|
Variables |
|
Group A N=117 (48.75%) |
Group B N=123 (51.25%) |
Test of significance |
P-Value |
|
Age (years) |
|
42.6±3.8 |
44.2±4.5 |
St. “t” =1.71 |
0.09 (NS) |
|
Gender |
Male |
27 (23.1%) |
33 (26.8%) |
X2=0.017 |
0.89 (NS) |
|
Female |
90 (76.9%) |
90 (73.2%) |
|
|
|
|
Duration of symptoms (days) |
4.8±2.01 |
3.5±1.39 |
ZMWU= 1.09 |
0.27 (NS) |
|
|
Treated limbs |
Left-sided |
96 (82.1%) |
87 (70.7) |
X2=2.88 |
0.23 (NS) |
|
Right-sided |
6 (5.1%) |
21 (17.1%) |
|||
|
Bilateral |
15 (12.8%) |
15 (12.2%) |
|||
Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
None of the patients in group (A) experienced allergic reactions nor contrast induced nephropathy (CIN). CDT was performed at a median of 1 day after randomization. CDT patients passed uneventful course without procedure related one month mortality. Mean degree of thrombus removal was 76.9% (mean preprocedure Marder score, 13.6; mean postprocedure score, 1.9; P <0.001).
Among patients who had complete lysis; 90 patients (76.9%); culprit stenotic lesions in proximal common iliac vein were encountered in 18 patients. Fifteen of these lesions were located on the left side and the other three were located on the right side. Balloon angioplasty was performed in all the 18 patients (12, 14 mm×60 mm Advance® ATB balloon (Cook Medical, Inc, USA)). Stenting of the lesion was performed in 15 patients (two in the right side and thirteen in the left side) with self-expanding wall stents (wallstent endoprothesis; Boston Scientific, USA). Left thirteen stents varied from 14 to 16mm diameter and their length varied from 70 mm (in 3 patients) to 90 mm (in the remaining 10 patients). But right two stents were of 14mm diameter & 60mm length.
IVC filter insertion was not routinely used during CDT. Ten cases (4.2%) were considered for filter insertion. This was because eight patients developed PE; five patients in group (A) and three patients in group (B) and the other two filters were used for fear of embolization in patients having free floating thrombus tail extending to the IVC; one in each group. Celect filter was used (Cook Medical Inc., USA) in 4 patients and B/BRAUN Filter (Vena Tech LP®, Vena Cava Filter system, France) in the remaining six patients. The choice of which filter was based on availability of the filter.
The five patients (4.3%) in the CDT group suffered pulmonary embolism. Three of them experienced sudden onset of retrosternal chest pain and dyspnea during passage of the wire through the thrombus. An IVC filter was immediately inserted through a right jugular venous puncture or contralateral femoral vein puncture followed by initiation of catheter-directed thrombolytic drug instillation in the iliofemoral segment. These 3 patients were then transferred to ICU for oxygen mask and an urgent CT pulmonary angiography revealed presence of subsegmental pulmonary embolism. The patients’ symptoms improved at ICU and the second session of the procedure was accomplished as scheduled. The other two patients experienced tachycardia, tachypnea and hypoxia, during infusion of thrombolytic agent in the intermediate care unit. Urgent CT pulmonary angiography revealed presence of submassive pulmonary embolism; IVC filter was immediately inserted through contralateral femoral venous puncture and patient symptoms improved on oxygen mask and second session of the procedure was accomplished as scheduled. Anticoagulant therapy was continued.
In the group (B); three patients (2.4%) had pulmonary embolism. These 3 patients had hemoptysis at day one postrandomization, CT pulmonary angiography revealed the presence of submassive PE; An IVC filter was immediately inserted through contralateral femoral venous puncture and These patients were transferred to ICU; Having no hemodynamic compromise. Anticoagulant therapy was continued with clinical improvement.
Daily monitoring of hemoglobin, INR and platelet count did not reveal or indicate occult bleeding in any of the patients undergoing CDT or led to modification of the therapy. A total of 9 (7.7%) clinically relevant non major bleeding complications were reported in the CDT group. There were four cases (3.4%) of groin hematoma (contralateral; related to puncture of IVC filter placement), four cases (0.86%) of popliteal fossa hematoma (related to the puncture sites) and a single case (0.86%) of mild hematuria. All were managed conservatively; Micronized purified flavonoid fraction (MPFF, 450 mg diosmin plus 50 mg hesperidin-Daflon 500 mg, Servier, France) was prescribed and none had necessitated blood transfusion.
But in group (B); six patients developed bleeding; of which 4 (3.25%) minor bleeding complications were reported and remaining 2 (1.64%) patients developed major bleeding. Patients with minor bleeding were managed conservatively; two of them had gynecological bleeding (menorrhagia without hemoglobin drop, they were prescribed Micronized purified flavonoid fraction (MPFF, 450 mg diosmin plus 50 mg hesperidin-Daflon 500 mg, Servier, France) and advised to use it regularly during their menses) and the other two cases developed gingival bleeding & epistaxis. Warfarin was resumed thereafter, with no further bleeding episodes during the follow-up period. The two patients (1.63%) experienced major bleeding complications; one patient (0.81%) had major upper GI bleeding (2 months following DVT, this patient stopped warfarin, INR was normalized through transfusion of fresh blood and frozen plasma. Upper GI endoscopy revealed erosive gastritis and bulb duodenitis, proton pump inhibitor was prescribed and LMWH resumed for one week followed by reinstitution of warfarin). The other patient (0.81%) had massive subcutaneous trunk hematoma and intramuscular hematoma of right upper and lower limbs with hemoglobin drop of 2 gm/dl in 3 days related to anticoagulant therapy; the patient stopped warfarin, INR was normalized through transfusion of fresh blood and frozen plasma. Warfarin was temporarily withheld and substituted by LMWH for one week followed by reinstitution of warfarin. Micronized purified flavonoid fraction (MPFF, 450 mg diosmin plus 50 mg hesperidin-Daflon 500 mg, Servier, France) was prescribed also for one week.
Mean duration of hospital stay was longer for group (B).Tab. (2), Graph (1).
Table 2: Post-intervention early outcomes: (N=270 limbs in 240 patients)
|
Variables |
Group A N=117 (48.75%) |
Group B N=123 (51.25%) |
Test of significance |
P-Value |
|
|
Pulmonary embolism |
5 (4.3%) |
3 (2.4%) |
ZPROP.=0.82 |
0.41 (NS) |
|
|
Bleeding |
Minor |
9 (7.7%) |
4 (3.25%) |
X2=1.18 |
0.27 (NS) |
|
Major |
0 |
2 (1.6%) |
|||
|
Hospital stay (days) |
3.2±1.45 |
5±2.03 |
St.”t”= 3.03 |
0.003 (S) |
|
Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
Given the concern of leg pain and swelling assessment during the 1st month; there was statistically significant difference between both groups; patients of group
(A) Complained less pain at 10 & 30 days; P-Value; 0.02 & 0.04 respectively. Also there was significant decrease in leg circumference in group (A) at 10 & 30 days; P-Value; 0.001 & 0.03 respectively. Table. (3).
Table 3: Leg pain and swelling assessment during the 1st month
|
Variables |
Group A N=117 (48.75%) |
Group B N=123 (51.25%) |
t-test |
P-Value |
|
|
Change in leg pain severity |
1st day |
5.9±1.38 |
6.8±1.09 |
1.71 |
0.09 (NS) |
|
10 days |
4.26±1.28 |
5.53±0.99 |
2.14 |
0.02 (S) |
|
|
30 days |
3.71±1.27 |
4.99±0.98 |
3.41 |
0.04 (S) |
|
|
Change in leg Swelling (cm) |
1st day |
39.7±4.1 |
40.2±3.5 |
1.83 |
0.08 (NS) |
|
10 days |
39.41±3.93 |
40.48±3.34 |
4.16 |
0.001 (HS) |
|
|
30 days |
38.94±3.93 |
39.91±3.34 |
3.21 |
0.03 (S) |
|
Data are presented as numbers & mean ± SD; ranges are in parenthesis and statistically significant difference by using unpaired t-test.
In group (A), six patients (nine limbs; 6.8%) suffered recurrent DVT. Three patients with incomplete thrombolysis had bilateral recurrent iliac and femoral vein partial thrombosis as identified by follow-up duplex study at 3months, yet the patients were not symptomatic. Therefore, anticoagulation was continued. Other three patients had complete thrombolysis with insetion of a wallstent into a left common iliac vein stenotic lesion. They had recurrent symptoms at the follow-up visit. CT venography revealed occluded iliac vein stent. In group (B), nine patients (fifteen limbs; 10.9%) had recurrent DVT. These patients were incompliant to anticoagulant therapy and complaining of symptoms of recurrence (90 days postrandomization), their INR was 1.4 to 2.1. Duplex ultrasound showed recurrent iliofemeropopliteal DVT. Tab. (4), Graph (1).
Table 4: Incidence of recurrent DVT in the 1st 6 months: (N=270 limbs)
|
Recurrent DVT |
Group A N=117 (48.75%); 132 limbs (48.9%) |
Group B N=123 (51.25%); 138 limbs (51.1%) |
ZPROP |
P-Value |
|
Unilateral DVT |
3 |
3 |
0.032 |
0.97 (NS) |
|
Bilateral DVT |
3 |
6 |
0.55 |
0.58 (NS) |
|
Total limbs |
9 (6.8%) |
15 (10.9%) |
0.68 |
0.49 (NS) |
Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
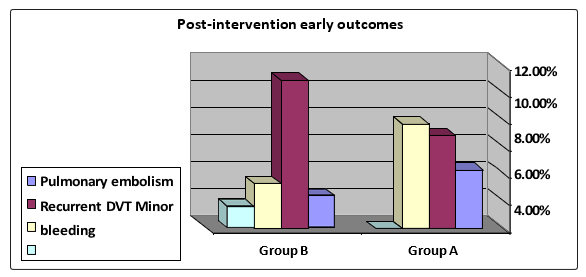
Graph 1: Post-intervention early outcomes
At six month; there was statistically significant differences between both groups; patency of the iliac vein segment was better and Femoral vein Reflux was less in group (A); P-Value; <0.001 (HS) and 0.003 (S) respectively. Table. (5).
Table 5: Post-thrombotic changes at 6th month in both groups: (N=270 limbs)
|
Variables |
Group A 132 limbs (48.9%) |
Group B 138 limbs (51.1%) |
ZPROP |
P-Value |
|
|
Iliac vein |
Patency |
120 (90.9%) |
63 (45.7%) |
4.6 |
<0.001(HS) |
|
Residual thrombi |
27 (20.5%) |
105 (76.1%) |
5.2 |
<0.001(HS) |
|
|
Wall thickening |
30 (22.7%) |
102 (73.9%) |
4.85 |
<0.001(HS) |
|
|
Femoral vein |
Reflux |
57 (43.2%) |
102 (73.9%) |
2.96 |
0.003 (S) |
|
Residual thrombi |
39 (29.6%) |
81 (58.7%) |
2.78 |
0.005 (S) |
|
|
Sclerosis |
42 (31.8%) |
87 (63.04%) |
2.96 |
0.003 (S) |
|
Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
At one year; Patency of iliac vein segment was still high in group (A); (P- Value <0.001 (HS)). Also other post-thrombotic changes were less in group (A) except residual thrombi in femoropopliteal vein; (P-Value= 0.87 (NS)). Tab. (6).
Table 6: Post-thrombotic changes at one year in both groups: (N=270 limbs)
|
Variables |
Group A 132 limbs (48.9%) |
Group B 138 limbs (51.1%) |
ZPROP |
P-Value |
|
|
Iliac vein |
Patency |
123 (93.2%) |
72 (52.2%) |
4.34 |
<0.001 (HS) |
|
Residual thrombi |
21 (15.9%) |
54 (39.1%) |
2.46 |
0.014 (S) |
|
|
Wall thickening |
36 (27.3%) |
78 (56.5%) |
2.81 |
0.0049 (S) |
|
|
Femoral vein |
Reflux |
69 (52.2%) |
114 (82.6%) |
3.08 |
0.002 (S) |
|
Residual thrombi |
24 (18.2%) |
27 (19.6%) |
0.17 |
0.87 (NS) |
|
|
Sclerosis |
45 (34.1%) |
93 (67.4%) |
3.16 |
0.0016 (S) |
|
Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
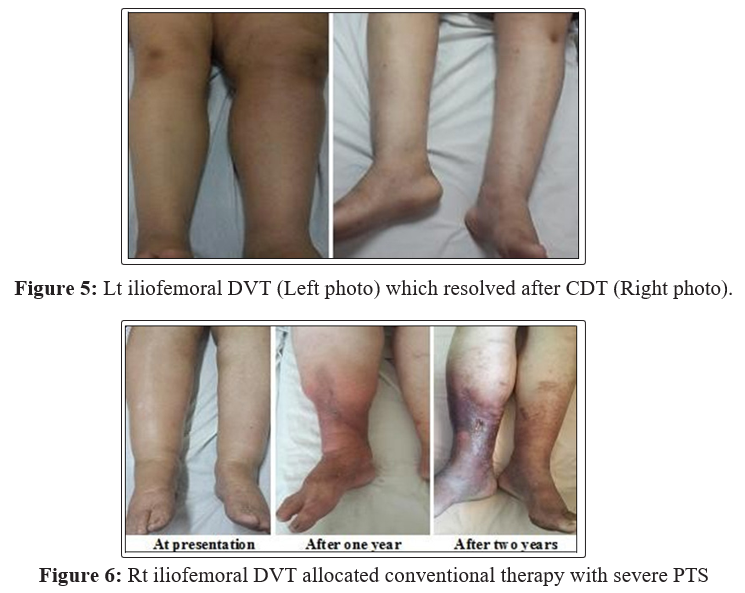
After two years, post-thrombotic changes were still significantly less in group (A) except; iliac vein segment residual thrombi and wall thickening; (P- Value =0.059 (NS) and 0.17 (NS)) respectively and residual thrombi in femoropopliteal vein lumen; (P-Value= 0.41 (NS)). Tab. (7), Fig. (5, 6)
Table 7: Post-thrombotic changes at 2 years in both groups: (N=270 limbs)
|
Variables |
Group A 132 limbs (48.9%) |
Group B 138 limbs (51.1%) |
ZPROP |
P-Value |
|
|
Iliac vein |
Patency |
126 (95.5%) |
81 (58.7%) |
4.12 |
<0.001(HS) |
|
Residual thrombi |
12 (9.1%) |
33 (23.9%) |
1.88 |
0.059 (NS) |
|
|
Wall thickening |
42 (31.2%) |
63 (45.7%) |
1.34 |
0.17 (NS) |
|
|
Femoral vein |
Reflux |
78 (59.1%) |
123 (89.1%) |
3.27 |
<0.001 (HS) |
|
Residual thrombi |
15 (11.4%) |
24 (17.4%) |
0.81 |
0.41 (NS) |
|
|
Sclerosis |
51 (38.6%) |
105 (76.1%) |
3.6 |
<0.001(HS) |
|
Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
By reviewing the incidence and severity of PTS using the Villalta score; patients in group (A) developed significantly less PTS at six months, one year and two years; P-Value; 0.024, 0.017 and 0.035 respectively. Moreover, severe cases were significantly less in group (A); at six months and at two years; P-Value; 0.035 and 0.042 respectively. Tab. (8), Graph (2).
Table 8: Severity grading of PTS using Villalta score in both groups: (N=270 limbs)
|
Variables |
Group A 132 limbs (48.9%) |
Group B 138 limbs (51.1%) |
ZPROP |
P-Value |
|
|
After 6 months |
33 (25%) |
66 (47.8%) |
2.24 |
0.024 (S) |
|
|
Villalta severity category |
Mild |
27 (81.8) |
27 (40.9) |
2.22 |
0.026 (S) |
|
Moderate |
6 (18.2) |
18 (27.3) |
0.57 |
0.56 (NS) |
|
|
Severe |
0 (0.0) |
21 (31.8) |
2.1 |
0.035 (S) |
|
|
After one year |
39 (29.6%) |
75 (54.4%) |
2.38 |
0.017 (S) |
|
|
Villalta severity category |
Mild |
30 (76.9) |
30 (40.0) |
2.16 |
0.031 (S) |
|
Moderate |
6 (15.4) |
33 (44.0) |
1.76 |
0.078 (NS) |
|
|
Severe |
3 (7.7) |
12 (16.0) |
0.72 |
0.47 (NS) |
|
|
After two years |
54 (40.9%) |
87 (63.0%) |
2.1 |
0.035 (S) |
|
|
Villalta severity category |
Mild |
36 (66.7) |
30 (34.5) |
2.15 |
0.031 (S) |
|
Moderate |
16 (26.6) |
33 (37.9) |
0.81 |
0.57 (NS) |
|
|
Severe |
2 (3.7) |
24 (27.6) |
1.93 |
0.042 (S) |
|
Villalta severity category; Mild (score 5 to 9), Moderate (score 10 to 14) or Severe (score >14) (S= Significant, HS= Highly Significant, NS= Non Significant)Data are presented as numbers & mean ± SD; percentages & ranges are in parenthesis.
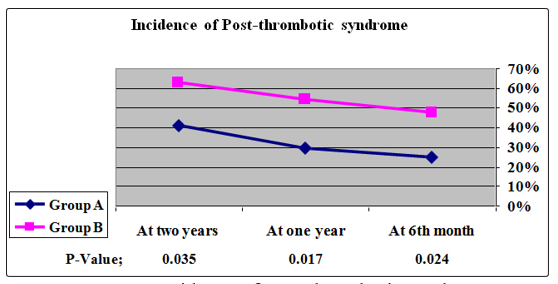
Graph 2: Incidence of Post-thrombotic syndrome
Post-thrombotic syndrome severely impacts the quality of life of affected individuals; in the current study; Patients of group (A) experienced significantly better Quality of life; P-Value; 0.003. Tab. (9).
Table 9: Quality of life (Symptoms) during 24 months follow up
|
Variables |
Group A N=117 (48.75%) |
Group B N=123 (51.25%) |
t-test |
P-Value |
|
Change in VEINES-QOL |
25±2.03 |
23.2±1.45 |
3.03 |
0.003 (S) |
(VEINES-QOL); Venous Insufficiency Epidemiological & Economic Study Quality of Life.
Data are presented as numbers & mean ± SD; ranges are in parenthesis and statistically significant difference by using unpaired t-test.
Discussion
Despite advances in the diagnosis and management of thromboembolic disease, it remains a potentially life threatening disorder affecting hospitalized and healthy individuals. In addition to early risk of pulmonary embolism, late morbidity may develop from post thrombotic syndrome [19,20].
Early removal of thrombus may improve deep venous flow and reduce valvular reflux and incidence of PTS. Early thrombus removal can be achieved by surgical thrombectomy, systemic thrombolysis, catheter-directed thrombolysis, and/or pharmacomechanical thrombolysis [21]. In the current study population, 180/240 patients (75%) were females. This was similar to studies done by Elsharawy and Elzayat,Bækgaard, Protack and Manninen; 68.57%, 77.22%, 61% and 54% of patients were females respectively [22-25]. On the contrary, only 37% and 49.06% of patients enrolled in the CaVenT trial and Lee et al. were females respectively [26,27].
Left-sided DVT was reported in this study in 183 patients (76.25%) which was less than that mentioned in the study of Sillesen et al., 84.44% of patients and was more than that reported in CaVenT trial; 58.52% of patients [13,28].
Access to venous system was gained through sonographic guided ipsilateral popliteal vein in prone position. This was similar to access done by Beakgaard and co-workers stipulated presence of at least open distal half of popliteal vein to act as an access site [23].
By reviewing success of thrombolysis; it was noticed that complete lysis was achieved in 90 patients (76.9%); These results were comparable to that mentioned by Gomaa et al. who reported that Complete lysis was achieved in 23/28 patients (82.2%) and partial lysis was achieved in 4/28 (14.3%) [29]. Also the results were comparable with studies done by who mentioned complete lysis that was achieved in 61%, 88.89%, 93% and 80% respectively [13,22,26,30].
Alteplase was used in this study because streptokinase had been abandoned due to its allergic complications [31]. Our results were significantly better than result of Mewissen et al. who achieved complete lysis rate in only 31% using urokinase. This discrepancy is due to higher fibrin specificity of Alteplase than urokinase [32].
The mean duration of symptoms was 4.8±2.01 days (1-14 days); meta- analysis performed by Casey et al, who stated that no significant effect of duration between the onset of symptoms and the time of delivering intervention within the 14-day therapeutic window [33]. The timing of CDT has been a matter of debate; the clinical practice guidelines of the society for vascular surgery and the American venous forum accepted 14 days as the cut-off for CDT in DVT treatment [21].Also ATTRACT and CaVenT trials used cut-off limits of 14 and 21 days from the start of symptoms, respectively [34]. On the other hand, Dumantepe and co-workers; by using ultrasound accelerated thrombolysis; reported successful results in DVT patients of more than 90 days; Of their 12 patients, six had complete lysis while five had partial lysis with an average duration of 26 hours [35].
Among patients of group (A); the culprit stenotic lesions in the proximal common iliac vein were encountered in 18 patients; Balloon angioplasty was performed in all but Stenting was performed in 15/18 patients (83.33%); These results were comparable to study done by Sillesen et al., 30/45 occluded veins (67%) revealed underlying stenoses in iliac veins; All were treated with balloon angioplasty and stenting [13]. In CaVenT trial, 23/90 patients (25.56%) received balloon angioplasty, 15/23 patients (65.2%) received venous stents [26].
Catheter directed thrombolysis has two drawbacks: it is invasive with bleeding risk. During the current study; A total of 9 patients (7.7%) clinically relevant non major bleeding complications were reported in CDT group which is less than found by Gomaa et al.; only echomosis at puncture site in 10/28 patients (35%) [29]. Sillesen et al.; 16% and Verhaegue et al.; 12 patients (50%) bleeding at the puncture site with 6 patients needing blood transfusion [13].
This was mostly due to use of a low to medium dose of alteplase (1 mg/h) in comparison to Verhaege et al. who used high dose of 3 mg/h [36]. But this bleeding complications were similar to that reported by CaVenT trial; 9% at puncture site [26].
By reviewing incidence of pulmonary embolism; five patients (4.3%) in CDT group versus three patients (2.4%) in group (B). Nevertheless, PE had no effect on conductance and subsequent follow-up. Enden and colleagues; in early results of CaVenT trial; no pulmonary embolism occurred due to CDT [37].
In group (A), six patients (nine limbs; 6.8%) suffered recurrent DVT versus nine patients (fifteen limbs; 10.9%) in group (B); this result was less than that reported by CaVenT trial; 34 patients (19%) had recurrent DVT with no difference between treated groups [28]. Based on findings of the current study and findings of CaVenT trial, it can be assumed that addition of CDT to standard anticoagulant therapy does not have additional protection against recurrent DVT.
Given the concern of leg pain and swelling assessment during the 1st month; there was statistically significant difference between both groups. This result was near similar to that approved by ATTRACT Trial investigators who reported that; Change in leg pain severity; Baseline to day 10 and day 30; P-Value; 0.02 & 0.03 respectively and Change in leg circumference; Baseline to day 10 and day 30; P- Value; 0.02 & 0.05 respectively [38].
As regards of post thrombotic morbidity; after six months; patency of iliac vein segment was significantly better and femoral vein Reflux was significantly less in group (A). These results were comparable to that mentioned by CaVenT trial; after six months follow up, patency of iliofemoral vein segment was found in 64% of patients in CDT group and 35.8% of patients in control group while femoral venous insufficiency and other post-thrombotic changes did not differ significantly in treated arms [37]. At one year; Patency of iliac vein segment was still high and other post-thrombotic changes were less in group (A); as mentioned by Mewissen et. al who reported that vein patency might be twice as good after total clot removal [32].
It is assumed that both recanalization and development of reflux are continuing processes, mainly established during the 1st 2 years following DVT. At two year of the current study, post-thrombotic changes were still less in group (A); as mentioned in the CaVenT trial; deep vein reflux reduced after 2 years and iliofemoral patency was improved after 6 months to 2 years [22]. Also this result was comparable to studies done by AbuRahma et. al.; two years follow-up period symptom resolution was 78% in CDT group versus 30% in group treated with anticoagulation and Ly et. al.; Reflux was found in 32% after 24 months [39,40].
A limitation in clinical studies on post-thrombotic syndrome is the absence of a gold standard for its diagnosis. Several scoring systems have been used for diagnosis and severity grading of chronic venous disorders. In the current study, Villalta score had been used to diagnose and to grade PTS. After follow-up period of 24 months; patients in group (A) developed less post-thrombotic syndrome at six months, at one year and at two years; P-Value; 0.024, 0.017 and 0.035 respectively; this result was near similar to CaVenT trial; After two-year follow-up period, 43% of patients in CDT developed post- thrombotic syndrome compared with 71% in the control group [29]. Our results regarding PTS were against ATTRACT trial; might be due to our early intervention in most cases in the 1st few days of DVT onset and our patients were younger and some patients of the ATTRACT trial had DVT in femoropopliteal segment only [38].
In a Cochrane review, Watson et al. performed a meta-analysis of 17 RCT totaling 1103 patients. They found that thrombolysis offers potential advantages over standard treatment, by reducing the proportion of patients with PTS by a third in the longer term, particularly in patients with iliofemoral vein thromboses [41].
Given the special concern of Post-thrombotic syndrome that severely impacts quality of life of affected individuals by lost productivity, loss of functional independence and forced retirement; in the current study; Patients of group (A) experienced better Quality of life due to less PTS patients; P-Value; 0.003; This was reported by Kahn et al.; In a multicenter cohort study that recruited 357 patients with acute DVT; PTS was the principal determinant of health-related quality of life at 2 years’ follow-up [5]. But ATTRACT Trial investigators reported that; Change in VEINES disease-specific quality of life; Symptoms, baseline to 2 years were insignificant in both groups, P-Value; 0.17 [38]. The best endovascular thrombolytic approach remains unclear. Adjunctive mechanical techniques might have an impact on treatment time and possibly the clinical outcome compared with the catheter-directed thrombolysis only approach.
One of limitations of our study that we did not use adjunctive mechanical techniques.
Conclusion
Addition of catheter-directed thrombolysis in the treatment of acute iliofemoral deep vein thrombosis; was safe and tolerated by most of the patients with better effect to reduce leg pain & circumference. It was considered a protecting weapon to prevent post-thrombotic syndrome and so improve quality of life and was related to achievement of higher iliac vein patency and less reflux.
References
- Anthony J, Comerota A and Aziz F (2018) Acute Lower Extremity Deep Venous Thrombosis; Rutherford’s vascular surgery and endovascular therapy 9th by Anton N. Sidawy, Bruce A. Perler, 2: 6398-6408.
- Augustinas P, Ouriel K (2004) Invasive approaches to treatmentof venous thromboembolism. Circulation, 110: 1-34.
- Goldhaber SZ, Elliot CG (2003) Acute pulmonary embolism: part II: risk stratification, treatment and prevention. Circulation, 108: 2834-2838.
- Kahn S, Hirsch A, Shrier I (2002) Effect of postthrombotic syndrome on health- related quality of life after deep venous thrombosis. Arch Intern Med, 162: 1144-1148.
- Kahn S, Shrier I, Julian J, Ducruet T, Arsenault L, et al. (2008) Determinants and time course of the postthrombotic syndrome after acute deep venous thrombosis. Ann Intern Med, 149: 698-707.
- Delis K, Bountouroglou D and Mansfield A (2004) Venous claudication in iliofemoral thrombosis: long-term effects on venous hemodynamics, clinical status, and quality of life. Ann Surg 239:118-126.
- Douketis JD, Crowther MA, Foster GA and Ginsberg JS (2001) Does the location of thrombosis determine the risk of disease recurrence in patients with proximal deep vein thrombosis? Am J Med 110: 515-519.
- Baldwin M, Moore H, Rudarakanchana N, Gohel M and Davies A (2013) Post- thrombotic syndrome: a clinical review. J Thromb Haemost 11: 795-805.
- Comerota AJ (2010) Randomized trial evidence supporting a strategy of thrombus removal for acute DVT. Semin Vasc Surg 23: 192-198.
- Du GC, Zhang MC and Zhao JC (2015) Catheter-directed thrombolysis plus anticoagulation versus anticoagulation alone in the treatment of proximal deep vein thrombosis; a meta- analysis. Vasa, 44: 195-202.
- Engelberger R, Spirk D and Willenberg T (2015) Ultrasound assisted versus conventional catheter-directed thrombolysis for acute iliofemoral deep vein thrombosis. Circ Cardiovasc Interv 8.
- Lin P, Ochoa L and Duffy P (2010) Catheter-directed thrombectomy and thrombolysis for symptomatic lower- extremity deep vein thrombosis: review of current interventional treatment strategies. Perspect Vasc Surg Endovasc Ther, 22: 152-63.
- Sillesen H, Just S, Jorgensen M and Baekgaard N (2005) Catheter directed thrombolysis for treatment of ilio-femoral deep venous thrombosis is durable, preserves venous valve function and may prevent chronic venous insufficiency. Eur J Vasc Endovasc Surg 30: 556-562.
- Kearon C, Akl E and Ornelas J (2016) Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest 149: 315-352.
- Marder V, Soulen R and Atichartakarn V (1977) Quantitative venographic assessment of deep vein thrombosis in the evaluation of streptokinase and heparin therapy. J Lab Clin Med 89: 1018-1029.
- Kahn S, Partsch H, Vedantham S, Prandoni P and Kearon C (2009) Definition of post-thrombotic syndrome of the leg for use in clinical investigations: a recommendation for standardization. Journal of Thrombosis and Hemostasis 7: 879-883.
- Lamping D, Schroter S, Kurz X, Kahn S and Abenhaim L (2003) Evaluation of outcomes in chronic venous disorders of the leg: development of a scientifically rigorous, patient- reported measure of symptoms and quality of life. J Vasc Surg 37: 410-419.
- Khothari CR (2004) Research Methodology: Methods and Techniques, New Age International, New Delhi.
- Bosson J, Labarere J and Sevestre M (2003) Deep vein thrombosis in elderly patients hospitalized in sub acute care facilities: a multicenter cross-sectional study of risk factors, prophylaxis and prevalence. Arch Intern Med 163: 2613-2618.
- Kearon C (2003) Natural history of venous thromboembolism.Circulation 107: 122-1230.
- Meissner M, Gloviczki P, Comerota A, Dalsing M, Eklof B, et al. (2012) Society for Vascular, S. and American Venous, F.: Early thrombus removal strategies for acute deep venous thrombosis: clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg 55: 1449-1462.
- Elsharawy M and Elzayat E (2002) Early results of thrombolysis vs. anticoagulation in iliofemoral venous thrombosis. A randomized clinical trial. Eur J Vasc Endovasc Surg 24: 209e14.
- Baekgaard N, Broholm R, Just S, Jorgensen M and Jensen L (2010) Long- term results using catheter-directed thrombolysis in 103 LL with acute iliofemoral venous thrombosis. Eur J Vasc Endovasc Surg 39: 112-117.
- Protack C, Bakken A, Patel N, Saad W, Waldman D and Davies M (2007) Long-term outcomes of catheter directed thrombolysis for lower extremity deep venous thrombosis without prophylactic inferior vena cava filter placement. J Vasc Surg 45: 992-997.
- Manninen H, Juutilainen A, Kaukanen E and Lehto S (2012) Catheter- directed thrombolysis of proximal lower extremity deep vein thrombosis: a prospective trial with venographic and clinical follow-up. Eur J Radiol 81: 1197-1202.
- Enden T, Haig Y, Kløw N, Slagsvold C, Sandvik L, et al. (2012) Long-term outcome after additional catheter-directed thrombolysis versus standard treatment for acute iliofemoral deep vein thrombosis (the CaVenT study): a randomised controlled trial. The Lancet 379: 31-38.
- Lee CY, Lai ST, Shih CC and Wu TC (2013) Short-term results of catheter- directed intrathrombus thrombolysis versus anticoagulation in acute proximal deep vein thrombosis. J Chin Med Assoc 76: 265-70.
- Haig Y, Enden T, Grøtta O, Kløw N, Slagsvold C, et al. (2016) Post-thrombotic syndrome after catheter-directed thrombolysis for deep vein thrombosis (CaVenT): 5-year follow-up results of an open-label, randomised controlled trial. The Lancet Hematology 3: e64-e71.
- Gomaa M, Fahmy H and Farouk A (2013) Catheter direct thrombolysis: Role of actilyse in treatment of acute deep venous thrombosis in The Egyptian Journal of Radiology and Nuclear Medicine 44: 57-62.
- Chang R, Chen C and Kam A (2008) Deep vein thrombolysis of lower extremity; direct intra clot injection of alteplase, once daily with systemic anticoagulation- result of pilot study. Radiology 246: 619-629.
- Grunwald MR and Hofmann LV (2004) Comparison of urokinase, alteplase, and reteplase for catheter-directed thrombolysis of deep venous thrombosis. J Vasc Interv Radiol 15: 347e52.
- Mewissen MW, Seabrook GR and Meissner MH (1999) Catheter-directed thrombolysis for lower extremity deep venous thrombosis: report of a national multicenter registry. Radiology 211: 39-49.
- Casey E, Murad M, Zumaeta-Garcia M, Elamin M, Shi Q, et al. (2012) Treatment of acute iliofemoral deep vein thrombosis. J Vasc Surg 55: 1463-1473.
- Huynh K (2016) Venous thromboembolism: 5-year follow-up data from CaVenT. Nat Rev Cardiol 13: 124.
- Dumantepe M, Tarhan I and Ozler A (2013) Treatment of chronic deep vein thrombosis using ultrasound accelerated catheter-directed thrombolysis. Eur J Vasc Endovasc Surg 46: 366-371.
- Verhaeghe R, Stockx L and Lacroix H (1997) Catheter-directed lysis of ileo- femoral vein thrombosis with use of rt.PA. Eur Radiol 7: 996-1001.
- Enden T, Klow N, Sandvik L, Slagsvold C, Ghanima W, et al. (2009) Catheter-directed thrombolysis vs. anticoagulant therapy alone in deep vein thrombosis: results of an open randomized, controlled trial reporting on short-term patency. J Thromb Haemost 7: 1268-1275.
- Vedantham S, Goldhaber S, Julian J, Kahn S, Jaff M, et al. (2017) Pharmacomechanical Catheter-Directed Thrombolysis for Deep-Vein Thrombosis; N Engl J Med 377: 2240-2252.
- AbuRahma AF, Perkins SE, Wulu JT and Ng HK (2001) Iliofemoral deep vein thrombosis: conventional therapy versus lysis and percutaneous transluminal angioplasty and stenting. Ann Surg 233: 752e60.
- Ly B, Njaastad A, Sandbæk G, Solstrand R, Rosales A (2004) Catheter-based thrombolysis of iliofemoral venous thrombosis. Tidsskr Nor Lægeforen 124: 478e80.
- Watson L, Broderick C and Armon M (2014) Thrombolysis for acute deep vein thrombosis. Cochrane Database Syst Rev, 1.




