Research Article - (2026) Volume 5, Issue 1
The Role of Immunotherapy in Solid Malignancies: Biological Rationale, Clinical Evidence and Future Directions
Received Date: Dec 15, 2025 / Accepted Date: Jan 20, 2026 / Published Date: Jan 28, 2026
Copyright: ©2026 Sanjaya K Upadhyaya. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Upadhyaya, S. K. (2026). The Role of Immunotherapy in Solid Malignancies: Biological Rationale, Clinical Evidence and Future Directions. Insights Herbal Med, 5(1), 01-09.
Abstract
Immunotherapy—especially immune checkpoint inhibitors (ICIs)—has reshaped the therapeutic landscape of solid tumors by restoring antitumor immunity through blockade of the programmed cell death protein 1/programmed death- ligand 1 (PD-1/PD-L1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) axes. The mechanistic foundation of ICI efficacy includes reversal of T-cell exhaustion, enhancement of antigen presentation, and remodeling of the tumor immune microenvironment (TME). Clinically, immune checkpoint blockade has expanded from early successes in melanoma and lung cancer to become integral across gastrointestinal, hepatobiliary, renal, gynecologic, breast, and molecularly selected colorectal cancers, with accelerating incorporation into perioperative and curative-intent strategies. Yet, broad benefit remains constrained by primary and acquired resistance, imperfect biomarkers, and immune- related adverse events (irAEs). This review synthesizes biologic principles, pivotal and contemporary trial evidence, and evolving clinical applications across major solid tumor groups, with emphasis on recent perioperative advances (2023–2025), circulating tumor DNA (ctDNA)-guided concepts, microbiome influences, combination immunotherapy strategies, and emerging cellular and adoptive immunotherapies. We highlight practical biomarker frameworks, toxicity recognition/management, and future directions that include next-generation checkpoints, engineered cell therapies, microbiome modulation, and adaptive treatment paradigms to extend durable benefit safely and equitably.
Keywords
Immune Checkpoint Inhibitors, PD-1, PD-L1, CTLA-4, Tumor Microenvironment, Biomarkers, ctDNA, Microbiome, Neoadjuvant Immunotherapy, Perioperative Immunotherapy, Adoptive Cell Therapy, TIL, CAR-T, Immune-Related Adverse Events
Introduction
Cancer progression is inseparable from immune escape. While early conceptual models proposed immune surveillance as a barrier to malignant transformation, tumors evolve “immune editing” strategies that permit growth despite host immunity. Modern immunotherapy leverages this biology: instead of directly killing tumor cells (as with cytotoxic therapy), it aims to re-enable effective immune recognition and elimination.
The clinical turning point came with immune checkpoint blockade. Sharma and Allison articulated the therapeutic logic that inhibitory checkpoints are actionable “brakes” on antitumor T-cell responses, and that checkpoint blockade can produce deep, durable remissions distinct from chemotherapy response patterns [1]. Ribas and Wolchok further emphasized that the signature of checkpoint therapy is the potential for durability, including long-term survival in subsets of patients across multiple tumor types, and the need to reframe response kinetics and endpoints accordingly [2].
Mechanistically, immunotherapy can be anchored to the “cancer- immunity cycle,” in which effective antitumor immunity requires coordinated antigen release, presentation, priming, trafficking, infiltration, recognition, and killing [3]. Tumors can interrupt this cycle at many steps, which explains heterogeneity in response and provides a rationale for combination approaches that target multiple barriers simultaneously. The centrality of the TME—its immune composition, stromal architecture, and suppressive networks— was comprehensively described by Binnewies and colleagues, framing “hot,” inflamed tumors as more ICI-responsive and “cold” or immune-excluded tumors as resistant [4].
Contemporary review syntheses across tumor types underscore that immunotherapy has evolved from “a drug class” into a treatment platform—combined with chemotherapy, anti- angiogenic therapy, radiation, targeted therapies, cellular therapy, and novel checkpoints to broaden efficacy [5-7]. This review integrates biologic rationale with clinical evidence, focusing on recent practice-changing advances and the next phase of immuno- oncology.
Biological Rationale: From Checkpoints to Systems Immunology
The PD-1/PD-L1 and CTLA-4 Axes in T-cell Regulation
CTLA-4 primarily modulates early T-cell priming in lymphoid tissues, while PD-1 regulates effector function within peripheral tissues and the tumor bed. When PD-1 binds PD-L1/PD-L2, T-cell signaling is dampened, promoting exhaustion phenotypes and immune escape. Blockade of these inhibitory interactions reactivates effector T-cells but can also break peripheral tolerance, leading to irAEs [1,2].
A practical clinical implication is that different checkpoints map onto different immunologic compartments: CTLA-4 blockade tends to amplify priming and repertoire breadth; PD-1/PD-L1 blockade tends to enhance intratumoral effector function. This compartmentalization supports dual blockade strategies in selected cancers and informs toxicity expectations [8,9].
The Tumor Immune Microenvironment and the “Hot– Cold” Continuum
The TME comprises tumor cells, immune cells (T cells, macrophages, dendritic cells, neutrophils), stromal fibroblasts, vasculature, and extracellular matrix. Immune-inflamed tumors often exhibit interferon signaling, antigen presentation, and pre-existing CD8+ infiltration, enabling robust ICI activity. By contrast, immune-desert tumors may lack antigenic stimuli or fail T-cell priming; immune-excluded tumors may have T-cells trapped in stroma without tumor penetration [4].
Recent integrative frameworks emphasize multidimensional immune profiling—cellular abundance, spatial localization, and functional states—as essential to predicting ICI benefit. A 2025 multidimensional synthesis of tumor-infiltrating lymphocytes (TILs) highlights how quantity alone is insufficient; spatial patterns and functional exhaustion markers often better correlate with outcomes in the modern immunotherapy era [10].
Neoantigens And Tumor Mutational Burden: Why Some Tumors Respond Dramatically
Neoantigens arise from somatic mutations that create novel peptides presented by HLA molecules. These non-self-epitopes can be recognized by T cells, enabling immune-mediated tumor control. A 2025 synthesis focusing on neoantigens and tumor mutational burden (TMB) reinforces the mechanistic linkage between mutation-derived antigenicity and ICI responsiveness, while also emphasizing limitations—TMB is a crude surrogate that does not capture antigen clonality, HLA presentation, or immune context [11].
Clinically, TMB has informed pan-tumor treatment concepts, and regulatory frameworks have increasingly reflected biomarker- driven approvals and label indications. A practical reference point for clinicians is the FDA Orange Book as a regulatory record for approved products and indications, which helps track evolving immunotherapy access and combinations [12].
Immunotherapy Modalities Beyond Checkpoint Blockade
Combination Immunotherapy: Principles and Practice
Combination strategies aim to convert “cold” tumors to “hot,” enhance antigen release, improve T-cell trafficking, and suppress immunosuppressive networks. Reviews of immunotherapy combinations highlight rationale-based pairing: ICI plus chemotherapy (immunogenic cell death), ICI plus anti-angiogenic therapy (vascular normalization and immune trafficking), dual ICI (breadth + effector restoration), and ICI plus targeted therapy (oncogenic pathway/antigen modulation) [8,13-15].
Adoptive and Engineered Cellular Therapies in Solid Tumors
Cell-based therapies have transformed hematologic malignancies, but solid tumors pose barriers including antigen heterogeneity, trafficking limitations, and suppressive TME. Comprehensive recent reviews detail evolving platforms: CAR-T, CAR-NK, engineered TCR therapies, and TIL therapy, alongside strategies to improve persistence and penetration [16-19].
A 2025 synthesis on combined ICIs and TIL strategies emphasizes the emerging logic that checkpoint blockade may amplify the function of transferred tumor-reactive lymphocytes, potentially yielding synergistic intratumoral activation [20]. While still maturing clinically, these approaches reflect the “next wave” of immunotherapy—especially for refractory solid tumors.
Gastrointestinal Malignancies: From Metastatic Standards to Perioperative Immunotherapy
Gastric and Gastroesophageal Junction Adenocarcinoma
CheckMate-649 established nivolumab plus chemotherapy as a first-line standard for advanced gastric/GEJ and esophageal adenocarcinoma, improving overall survival compared with chemotherapy alone [21]. Contemporary GI-focused reviews emphasize how PD-L1 enrichment, histologic/biologic heterogeneity, and evolving perioperative trials shape modern management [5-22].
Perioperative immunotherapy has accelerated recently. MATTERHORN evaluated perioperative durvalumab with FLOT in resectable gastric/GEJ cancers, reflecting a broader paradigm shift: bringing ICI earlier to potentially improve cure rates [23] KEYNOTE-585 similarly studied perioperative pembrolizumab with chemotherapy, expanding evidence supporting ICI integration in curative-intent settings [24].
Esophageal Cancer
KEYNOTE-590 demonstratedsurvivalbenefitwithpembrolizumab plus chemotherapy in advanced esophageal cancer, supporting chemo-immunotherapy as a key first-line approach in appropriate subgroups [25]. Evolving perioperative strategies have also been shaped by comparative analyses and trial syntheses; the ESOPEC trial commentary highlights refinements in multimodality therapy and underscores how perioperative regimen selection is increasingly data-driven [6].
Colorectal Cancer: MSI-H, Hypermutation, and ctDNA- Enabled Thinking
KEYNOTE-177 established pembrolizumab as first-line therapy for MSI-H metastatic colorectal cancer, with superior progression-free survival compared with chemotherapy and improved tolerability profiles [26]. Beyond MSI, ultra-hypermutated POLE/POLD1 tumors can behave like “immunotherapy-sensitive” disease due to abundant neoantigens. The 2025 report demonstrating ctDNA response and durable benefit after immune checkpoint blockade in POLE-mutant early-onset colon cancer illustrates how molecular phenotype and dynamic ctDNA monitoring can converge to guide precision immunotherapy [7].
Pancreatic Cancer: Overcoming Cold Biology
Pancreatic ductal adenocarcinoma remains among the most immunotherapy-resistant tumors due to low baseline immunogenicity, dense desmoplastic stroma, immune exclusion, and suppressive myeloid networks. Comprehensive pancreatic management reviews emphasize the urgent need for rational combinations—stroma modulation, vaccine/oncolytic priming, radiation synergy, and multi-agent immunotherapy designs—to generate meaningful activity beyond rare MSI-H cases [27].
Hepatobiliary Cancers: Anti-Angiogenic Synergy and Molecular Stratification
Hepatocellular Carcinoma
IMbrave150 changed first-line therapy for unresectable hepatocellular carcinoma by demonstrating improved survival with atezolizumab plus bevacizumab compared with sorafenib [28]. The biological rationale—simultaneously targeting immune suppression and abnormal tumor vasculature—has become a prototype for combination immunotherapy in other tumors as well. The HIMALAYA regimen (durvalumab with a single priming dose of tremelimumab) further reinforced that checkpoint strategies can produce durable benefit when appropriately designed [29].
Cholangiocarcinoma and Precision Immunotherapy Frameworks
Cholangiocarcinoma is molecularly heterogeneous. A multidisciplinary synthesis on intrahepatic cholangiocarcinoma emphasizes that immunotherapy decision-making should be integrated with molecular testing and targeted therapy planning, moving toward biomarker-informed selection rather than one- size-fits-all approaches [30]. Recent meeting-level updates (ASCO 2025 abstracts) suggest expanding immunotherapy combinations and trial activity in hepatobiliary and GI malignancies, reflecting ongoing innovation even where benefit has historically been modest [31,32].
Breast Cancer: Immunotherapy as a Curative-Intent Strategy and Beyond TNBC
Early Triple-Negative Breast Cancer (TNBC)
KEYNOTE-522 established pembrolizumab in combination with chemotherapy in the neoadjuvant setting followed by adjuvant pembrolizumab, improving event-free survival and supporting immunotherapy as a component of curative-intent TNBC management [33]. Reviews focused on immunotherapy in breast cancer emphasize that benefit is context-dependent, shaped by tumor biology, PD-L1/TIL landscape, and optimal integration with chemotherapy backbones [34].
Hormone Receptor–Positive Disease: Signals and Outliers
While ER+/HER2− breast cancer has typically been less responsive to checkpoint blockade, case-level evidence of exceptional responses can be biologically instructive. The reported exceptional response to pembrolizumab in metastatic ER+/HER2− breast cancer with liver metastases challenges assumptions and highlights that immune responsiveness may emerge in selected contexts— through tumor mutational landscapes, microenvironmental features, or prior therapy–induced immunogenic shifts [35].
Renal Cell Carcinoma: ICI Backbones, Sequencing, and Adjuvant Benefit
RCC has become a flagship for immunotherapy combinations. CheckMate-214 established nivolumab plus ipilimumab as a standard for metastatic RCC with durable overall survival benefit [36]. KEYNOTE-426 showed that pembrolizumab plus axitinib improves outcomes relative to sunitinib, cementing ICI plus VEGF-targeting therapy as a major strategy [37]. A contemporary RCC-focused review emphasizes the central clinical questions now: sequencing, resistance patterns, patient selection, and the optimal use of dual ICI versus ICI/VEGF combinations [38]. Importantly, immunotherapy has moved into the adjuvant setting. KEYNOTE-564 demonstrated overall survival benefit with adjuvant pembrolizumab after nephrectomy in high-risk resected RCC [39].
Lung Cancer and Head & Neck Cancer: The Perioperative Revolution
Resectable NSCLC
Perioperative immunotherapy has become one of the most rapidly evolving domains in thoracic oncology. Neoadjuvant nivolumab plus chemotherapy in CheckMate-816 improved pathologic complete response and event-free survival, demonstrating that immune priming in the presence of an intact tumor can translate into meaningful clinical benefit [40]. KEYNOTE-671 evaluated perioperative pembrolizumab plus chemotherapy, while AEGEAN studied perioperative durvalumab plus chemotherapy [41,42].
Head and Neck Squamous Cell Carcinoma (HNSCC)
HNSCC is immunogenic in many cases, and checkpoint blockade has become embedded in advanced disease management. Recent evidence supporting neoadjuvant and adjuvant pembrolizumab in resectable HNSCC (KEYNOTE-689) underscores the broader movement of immunotherapy into earlier disease stages in multiple tumor types [43].
Gynecologic Malignancies: Chemo-Immunotherapy Becomes Foundational
In endometrial cancer, randomized trials have rapidly changed practice. RUBY demonstrated benefit with dostarlimab plus chemotherapy in advanced or recurrent settings [44]. NRG- GY018 established pembrolizumab plus chemotherapy as another effective approach, with continued updates refining how molecular subtypes (e.g., dMMR vs pMMR) influence magnitude of benefit [45]. In locally advanced cervical cancer, pembrolizumab plus chemoradiotherapy improved outcomes, representing an important proof-of-concept for combining immunotherapy with definitive local therapy [46].
Biomarkers and Monitoring: From Enrichment to True Prediction
PD-L1, MSI/dMMR, and Beyond
Current clinical practice uses several biomarker classes:
• PD-L1: widely used but imperfect; varies by assay, scoring, and tumor type; often enriches rather than predicts [25,34].
• MSI/dMMR: one of the most robust predictors of ICI response across tumors, exemplified by MSI-H CRC outcomes in KEYNOTE-177 [26].
• TMB/neoantigens: mechanistically compelling, but clinically noisy; best interpreted alongside immune context and antigen clonality [11].
• TILs and immune gene signatures: increasingly important as multidimensional biomarkers integrating spatial and functional properties [10].
ctDNA as a Dynamic Biomarker
ctDNA offers a real-time window into tumor burden, minimal residual disease, and early response. The POLE-mutant early- onset CRC report showing ctDNA response with ICI illustrates how ctDNA could help distinguish pseudo-progression from true progression, monitor depth of response, and potentially guide treatment duration [7].
Microbiome Influences
The gut microbiome influences systemic immunity and response to checkpoint blockade. Landmark studies demonstrated associations between microbiome composition and ICI outcomes in epithelial tumors and melanoma, supporting the concept that microbial ecosystems modulate immune tone and therapeutic responsiveness [47,48].
Resistance Mechanisms: Primary, Adaptive, and Acquired Failure
Even in highly immunogenic cancers, resistance limits population- level benefit. Primary resistance includes the absence of T-cell priming, immune desert/exclusion, or dominant suppressive myeloid populations [4]. Acquired resistance involves tumor evolution under immune pressure, including antigen loss, interferon signaling defects, and immune escape mutations. The classic NEJM report on acquired resistance to PD-1 blockade in melanoma describes mutations that disrupt interferon response and antigen presentation, providing mechanistic grounding for resistance management strategies [49].
Immune-Related Toxicities: Safety as a Core Competency
ICIs can cause inflammatory toxicities involving dermatologic, gastrointestinal, hepatic, endocrine, pulmonary, neurologic, and renal systems. Kidney injury is a clinically important toxicity domain. A clinicopathologic series defined features of ICI- associated acute kidney injury and underscored the importance of recognition and appropriate immunosuppression [50]. Case-level reports continue to provide granular insights into management, such as pembrolizumab-induced acute interstitial nephritis [51]. Endocrine toxicities are also common; thyroid dysfunction, in particular, may require careful evaluation and longitudinal management [52].
Future Directions: What Comes Next
Next-generation checkpoints and Rational Combinations
Expanding beyond PD-1/CTLA-4 is a major frontier. Contemporary analyses emphasize next-generation checkpoints, optimal sequence design, and strategies to balance efficacy with toxicity [53]. Reviews of “next-generation immunotherapy” highlight the convergence of biomarkers, novel targets, and combination principles including radiotherapy and targeted therapy [54].
Cellular Therapy for Solid Tumors: Accelerating Translation
Engineered immune cell therapies are rapidly advancing, with innovations aimed at persistence and penetration [16]. Multi- review syntheses in 2023–2025 outline an expanding toolkit: TCR-engineered products, CAR platforms, armored cells, and combinatorial antigen targeting [17-19].
Precision Immunotherapy: Integrated Biom
The next phase is about the right immunotherapy, for the right patient, at the right time. Molecular profiling enables selection of sensitive subgroups and rational combinations [30,22]. Innovation in single-cell and spatial biology, along with AI-enabled signature discovery, is expected to produce better predictive biomarkers [4,10,54].
Perioperative Immunotherapy: Redefining Cure Paradigms
Neoadjuvant and perioperative immunotherapy continue to expand across tumors, supported by both mechanistic rationale and large randomized trials [23,24,40-42,55,56]. These strategies may ultimately alter recurrence patterns and redefine long-term survivorship.
Conclusion
Immunotherapy has evolved into a foundational modality in solid tumor oncology, with immune checkpoint blockade transforming outcomes across multiple malignancies and expanding rapidly into perioperative and curative-intent contexts. The field’s current challenges—resistance, biomarker imperfections, and irAE management—are now matched by equally rapid innovation: rational combinations, microbiome-directed strategies, ctDNA- guided monitoring, and next-generation cellular therapies. Continued progress will require integrated precision approaches that unify tumor genomics, immune context, dynamic biomarkers, and toxicity-aware clinical decision-making so that durable benefit becomes the rule rather than the exception [57,58].
Tables
|
Tumor Type |
Trial |
Regimen |
Key Outcome / Clinical Impact |
|
Gastric / GEJ adenocarcinoma (advanced) |
CheckMate-649 [17] |
Nivolumab + chemotherapy [17] |
Overall survival (OS) benefit; established first-line chemo- immunotherapy [17] |
|
Gastric / GEJ (resectable, perioperative) |
MATTERHORN [23] |
Durvalumab + FLOT [23] |
Supports integration of perioperative immunotherapy in curative settings [23] |
|
Gastric / GEJ (resectable, perioperative) |
KEYNOTE-585 [24] |
Pembrolizumab + chemotherapy [24] |
Reinforces perioperative ICI strategy movement into curative settings [24] |
|
Esophageal cancer (advanced) |
KEYNOTE-590 [18] |
Pembrolizumab + chemotherapy [18] |
OS benefit; supports chemo- immunotherapy as first-line approach [18] |
|
Esophageal adenocarcinoma (perioperative) |
ESOPEC [7] |
Multimodality perioperative strategy refinement [7] |
Refines perioperative treatment sequencing and selection [7] |
|
Colorectal cancer (MSI-H, metastatic) |
KEYNOTE-177 [19] |
Pembrolizumab [19] |
PFS benefit; established first-line ICI standard for MSI-H mCRC [19] |
|
Colorectal cancer (POLE- mutated) |
Case evidence (ctDNA) [6] |
ICI with ctDNA monitoring [6] |
Demonstrated ctDNA clearance and durable response [6] |
|
Hepatocellular carcinoma (unresectable) |
IMbrave150 [20] |
Atezolizumab + bevacizumab [20] |
OS benefit; established synergy of ICI + anti-VEGF [20] |
|
Hepatocellular carcinoma (unresectable) |
HIMALAYA [35] |
Durvalumab + tremelimumab [35] |
Durable survival benefit; validates priming-dose CTLA-4 [35] |
|
Renal cell carcinoma (metastatic) |
CheckMate-214 [21] |
Nivolumab + ipilimumab [21] |
OS benefit; established dual ICI standard in risk groups [21] |
|
Renal cell carcinoma (adjuvant) |
KEYNOTE-564 [40] |
Pembrolizumab (post- nephrectomy) [40] |
OS benefit; supports adjuvant ICI in high-risk resected RCC [40] |
|
Triple-negative breast cancer (early) |
KEYNOTE-522 [34] |
Neoadjuvant + adjuvant pembrolizumab [34] |
Improved EFS/OS; moved ICI into curative-intent TNBC [34] |
|
Resectable NSCLC (neoadjuvant) |
CheckMate-816 [30] |
Nivolumab + chemotherapy [30] |
Improved pathologic response and event-free survival [30] |
|
Resectable NSCLC (perioperative) |
KEYNOTE-671 [28] |
Perioperative pembrolizumab + chemotherapy [28] |
Improved EFS; solidifies perioperative ICI paradigm [28] |
|
Resectable HNSCC |
KEYNOTE-689 [31] |
Neoadjuvant + adjuvant pembrolizumab [31] |
Demonstrated benefit; expands perioperative ICI concept [31] |
|
Endometrial cancer (advanced) |
RUBY [37] |
Dostarlimab + chemotherapy [37] |
Survival benefit; established chemo- immunotherapy standard [37] |
|
Cervical cancer (locally advanced) |
Pembrolizumab + CRT [39] |
Pembrolizumab + chemoradiotherapy [39] |
Improved survival; integrates ICI into definitive setting [39] |
Table 1: Key Immunotherapy Trials Across Solid Malignancies
|
Biomarker / Modulator |
Tumor Types Where Most Relevant |
Clinical Utility |
Key Supporting Evidence |
|
MSI-H / dMMR |
CRC, gastric, endometrial, pan-tumor |
Strong predictor; enables first- line ICI in MSI-H mCRC |
KEYNOTE-177 [19]; biomarker framework reviews [49] |
|
TMB / Neoantigens |
Pan-tumor (context-dependent) |
Surrogate of immunogenicity; predictive but imperfect |
Neoantigen/TMB synthesis [11]; biomarker review [49] |
|
POLE / POLD1 mutations |
CRC (esp. early-onset), endometrial |
Ultra-hypermutated phenotype; potential for dramatic benefit |
POLE ctDNA report [6]; neoantigen synthesis [11] |
|
PD-L1 expression |
NSCLC, HNSCC, gastric/GEJ, others |
Enrichment biomarker (not universally predictive) |
Biomarker review [49]; KEYNOTE-590 [18] |
|
TILs / spatial contexture |
Breast, melanoma, many solid tumors |
Prognostic and increasingly predictive via spatial analysis |
TIL multidimensional review [10] |
|
ctDNA dynamics |
Pan-tumor (emerging) |
Response monitoring, MRD risk stratification |
POLE ctDNA response report [6] |
|
Gut microbiome |
Pan-tumor (modulator) |
Modulates ICI efficacy; potential target |
Routy et al. [50]; Gopalakrishnan et al. [51] |
|
Interferon signaling |
Pan-tumor |
Mechanistic determinant of acquired resistance |
Zaretsky et al. [52] |
|
TME phenotype ("hot vs cold") |
Pan-tumor |
Framework for combination approaches |
Binnewies et al. [4] |
Table 2: Biomarkers Predicting Immunotherapy Response or Modulating Outcomes
Figures
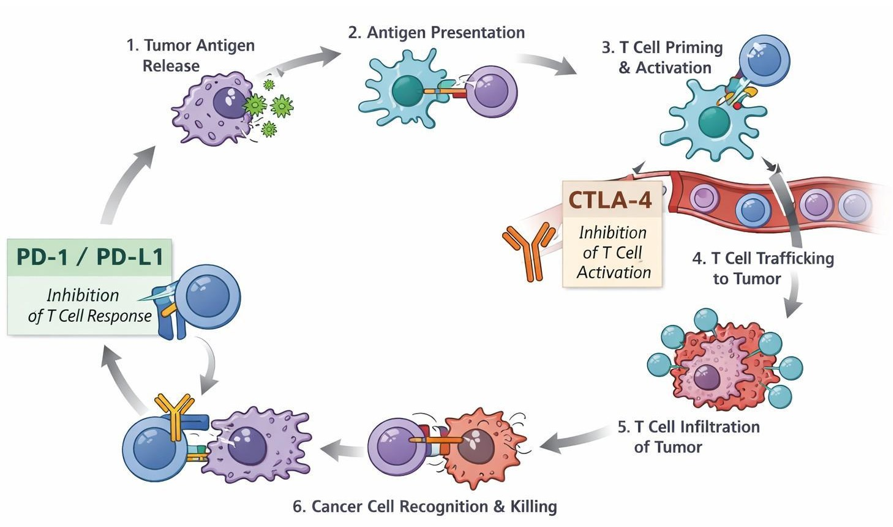
Figure 1: The Cancer-Immunity Cycle and Checkpoint Blockade Targets
This figure illustrates the cancer-immunity cycle, depicting the sequential steps required for effective antitumor immune responses, from tumor antigen release and presentation to T-cell priming, trafficking, tumor infiltration, recognition, and cancer cell killing. CTLA-4 primarily regulates the early priming phase in lymphoid tissues by inhibiting T-cell activation. In contrast, the PD-1/PD-L1 pathway suppresses effector T-cell function within the tumor microenvironment. Immune checkpoint inhibitors restore antitumor immunity by blocking CTLA-4 and PD-1/PD-L1 at their respective stages. This reactivation enables sustained immune- mediated tumor control across multiple solid malignancies.
References
- Sharma, P., & Allison, J. P. (2015). The future of immune checkpoint therapy. Science, 348(6230), 56-61.
- Ribas, A., & Wolchok, J. D. (2018). Cancer immunotherapy using checkpoint blockade. Science, 359(6382), 1350-1355.
- Chen, D. S., & Mellman, I. (2013). Oncology meets immunology: the cancer-immunity cycle. immunity, 39(1), 1-10.
- Binnewies, M., Roberts, E. W., Kersten, K., Chan, V., Fearon,D. F., Merad, M., ... & Krummel, M. F. (2018). Understanding the tumor immune microenvironment (TIME) for effective therapy. Nature medicine, 24(5), 541-550.
- Peshin, S., Bashir, F., Kodali, N. A., Dharia, A., Zaiter, S., Singal, S., & Moka, N. (2025). Immunotherapy in GI cancers: lessons from key trials and future clinical applications. Antibodies, 14(3), 58.
- Modi, S., Peshin, S., & Gim, G. (2024). ESOPECtrial: enhancing treatment approaches for esophageal adenocarcinoma. Journal of Cancer Metastasis and Treatment, 10, N-A.
- Ramachandran, R., Cannon, M., Peshin, S., Kundranda, M., & Scott, A. J. (2025). Unveiling ctDNA Response: Immune Checkpoint Blockade Therapy in a Patient with POLE Mutation-Associated Early-Onset Colon Cancer. Current Oncology, 32(7), 370.
- Bluthgen, M. V., Basté, N., & Recondo, G. (2020). Immunotherapy combinations for the treatment of patients with solid tumors. Future Oncology, 16(23), 1715-1736.
- Barbari, C., Fontaine, T., Parajuli, P., Lamichhane, N., Jakubski, S., Lamichhane, P., & Deshmukh, R. R. (2020). Immunotherapies and combination strategies for immuno- oncology. International journal of molecular sciences, 21(14), 5009.
- Lopez de Rodas, M., Villalba-Esparza, M., Sanmamed,M. F., Chen, L., Rimm, D. L., & Schalper, K. A. (2025). Biological and clinical significance of tumour-infiltrating lymphocytes in the era of immunotherapy: a multidimensional approach. Nature Reviews Clinical Oncology, 22(3), 163-181.
- Sun, S., Liu, L., Zhang, J., Sun, L., Shu, W., Yang, Z., ...& Zhang, Z. (2025). The role of neoantigens and tumor mutational burden in cancer immunotherapy: advances, mechanisms, and perspectives. Journal of Hematology & Oncology, 18(1), 84.
- Raut, G., & Sharma, S. (2025, June). Trends in FDA Drug Approvals: 2021-2024 Insights & Innovations. In Annales Pharmaceutiques Françaises. Elsevier Masson.
- Garg, P., Pareek, S., Kulkarni, P., Horne, D., Salgia, R., & Singhal, S. S. (2024). Next-generation immunotherapy: advancing clinical applications in cancer treatment. Journal of Clinical Medicine, 13(21), 6537.
- Kumar, A., Emdad, L., Das, S. K., & Fisher, P. B. (2024). Recent advances and progress in immunotherapy of solid cancers. Advances in Cancer Research, 164, 111-190.
- Sun, Q., Hong, Z., Zhang, C., Wang, L., Han, Z., & Ma, D. (2023). Immune checkpoint therapy for solid tumours: clinical dilemmas and future trends. Signal transduction and targeted therapy, 8(1), 320.
- Althafar, Z. M. (2025). Engineered immune cell therapies for solid tumors: pharmacological advances, clinical outcomes, and future directions. Frontiers in Pharmacology, 16, 1614325.
- Albarrán, V., San Román, M., Pozas, J., Chamorro, J., Rosero, D. I., Guerrero, P., ... & Soria, A. (2024). Adoptive T cell therapy for solid tumors: current landscape and future challenges. Frontiers in immunology, 15, 1352805.
- Zhao, T., You, J., Wang, C., Li, B., Liu, Y., Shao, M., ... &Zhou, C. (2025). Cell-based immunotherapies for solid tumors: advances, challenges, and future directions. Frontiers in Oncology, 15, 1551583.
- Wang, Y., Ding, Q., & Wei, J. (2025). Current advances and future directions of combined ICIs and TILs in solid tumors. Cancer Letters, 218145.
- Menon, S., Shin, S., & Dy, G. (2016). Advances in cancer immunotherapy in solid tumors. Cancers, 8(12), 106.
- Janjigian, Y. Y., Shitara, K., Moehler, M., Garrido, M., Salman, P., Shen, L., ... & Ajani, J. A. (2021). First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. The Lancet, 398(10294), 27-40.
- Peshin, S., Takrori, E., Kodali, N. A., Bashir, F., Gibson, M., & Singal, S. (2025). Therapeutic Frontiers in Gastroesophageal Cancer: Contemporary Concepts in Management and Therapy. International Journal of Molecular Sciences, 26(23), 11424.
- MATTERHORN Investigators, (2024). Perioperative durvalumab plus FLOT in resectable gastric and gastroesophageal junction cancer (MATTERHORN). N Engl J Med, 390(10): 900–915.
- Ajani, J. A. (2024). Perioperative pembrolizumab plus chemotherapy in gastric/GEJ adenocarcinoma (KEYNOTE-585). J Clin Oncol, 42(15), 1800.
- Sun, J. M., Shen, L., Shah, M. A., Enzinger, P., Adenis, A., Doi, T., ... & Kato, K. (2021). Pembrolizumab plus chemotherapy versus chemotherapy alone for first-line treatment of advanced oesophageal cancer (KEYNOTE-590): a randomised, placebo- controlled, phase 3 study. The Lancet, 398(10302), 759-771.
- André, T., Shiu, K. K., Kim, T. W., Jensen, B. V., Jensen,L. H., Punt, C., ... & Diaz Jr, L. A. (2020). Pembrolizumab in microsatellite-instability–high advanced colorectal cancer. New England Journal of Medicine, 383(23), 2207- 2218.
- Peshin, S., Takrori, E., Kodali, N. A., Bashir, F., & Singal,S. (2025). Advances in the management of pancreatic cancer: Current strategies and emerging therapies. International Journal of Molecular Sciences, 26(15), 7055.
- Finn, R. S., Qin, S., Ikeda, M., Galle, P. R., Ducreux, M., Kim, T. Y., ... & Cheng, A. L. (2020). Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. New England Journal of Medicine, 382(20), 1894-1905.
- Abou-Alfa, G. K., Lau, G., Kudo, M., Chan, S. L., Kelley, R. K., Furuse, J., ... & Sangro, B. (2022). Tremelimumab plus durvalumab in unresectable hepatocellular carcinoma. NEJM evidence, 1(8), EVIDoa2100070.
- Gujarathi, R., Peshin, S., Zhang, X., Bachini, M., Meeks,M. N., Shroff, R. T., & Pillai, A. (2025). Intrahepatic cholangiocarcinoma: Insights on molecular testing, targeted therapies, and future directions from a multidisciplinary panel. Hepatology Communications, 9(7), e0743.
- ASCO Annual Meeting 2025, 2025, Immunotherapy in hepatobiliary and GI malignancies. J Clin Oncol, 43(16_ suppl): e14663.
- ASCO Annual Meeting 2025, 2025, Emerging immunotherapy combinations in GI cancers. J Clin Oncol, 43(16_suppl): e20627.
- Schmid, P. (2024). Pembrolizumab for early triple-negative breast cancer (KEYNOTE-522). N Engl J Med, 390(5): 400.
- Jacob, S. L., Huppert, L. A., & Rugo, H. S. (2023). Role of immunotherapy in breast cancer. JCO Oncology Practice, 19(4), 167-179.
- Celi, C. V., Peshin, S., Dharia, A., Bashir, F., & Erica, L. (2025). Exceptional Response to Pembrolizumab in Metastatic ER+/HER2− Breast Cancer with Liver Metastases: A Case Report and Literature Review. Case Reports in Oncological Medicine, 2025(1), 7970572.
- Motzer, R. J., Tannir, N. M., McDermott, D. F., Arén Frontera, O., Melichar, B., Choueiri, T. K., ... & Escudier,B. (2018). Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. New England Journal of Medicine, 378(14), 1277-1290.
- Rini, B. I., Plimack, E. R., Stus, V., Gafanov, R., Hawkins, R., Nosov, D., ... & Powles, T. (2019). Pembrolizumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. New England Journal of Medicine, 380(12), 1116-1127.
- Peshin, S., Dharia, A., Moka, N., & Skelton IV, W. P. (2025). A Review of Immunotherapy in Renal Cell Carcinoma: Current Landscape and Future Directions. Cancers, 17(19), 3139.
- Choueiri, T. K., Tomczak, P., Park, S. H., Venugopal, B., Ferguson, T., Symeonides, S. N., ... & Powles, T. (2024). Overall survival with adjuvant pembrolizumab in renal-cell carcinoma. New England Journal of Medicine, 390(15), 1359- 1371.
- Forde, P. M., Spicer, J., Lu, S., Provencio, M., Mitsudomi, T., Awad, M. M., ... & Girard, N. (2022). Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. New England Journal of Medicine, 386(21), 1973-1985.
- Wakelee, H. A. (2024). Perioperative pembrolizumab plus chemotherapy in resectable NSCLC (KEYNOTE-671). Lancet, 403(10430): 900–915.
- Mitsudomi, T., Heymach, J. V., Reck, M., Taube, J. M., Gao, S., Horio, Y., ... & Harpole, D. (2023). OA12. 05 surgical outcomes with neoadjuvant durvalumab+ chemotherapy followed by adjuvant durvalumab in resectable NSCLC (AEGEAN). Journal of Thoracic Oncology, 18(11), S71-S72.
- Krishnamoorthy, M., Lenehan, J. G., & Maleki Vareki, S. (2021). Neoadjuvant immunotherapy for high-risk, resectable malignancies: scientific rationale and clinical challenges. JNCI: Journal of the National Cancer Institute, 113(7), 823-832.
- Keung, E. Z., Ukponmwan, E. U., Cogdill, A. P., & Wargo,J. A. (2018). The rationale and emerging use of neoadjuvant immune checkpoint blockade for solid malignancies. Annals of surgical oncology, 25(7), 1814-1827.
- Ferris, R. L. (2025). Neoadjuvant and adjuvant pembrolizumab in resectable head and neck squamous cell carcinoma (KEYNOTE-689). N Engl J Med, 392(1): 100.
- Powell, M. A., Auranen, A., Willmott, L. J., Gilbert, L., Black, D., Cibula, D., ... & Mirza, M. R. (2024). 37MO Dostarlimab plus chemotherapy in primary advanced or recurrent endometrial cancer (pA/rEC) in the RUBY trial: overall survival (OS) by MMR status and molecular subgroups. ESMO open, 9.
- Eskander, R. N., Sill, M. W., Beffa, L., Moore, R. G., Hope, J. M., Musa, F. B., ... & Aghajanian, C. (2023). Pembrolizumab plus chemotherapy in advanced endometrial cancer. New England Journal of Medicine, 388(23), 2159-2170.
- Tewari, K. S. (2024). Pembrolizumab plus chemoradiotherapy in locally advanced cervical cancer. Lancet, 403(10435): 1500.
- Arora, S., Velichinskii, R., Lesh, R. W., Ali, U., Kubiak, M., Bansal, P., ... & Boumber, Y. (2019). Existing and emerging biomarkers for immune checkpoint immunotherapy in solid tumors. Advances in therapy, 36(10), 2638-2678.
- Routy, B., Le Chatelier, E., Derosa, L., Duong, C. P., Alou,M. T., Daillère, R., ... & Zitvogel, L. (2018). Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science, 359(6371), 91-97.
- Gopalakrishnan, V., Spencer, C. N., Nezi, L., Reuben, A., Andrews, M. C., Karpinets, T. V., ... & Wargo, J. (2018). Gut microbiome modulates response to anti–PD-1 immunotherapy in melanoma patients. Science, 359(6371), 97-103.
- Zaretsky, J. M., Garcia-Diaz, A., Shin, D. S., Escuin- Ordinas, H., Hugo, W., Hu-Lieskovan, S., ... & Ribas, A. (2016). Mutations associated with acquired resistance to PD-1 blockade in melanoma. New England Journal of Medicine, 375(9), 819-829.
- Cortazar, F. B., Marrone, K. A., Troxell, M. L., Ralto,K. M., Hoenig, M. P., Brahmer, J. R., ... & Leaf, D. E. (2016). Clinicopathological features of acute kidney injury associated with immune checkpoint inhibitors. Kidney international, 90(3), 638-647.
- Peshin, S., Sonar, N., Sadiq, Z., & Kaur, G. (2024).Pembrolizumab-Induced Acute Interstitial Nephritis: Case Report of a Colorectal Cancer Patient. Int J Cancer Rest Ther, 9(2), 01-02.
- Ringel, M. D., Sosa, J. A., Baloch, Z., Bischoff, L., Bloom, G., Brent, G. A., ... & Wirth, L. J. (2025). 2025 American Thyroid Association management guidelines for adult patients with differentiated thyroid cancer. Thyroid®, 35(8), 841-985.
- Lisovska, N. (2022). Multilevel mechanism of immune checkpoint inhibitor action in solid tumors: History, present issues and future development. Oncology Letters, 23(6), 190.
- Zhang, M., Liu, C., Tu, J., Tang, M., Ashrafizadeh, M., Nabavi, N., ... & Liu, S. (2025). Advances in cancer immunotherapy: historical perspectives, current developments, and future directions. Molecular Cancer, 24(1), 136.
- Peshin, S., Modi, S., & Singh, S. (2024). Advancements in cancer immunotherapy: a comprehensive review of immune checkpoint inhibitors with a focus on pembrolizumab and emerging strategies. Medi Clin Case Rep J, 2(3), 430-434.