Review Article - (2025) Volume 10, Issue 3
The Possible Role of Glial Cells in Processing Non-Automatic Responses
Received Date: Jul 18, 2025 / Accepted Date: Aug 11, 2025 / Published Date: Aug 22, 2025
Copyright: ©2025 Roberto Dotti. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Dotti, R. (2025). The Possible Role of Glial Cells in Processing Non-Automatic Responses. Int J Psychiatry, 10(3), 01-11.
Abstract
Taking inspiration from recent findings about properties and role of the glial cells in variously modulating the activity of the nervous system – especially the CNS –, I sketch an ideal system formed by two structures that result functionally interconnected. On a first level, there is a wired network the operation of which should resemble that of a neural network, while on a second level an autonomous network – with structural and functional features similar to the ones of the presynaptic glial cells of the CNS – spreads over the former and, in determinate conditions, leads its functioning according to a global systemic need for internal activation equilibrium that can be somehow assimilated to homeostasis. So, what I want to highlight, although in a very simplified way for now, is that these networks cooperate with different roles and mechanisms in the constant pursuit of the integrity of the system as a whole and that, over the time, they are mutually subject to structural enhancements and functional reinforcements making them evolutionary entities in all respects.
My purpose is hence to outline the basic functioning of an ideal system that reacts to external and internal stimuli affecting it by activating its response networks in such a way that the activation state of the latter within certain physiological parameters (equilibrium state or cenesthesia) denotes the free-disturbance condition that can be pursued and achieved exactly through more or less complex activation patterns and the final effects resulting from them. In my perspective, such a system should formally represent what neurons and glial cells do – each according to their own functional properties – whenever some environmental or internal disturbance affects it by triggering a first reaction in terms of network activation beyond a certain equilibrium level and thus forcing it to organize responses, within the networks themselves, to return to an equilibrium state.
Introduction
Since, in order to grasp the criteria according to which the ideal system sketched here works, it is preliminarily helpful to determine how glial cells could interact with neurons in the physiology of the real nervous system, let us first see briefly and summarily what is to date known about glia and some of their specific properties that could in fact give them a significant role in managing and implementing higher neurological dynamics such as decision- making processes and other non-automatic response processes. For this purpose, we can consider the following morphological, physiological and functional features revealed over the past few decades1.
I) Glia transmit information by means of calcium waves (Ca2+); this ion is released inside the cells as the result of appropriate external chemical stimulations on specific receptors, and thanks to the work of internal second messengers, or conveyed from cell to cell through gap junction connections.
II) Glia communicate with neurons in various ways, both at the synaptic level and more generally at the structural level. This communication occurs mostly using the same transmitters used by neurons to communicate each other.
III) Glia have a modulatory power on the neural activity which they exert both at the synaptic level and at the network level, thus contributing in various ways to regulate the global functioning of neural transmission.
IV) Glia, specifically astrocytes, can communicate with many neurons even located distally from each other, thus constituting a sort of glial-neural hyper connection that adds to the one structured on the wiring of the neurons themselves.
V) The Ca2+ flow can travel in both directions within the cell. This is to say that, unlike what occurs within the neurons where the ionic flow only follows the body-terminal direction, glia have the advantage of being able to communicate in tandem with each other.
VI) Astrocytes exhibit a certain phenotypic variety that is most likely correlated to a functional ductility about which, however, little is known at present.
VII) Thanks to these (and other) properties, astrocytes seem to be in the condition to constitute a real ubiquitous communication network and, through it, to detect and control nervous processes occurring even distally from each other according to a unitary systemic operation.
The idea is that these general and fundamental properties, acquired by evolution, give glia the ability to develop a ubiquitous (wide- ranging) internal communication network and, thanks to it, to variously control neuronal activity in compliance with global systemic requests the satisfaction of which is imposed on the several districts active in a given temporal context in order to adequately respond to stimuli or disturbances and regain a state of equilibrium
N-G System
The N-G System described below is a purely ideal and formally defined system that should somehow reproduce what occurs inside the CNS, in response to perturbing stimuli of various origins, by virtue of a coordinated work of neurons and glia, each according to their specific functional competences.The N-G System is formed by:
i) a wired network N = [N →], in which each connection n → m between two elements of N is: (a) unidirectional 2; (b) excitatory (+) or inhibitory (-) in output with respect to the element m reached by → and (c) subjected to potentiation and weakening depending on its use and
ii) a network G = [G ↔], in which: (a) each connection g ↔ g between two elements of G is bidirectional; (b) each g is structurally and functionally linked to multiple elements n by means of →; (c) each g can be activated by → and it can in turn exert a modulatory effect on → that can be either exciting or inhibiting in accordance with the operational requests of the context; (d) G is divided into groups of g interconnected by↔ (denoted by G*) that form ubiquitous communication networks spread across the n→ connections to which their components g are structurally and functionally linked and (e) each G* can be subjected to structural extensions depending on the effective dynamics occurring over the time between the g and n structurally connected and functionally interacting3. The fine structure of the N-network is then established through the following definitions
[Def. 1]
Let us define line in N a structure L such that:
i) if n and m are elements of N then L = n → m is a line in N;
ii) if L1 and L2 are lines in N then L = L1 → L2 is a line in N;
iii) if L1 …. Ln are lines in N then L = L1 → Ln & L2 → Ln & ….& Ln-1 → Ln is a line in N;
iv) if L1 and L2 are lines in N then L = L1 → L2 & L2 → L1 is a line in N – i.e. a line can be formed by lines reciprocally connected;
v) nothing else is a line in N
[Def. 2]
Given two lines L and L’ in N, let us define L’ convergent on L – shortly L’-convL – when:
i) L’ → L (in this case L’ is said immediately convergent on L) or
ii) for some L°convL, L’ → L°..
Given a line L, let us define convergence point of L – shortly convL – the line L* in L such that, for every line L’ in L, L* is not convergent on L’. Intuitively, L* represents the final result of the entire wiring of L.
[Def. 3]
Every line L has an activation level that can vary from 0 to a maximum4. Symbolically expressed as:
0 ≤ actL ≤ max.
Such a value depends on the global activation state of N at a given time tx. Thus we can put actLtx as the activation level of L at the time tx.
[Def. 4]
Given L and L’ such that L’ → L and given actLtx, let us put act*L’actLtx as the minimum activation level that L’ must have to change the activation level of L at the time tx.
[Corollary to Def. 4]
In general, if L’convL, the symbolic expression act L’ actLtxdenotes the minimum activation level that L’ must have to change the activation level of L at the time tx.
Based on what has been just defined, we can go on and say that the N-G System basically works with the aim of maintaining a state of activation internal to N in which every line L in N:
i) discharges its activation out of L itself or
ii) exhausts the amount of activation within itself;
iii) a stagnation tolerance value stag* inside L is admitted.
Such a condition is called here coenaesthesia and it has to be spontaneously reached within a determinate time before the system enters a state of disorder to overcome which it must elaborate appropriate and more or less complex reactive responses. This is to say that N-G does not admit any prolonged and localized stagnation of activation, above a given tolerance value. I.e., for every L:
stagL ≤ stag*5.
We have to emphasize that stagnation does not equate to prolonged activation flow in L. The latter represents the dynamics in which the several L’ conv L rhythmically acquire and discharge or exhaust activation, which does not cause any overall disorder in the N-G system. A stagnation in L occurs when, for some reason, there is no activation turnover and this condition can essentially take three different forms:
1) an activation conflict between two lines immediately convergent on convL; which can occur when these two lines are both excitatory on convL but reciprocally inhibiting and they have the same activation level;
2) a persistent activation on convL which however turns out to be too weak to find a discharge or exhaustion through the latter and
3) an amount of incoming activation that exceeds the discharge or exhaustion capabilities of the convL.
When a line L fails to discharge or exhaust within a certain time, and thus enters a state of disorder at a certain time tx (symbolically denoted by DLtx), the N-G system responds by triggering dynamics of reorganization of the global activation framework inside its structure N in order to regain k (coenaesthesis) and it does this involving not only the level of neural processing but, if it comes out to be necessary, also the glial one, each according to different operating times.
The first step to overcome the disorder state is a competence of N itself. This is because, in the real model provided by the animal CNS, being the neural information processing and transmission times much faster than the glial ones, it is physiologically normal for neurons to react first. So, faced to a DN’6 , N starts to seek some solution consisting in a rearrangement of the global activation lines that is able to unlock the activation impasse occurring in L through a feedback path having a final discharging or exhausting effect on L itself. By virtue of its rather rigid wiring, N can act in a one-way direction only starting by L and following the connection pathways signed by →. This structurally constrained modus operandi inevitably imposes that N only searches paths being somehow already marked inside the wiring →, although not yet factually used, and strictly centripetal from L. It means that a reaction to DL leaded by N can only be aimed at finding solution strategies arising from the involvement of centered on L →-paths that result to be more or less use-strengthened at the time of processing. Thus, in psychological classification, a response pursued in this way could be included in instinct, automatism or in some form of response processing immediately deriving from these. In this dimension, there seems to be very little room for a search for responses that are somehow creative and potentially alternative to each other. The N-processing – similar to neural one – works in a rather rigid or stereotyped way and can thus only slightly extend the repertoire of possible responses in the ongoing context. As in action-oriented decision-making or pure speculative thinking, the N-G system also requires a greater procedural flexibility to obtain a wider range of possible responses to its inner problems – what is identified here as L-line activation disorder.
Principle Of N-Dominance (Over G)
Before seeing when and how the G component comes into play to deal with a DL, we need to make explicit a hierarchy existing within the N-G system7. N exerts a functional dominance over G and this dominance occurs as:
1) a faster processing speed: each activation in N precedes and triggers a related activation in G – each n is functionally related to an g and each g can be related to more than one n8 – ;
2) temporally prioritized approach of N to disorder states within itself; 3) dependence of any modulatory activity of G on N on any other autonomous activation state occurring in N that affects the former. The third constrain simply expresses the inner dynamics according to which G can modulate N-activities being somehow already started and defined but it cannot determine anything in N motu proprio. This is why any autonomous or independent N activity can interrupt or modify an ongoing G-modulatory activity at any time of processing, forcing G to reorganize its functional responses.
Therefore, as said above, the system N-G primarily reacts to a disorder state in a line L in N at the time tx – i.e. a state in which stagL > stag* – tackling the problem in N itself, according to the processing mechanisms conditioned by the wiring of N and in N-times being much faster than the ones of G. Shortly and symbolically:
DLtx => N-reaction in NT (with NT just donating N-times)
However, as soon as N is activated to react to DL, the g-components of G structurally and functionally linked to the n-components of N forming conv L9 are also activated and their activations follow the evolution of the N-processing that searches for a solution to DL. If, after a certain amount of time, N is not able to find a solution, entering into a sort of impasse, and if this time is enough for G to trigger its own internal communication10 , then the latter comes into play and, through a significantly different procedure strategy, faces the problem. Starting from the DL with respect to which N failed, a specific G*L network begins its task. Shortly and symbolically:
DLtx + NT = GT N-failure => G*L (with GT denoting G-times)
What G*L exactly is and how it functions just represent the core of the ideal N-G system described here. In this regard, it has to be firstly emphasized the supporting structure of G*L consists of. So, we can say that G*L is a g-network formed by:
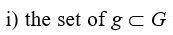
that are structurally and functionally linked to the n→ connections basically forming L in all its convergences and
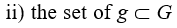
that are structurally connected – by means of ↔ – to the basic g-units of G*L11 and that can actually, at the time DLtx+NT=GT , communicate with them12.
Point ii) establishes something that is crucial for the structural and functional nature of a G*L network. It expresses the temporally determined limitation of G*L – which can however receive extension and enhancement through its actual use, as we will see shortly. More specifically, point ii) says that, at the time when DL occurs and the G-processing comes into play, the basic g-units – i.e. the g-units linked to the N-center from which G*L develops – are able to effectively communicate with only a part of the g-units in G with which are structurally connected. They, as their ideal glial counterparts, begin their functional life with a fundamentally defined endowment (genotypic heritage), but it is then through practice that the potential can be more or less and in somewhat different forms actualized (phenotypic expression).
Thus, if this is the basic architecture of a G*L network, we have now to see how it works once it starts its task temporally and in the merit consequent to that carried out by N without success. In this regard, we can first say that G, just like N, does not tolerate any prolonged and stagnant activation states within itself. It spontaneously looks for its coenaesthesis and a DL inevitably causes stagnation of activation in the g linked to the n → composing L, that is, it causes a departure from coenaesthesis in a certain area of G. A G*L network therefore has the precise task of seeking to break the deadlock in N in order to regain its own inner equilibrium and to do so by the tools that G is equipped with.
Let us now see according to which procedural mechanisms a G*L network acts, reiterating that it comes into operation exactly starting from a precise DL left by N without any satisfactory coenaesthesis solution, that is, from a DL in which the L to be considered can also be an unsuccessful reworking of a first problematic L’. Anyhow, in compliance with the principle of N-dominance, G*L takes control of the process starting from:
DLtx + NT = GT
Imagining how real glial cells (specifically astrocytes) might actually behave in such a dynamic – based on what is known to date about their physiological characteristic and functional properties13 – we can describe the progression of the work a G*L network by dividing it into several phases, each with particular implementation modes and determined aims. What is now being explained refers, for the sake of simplicity, to a single ideal focus L of activation disorder, but the discussion could obviously also be extended to a condition in which there are multiple foci of activation disorder existing simultaneously in the N-G system.
Phase I
In the first phase, the G*L network carries out a work of selection among several possible intervention directions, in order to then operate in a targeted manner in attempt to resolve the activation disorder contained in L. This phase is in turn divided into several procedural steps.
Step 1 – As first step, the G*L network has to search the several pathways n1 → n2 → … which can be identified by the g-units being actually communicating – through ↔ – with the coordination center constituted by the g-units linked to the n → in L14. The number and length of the pathways n → … just identified depends precisely on the current extent of communication within G*L and originating from the L-linked g-units.
Step 2 – In the first step G*L defines the map of all the pathways n→ … that can be reached by the g↔-network centered on L and its current extension in communication capacity. The second step of the G*L process is to identify the lines L1 … Ln that:
i) can be formed starting from the n→ …-map previously defined and
ii) can have some feedback on L, i.e. can somehow modify the activation state of L. This is to say that G*L seeks the inherent-map lines that could provide some effect on L in case of activation.
Step 3 – The third step requires that the structure G in general – and thus even a G*L network in particular – owns the specific property to distinguish for every basic connection n → m in N whether it is excitatory (+) or inhibitory (-), i.e. whether it tends to activate or to negatively modulate what comes next within the same pathway15 →15. By virtue of this capacity, G*L makes a selection of the Lx identified in the previous step. Specifically, it has to select only the Lx such that:
DLx ≤ DL
i.e. it has to give up, within the limits of its tools and according to its current communication power, any potential feedback that can increase the ongoing disorder condition. This selection can be done just following the →-wiring that forms the Lx and assessing the possible global effect of the single +/− →-connections present in it in light of the general activation state of N in which the whole G also is involved due to its close structural and functional relationship16. The processing criterion underlying this operation is trivially to avoid, in the phase of active intervention of G*L on N – that we will see later –, favoring lines that would worsen the situation rather than alleviate17.
Step 4 – The fourth step is strategically tied to the third one and its implementation requires a further faculty with which G is provided – again in analogy with its biological counterpart represented by astrocytic cells –. It is hence imagined that the g-components of G, each of which is linked to more than one n → m connection of N, are equipped with the capacity to detect and register the strength of the n → m activation transmission, i.e. the ease with which the activation flow passes from n to m18. This property is used by G*L to perform an additional selection on the lines Lx chosen in compliance with the criterion described just above. In fact, the several Lx selected in that way do not cause more disorder if assessed independently of each other. However, it can occur that two or more lines cause an increase in disorder if simultaneously considered or that the one negatively interferes with the other, that is:
DLx & Ly & …> DL or
actLx & actLy & … < actLx aut actLyaut 19 …
Such a danger has thus to be averted. To this end, G*L has to select among the lines that, if activated together, would cause disorder or would reciprocally interfere and give priority to that or those that appear to be stronger, i.e. easier to bring to a state of activation sufficient to have some effect on L20. If two or more lines to be selected according the criterion of strength in connections come out to be equally strong then G*L can resort to a random choice, without thereby modifying anything, as far as it can be established in this phase of mapping and preliminary selection. The first phase, that ends with the identification of a certain number of lines L1 … Ln having determined characteristics, gives then way to a second phase in which the G*L network assumes an active role on N.
Phase II
In this second phase of the G*L’s work, G’s claimed property to modulate the →-connections in N, both positively and negatively, comes into play21. The aim of G*L is to:
i) induce the lines L1 … Ln previously chosen to switch from actLx < act* to actLx ≥ act*, i.e. lead the various Lx lines until an activation state such that they can change22 the activation state of L through some feedback and
ii) keep any line L’ identified as potentially disordering or disturbing, according to a previous selection, in an activation state actL’ < act* 23.
The first task consists of a positive modulation24 on the →-connections in Lx in order to increase its activation level, while the second and parallel work is configured as a negative modulation25 exerted on the →-connections that do not belong to any Lx and that contribute to forming some L’ given up as potentially disordering or disturbing.
The G*L modulation process, just because it is defined on the basis of what glial cells could do in the CNS, needs to be clarified in more detail.
First of all, by virtue of the principle of N-dominance, what G*L does can at any moment be subverted by what independently of G*L – in a spontaneous way or induced by external factors – occurs within the activation dynamics affecting N. This means that both the positive modulations and the negative ones could be interrupted at every time of the G*L-processes if the activation framework in N network changes in such a way as to make the operations performed by G to solve an activation disorder problem in L no longer relevant due to modified needs of the system itself centered on N. G, with its local G*.. networks, always chases the events in N and starts each time from what N leaves unsolved.
A second point to be highlighted is that G*L is not able to generate an immediate activation on the →-connections just as is not able to produce a deactivation26. Therefore, G*L positively modulates each →- connection forming a line Lx so that N itself finally reaches an activation level sufficient to provide feedback on L. In parallel, G*L negatively modulates all the →-connections extraneous to each Lx and converging on some potentially disturbing L’ so that they remain at an activation level not sufficient to modify what occurs in Lx by the positive work of G*L itself, but it cannot block a possible activation exploit arising autonomously from N.
Furthermore, and very importantly, a G* network is functionally limited, that is – as said above – it has an actual communication range that only partially covers the potentially available ↔-connection structure in which it is inserted27. For this reason, the mapping through which G*L identifies the lines L1 … Ln to be supported reaches as far as the g-units centered on L can actually communicate. It follows from this premise that any Lx could in turn be convergent on some other L° – i.e. Lx-convL° – not identified by G*L at the time of processing and such that, once activated thanks to the G*L modulation, it gives feedback also unexpected by G*L mapping – i.e. L° originates effects that escape the selection process of G*L due to the current limitations of communication among g-units –.
Well, pointed up these essential details, we have enough items about the matter to see what specific results the integrated processes of mapping-selection and modulation carried out by the G*L network can produce. Remembering that a G*L modulation is a slow job that tends to stimulate N to eventually and definitively trigger the activation of the Lx previously chosen, we can imagine that such a job exactly starts from the sources →28 of every L – and then continues along the directions imposed by the →-wiring of Lx – as soon as the selection of all the acceptable lines is concluded according to the criteria of completeness and caution seen before29 and we can also imagine that the several Lx get activation at different times depending their structural complexity and internal connection strength. However, once Lx is activated, it can happen that Lx converges on some other L° – i.e. Lx-convL° – and so does every other Ly by determining a new and possibly unexpected30 global activation framework in N – that we can symbolically put as G*N –. This possibility makes the final pattern achieved by a G*L-mapping-modulation cycle the following:
Lx & Ly & .. convG*N.
It is to be pointed out that a G*N can be achieved by a single line or by multiple lines. This depends on the times need by G*L modulation to bring the various lines to an activation level act ≥ act*. If the times are such as to trigger N-responses in parallel then two or more lines can originate a single G*N. In any case, the end of a G*L mapping-modulation cycle, barring independent and alternative N-activities occurring during the G*L processing time31, comes when some G*N is triggered at a sufficient N-activation level, thus falling again within the exclusive competence of the N-processing with its TN execution time A G*N can then essentially present three distinct and alternative outcomes in terms of the general activation state resulting in N and including a new activation state just for L32 , namely:
a) stagG*N ≤ stag*;
b) stagL > stagG*N > stag* or
c) stagG*N ≥ stagL > stag*.
Keeping in mind that processing is now back under the control of N, we can think of G as entering a standby state waiting for exactly what N can decide. In the first case, in accordance with the principle of regained coenaesthesis, N will tend to exhaust any targeted reactive process and, for the same reason, G will also cease its own activity. Instead, in the second and third cases, N will be forced to continue the search for an internal reorganization to overcome the activation impasse still in place, although with different pressure in the two respective contexts. So, both in the case b) and in the case c) N takes control of the situation but does so starting from a different framework compared to the temporally previous one represented by DL. A different framework for two reason. First, the origin of the coenaesthesis disorder is now somewhere in G*N and no longer or no longer necessarily centered right and only on L. Furthermore, and most importantly, both the N-wiring and G-connection network have undergone modifications due just to the N-G*L processing cycle recently concluded. The lines L1 … Ln positively modulated by G*L, regardless of whether they have reached full activation or not, have anyway been reinforced, through use, in their →-connections and are therefore now more identifiable, exploitable and avoidable – depending on contextual convenience – by N itself that hence obtains a functional improvement from its systemic counterpart G. On the other hand, any possible further activation produced by N and going beyond the functional limits of G*L – i.e. an event like the one represented here as G*N – brings with it a parallel activation of the g-units linked to the →-connections affected by the process. This in turn determines an expansion of the effectively available communication network among g-units that were previously only structurally connected by ↔. As if N, through G*N, acted as a radio bridge between the g-units of G*L and those linked to G*N. Schematically:
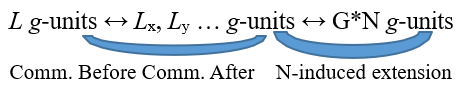
But that’s not all. The very use of communication among g-units in G*L makes the communication itself stronger, in the sense that the information, conveyed through the ↔-connections, can flow more easily and therefore, with the same amount of activation, the message can reach further and spread with a wider radius through the pathways defined by ↔. In this way g-units, that were not reachable from other g-units before a certain practical work, subsequently become reciprocally accessible precisely thanks to the functional reinforcement deriving from use33. As we can notice, the G structure in its whole gets a functional enrichment from two sources and both coming from the activity of its G*.. networks: on the one hand, G is passively improved by the extension G*N occurring in N and promoted by the G*.., on the other hand, as just highlighted, an autonomous strengthening originates from the actual activity of any G*.. itself in performing its own coenaesthesis task. So, in case of a second G cycle starting from G*N at the time tx + TN = TG – i.e. a cycle denoted as G*(G*N)tx + TN = TG –, G*(G*N) network will be in the condition to count on a wider and hence functionally better potential of effective communication among g-units.
Each coenaesthesis cycle in the N-G system of the N-G*.. –N type determines a mutually stimulated evolution in the two subsystems N and G that forms it.
Final Remarks
To conclude we can simply reiterate what the N-G system wants to be and try to show what utility it could have. First of all, the structure of N-G is ideally and roughly derived from what is hypothesized to be a possible functional interaction between the glial cells and neurons in the CNS aimed at seeking and implementing non-automatic responses to events that affect the system itself in a somewhat unbalancing way. We say possible interaction because little is still known about the conjugated systemic behavior of glia and neurons. To date many scientific clues suggest that such a functional synergy exists, however much still needs to be investigated to confirm this reasonable suggestion. Thus, within a so defined framework, the N-G system is conceived as a theoretical and oversimplified model in which a idealized neural-glial cooperation works in order to preserve or regain a state of dynamic equilibrium inside both components and thus inside the system in its whole. This is to say that N and G are thought as interdependent agents in pursuing their coenaesthesis goal. The model offered here is not intended to represent any specific reactive dynamics that can occur in a biological organism grappling with the problem of finding adequate responses to overcome a somehow disturbing condition. More generally, the N-G system aims at providing a formal image of the mechanisms that might underlie higher processes such as problem solving, decision making or abstract thinking. Of course, without any claim to representative exhaustiveness. What would be desirable is that such a model could be helpful, in a heuristic perspective, for designing targeted research both in the field of neurobiological sciences and in that of artificial neural-(glial)-inspired systems. For my part, my next goal is to outline a mathematical, and therefore procedurally precise, formulation of the functioning of a G*.. network, as it has been conceived here in its active dependence – if I may exploit this oxymoron – with respect to what occurs in N [1-95].
Declarations
Ethical approval: no humans or animals were used in any way to develop this work.
Funding
no funding was received for this work.
References
- Aguado, F., Espinosa-Parrilla, J. F., Carmona, M. A., & Soriano, E. (2002). Neuronal activity regulates correlated network properties of spontaneous calcium transients in astrocytes in situ. Journal of Neuroscience, 22(21), 9430- 9444.
- Allen, N. J., & Eroglu, C. (2017). Cell biology of astrocyte- synapse interactions. Neuron, 96(3), 697-708.
- Armbruster, M., Naskar, S., Garcia, J. P., Sommer, M., Kim, E., Adam, Y., ... & Dulla, C. G. (2022). Neuronal activity drives pathway-specific depolarization of peripheral astrocyte processes. Nature neuroscience, 25(5), 607-616.
- Ameroso, D., Meng, A., Chen, S., Felsted, J., Dulla, C. G., & Rios, M. (2022). Astrocytic BDNF signaling within the ventromedial hypothalamus regulates energy homeostasis. Nature metabolism, 4(5), 627-643.
- Araque, A., Martin, E. D., Perea, G., Arellano, J. I., & Buno,W. (2002). Synaptically released acetylcholine evokes Ca2+ elevations in astrocytes in hippocampal slices. Journal of Neuroscience, 22(7), 2443-2450.
- Araque, A., Carmignoto, G., Haydon, P. G., Oliet, S. H., Robitaille, R., & Volterra, A. (2014). Gliotransmitters travel in time and space. Neuron, 81(4), 728-739.
- Baddeley, A. D. (2015). Working memory in second language learning. Working memory in second language acquisition and processing, 17-28.
- Baldwin, K. T., & Eroglu, C. (2017). Molecular mechanisms of astrocyte-induced synaptogenesis. Current opinion in neurobiology, 45, 113-120.
- Bate, C., Boshuizen, R., & Williams, A. (2005). Microglial cells kill prion-damaged neurons in vitro by a CD14 dependent process. Journal of neuroimmunology, 170(1-2), 62-70.
- Becker, S., Nold, A., & Tchumatchenko, T. (2022). Modulation of working memory duration by synaptic and astrocytic mechanisms. PLoS Computational Biology, 18(10), e1010543.
- Benini, A. (2023). Neurobiologia della volontà. RaffaelloCortina Editore.
- Bernardinelli, Y., Randall, J., Janett, E., Nikonenko, I., König, S., Jones, E. V., ... & Muller, D. (2014). Activity-dependent structural plasticity of perisynaptic astrocytic domains promotes excitatory synapse stability. Current Biology, 24(15), 1679-1688.
- Berridge, M. J., Bootman, M. D., & Roderick, H. L. (2003). Calcium signalling: dynamics, homeostasis and remodelling.Nature reviews Molecular cell biology, 4(7), 517-529.
- Bezzi, P., & Volterra, A. (2001). A neuron–glia signalling network in the active brain. Current opinion in neurobiology, 11(3), 387-394.
- Bhandare, A., van de Wiel, J., Roberts, R., Braren, I., Huckstepp, R., & Dale, N. (2022). Analyzing the brainstem circuits for respiratory chemosensitivity in freely moving mice. Elife, 11, e70671.
- Brenneman, D. E., Hauser, J., Spong, C. Y., & Phillips, T. M. (2000). Chemokines released from astroglia by vasoactive intestinal peptide. Mechanism of neuroprotection from HIV envelope protein toxicity. Annals of the New York Academy of Sciences, 921, 109-114.
- Broadhead, M. J., & Miles, G. B. (2020). Bi-directional communication between neurons and astrocytes modulates spinal motor circuits. Frontiers in Cellular Neuroscience, 14, 30.
- Bullock, T. H., Bennett, M. V., Johnston, D., Josephson, R., Marder, E., & Fields, R. D. (2005). The neuron doctrine, redux. Science, 310(5749), 791-793.
- Bushong, E. A., Martone, M. E., Jones, Y. Z., & Ellisman,M. H. (2002). Protoplasmic astrocytes in CA1 stratum radiatum occupy separate anatomical domains. Journal of Neuroscience, 22(1), 183-192.
- Chadwick, D. J., & Goode, J. A. (Eds.). (2006). PurinergicSignalling in Neuron-glia Interactions. John Wiley & Sons.
- Chung, W. S., Allen, N. J., & Eroglu, C. (2015). Astrocytes control synapse formation, function, and elimination. ColdSpring Harbor perspectives in biology, 7(9), a020370.
- Curreli, S., Bonato, J., Romanzi, S., Panzeri, S., & Fellin, T. (2022). Complementary encoding of spatial information in hippocampal astrocytes. PLoS biology, 20(3), e3001530.
- Czeisler, C. M., Silva, T. M., Fair, S. R., Liu, J., Tupal, S., Kaya, B., ... & Otero, J. J. (2019). The role of PHOX2B-derived astrocytes in chemosensory control of breathing and sleep homeostasis. The Journal of physiology, 597(8), 2225- 2251.
- Damasio, A. (2010). Self Comes to Mind. Pantheon, a Division of Random House. Pantheon: New York, NY, USA.
- Damulewicz, M., Doktór, B., Baster, Z., & Pyza, E. (2022). The role of glia clocks in the regulation of sleep in Drosophila melanogaster. Journal of Neuroscience, 42(36), 6848-6860.
- Delgado, L., & Navarrete, M. (2023). Shining the light on astrocytic ensembles. Cells, 12(9), 1253.
- De Pitta, M., & Brunel, N. (2016). Modulation of synaptic plasticity by glutamatergic gliotransmission: a modeling study. Neural plasticity, 2016(1), 7607924.
- Diamond, J., & Diamond, J. M. (2006). The third chimpanzee: the evolution and future of the human animal: HarperPerennial. New York City, NYC, USA.
- Doron, A., Rubin, A., Benmelech-Chovav, A., Benaim, N., Carmi, T., Refaeli, R., ... & Goshen, I. (2022). Hippocampal astrocytes encode reward location. Nature, 609(7928), 772- 778.
- Dzyubenko, E., Prazuch, W., Pillath-Eilers, M., Polanska, J., & Hermann, D. M. (2021). Analysing intercellular communication in astrocytic networks using “Astral”. Frontiers in Cellular Neuroscience, 15, 689268.
- Ferraguti, M., Castellacci, C., & Allegrucci, G. (2011).Evoluzione: modelli e processi. Pearson.
- Fields, R. D. (2005). Making memories stick. Scientific American, 292(2), 74-81.
- Finley, J. R., Naaz, F., & Goh, F. W. (2018). Memory and technology. Cham: Springer International Publishing.
- Fields, R. D. (2006). Beyond the neuron doctrine. ScientificAmerican Mind, 17(3), 20-27.
- Fields, R. D. (2008). White matter in learning, cognition and psychiatric disorders. Trends in neurosciences, 31(7), 361- 370.
- Dotti, R. (2025). Neurons and Glia Cells: Some Mention of their General Interrelationships. Studies in Philosophy of Science and Education, 6(1), 1-40.
- Fields, R. D. (2009). The other brain: From dementia to schizophrenia, how new discoveries about the brain are revolutionizing medicine and science. Simon and Schuster.
- Finke, R. A. (1989). Principles of mental imagery. The MIT Press.
- Dominici, P. (2019, July). Educating for the Future in the Age of Obsolescence. In 2019 IEEE 18th International Conference on Cognitive Informatics & Cognitive Computing (ICCI* CC) (pp. 278-285). IEEE.
- Gordleeva, S. Y., Tsybina, Y. A., Krivonosov, M. I., Ivanchenko, M. V., Zaikin, A. A., Kazantsev, V. B., & Gorban, A. N. (2021). Modeling working memory in a spiking neuronnetwork accompanied by astrocytes. Frontiers in Cellular Neuroscience, 15, 631485.
- Haber, M., & Murai, K. K. (2006). Reshaping neuron–glial communication at hippocampal synapses. Neuron glia biology, 2(1), 59-66.
- Halassa, M. M., & Haydon, P. G. (2010). Integrated brain circuits: astrocytic networks modulate neuronal activity and behavior. Annual review of physiology, 72(1), 335-355.
- Han, J., Kesner, P., Metna-Laurent, M., Duan, T., Xu, L., Georges, F., ... & Zhang, X. (2012). Acute cannabinoids impair working memory through astroglial CB1 receptor modulation of hippocampal LTD. Cell, 148(5), 1039-1050..
- Hirase, H., Qian, L., Barthó, P., & Buzsáki, G. (2004). Calcium dynamics of cortical astrocytic networks in vivo. PLoS biology, 2(4), e96.
- Hirrlinger, J., Hülsmann, S., & Kirchhoff, F. (2004). Astroglial processes show spontaneous motility at active synaptic terminals in situ. European Journal of Neuroscience, 20(8), 2235-2239.
- Hirrlinger, J., & Nimmerjahn, A. (2022). A perspective on astrocyte regulation of neural circuit function and animal behavior. Glia, 70(8), 1554-1580.
- Houades, V., Koulakoff, A., Ezan, P., Seif, I., & Giaume, C. (2008). Gap junction-mediated astrocytic networks in the mouse barrel cortex. Journal of Neuroscience, 28(20), 5207- 5217.
- Iino, M., Goto, K., Kakegawa, W., Okado, H., Sudo, M., Ishiuchi, S., ... & Ozawa, S. (2001). Glia-synapse interaction through Ca2+-permeable AMPA receptors in Bergmann glia. Science, 292(5518), 926-929.
- Ishibashi, T., Dakin, K. A., Stevens, B., Lee, P. R., Kozlov, S. V., Stewart, C. L., & Fields, R. D. (2006). Astrocytes promote myelination in response to electrical impulses. Neuron, 49(6), 823-832.
- Jones, T. A., & Greenough, W. T. (2002). Behavioral experience-dependent plasticity of glial-neuronal interactions. The Tripartite Synapses: Glia in Synaptic Transmission, 248- 265.
- Kandell, E. R., Koester, J. D., Mack, S. H., & Siegelbaum,S. (Eds.). (2021). Principles of neural science. McGraw-Hill.
- Khakh, B. S., & McCarthy, K. D. (2015). Astrocyte calcium signaling: from observations to functions and the challenges therein. Cold Spring Harbor Perspectives in Biology, 7(4), a020404.
- Dotti, R. Glia Cells and Will. Available at SSRN 4959809.
- Koch, C. (2004). The quest for consciousness: A neurobiological approach.
- LeDoux J.; The Emotional Brain: The Mysterious Underpinnings of Emotional Life. Simon & Schuster 1998.
- Lehre, K. P., & Rusakov, D. A. (2002). Asymmetry of glia near central synapses favors presynaptically directed glutamate escape. Biophysical journal, 83(1), 125-134.Libet B.; Neurophysiology of Consciousness. Springer, New York 1993.
- Lopez-Ortiz, A. O., & Eyo, U. B. (2024). Astrocytes andmicroglia in the coordination of CNS development and homeostasis. Journal of Neurochemistry, 168(10), 3599-3614.
- Marella, M., & Chabry, J. (2004). Neurons and astrocytes respond to prion infection by inducing microglia recruitment. Journal of Neuroscience, 24(3), 620-627.
- Markham, J. A., & Greenough, W. T. (2004). Experience- driven brain plasticity: beyond the synapse. Neuron glia biology, 1(4), 351-363.
- Matsui, K., & Jahr, C. E. (2003). Ectopic release of synaptic vesicles. Neuron, 40(6), 1173-1183.
- Matsui, K., & Jahr, C. E. (2004). Differential control of synaptic and ectopic vesicular release of glutamate. Journal of Neuroscience, 24(41), 8932-8939.
- Mauch, D. H., Nagler, K., Schumacher, S., Göritz, C., Muller,E. C., Otto, A., & Pfrieger, F. W. (2001). CNS synaptogenesis promoted by glia-derived cholesterol. Science, 294(5545), 1354-1357.
- Mesejo, P., Ibánez, O., Fernández-Blanco, E., Cedrón, F., Pazos, A., & Porto-Pazos, A. B. (2015). Artificial neuron–glia networks learning approach based on cooperative coevolution. International journal of neural systems, 25(04), 1550012.
- Mu, Y., Bennett, D. V., Rubinov, M., Narayan, S., Yang, C. T., Tanimoto, M., ... & Ahrens, M. B. (2019). Glia accumulate evidence that actions are futile and suppress unsuccessful behavior. Cell, 178(1), 27-43.
- Murai, K. K., Nguyen, L. N., Irie, F., Yamaguchi, Y., & Pasquale, E. B. (2003). Control of hippocampal dendritic spine morphology through ephrin-A3/EphA4 signaling. Nature neuroscience, 6(2), 153-160.
- Nagai, J., Yu, X., Papouin, T., Cheong, E., Freeman, M.R., Monk, K. R., ... & Khakh, B. S. (2021). Behaviorally consequential astrocytic regulation of neural circuits. Neuron, 109(4), 576-596.
- Nett, W. J., Oloff, S. H., & Mccarthy, K. D. (2002). Hippocampal astrocytes in situ exhibit calcium oscillations that occur independent of neuronal activity. Journal of neurophysiology, 87(1), 528-537.
- Oberheim, N. A., Wang, X., Goldman, S., & Nedergaard, M. (2006). Astrocytic complexity distinguishes the human brain. Trends in neurosciences, 29(10), 547-553.
- Oberheim, N. A., Goldman, S. A., & Nedergaard, M. (2011). Heterogeneity of astrocytic form and function. Astrocytes: Methods and protocols, 23-45.
- Oliet, S. H., Piet, R., & Poulain, D. A. (2001). Control of glutamate clearance and synaptic efficacy by glial coverage of neurons. Science, 292(5518), 923-926.
- Parri, H. R., Gould, T. M., & Crunelli, V. (2001). Spontaneous astrocytic Ca2+ oscillations in situ drive NMDAR-mediated neuronal excitation. Nature neuroscience, 4(8), 803-812.
- PATTON, A. P., SMYLLIE, N. J., CHESHAM, J. E., & HASTINGS, M. H. (2022). ASTROCYTES SUSTAIN CIRCADIAN OSCILLATION AND BIDIRECTIONALLY DETERMINE CIRCADIAN PERIOD, BUT DO NOT REGULATE CIRCADIAN PHASE IN THE SUPRACHIASMATIC NUCLEUS. JOURNAL OF NEUROSCIENCE, 42(28), 5522-5537.
- PEREA, G., & ARAQUE, A. (2005). PROPERTIES OFSYNAPTICALLY EVOKED ASTROCYTE CALCIUM SIGNAL REVEAL SYNAPTIC INFORMATION PROCESSING BY ASTROCYTES. JOURNAL OF NEUROSCIENCE, 25(9), 2192-2203.
- Perea, G., Navarrete, M., & Araque, A. (2009). Tripartite synapses: astrocytes process and control synaptic information. Trends in neurosciences, 32(8), 421-431.
- Piet, R., Vargová, L., Syková, E., Poulain, D. A., & Oliet,S. H. (2004). Physiological contribution of the astrocytic environment of neurons to intersynaptic crosstalk. Proceedings of the National Academy of Sciences, 101(7), 2151-2155.
- Reist, N. E., & Smith, S. J. (1992). Neurally evoked calcium transients in terminal Schwann cells at the neuromuscular junction. Proceedings of the National Academy of Sciences, 89(16), 7625-7629.
- Rips, L. J. (2011). Lines of thought: Central concepts in cognitive psychology. Oxford University Press.
- Robitaille, R. (1998). Modulation of synaptic efficacy and synaptic depression by glial cells at the frog neuromuscular junction. Neuron, 21(4), 847-855.
- Rose, C. R., Blum, R., Pichler, B., Lepier, A., Kafitz, K. W., & Konnerth, A. (2003). Truncated TrkB-T1 mediates neurotrophin-evoked calcium signalling in glia cells. Nature, 426(6962), 74-78.
- Rossi, G. (2020). Origin and Evolution of Glia. BM&L-Italy, 20200516.
- Sajedinia, Z., & Hélie, S. (2018). A new computational model for astrocytes and their role in biologically realistic neural networks. Computational intelligence and neuroscience, 2018(1), 3689487.
- Santello, M., Toni, N., & Volterra, A. (2019). Astrocyte function from information processing to cognition and cognitive impairment. Nature neuroscience, 22(2), 154-166.
- Song, H., Stevens, C. F., & Gage, F. H. (2002). Astroglia induce neurogenesis from adult neural stem cells. Nature, 417(6884), 39-44.
- Stork, T., Sheehan, A., Tasdemir-Yilmaz, O. E., & Freeman,M. R. (2014). Neuron-glia interactions through the Heartless FGF receptor signaling pathway mediate morphogenesis of Drosophila astrocytes. Neuron, 83(2), 388-403.
- Su, Y., Wang, X., Yang, Y., Chen, L., Xia, W., Hoi, K. K.,... & Niu, J. (2023). Astrocyte endfoot formation controls the termination of oligodendrocyte precursor cell perivascular migration during development. Neuron, 111(2), 190-201.
- Sul, J. Y., Orosz, G., Givens, R. S., & Haydon, P. G. (2004). Astrocytic connectivity in the hippocampus. Neuron glia biology, 1(1), 3-11.
- Theodosis, D. T., Poulain, D. A., & Oliet, S. H. (2008). Activity-dependent structural and functional plasticity of astrocyte-neuron interactions. Physiological reviews, 88(3), 983-1008.
- Tognatta, R., Karl, M. T., Fyffe-Maricich, S. L., Popratiloff, A., Garrison, E. D., Schenck, J. K., ... & Miller, R. H. (2020). Astrocytes are required for oligodendrocyte survival and maintenance of myelin compaction and integrity. Frontiers in cellular neuroscience, 14, 74.
- Volterra, A., & Meldolesi, J. (2005). Astrocytes, from brain glue to communication elements: the revolution continues. Nature Reviews Neuroscience, 6(8), 626-640.
- Waldrop, M. M. (1993). Complexity: The emerging science at the edge of order and chaos. Simon and Schuster.
- Wheeler, M. A., Clark, I. C., Lee, H. G., Li, Z., Linnerbauer,M., Rone, J. M., ... & Quintana, F. J. (2023). Droplet-based forward genetic screening of astrocyte–microglia cross-talk. Science, 379(6636), 1023-1030.
- Yu, X. (2022). Hippocampal astrocytes represent navigation space. PLoS biology, 20(3), e3001568.
- Zhang, J. M., Wang, H. K., Ye, C. Q., Ge, W., Chen, Y.,Jiang, Z. L., ... & Duan, S. (2003). ATP released by astrocytes mediates glutamatergic activity-dependent heterosynaptic suppression. Neuron, 40(5), 971-982.
- Zhang, Y. M., Qi, Y. B., Gao, Y. N., Chen, W. G., Zhou, T.,Zang, Y., & Li, J. (2023). Astrocyte metabolism and signaling pathways in the CNS. Frontiers in neuroscience, 17, 1217451.
- Zonta, M., Angulo, M. C., Gobbo, S., Rosengarten, B., Hossmann, K. A., Pozzan, T., & Carmignoto, G. (2003). Neuron-to-astrocyte signaling is central to the dynamic control of brain microcirculation. Nature neuroscience, 6(1), 43-50.
Notes
1At the end of the text, some essential references concerning the main findings on the issue are provided.
2 That is, the command transmission may only occur in one direction.
3 The exact meaning of these definitions will become clearer shortly.
4 It is not relevant here to specify the way in which the activation level can be established. For our scope, it is enough to know that such a level is not unique but can vary on a limited continuum of values from non-activation to maximum activation.
5 stagL just denotes the amount of stagnant activation in the line L that must always be lower than stag* representing the value of stagnation tolerated by the system – to be acceptable.
6 A disorder state in some line L in the sense specified above.
7 Even in this respect, the reference is the organization of the neural-glial biological system.
8 According to the structure of the tripartite synapsis.
9 The convergence point of a line L is exactly the point in which activation discharge or exhaustion either occur or cause disorder DL.
10 The G-times are slower than the N-ones.
1 In the sense specified at point i).
2 The symbol ↔ denotes the connection type characterizing G. that in the biological glial system should correspond to the gap-junction connection network thanks to which astrocytes communicate with each other through Ca2+ waves of varying frequency and amplitude.
13 I repeat once again that the system presented here is intended to be an ideal and simplified model of how the neural-glial system could really work in shaping complex and non-automatic responses to events that are somehow problematic for the integrity and equilibrium of the system itself. Of course, this is only a hypothetical construction based on what is known about the functional interaction between glial cells and neurons and therefore more than one crucial point of this theoretical system should be carefully investigated on an experimental level. My hope is that an ideal model like this can at least serve as a heuristic indication of how and where one can then empirically and in appropriate contexts further research in order to better understand whether the glial cells are really able to actively and with a significant role intervene in the onset of higher-order nervous processes.
4 We have to remember that ↔ establishes connections among g but the actual communication ability depends on the use and it is subjected to both autonomous and →-correlated enhancement and extension.
5 Such a property recalls the glial one – not yet tested but reasonably hypothesized – of functionally reacting in a different way depending on whether excitatory rather than inhibitory neurotransmitters are present in the shared synapses.
6 The idea, derived from the prerogatives of astrocytes in CNS, is that G, thanks to its faculty to ubiquitously map the N network and the activation foci in it as well as to communicate information within itself in both centrifugal and centripetal direction from any point, is able to perform a real tomography of the states in which N or parts of it are found.
7 This selection criterion could be defined as minimal risk in changing.
8 In this case too, the reference is given by what is reasonably hypothesized about the functional properties of perisynaptic glia. Indeed, it is highly plausible that astrocytes can assess how easily a presynaptic terminal triggers an action potential in the postsynaptic target and consequently set their functional behaviour according to the contextual necessity.
9 Aut denotes the exclusive disjunction.
20Even this criterion of economy recalls what emerges in the biological systems where the organization of responses in normally aimed at the least possible expenditure of energy to achieve the goal.
21 Again, this property of G is derived from what is known about the actual capacity of some glial cells to influence and modify in some way the neural activation dynamics.
22 A change in the desired way.
23G*L builds a sort of Faraday Cage around the Lx.
24 What corresponds to an excitatory effect in neuronal terms.
25 What corresponds to an inhibitory effect in neuronal terms.
26Even for this functional limit, the archetype is the interaction between glia and neurons in the NS. The former can in fact act at most with an agonistic or, vice versa, antagonistic role on the synapses. However, they cannot generate an action potential by themselves nor determine an inhibition. They can just favour one or the other process so that neurons can then definitively complete it.
27 A time-bound limit that can be reduced through use.
28 That is, from the most elementary components of Lx identified by G*L.
29 As said, G*L pushes its search in depth by following the n→ connections as far as its g-units are able to communicate with each another and thus investigate what is relevant to a good choice. The lines Lx identified in this way are therefore the most articulated and reciprocally checked possible within the communication limits of G*L at the processing moment
30 Possibly unexpected because it can also happen that nothing substantially different is triggered by the activation of Lx.
31 Always for the principle of N-dominance.
32 The G*L processing is structured in such a way as to provide feedback on the activation state of L.
33Even this evolutionary property attributed to the G structure is thought to be representative of what appears to belong to glial cells in the nervous system. In fact, just like neurons, glia – astrocytes at least – also seem to own the prerogative of improving their functional performances depending on the practice as such and on the specific type of activity they are called to carry out, support or assist. Therefore, like the neuronal system functionally improves itself both by making synaptic transmission easier and by broadening and enriching its own circuit architecture, glia also functionally strengthen themselves by increasing the communication power within their gap-junctions-network, that is, by making the flow of calcium ions gradually less subject to structural constraints and freer to spread centrifugally from any internal source of emission.


