Research Article - (2025) Volume 4, Issue 1
The Evidence for Office-Based Carpal Tunnel and The Use of UltraSound: A Systematic Review and Meta-Analysis
Received Date: Dec 12, 2024 / Accepted Date: Jan 20, 2025 / Published Date: Jan 24, 2025
Copyright: ©Â©2025 Alexis Chow, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Chow, A., Fares, J., Diyya, A., Samuel, M., Sharobim, M. (2025). The Evidence for Office-Based Carpal Tunnel and The Use of UltraSound: A Systematic Review and Meta-Analysis. J Surg Care. Vol 4(1), 01-07.
Abstract
Introduction Carpal Tunnel Syndrome (CTS) is a common condition characterized by compression of the median nerve at the wrist, leading to neuropathy and associated symptoms that can significantly impair hand function and quality of life. Recent interest has focused on the use of ultrasound-guided carpal tunnel release (CTR-US) as a treatment modality, given its potential to facilitate faster recovery, minimize complications, and enhance patient satisfaction. This systematic review aims to evaluate the efficacy and safety of in-office CTR-US in managing CTS in comparison to interventions performed without ultrasound.
Methods A systematic review and meta-analysis were conducted in SPSS v29 using PRISMA 2020 guidelines. Five databases (PubMed, Embase, Scopus, Cochrane, and Web of Science) were searched yielding a total of 713 articles and 5 after the exclusion criteria was applied. The review included clinical trials regarding office based carpal tunnel release (CTR) surgeries with and without ultrasound guidance. Outcome measures included the QuickDASH at 2 weeks and 1 year.
Results QuickDASH improved with CTR in both groups at 2 weeks and a year. There was a large effect size difference, 4.42, at 2 weeks and at 1 year, 2.15, between CTR-US and CTR without US, indicating clinical significance. However, the results were not statistically significant, p>0.05.
Conclusion The findings show that carpal tunnel release is effective at improving QuickDash scores at 2 weeks and 4 years post treatment. Though more research is required, CTR-US may provide superior clinical outcomes when compared to CTR without US.
Keywords
Carpal Tunnel Syndrome (CTS), Ultrasound-Guided Carpal Tunnel Release (Ctr-Us), Carpal Tunnel Release (Ctr), Median Nerve Compression, Quickdash, Minimally Invasive Procedures, In-Office Procedures, Surgical Intervention
Introduction
Carpal Tunnel Syndrome (CTS) is a prevalent and disabling condition caused by the compression of the median nerve at the wrist. It is characterized by symptoms such as pain, numbness, tingling, and weakness in the hand, which can significantly impair hand function and reduce quality of life. The condition affects a wide range of individuals, from those with occupations requiring repetitive hand movements to patients with underlying conditions like diabetes or obesity that increase the risk of CTS [1]. If left untreated, CTS can lead to persistent functional limitations and even permanent nerve damage, making early diagnosis and appropriate intervention crucial for optimal outcomes. Historically, conservative treatments such as wrist splinting, steroid injections, and physical therapy are first-line approaches for CTS. However, when these non-surgical measures fail to alleviate symptoms, surgical intervention becomes necessary. The standard surgical procedure for CTS is carpal tunnel release (CTR), which involves cutting the transverse carpal ligament to relieve pressure on the median nerve [2]. While CTR is generally effective, it requires a careful balance of technique to minimize complications such as nerve injury, scarring, and prolonged recovery time.
In recent years, there has been growing interest in ultrasound-guided carpal tunnel release (CTR-US) as an alternative to traditional, open surgical methods. The primary advantage of ultrasound guidance is that it allows for real-time visualization of the median nerve, surrounding tissues, and the carpal tunnel, enabling surgeons to perform the procedure with greater precision [3]. Ultrasound guidance has been associated with a reduced risk of injury to critical structures, such as the median nerve and flexor tendons, and offers the potential for less invasive surgery, faster recovery, and enhanced patient satisfaction. These factors have led to increasing adoption of CTR-US in both clinical and office-based settings. In particular, the frequency of in-office procedures for simple operations like carpal tunnel release under ultrasound guidance (CTR-US) has grown significantly in recent years. This shift toward office-based care reflects broader trends in healthcare, emphasizing minimally invasive procedures that reduce recovery time and optimize patient convenience. As part of this trend, in-office CTR-US procedures have gained popularity due to their lower costs, efficiency, and convenience [4,5].
Despite its promising benefits, the clinical effectiveness and safety of CTR-US relative to traditional CTR without ultrasound guidance remain unclear. Some studies suggest that CTR-US may lead to improved outcomes, such as faster recovery times and better functional results, while others show minimal differences between the two techniques. There is a pressing need for more rigorous studies to evaluate these claims systematically. The aim of this systematic review and meta-analysis is to evaluate the efficacy and safety of in-office ultrasound-guided carpal tunnel release (CTR-US) compared to traditional carpal tunnel release (CTR) performed without ultrasound. Specifically, we focus on two primary outcome measures: the improvement in QuickDASH (Disabilities of the Arm, Shoulder, and Hand) scores at 2 weeks and 1year post-surgery [6]. By synthesizing the available evidence, this review seeks to provide valuable insights into whether CTR-US offers superior clinical outcomes in managing CTS, potentially guiding future treatment practices and clinical decision-making. This study will also highlight any safety concerns or complications associated with both procedures, providing a comprehensive evaluation of their relative advantages and risks in the treatment of CTS.
Methods
A systematic review and meta-analysis were conducted using SPSS v29 and following the PRISMA 2020 guidelines. The review aimed to assess the efficacy and safety of in-office CTR-US for managing carpal tunnel syndrome (CTS) in comparison to interventions performed without ultrasound.
Literature Search
Five metabases (PubMed, Embase, Scopus, Cochrane Library, and Web of Science) were searched for relevant articles on the topic. Key terms were identified and incorporated into a Boolean search string: ("in-office" OR "office-based" OR "office" OR "outpatient") AND ("carpal tunnel release" OR "carpal tunnel surgery"). Only manuscripts published within the last 10 years were considered. The search initially yielded 713 articles, from which 437 duplicates were removed using Rayyan.ai. The remaining 276 articles were reviewed by two authors to determine eligibility based on the inclusion and exclusion criteria. Ultimately, five studies met the specified criteria. The screening process is summarized in Figure 1.
Figure 1: PRISMA Flow Chart
The flowchart illustrates the screening process for article inclusion and exclusion for the study. The initial screening was done by AC and AD. MS broke any ties.
Inclusion and Exclusion Criteria
The included studies were prospective cohort studies, descriptive clinical studies, and single-center cohort studies focusing on ultrasound-guided carpal tunnel release. Only articles published within the last 10 years were considered. Exclusion criteria included studies that did not involve carpal tunnel release or were not performed in an office setting. Articles older than 10 years were excluded due to the advent of newer, more efficient noninvasive techniques and tools.
Data Collection and Analysis
QuickDASH scores from each of the included studies were documented at baseline, 2 weeks, and 1 year. Statistical Analysis was done with SPSS v29 to perform a meta-analysis of 3 studies that used ultrasound and 2 studies that did not [7]. Hedge’s G was used to determine the effect size of each of the articles and to assess the differences between baseline, 2 weeks, and 1 year. A p-value of <0.05 was used to determine if the study was statistically significant. Cochran’s I2 ratio was used to determine homogeneity and heterogeneity, with a value of I2<40% indicating that differences in effect sizes could be due to random chance alone.
Results
Included Studies
After examining the articles for inclusion and exclusion criteria, five studies were included for data analysis. A summary of the baseline characteristics of population, study designs, and surgeon experience levels with ultrasound is included in Table 1 [8-11]. The study included one retrospective review, two prospective studies, one non-randomized control trial, and one randomized control trial. 2 studies investigated in-office surgery without ultrasound and 4 studies investigated in-office surgery with ultrasound.
|
TITLE |
STUDY ID (FIRST AUTHOR, YEAR) |
COUNTRY |
STUDY DESIGN |
RECRUITMENT PERIOD |
INTERVENTION |
#OF HANDS |
# OF PATIENTS |
AGE MEAN (RANGE) |
GENDER, %FEMALE |
SURGEON ULTRASOUND EXPERIENCE LEVEL |
INCLUSION CRITERIA |
OUTCOMES COLLECTED |
OUTCOMES MEASURED |
|
Office-Based Carpal Tunnel Release Using Ultrasound Guidance in a Community Setting: Long-Term Results |
Bergum et al 2022 |
USA |
Prospective single-center study |
March 2019 - August 2020 |
Office-based carpal tunnel release with US guidance |
123 |
88 |
66 (30-89) |
57% |
Surgeon #1: 10 years, Surgeon #2: 1 year |
Patients who completed the one year followup after undergoing CTR |
*BCTQ, **QDASH, complications |
Pre-op, 2 wks, mo, 3 mo, 1 yr |
|
Minimally Invasive Ultrasound-Guided Carpal Tunnel Release Improves Long-Term Clinical Outcomes in Carpal Tunnel Syndrome |
Kamel et al 2020 |
USA |
Retrospective Review |
July 2017 - April 2019 |
Office-based carpal tunnel release with US guidance |
69 |
51 |
60.7 (21-80) |
54% |
25 years experience with US-guided injections |
Patients who met the clinical and imaging criteria and elected to undergo carpal tunnel release |
*BCTQ, **QDASH, satisfaction |
Pre-op, 2 wks, 1 yr |
|
Early Postoperative Improvement in Sleep and Pain After Carpal Tunnel Release |
Niedermeier et al 2018 |
USA |
Prospective study |
June 2017 - December 2017 |
Outpatient Carpal Tunnel Release without US (some open, some endoscopic) |
61 |
61 |
52.1 (26-74) |
77% |
N/A |
Patients who met the clinical and imaging criteria and elected to undergo carpal tunnel release |
**QDASH, Visual Analog Scale, ***PSQI |
Pre-op, 2 wks, 6 wks |
|
Endoscopic Versus Open Carpal Tunnel Release |
Vasiliadis et al 2009 |
Greece, Sweden |
Non-randomized control trial |
April 2006 - April 2007 |
Outpatient open vs endoscopic tunnel release |
35 vs 37 |
35 vs 37 |
54.9 vs 53.1 |
80% vs84% |
N/A |
Patients with positive Phalen test or Tinel sign, confirmed with EMG without underlying arthritis, endocrinopathy, or previous hand surgeries |
*BCTQ, **QDASH, grip strength, key pinch, pinch strength, 2 point discrimination |
Pre-op, 2 days, 1 wk, 2 wks, 1 year |
|
Ultra-Minimally Invasive Ultrasound-Guided Carpal Tunnel Release |
Rojo-Manuat e et al 2016 |
Spain |
Randomized Control Trial |
January 2010 - December 2010 |
Ultra Minimally Invasive carpal tunnel release with US guidance vs Mini open carpal tunnel release without US guidance |
46 vs 46 |
46 vs 46 |
58.21 (29-83) vs 59.32 (30-84) |
58.6% vs 63.0% |
Not indicated |
Patients who met the clinical and imaging criteria and elected to undergo carpal tunnel release and were prospectively solicited |
**QDASH, Visual Analog Scale, Time to full wrist extension and flexion, discontinuation of oral analgesic use, return to daily activities |
Pre-op, 1 wk, 3 wks, 6 wks, 3 mo, 6 mo, 1 year |
|
*Boston Carpal Tunnel Questionnaire scores, **Quick Disabilities of the Shoulder and Hand, ***Pittsburgh Sleep Quality Index |
|||||||||||||
Table 1: Summary of Included Studies
QuickDASH
Four studies (pre: n=285, post:n=275) indicated the QuickDASH results at 2 weeks post-operation. Both the US group and non-US group both showed improvement in the QuickDASH at 2 weeks [6-9]. There was a significant difference in effect size, 4.42, between the US-guided surgery group and the non-US surgery group, demonstrating clinical significance. The group that received ultrasound-guided surgery had a larger decrease in QuickDASH scores, 17.5±14.1, compared to the difference in QuickDASH scores in the non-US group, 7.9±20.4. The average change in QuickDASH for the CTR-US group met the minimal clinically important difference (MCID) whereas the non-US group did not [12]. There was also a large difference in effect sizes between the two groups, 4.45, indicating clinical significance, as shown in Figure 2.
However, the results were not statistically significant (p>0.05). The results for the QuickDASH at 2 weeks showed high heterogeneity, particularly in the group that had CTR-US surgery, which had substantial heterogeneity (I2=99.6%), while the non-ultrasound guided group had minimal heterogeneity (I2=24.3%).8-12 This indicates that there were differences within the ultrasound group that could not be attributed to random chance alone.
Figure 2: Forest Plot of Effect Sizes for QuickDASH at 2 Weeks
The ultrasound group had a larger effect size (4.85) than the non-ultrasound group (0.43), indicating clinical benefit of using ultrasound. However, results were not statistically significant.
Five studies (pre: 316, post: 306) included the QuickDASH results at 1year follow-up [8-12]. Both the CTR-US and non-US group showed improved QuickDASH scores at 1 year follow-up. The ultrasound-guided surgery group had a significantly larger effect size than the non-US surgery group, with an effect size difference of 2.16, as shown in Figure 3 indicating clinical significance. No statistical significance was found between the two groups (p>0.05). Both of the groups met the MCID with minimal difference in the MCID for both groups. There was substantial heterogeneity in the effect sizes of both groups as shown in Figure 4.
Figure 3: Forest Plot of Effect Sizes of QuickDASH at 1 Year
The ultrasound group had a larger effect size (11.34) compared to the group without ultrasound (9.18), but the results were not statistically significant.
Figure 4: Funnel Plot of Effect Sizes of QuickDASH at 1 Year
There was significant heterogeneity in both the ultrasound and non-ultrasound group, with most studies outside the 95% confidence interval.
Risk of Bias and GRADE Assessment
Based on the ROBINS-I criteria, there was risk of bias in the included studies due to various reasons [13]. Of note, Bergum et al. 2022 had serious risk of bias due to missing data because of the lack of explanation for data in a large proportion of participants, 77 patients, in addition to varying numbers of patients at follow¬up timepoints. There were more moderate risks of bias in Kamel et al. 2020 due to the retrospective nature of the review. These included bias in confounding, selection of participants, missing data, and measurements of outcomes. There was low risk of bias in the control trials, Rojo-Manuate et al. 2016 and Vasialidis et al. 2009. Overall, there was risk associated with the data included in the meta-analysis with the largest concern being missing data, shown in Figure 5.
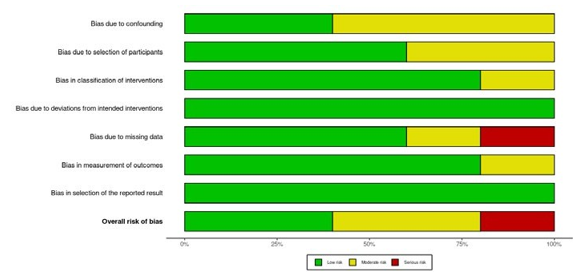
Figure 5: Risk of Bias in Included Studies
Based on the ROBINS-I criteria, there was serious risk of bias due to missing data.
There was also moderate risk of bias due to confounding, classification of interventions, and measurement of outcomes. Overall, there was a moderate certainty of evidence based on the grading of recommendations, assessment, development, and evaluations (GRADE) criteria [14]. Lowering the quality of evidence is the moderate to serious risk of bias, high heterogeneity, and imprecision in the studies due to varying ultrasound skill levels and differing surgical technique. However, the effect size between the studies is very large and indicates that there may be clinical significance.
Discussion
This meta-analysis examined the effectiveness of ultrasound-guided carpal tunnel release (CTR-US) compared to traditional carpal tunnel release (CTR) without ultrasound. The key finding was that both CTR-US and traditional CTR improved QuickDASH scores at 2 weeks and 1 year. Large effect sizes favoring CTR-US were observed at both 2 weeks (4.42) and 1 year (2.15), suggesting potentially superior clinical outcomes with CTR-US. However, despite these large effect sizes, the difference between the two procedures was not statistically significant (p > 0.05). This lack of statistical significance is likely attributable to the small number of included studies (n=5), which limited the statistical power of the analysis. Consequently, further research with larger sample sizes is needed to confirm this potential benefit.
An effect size quantifies the magnitude of the difference between groups or the magnitude of change within a group over time, indicating the practical significance of an observed effect. In this meta-analysis, the effect sizes compare the difference in QuickDASH score improvement between CTR-US and traditional CTR. The observed large effect sizes suggest that CTR-US leads to greater improvements in QuickDASH scores compared to standard CTR, both in the short term (2 weeks) and long term (1 year). The larger effect size at 2 weeks indicates that this difference is more pronounced in the immediate postoperative period. As previously stated, this difference was not statistically significant (p > 0.05) due to the limited number of included studies. This small sample size reduces statistical power, increasing the likelihood of a Type II error (failing to detect a true effect due to increased variability and insufficient population representation), thus limiting the ability to demonstrate statistically significant results.
Recent research on CTR-US has reported similar findings of improved QuickDASH scores over time [6]. However, this improvement is not unique to CTR-US, as mini-open CTR has also been shown to result in improved patient-reported outcomes [15]. A key distinction between open and CTR-US is the increased risk of complications, notably pillar pain and scar tenderness, associated with open CTR [15].
This meta-analysis has several strengths, including a systematic and comprehensive literature search using multiple databases, adherence to PRISMA guidelines, and the use of a standardized outcome measure (QuickDASH). The most significant limitation is the small number of included studies, which limits generalizability and statistical power. Other limitations include the moderate level of evidence due to the lack of control trials directly comparing CTR-US and CTR without US and the heterogeneity between the studies. The heterogeneity between the studies may be due in part to the small number of studies, but also because of the varying levels of experience with ultrasound, as ultrasound usage is user-dependent and the learning curve is steep [16]. This is evident with the particularly high heterogeneity in the CTR-US group at 2 weeks and 1 year. More studies on how ultrasound experience affects these results are indicated.
Despite the lack of statistical significance, the clinically significant effect sizes suggest that CTR-US may offer benefits to both surgeons and patients. For surgeons, CTR-US may allow for in-office procedures, eliminating the need for surgical center and anesthesia personnel for this procedure. This could also provide quicker access to treatment for both surgeons and patients by removing reliance on surgical center availability. Patients may benefit from improved short-term outcomes [11]. However, larger, well-designed randomized controlled trials (RCTs) with sufficient statistical power are necessary to confirm these potential benefits. Future research should investigate long-term outcomes of in-office CTR-US, explore the use of different ultrasound techniques and devices, compare cost-effectiveness with other CTR approaches, and directly compare CTR-US with endoscopic CTR to further refine treatment strategies for this common condition.
Conclusion
The findings show that carpal tunnel release, regardless of technique, is effective at improving QuickDash scores at 2 weeks and 4 years post treatment. When comparing the techniques of CTR-US to CTR without US, there was a large effect size difference between the two methods at 2 weeks and 1 year, indicating clinical significance supporting CTR-US. However, the results were not statistically significant, p>0.05. Though CTR-US may provide superior clinical outcomes when compared to CTR without US, more research is required to further support this. These findings can be confirmed with larger randomized controlled trials and the exploration of additional outcome measures such as patient-reported outcomes and return-to-work rates. Additional studies in the future should also aim to incorporate a larger sample size and a longer follow-up in their protocol to yield results that can better aid in establishing definitive guidelines.
References
- Papanas, N., Stamatiou, I., & Papachristou, S. (2022). Carpal tunnel syndrome in diabetes mellitus. Current Diabetes Reviews, 18(4), 16-19.
- Osiak, K., Elnazir, P., Walocha, J. A., & Pasternak, A. J. F. M. (2022). Carpal tunnel syndrome: state-of-the-art review. Folia morphologica, 81(4), 851-862.
- David, I. (2020). Sonography-Guided Carpal Tunnel Release.Hand clinics, 38(1), 75–82.
- Billig, J. I., Nasser, J. S., Chen, J. S., Lu, Y. T., Chung, K. C.,Kuo, C. F., & Sears, E. D. (2020). Comparison of safety andinsurance payments for minor hand procedures across operative settings. JAMA network open, 3(10), e2015951-e2015951.
- Starr, B. W., Davenport, R. O., Granzow, D., Johnson, S. P., & Lien, J. R. (2023). Optimizing the use of operating rooms by transitioning common hand surgeries into the office setting. The Journal of Hand Surgery, 48(3), 217-225.
- Galardini, L., Coppari, A., Pellicciari, L., Ugolini, A., Piscitelli, D., La Porta, F., ... & Vercelli, S. (2024). Minimal Clinically Important Difference of the Disabilities of the Arm, Shoulder and Hand (DASH) and the Shortened Version of the DASH (QuickDASH) in People With Musculoskeletal Disorders: A Systematic Review and Meta-Analysis. Physical Therapy, 104(5), pzae033.
- IBM Corp. Released 2023. IBM SPSS Statistics for Windows, Version 29.0.2.0 Armonk, NY: IBM Corp
- Bergum, R. A., & Ciota, M. R. (2022). Office-based carpal tunnel release using ultrasound guidance in a community setting: long-term results. Cureus, 14(7).
- Kamel, S. I., Freid, B., Pomeranz, C., Halpern, E. J., & Nazarian, L. N. (2021). Minimally invasive ultrasound-guided carpal tunnel release improves long-term clinical outcomes in carpal tunnel syndrome. American Journal of Roentgenology, 217(2), 460-468.
- Niedermeier, S. R., Pettit, R. J., Frantz, T. L., Colvell, K., & M. Awan, H. (2020). Early postoperative improvement in sleep and pain after carpal tunnel release. Hand, 15(3), 311-314.
- Vasiliadis, H. S., Xenakis, T. A., Mitsionis, G., Paschos, N., & Georgoulis, A. (2010). Endoscopic versus open carpal tunnel release. Arthroscopy: The Journal of Arthroscopic & Related Surgery, 26(1), 26-33.
- Rojo-Manaute, J. M., Capa-Grasa, A., Chana-Rodríguez, F., Perez-Mañanes, R., Rodriguez-Maruri, G., Sanz-Ruiz, P.,... & Vaquero-Martín, J. (2016). Ultraâ?minimally invasive ultrasoundâ?guided carpal tunnel release: a randomized clinical trial. Journal of Ultrasound in Medicine, 35(6), 1149-1157.
- Sterne, J. A., Hernán, M. A., Reeves, B. C., SavoviÄ?, J.,Berkman, N. D., Viswanathan, M., ... & Higgins, J. P. (2016). ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. bmj, 355.
- Guyatt, G. H., Oxman, A. D., Vist, G. E., Kunz, R., Falck-Ytter, Y., Alonso-Coello, P., & Schünemann, H. J. (2008). GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. Bmj, 336(7650), 924-926.
- Hammert, W. C., Chung, K. C., & Miller, L. E. (2024). Best-evidence systematic review and meta-analysis of mini-open carpal tunnel release. Journal of Hand Surgery Global Online, 6(1), 35-42.
- Smith, M. J., Rogers, A., Amso, N., Kennedy, J., Hall, A., & Mullaney, P. (2015). A training, assessment and feedback package for the trainee shoulder sonographer. Ultrasound, 23(1), 29-41.