Research Article - (2022) Volume 7, Issue 1
The Efficacy and Safety of the Off Label Dose of Direct Oral Anti Coagulants Treatment in Patients with Atrial Fibrillation in China
2Department of Endocrinology and Metabolism, Huashan Hospital, Shanghai Medical College, Fudan University, Shanghai, 200040, China
#Equally contribution
Received Date: Mar 10, 2022 / Accepted Date: Mar 19, 2022 / Published Date: Mar 26, 2022
Copyright: ©Jian Li, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Peng Zhou, Meng Wang, Rongchen Liu, Yangjie Yu, Wei Chen, Yunzhi Ma, Hengyang Wang, Maieryemu Waresi, Vladimir Melnikov, Xinping Luo, Jian Li (2022) The Efficacy and Safety of the Off-Label Dose of Direct Oral Anti-Coagulants Treatment in Patients with Atrial Fibrillation in China. Cardio Open, 7(1): 233-238.
Abstract
Background: Atrial fibrillation (AF) is the most common arrhythmia in clinical practice. Anti-coagulation therapy, especially direct oral anti-coagulants (DOACs) is an indispensable therapy for AF. Considering the increased risks of bleeding, the off-label dose of DOACs has been applied in East Asia. However, the efficacy and safety of off-label dose of DOACs is controversial. Here, we investigate its efficacy and safety in AF patients in China.
Methods: This is a retrospective cohort study. Patients diagnosed with AF between January 2015 and December 2015 were recruited. Participants were divided into three groups according to DOACs dose: standard dose of anti-coagulation (SAG) group, off-label dose of DOACs (OFL) group and non-anti-coagulation (NCG) group. Primary endpoint event was all-cause mortality. Secondary endpoint events included thromboembolic events, acute coronary syndrome, major bleeding and any rehospitalization. All endpoints were collected in 2020.
Results: 296 patients were included in this study. All-cause mortality in NCG (52%) group was the highest among these three groups (p<0.001), and there was no significant difference between OFL (14%) and SAG group (19%, p=0.601). Compared to SAG group (1.8%), patients in OFL group (16.2%) had higher risk of stroke and thromboembolic events (p<0.001). The incidence of other events including major bleeding (p=0.597) were similar between SAG group (8.1%) and OFL group (5.4%).
Conclusions: Off-label dose of DOACs shows no overall benefits in the occurrence of thromboembolic risk compared to the standard dose of DOACs in AF patients in China. Collectively, standard dose of DOACs should be advocated in China.
Keywords
Atrial Fibrillation, Anti-Coagulation Therapy, Stroke
Introduction
Atrial fibrillation (AF) is the most common sustained arrhythmia worldwide [1,2]. The prevalence of AF in China is approximate- ly 1% [3]. AF occurrence increases significantly with age, nearly 1 in 25 people over the age of 60 and 1 in 10 over the age of 80 affected by this arrhythmia [4]. AF causes thromboembolic events, such as stroke which brings a significant socioeconomic burden for healthcare systems worldwide [3]. Treatment of AF includes three-tiered: anti-coagulation, better symptom control and control of comorbidities [5,6]. Anti-coagulation therapy, in- cluding vitamin K antagonists (VKAs) and direct oral anticoag- ulants (DOACs), is still considered as the first-line drug therapy in AF management. Studies have reported that the overall effi- cacy and safety of DOACs medications are superior to that of VKAs [7]. DOACs have been considered as the first-line drug therapy for AF [8,9].
The standard dose of DOACs in AF treatment includes dabiga- tran 150 mg twice daily, rivaroxaban 20 mg once daily, apixaban 5 mg twice daily or edoxaban 60 mg once daily [5]. Considering of the increased risks of bleeding and anti-coagulation related intracranial hemorrhage in East Asia including China, a compro- mised dosage of DOACs, such as low-dose rivaroxaban (10-15 mg once daily) or dabigatran (110-150 mg once daily), has been applied for anti-coagulation therapy in East Asia [10-12]. Sever- al studies from Taiwan have demonstrated that the off-label dos- age of DOACs demonstrate better efficacy and safety compared to VKAs, and exhibit similar efficacy compared to the standard dosage of DOACs [13,14]. However, other studies indicate that off-label dose of DOACs leads to higher thrombosis risk com- pared to the standard dosage of DOACs [11]. Thus, the benefit of this off-label dose was still controversial and need more ex- ploration [15].
Here, we carried out this retrospective cohort study to investi- gate the efficacy and safety of off-label DOACs in AF patients in China. Patients (n=296) with AF who were hospitalized in Huashan Hospital, Fudan University, from January 2015 to De- cember 2015 were recruited in our study. We compared the prog- nosis among patients without anti-coagulation therapy, patients with off-label anti-coagulation therapy and ones with standard anti-coagulation therapy during last 5 years. Our data showed that patients treated with the off-label dose of anti-coagulation therapy had higher thromboembolic risk compared to ones with standard anti-coagulation therapy, indicating that standard dose of DOACs should be advocated in AF treatment.
Methods
Ethics Approval
All procedures performed in studies involving human partici- pants were in accordance with the ethical standards of the insti- tutional research committee (KY2019-552) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Patients and Study Design
This was a retrospective cohort study of in-patients between Jan- uary 2015 and December 2015 at a tertiary referral center in the East of China. Subjects were selected based on the
Inclusion Criteria
(1) Patients who were diagnosed with AF by at least 30 seconds electrocardiograph or 24h-Holter before or during hospitaliza- tion. (2) Patients with estimated glomerular filtration rate ≥ 50 ml/ min/1.73m2.
Exclusion Criteria
(1) Patients who used warfarin irregularly (2) Patients using warfarin with time in therapeutic range of in- ternational normalized ratio (TTR) < 50% (TTR was calculated by the percentage of tests within the target international normal- ized ratio (INR) (tests ratio)) [16]. (3) Patients whose anti-coagulation therapy was changed during last 5 years. All recruited patients were followed up in 2020.
Participants included in our study were separated into three groups according to the dose of anti-coagulation therapy re- ceived: standard dose of anti-coagulation (SAG) group, off-label dose of anti-coagulation (OFL) group and non-anti-coagulation (NCG) group. Patients in SAG group received standard dosage of anti-coagulation therapy, including rivaroxaban (20 mg once daily) or dabigatran (either 110 mg or 150 mg twice daily), or warfarin (TTR > 50%). Patients in OFL group received rivar- oxaban (< 20 mg daily) or dabigatran (either 110 mg or 150 mg once daily). Patients without anti-coagulation therapy were clas- sified into NCG group. The reason why these patients did not re- ceive anti-coagulation treatment was because most of these pa- tients were admitted to the hospital for surgery and the surgeon lacked knowledge of anticoagulation drugs. Primary endpoint event in this study was defined as all-cause mortality. Second- ary endpoint events included stroke and other thromboembolic events, acute coronary syndrome, bleeding (major bleeding as intracranial hemorrhage and gastrointestinal hemorrhage) and any re-hospitalization. All endpoints were collected during fol- low-up in 2020.
Statistical Analyses
All analyses were carried out using SPSS 24.0. Descriptive statistics were used to evaluate patient related secondary ob- jectives. Two-sided Student’s t-test, Chi square test and Ka- plan-Meier method were applied in our study All statistical tests were two-sided and a p value < 0.05 was considered statistically significant.
Results
Baseline Clinical Characteristics
Totally, 296 patients with AF were included in this study. These patients are divided into three groups: 148 patients in non-an- ti-coagulation of DOACs (NCG) group, 37 patients in off-label dose of DOACs (OFL) group and 111 patients in standard dose of DOACs (SAG) group. As shown in Figure 1, 95% (35/37) patients used rivaroxaban (25 patients using 15mg once daily, 10 patients using 10mg once daily and 5 patients using 7.5mg once daily) in OFL group, while only 5% (2/37) patients were treated with dabigatran (110mg once daily). In SAG group, 75% (83/111) patients were treated with warfarin, 22% (24/111) with dabigatran, and 3% (3/111) with rivaroxaban.
Figure 1: The proportion of anti-coagulation drugs used to treat AF patients in OFL group and SAG group (a) OFL = Off-Label Group, (b) SAG = Standard Anti-Coagulation Group
The baseline clinical characteristics of these three groups were summarized in Table. Compared to OFL group (71±10 years) and SAG group (69±13 years), age in NCG group was the old- est (73±12 years, p = 0.036). There was no statistical significant difference in gender among three groups (p=0.331). The propor- tion of complications including hypertension, diabetes mellitus, heart failure and vascular disease (including prior myocardial infarction, aortic plaque and peripheral arterial disease) were all similar among these three groups Table.
Table: The Baseline Characteristics of these 296 Patients
|
|
NCG group |
OFL group |
SAG group |
P value |
|
Number of patients (n) |
148 |
37 |
111 |
- |
|
Age (years) |
73±12 |
71±10 |
69±13 |
0.036 |
|
Male (n, %) |
48 |
57 |
59 |
0.331 |
|
Hypertension (%) |
60 |
62 |
54 |
0.473 |
|
CHF (%) |
17 |
22 |
30 |
0.074 |
|
DM (%) |
31 |
38 |
20 |
0.114 |
|
Vascular disease (%) |
31 |
30 |
20 |
0.114 |
|
Prior stroke/SE (%) |
22 |
35 |
43 |
0.014 |
|
CHA2DS2-VASc score |
3.6±1.8 |
3.7±1.7 |
3.5±1.7 |
0.760 |
|
HAS-BLED score |
2.6±1.2 |
2.3±1.3 |
2.0±1.1 |
0.001 |
|
Anti-platelet therapy (%) |
49 |
35 |
10 |
<0.001 |
|
SAG: Standard Anti-Coagulation Group, OFL: Off-Label Group, NCG: Non-Anticoagulation Group, CHF: Congestive Heart Failure, DM: Diabetes Mellitus, SE: Systemic Embolism. |
||||
The proportion of prior stroke or systemic embolism of the NCG group, OFL group and SAG group were 22% (33/148), 35% (13/37) and 43% (48/111), respectively. Less patients in NCG group had stroke history (22% in NCG group, 35% in OFL group, 43% in SAG group, p=0.014). The CHA2DS2- VASc score, as the main evaluation tool of stroke in AF patients, was 3.6±1.8, 3.7±1.7, 3.5±1.7 in NCG group, OFL group and SAG group, respectively. There is no statistical difference in the CHA2DS2-VASc score among these groups. A larger proportion of patients in NCG group were treated with anti-platelet drugs (49% in NAG, 35% in OFL, 10% in SAG, p<0.001).
Off-Label Dose of DOACs did not Increase the All- Cause Mortality of AF Patients Compared with Stan- dard Dose of DOACs
We collected and analysed the clinical events of these 296 pa- tients in our last follow-up in 2020. Primary endpoint event was defined as all-cause mortality. The all-cause mortality was 52% (77/148) in NCG group, 14% (5/37) in OFL group and 19% (21/111) in SAG group. As shown in Figure 2, all-cause mortali- ty in NCG group was the highest among these three groups (p < 0.001), and there was no significant difference between OFL and SAG group (p = 0.601).
Figure 2: The Kaplan-Meier Curve of all-cause mortality among the three groups SAG = standard anti-coagulation group, OFL = Off-Label Group, NCG = Non-Anticoagulation Group
Off-Label Dose could not Decrease the Risk of Stroke in AF Patients
During the 5 years period, 15 out of 148 (10.13%) patients in NCG group suffered from cerebral infarction and 1 out of 148 (0.67%) patient had femoral artery embolism. 6 out of 37 (16.21%) patients in OFL group suffered from stroke. 1 out of 111 (0.9%) patient in SAG group had stroke and 1 out of 111 (0.9%) had femoral artery embolism. As shown in Figure 3, compared to SAG group, patients in OFL group had higher risk of stroke and thromboembolic events (p < 0.001).
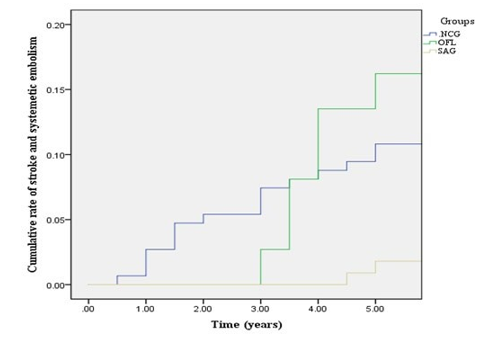
Figure 3: Cumulative rate of stroke and systemic embolism events SAG = standard anti-coagulation group, OFL = Off-Label Group, NCG = Non-Anticoagulation Group
The incidence of other events including major bleeding (p = 0.597), re-hospitalization (p = 0.213) and cardiovascular mor- tality (p = 0.640) were all similar between SAG group and OFL group. In OFL group, 2 out of 37 (5.40%) patients had gastro- intestinal hemorrhage. In SAG group, 6 out of 111 (5.40%) pa- tients had gastrointestinal hemorrhage and 2 out of 111 (1.80%) patients had cerebral hemorrhage, while 1 out of 111 (0.90%) patient had vesical hemorrhage. Expectedly, major bleeding did not occur in patients of NCG group. Re-hospitalization (77% in NCG group, 69% in OFL group and 57% in SAG group, p < 0.001) and cardiovascular death (35% in NCG group, 14% in OFL group and 15% in SAG group, p < 0.001) were more com- mon in NCG group among the three groups Figure 4.
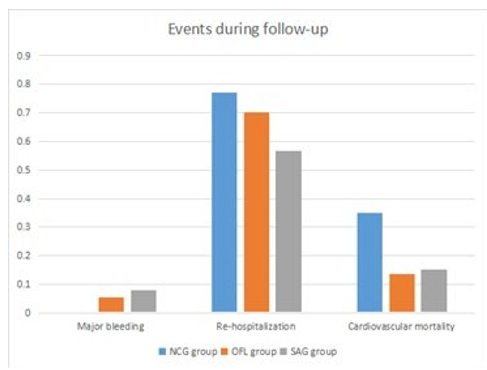
Figure 4: Adverse events (major bleeding, re-hospitalization and cardiovascular mortality) from 2015 to 2020 SAG = Stan- dard Anti-Coagulation Group, OFL = Off-Label Group, NCG = Non-Anticoagulation Group
Next, we conducted a comprehensive analysis of the anti-coag- ulant effect among these three groups by combining all primary and secondary events. The total cardiovascular events and mor- tality rate were not significantly different between OFL group and SAG group (P=0.863), while were lower than NCG group (p < 0.001), Figure 5.
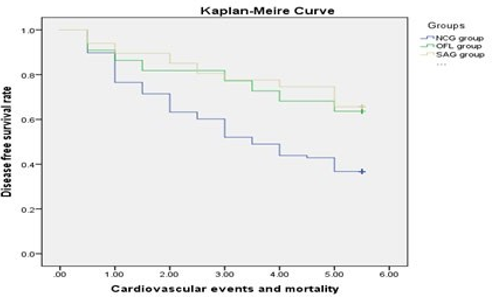
Figure 5: The Kaplan-Meier Curve of the total cardiovascular events and mortality rate among the three groups SAG = stan- dard anti-coagulation group, OFL = Off-Label Group, NCG = Non-Anticoagulation Group
Discussion
Previous studies have indicated that the risk of anti-coagulation related intracranial hemorrhage in Asians with AF is substantial- ly higher than Caucasians with AF [9]. Therefore, some compro- mised dosage of DOACs are clinically applied for the treatment of AF in East Asia, which is so called “Asian dose”. Around 3 in 10 Asian AF patients were treated with off-label dosing NOACs in daily practice [17]. Low dose of rivaroxaban (10 mg or 15 mg daily) have been proposed to decrease the risk of ischemic stroke/systemic embolism, intracranial hemorrhage, gastroin- testinal bleeding compared to warfarin [13]. Compared to stan- dard-dose rivaroxaban, rivaroxaban 10 mg daily in Taiwanese with AF was associated with similar risks of thromboembolism and bleeding to rivaroxaban 15 mg or 20 mg daily [14].
However, Cheng et al. have pointed out that rivaroxaban 10 mg daily was associated with an increased risk of ischemic stroke with 2.75 times compared to rivaroxaban 15 mg or 20 mg daily [11]. The standard dose of DOACs results in a relatively mild stroke in the patients compared to the off-label dose (rivarox- aban under 20 mg daily and dabigatran under 150 mg twice dai- ly) [18]. Off-label dose-reduced treatment was associated with a higher rate of all-cause mortality and stroke without a safe- ty benefit [19]. Similar outcomes were found in our study. The OFL group and SAG group were of similar all-cause mortality. However, we found that thrombosis risk was significantly higher in OFL group compared to SAG group. Interestingly, thrombo- sis risk in OFL group was even greater than NCG group. This might be partly because of comparatively shorter survival period in NCG group. In addition, our results indicated that off-label dose of anti-coagulation therapy, indeed, did not lead to a lower bleeding risk compared to the standard anti-coagulation therapy.
Here, our results show that the CHA2DS2-VASc score was com-parable among the groups, indicating similar thrombosis risk in three groups. The proportion of prior stroke and systematic em- bolism history was higher in OFL and SAG group compared to NCG group. The age was the oldest in NCG group. The above phenomena is highly reasonable in clinic as the anti-thrombot- ic therapy is more active for stroke AF patients [19]. For elder patients, anti-coagulation needs to be more prudent due to the higher bleeding risk as well [20]. The HAS-BLED score of NCG group was higher compared to other groups, acting as a possible reason why anti-coagulation therapy was not used for these pa- tients. Previous studies have demonstrated that anti-coagulation therapy could reduce the all-cause mortality of AF patients [21]. Here, our study demonstrates that off-label anti-coagulation therapy can increase the survival rate of AF patients compared to no anti-coagulants. We supposed that the major explanation for this finding may be the reduction of coronary events, which was similar to a previous study [21]. Low dose of rivaroxaban could reduce thrombus formation and platelet-dependent thrombin generation in patients with coronary heart disease [22]. Howev- er, off-label dose of DOACs brings higher bleeding risk without any improvements in thromboembolic events risk compared to anti-platelet therapy.
There were several limitations in our study. The sample size was relatively small, which may be due to the loss of follow-up. In addition, the study was an observational study. More evidence such as randomized clinical trials are needed.
Conclusion
Off-label dose of DOACs partly contributes to the decreased mortality in AF patients compared to no anti-coagulation ther- apy. However, it indeed does not decrease thromboembolic risk of AF patients compared to the standard dose of DOACs in Chi- na. Standard dose of DOACs is more appropriate for Chinese AF patients in prevention of stroke and systematic embolism.
Acknowledgement
The authors thank information department of Huashan Hospital affiliated to Fudan University and Xunshu Company for provid- ing patients’ information.
Authors Contributions
Jian Li and Xinping Luo designed the study. Peng Zhou, Rong- chen Liu, Yangjie Yu, Wei Chen, Yunzhi Ma, Maieryemu Ware- si, Hengyang Wang collected the data. Peng Zhou analysed the data. Peng Zhou and Vladimir Melnikov wrote the manuscript. Meng Wang critically revised for important intellectual content. All authors had full access to the data, contributed to the study, approved the final version for publication, and take responsibili- ty for its accuracy and integrity
References
-
- Wong, C. X., Brown, A., Tse, H. F., Albert, C. M., Kalman,J. M., & et al. (2017). Epidemiology of atrial fibrillation: the Australian and Asia-Pacific perspective. Heart, Lung and Circulation, 26(9), 870-879.
-
- Ceornodolea, A. D., Bal, R., & Severens, J. L. (2017). Epi- demiology and management of atrial fibrillation and stroke: review of data from four European countries. Stroke re-search and treatment, 2017, 1-12.
-
- Guo, Y., Tian, Y., Wang, H., Si, Q., Wang, Y., & et al. (2015). Prevalence, incidence, and lifetime risk of atrial fibrillation in China: new insights into the global burden of atrial fibril- lation. Chest, 147(1), 109-119.
- Bhatt, H. V., & Fischer, G. W. (2015). Atrial fibrillation: pathophysiology and therapeutic options. Journal of cardio- thoracic and vascular anesthesia, 29(5), 1333-1340.
- Hindricks, G., Potpara, T., Dagres, N., Arbelo, E., Bax, J. J., & et al. (2021). 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collab- oration with the European Association for Cardio-Thorac- ic Surgery (EACTS) The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. European heart journal, 42(5), 373-498.
- Yasuda, K., Fukuda, S., Nakamura, M., Ohtani, R., Kuwata, Y., & et al. (2018). Predictors of cardio embolic stroke in Japanese patients with atrial fibrillation in the Fushimi AF registry. Cerebrovascular diseases extra, 8(2), 50-59.
- Durand, M., Schnitzer, M. E., Pang, M., Carney, G., Elton- sy, S., & et al. (2020). Comparative effectiveness and safety of direct oral anticoagulants versus vitamin K antagonists in nonvalvular atrial fibrillation: a Canadian multicentre observational cohort study. Canadian Medical Association Open Access Journal, 8(4), E877-E886.
- Bai, Y., Guo, S. D., Shantsila, A., & Lip, G. Y. (2018). Mod- elling projections for the risks related with atrial fibrillation in East Asia: a focus on ischaemic stroke and death. EP Eu- ropace, 20(10), 1584-1590.
- Chan, Y. H., See, L. C., Tu, H. T., Yeh, Y. H., Chang, S. H.,& et al. (2018). Efficacy and safety of apixaban, dabigatran, rivaroxaban, and warfarin in Asians with nonvalvular atri- al fibrillation. Journal of the American Heart Association, 7(8), e008150.
-
- Shen, A. Y. J., Yao, J. F., Brar, S. S., Jorgensen, M. B., & Chen, W. (2007). Racial/ethnic differences in the risk of intracranial hemorrhage among patients with atrial fibrilla- tion. Journal of the American College of Cardiology, 50(4), 309-315.
- Cheng, W. H., Chao, T. F., Lin, Y. J., Chang, S. L., Lo, L.W., & et al. (2019). Low-dose rivaroxaban and risks of ad- verse events in patients with atrial fibrillation. Stroke, 50(9), 2574-2577.
-
- Cho, M. S., Yun, J. E., Park, J. J., Kim, Y. J., Lee, J., & et al. (2019). Outcomes after use of standard-and low-dose non– vitamin k oral anticoagulants in Asian patients with atrial fibrillation. Stroke, 50(1), 110-118.
- Lee, H. F., Chan, Y. H., Tu, H. T., Kuo, C. T., Yeh, Y. H.,& et al. (2018). The effectiveness and safety of low-dose rivaroxaban in Asians with non-valvular atrial fibrillation. International journal of cardiology, 261, 78-83.
-
- Lin, Y. C., Chien, S. C., Hsieh, Y. C., Shih, C. M., Lin, F. Y., & et al. (2018). Effectiveness and safety of standard-and low-dose rivaroxaban in Asians with atrial fibrillation. Jour- nal of the American College of Cardiology, 72(5), 477-485.
- Joung, B. (2017). Real-world data and recommended dos- age of non-vitamin K oral anticoagulants for Korean pa-tients. Korean Circulation Journal, 47(6), 833-841.
- Caldeira, D., Cruz, I., Morgado, G., Stuart, B., Gomes, A. C., & et al. (2015). Is the time in therapeutic range using the ratio of tests equivalent to the Rosendaal method. Blood Coagulation & Fibrinolysis, 26(8), 972-976
- Chan, Y. H., Chao, T. F., Chen, S. W., Lee, H. F., Yeh, Y.H., & et al. (2020). Off-label dosing of non–vitamin K an- tagonist oral anticoagulants and clinical outcomes in Asian patients with atrial fibrillation. Heart rhythm, 17(12), 2102- 2110.
- Jung, Y. H., Choi, H. Y., Lee, K. Y., Cheon, K., Han, S. W.,& et al. (2018). Stroke severity in patients on non-vitamin K antagonist oral anticoagulants with a standard or insufficient dose. Thrombosis and Haemostasis, 118(12), 2145-2151.
- Arbel, R., Sergienko, R., Hammerman, A., Green- berg-Dotan, S., Batat, E., & et al. H. (2019). Effectiveness and safety of off-label dose-reduced direct oral anticoagu- lants in atrial fibrillation. The American Journal of Medi- cine, 132(7), 847-855.
- Bauersachs, R. M., & Herold, J. (2020). Oral anticoagula- tion in the elderly and frail. Hämostaseologie, 40(01), 074- 083
- Arbel, R., Sergienko, R., Hammerman, A., Dotan-Green- berg, S., Batat, E., & et al. H. (2019). Direct oral anticoag- ulation and mortality in moderate to high-risk atrial fibrilla- tion. Heart, 105(19), 1487-1492
- Geisler, T., Muenzer, P., Alnaggar, N., Geue, S., Tegtmey- er, R., & et al. (2018). Inhibitory Mechanisms of Very Low Dose Rivaroxaban in Non-St-Elevation Myocardial In- farction. Journal of the American College of Cardiology, 71(11S), A1398-A1398.




