Research Article - (2025) Volume 10, Issue 3
Structural and Functional Synergistic Collagen-Based Formula: in Vitro Characterization and Clinical Evaluation of Dermatological Outcomes
2Health & Biomedicine Department, Leitat Technological Center, E-08028 Barcelona, Spain
3Laboratorio Dr. Goya, 28805 Alcala de Henares, Spain
Received Date: Jul 13, 2025 / Accepted Date: Aug 11, 2025 / Published Date: Aug 20, 2025
Copyright: ©Â©2025 Aleix Cuenca., et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Cuenca, A., Soto-Fernandez, C., Iascu, A., Bravo, Y., Romero-Rueda. J. (2025). Structural And Functional Synergistic Collagen-Based Formula: in Vitro Characterization and Clinical Evaluation of Dermatological Outcomes. Int J Clin Expl Dermatol, 10(3), 01-17
Abstract
Introduction: Dermal fibroblasts senescence impairs collagen turnover and decreased hyaluronic acid (HA) levels increase dryness and weaken barrier integrity, contributing to skin aging. Hydrolyzed fish collagen has been shown to stimulate extracellular matrix (ECM) synthesis, promoting collagen synthesis and potentially reducing signs of aging.
Aims: To characterize a synergistic formulation in vitro and assess its clinical effects on skin hydration and signs of ageing.
Methods: Human dermal fibroblast proliferation was assessed after treatment with a nutraceutical mixture of active ingredients with expected synergistic effects, containing fish collagen and antioxidants, including HA, Coenzyme Q10, and Vitamin C (INDIBA Sicily Collagen). Type I and III collagen accumulation in the ECM was quantified by ELISA. Skin ageing-related gene expression was quantified by real-time PCR. A single center, prospective cohort study including 22 females (>35 years) assessed the effects of daily oral administration of the nutraceutical over 56 days on skin hydration, wrinkle density and number, and other dermatological parameters.
Results: The synergistic formula significantly stimulated fibroblast proliferation, as well as HA, and collagen I and III production, with comparable effects to those of TGF-1. Clinically, the nutraceutical was well tolerated and led to a significant increase in skin hydration, but no wrinkle density or facial sagging improvement. Participants reported a high level of satisfaction, particularly regarding skin nourishment and moisturization.
Conclusion: The synergistic formula stimulates fibroblast proliferation, HA, and collagen production, with increased skin hydration observed clinically. Further research is required to explore its longer-term anti-aging efficacy and potential as a woundhealing adjuvant.
Keywords
Fish Collagen, Hyaluronic Acid, Collagen Type 1, Collagen Type Iii, Fibroblast Proliferation, Skin Aging, Nutraceuticals, Skin HydrationBackground
With chronological aging, human skin undergoes a physiological decline that affects its mechanical, protective, and wound healing capacities [1]. In the dermis, fibroblasts, the most abundant cells, synthesize, organize, and maintain the extracellular matrix (ECM), a collagen-rich network that also contains elastin and glycosaminoglycans (GAGs). Dermal fibroblast replicative senescence plays a major role in the skin aging process, via irreversible cell-cycle arrest and the acquisition of an aberrant secretory phenotype, leading to chronic inflammation and defective ECM turnover [2]. Increased degradation of the ECM, alongside the reduced biosynthesis of new ECM components, results in further breakdown and diminished production of key ECM components [3]. At the ultrastructural level, the aged dermal collagen network becomes increasingly fragmented, with shorter and disorganized fibrils. Additionally, elastin fibers degrade and lose elasticity, contributing to impaired dermal structural integrity [4].
And yet, the predominant component of the dermal ECM, also present in the epidermis, is hyaluronic acid (HA). The most notable histochemical change observed in senescent skin is the significant reduction of HA: the skin’s HA content starts to diminish as early as twenty years of age, decreasing to half by the age of fifty. This reduction in HA levels lead to a defective skin endogenous moisturizing capacity and contributes to the appearance of fine lines and wrinkles, dryness, roughness, and overall reduced barrier integrity, as well as thinning of the epidermis [5]. The reasons behind this age-related change in HA homeostasis are unclear; however, the clinical manifestations of this reduction in HA levels accompany the progressive loss of collagen and elastin. All these cellular and molecular alterations contribute to the apparent dehydration, atrophy, and loss of elasticity that characterizes aged skin.
Synergistic oral formulations with structural and functional active ingredients are emerging as efficient anti-aging nutraceuticals due to their capacity to stimulate the synthesis of ECM components, including intrinsic collagen and HA, while also reducing collagen degradation [6]. Collagen peptides are the hydrolysis product of native collagen, characterized by improved bioavailability and solubility, and reduced allergenic properties compared to native collagen. Additionally, hydrolyzed fish collagen (HFC) is metabolically compatible and free of religious constraints. A key aspect of this formula is the synergistic combination of collagen- stimulating components, such as HFC, with powerful antioxidants, including Coenzyme Q10 and Vitamin C, which have been proven to effectively reduce the increased risk of oxidative stress connected with collagen synthesis [7,8]. In this study, we evaluated the potential of a newly marketed synergistic fish collagen-based formula (INDIBA Sicily Collagen) enriched with vitamins, HA and coenzyme Q10, to reduce signs of skin aging. We assessed the in vitro effects of the formula by examining its ability to stimulate the proliferation of aged human dermal fibroblasts (HDFs), enhance the production and deposition of ECM proteins, and influence the expression of aging-related genes. In addition, we conducted in vivo dermatological and biometric assessments to determine its tolerability and effectiveness as a nutraceutical, along with a subjective evaluation of its organoleptic properties.
Methods
The present study used both in vitro and clinical approaches to characterize the properties and dermatological outcomes of the nutraceutical formula (INDIBA Sicily Collagen). The composition of the nutraceutical formulation is detailed in Supplementary Table 1 (Annex 1).

Table 1: Exclusion criteria for the clinical study
In vitro Analysis of the Synergistic Formula
Sterility Assessment of the Formulation
To ensure cell viability and prevent potential cell culture disorders related to microbiological contamination during in vitro tests, the sterility of the formulation was previously assessed by microbiological tests. To this end, the product was directly diluted in both HDF culture medium (see supplementary Methods) and microbial Luria Bertani broth (LB) medium, in sterile conditions using a BIOII cabinet (BIOIIA, TELSTAR, USA). The preparations were incubated for 5 days at 37 ºC, with daily monitoring to detect bacterial growth. This monitoring process was conducted both macroscopically, by observing the turbidity level of the preparation, and microscopically through direct examination under an optical microscope to identify the presence of microorganisms.
Cell Proliferation Assay
In order to assess the capacity of the oral formulation to promote skin cell proliferation, the proliferative rate of the HDFs after treatment with INDIBA Sicily Collagen was evaluated by the Alamar BlueTM Cell Proliferation assay. Briefly, both neonatal and aged HDF (see Supplementary methods, Annex 1) were seeded in 96-well culture plates and maintained in their specific growth medium for 24 h in standard culture conditions (SCC). Thereafter, cells were starved overnight with a serum-deprived medium (DMEM assay medium supplemented with 0.1% (v/v) Fetal bovine serum, Supplementary methods, Annex 1) and exposed to eight different concentrations of the synergistic fish collagen-based formula (from 10 to 0.08 mg/mL) for 24 and 72 hours. In parallel, HDFs were treated with 0.02% (w/v) sodium dodecyl sulfate (SDS, Merck, Sigma) as a positive control for cytotoxicity, while non-treated (NT) cells were maintained in assay medium. After removing the culture medium containing the tested product, cells were incubated with 100 μL/well of 10% (v/v) Alamar BlueTM (Invitrogen, Waltham, Massachusetts, USA) solution for 2 - 3 hours in SCC and protected from light. Fluorescence (Excitation wavelength: 530-560 nm / Emission wavelength: 590 nm) was measured with a Safire2 multi-detection plate reader (Tecan; ZU, Switzerland), and cell proliferation rate was calculated as the differences in relative fluorescence units (RFU) normalized to NT cells (basal level, 100%). Treatments reducing proliferation over 80% were considered cytotoxic.
Evaluation of the Stimulation of Extracellular Matrix Protein Production
Serum-deprived (0.1% FBS) HDFs were exposed to 0.32 to 1.25 mg/mL of the synergistic formula for 72 h in SCC. In parallel, HDFs were exposed to Transforming Growth Factor Beta-1 (TGF- 1) 10 ng/mL (Peprotech, USA), a widely recognized physiological driver of collagen production and secretion into the ECM9, used as a positive control for the induction of ECM protein synthesis, while NT cells were maintained in assay medium (Supplementary methods, Annex 1) as the negative control. Following two washes with Hank's balanced salt solution (HBSS), cells were fixed with 3% paraformaldehyde (PFA) in Phosphate-buffered saline (PBS). The accumulation of the ECM proteins Type I and III collagens was quantified by indirect ELISA using the cell layer as the solid phase10. Briefly, human primary anti-collagen I and III (Sigma) were used, which were detected using a biotinylated secondary antibody (Vector Laboratories; USA) and a streptavidin-conjugated horseradish peroxidase (streptavidin-HRP, Sigma) to amplify the signal. The HRP-substrate OPD (SIGMAFAST OPD, Sigma) was then added, and the absorbance (429 nm) was measured with a Synergy Plate Reader (BioTek, Winooski, USA). Absorbance values were corrected by subtracting background signal values obtained from control wells where the primary antibody was omitted. The stimulated accumulation of type I and III collagens in the ECM was calculated as the difference in absorbance of formulation-treated cells compared to NT cultures.
Analysis of Skin Aging-related Genes
The antiaging capacity of the synergistic formula was evaluated by quantitative analysis of the expression of five target genes using real-time quantitative polymerase chain reaction (RT-qPCR). Overnight serum-starved aged HDFs at 80% confluence were exposed to three different concentrations of the synergistic formula and incubated for 24 h in SCC. In parallel, aged HDFs maintained in assay medium (2% FBS) were used as a negative control (NT), and quercetin (10 μM, Sigma), a natural antioxidant with inhibitory capacity on aging-induced cellular markers, was used as positive control [11]. After treatment, cells were washed twice with HBSS and lysed with RIPA buffer. Total RNA was extracted using the RNeasy Kit (Qiagen Hilden, Germany) according to the manufacturer’s instructions. Samples were treated with DNase-I (Qiagen) and cDNA synthesis was performed using PrimeScript RT Master Mix Perfect Real Time (Takara Bio, Kusatsu, Japan) in a MiniAmp Thermal Cycler (Applied Biosystems, Waltham, Massachusetts, USA). Primer sequences used for RT-qPCR are shown in Supplementary Table 2 (Annex 1). The five target genes evaluated are key genes involved in skin aging. COL1A2 encodes collagen type I α2 chain and its expression declines with aging [12]. HAS2 encodes the enzyme Hyaluronan synthetase 2, and its downregulation is associated with decreased secretion of hyaluronic acid by older human fibroblasts [13,14]. Matrix metalloproteinase 1 (MMP1) regulates collagen turnover in aged skin by initiating the cleavage of several types of collagens. Age- dependent decrease in Sirtuin 1 (SIRT1) expression correlates with reduced fibroblast proliferative capacity and a lower number of skin fibroblasts [15]. Finally, Interleukin 6 (IL6) was analyzed as a key marker of inflammation. RT-qPCR was carried out in a Light Cycler 480 (Roche, Basel, Switzerland) system using KiCqStart® SYBR® Green (Sigma) and TB Green (Takara). Data analysis was performed according to the 2-ΔΔCq method as previously described,16 using the GAPGH gene for expression normalization.
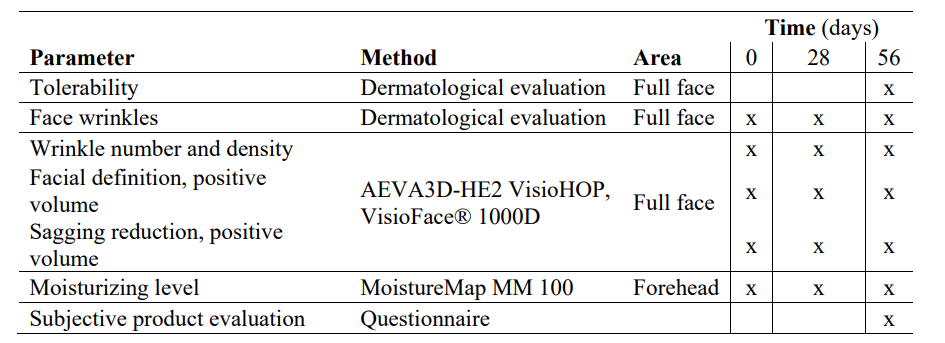
Table 2: Clinical study schedule
Clinical Evaluation
Study Design and Population
A single-center, prospective, cohort study was performed to evaluate the dermatological effects of daily oral administration of the synergistic fish collagen-based formula (INDIBA Sicily Collagen) for 56 days. Twenty-two healthy female participants, aged > 35 years with signs of skin aging, including dull and sagging skin, presence of wrinkles and fine lines, deformation of the facial contour, and nasolabial folds were included. Additional inclusion criteria were good general physical and mental health. Exclusion criteria are presented in Table 1.
Objectives, Assessments, and Variables
Data collection was performed at Dr. Goya analysis (Madrid, Spain). The main objectives of the clinical evaluation were to determine the tolerability and efficacy of the synergistic oral collagen-based formula (10 g/daily) in restoring skin-aging-related parameters after 28 and 56 days of administration. To standardize skin conditions among the participants, the week before the trial began, participants were instructed to avoid applying skincare products and/or topical drugs in the experimental areas and to perform a correct pre-wash. We recorded the incidence of adverse reactions (ARs) to evaluate the formula's tolerability. The efficacy outcomes consisted of change in the number of wrinkles, wrinkle density (%), positive volume (mm3) of the face and oval (as a sagging indicator), and near-surface hydration (moisturizing efficacy) between the initial day (D0) and 56 days (D56) of continuous oral intake of the synergistic formula (INDIBA Sicily Collagen). The corresponding variables were assessed at D0, at 28 days (D28), and at D56 using a biometric evaluation, while wrinkle number and density were clinically evaluated by an external dermatologist. Moreover, data regarding the participants' perception of the organoleptic characteristics of the formulation, degree of satisfaction with the product and subjective efficacy were collected at the end of the study using specific questionnaires.
Skin Tolerance Assessment
The tolerability of the nutraceutical formulation was monitored by recording any ARs occurring during the clinical study according to the dermatologist's criteria. At the end of the study, a dermatologist evaluated the appearance of skin ARs, including erythema, xerosis/desquamation, edema, exudation, comedogenicity, and pigmentation alterations, on a three-point Likert scale (mild/ moderate/severe). Relatedness to the synergistic formula was recorded as a six-point Likert scale: not related, improbable, possible, probable, certain, or not assessable.
Biometric Measurements
Biometric evaluation included measurements of the topography of the facial skin with AEVA3D- HE2 VisioHOP (Eotech, Marcoussis, France). Before measurement, participants were acclimatized for 10 minutes in a controlled environmental room at 20 ºC ± 2 ºC and 40%-60% relative humidity. The number of wrinkles, wrinkle density (%), and positive volume (mm3) of the face and oval (as a sagging indicator) were recorded. Moisturizing efficacy was evaluated by microtopography of the distribution of near-surface hydration in the skin (GrayIndex scale, arbitrary units, AU), based on the different capacitance in a conductive material (water-containing) and non-conductive material, with MoistureMap MM 100 (Courage & Khazaka electronic), using 3D facial photographs taken with AEVA3D- HE2 VisioHOP (EOTECH) and VisioFace®1000D (Courage + Khazaka electronic GmbH, Germany).
Dermatological Assessment
An external dermatologist evaluated the wrinkles on the forehead, frown, crow’s feet, and the area around the nasolabial fold using a six-point scale (0, none; 1, perceivable; 2, shallow wrinkles; 3, moderately deep wrinkles; 4, deep, well-defined edges; and 5, very deep, redundant folds).
Subjective Evaluation
Subjective evaluation of organoleptic characteristics, product packing, and efficacy were assessed using questionnaires completed by the participants at the end of the study. The questionnaire included 5 items regarding organoleptic characteristics and general opinion about the nutraceutical, with answers on a seven- point Likert scale ranging from 1, “really dislike it,” to 7, “like it a lot;” 4 items evaluating the product taste and aftertaste on a four-point scale ranging from 1, “a lot,” to 4, “none;” and 7 items evaluating the subjective opinion on packaging using a seven- point Likert scale ranging from 1, “really dislike it,” to 7, “like it a lot.” Moreover, a five-point Likert scale was used to rate 30 items evaluating the subjective efficacy of the nutraceutical, ranging from 1, “totally disagree,” to 5, “totally agree.” The clinical study schedule is summarized in Table 2.
Statistical Analysis
All biometric variables were recorded over multiple time points for each participant, resulting in repeated and correlated measurements. To appropriately account for this data structure, linear mixed-effects models (LMM) were used for continuous variables, incorporating random effects at the subject level to allow for individual variability in the intercepts. For categorical or ordinal outcomes, generalized linear mixed-effects models (GLMM) were applied.
Prior to any statistical analysis, data were preprocessed. This included a comprehensive data cleaning step and identification of outliers. In cases of missing values, whether due to participant dropout or instrument failure, or outliers behaving inconsistently with the overall trend, the corresponding participant was excluded from the analysis. This approach ensured a consistent sample size across all time points and minimized analytical noise. All exclusions were documented and reported in the results section corresponding to each parameter.
Descriptive statistics were calculated separately for numerical and categorical variables. For numerical variables, the following measures were reported: mean, standard deviation (SD), 95% confidence interval at each time point, difference from baseline, percentage variation from baseline, percentage of participants showing improvement, and maximum individual improvement observed. These variables were visualized using box plots, which also displayed medians, means, outliers, and the overall trend across time points.
For categorical or scaled variables, descriptive statistics included mean, median, mode, standard deviation, minimum, maximum, percentage variation from baseline, and the predicted probability distribution. Bar charts were used to visualize the cumulative frequency of each response item over time.
Assumption checks were performed prior to model fitting. Normality and homogeneity of variances were assessed to determine whether parametric or non-parametric methods were appropriate. If assumptions were met, ANOVA with Tukey's post hoc test was applied; otherwise, Kruskal-Wallis tests followed by pairwise Wilcoxon tests were used. A two-sided significance level of 0.05 was established for all inferential analyses.
In the case of comparative studies involving multiple experimental groups (e.g., placebo vs. treatment), all analyses were conducted both separately and jointly across groups, and the percentage variation between groups at each time point was reported. All statistical analyses were performed under blind conditions to avoid bias, meaning that the analyst was unaware of group assignments during the evaluation process.
Results
In Vitro Characterization
Dermal Fibroblast Proliferation
The treatment with the synergistic formula (INDIBA Sicily Collagen) at 0.08 to 0.31 mg/mL enhanced the proliferation of both HDF and aged HDF after 24 and 72 hours of exposure, compared to NT cells (p<0.0001) (Figure 1). At higher concentrations (0.62 to 1.25 mg/mL), the synergistic formula significantly promoted the proliferation of HDFs after 24 hours of treatment (p<0.0001), showing no effect at 72 hours. On the other hand, at concentrations above 2.5 mg/mL, all cell types showed a trend toward reduced proliferation at both time points, particularly HDFs (p <0.05) and with a slightly smaller effect on aged HDFs compared to HDFs.
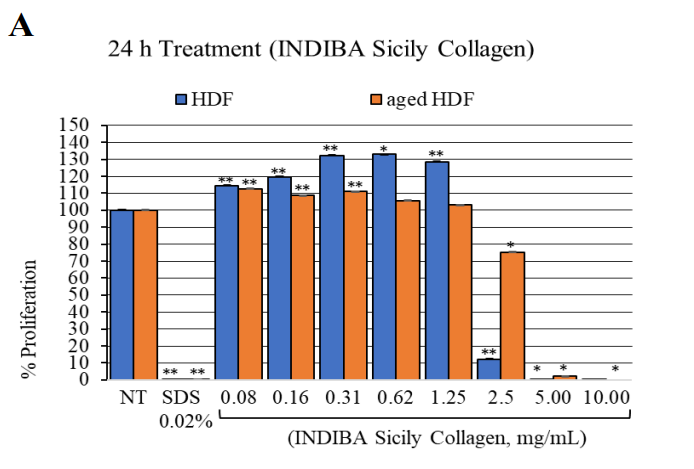
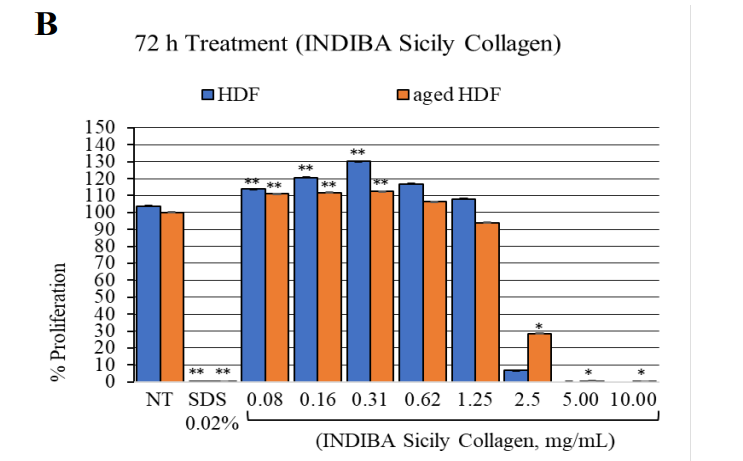
Figure 1: HDF and aged HDF proliferation at 24 h (A) and 72 h (B) after treatment with the nutraceutical (INDIBA Sicily Collagen).**, p<0.0001; *, p<0.04.
Production Enhancement of Extracellular Matrix Proteins
The synergistic formula significantly increased the production of Type I collagen in HDF across all tested concentrations (p <0.005 for all concentrations) (Figure 2). At the lowest concentration of 0.32 mg/mL, the formula induced Type I collagen accumulation at the ECM in HDFs at comparable levels to those obtained with TGF-1, with a mean (SD) increase of 19.90% (6.46) and 16.59% (4.38), respectively, compared to NT cells. Notably, at 0.62 mg/ mL, the formula led to a significantly greater Type I collagen accumulation of 34.55% (10.84), surpassing the levels induced by TGF-1 (16.6% [4.38]). Likewise, at this concentration of 0.62 mg/mL, the formula significantly increased Type III collagen accumulation, with a mean (SD) increase of 53.43% (28.53) (p=0.015), whereas TGF-1 induced a modest non-significant increase of 17.74% (5.52) (p=0.5).

Figure 2: Nutraceutical (INDIBA Sicily Collagen) induced Extracellular Matrix proteins (ECMp) accumulation. ***, p<0.0003; *, p<0.05.
Modulation of Aging-Related Gene Expression
Next, we examined the effect of the synergistic formula on the expression of key genes involved in skin aging, including HAS2, COL1A2, SIRT1, IL6, and MMP1. Aged HDFs were treated with increasing concentrations of the formula or 10 g/mL quercetin, which served as a negative regulator of ECM protein production (Figure 3). All tested concentrations of the formula significantly upregulated HAS2 expression compared to baseline levels (p<0.0035) (Figure 3A), suggesting a potential increase in HA synthesis in aged HDFs. No significant effects were observed on either COL1A2 or SIRT1 expression. On the other hand, the highest concentration tested significantly increased MMP1 expression (p=0.001), which may indicate enhanced fragmentation of collagen (Figure 3B). These assumptions should be confirmed with additional functional studies.
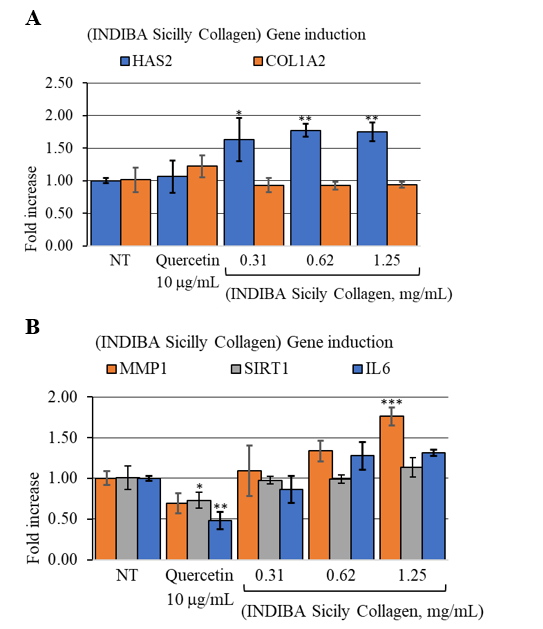
Figure 3: Nutraceutical (INDIBA Sicily Collagen) induced gene expression fold change of (A), ECMp genes HAS2 and COL1A2. **, p<0.0021; *, p<0.0035. (B), collagen remodeling gene MMP1, oxidative stress response, SIRT1 and inflammation marker IL6. ***, p=0.001.
Clinical Evaluation
Demographic and Clinical Characteristics of Study Participants
This prospective cohort study recruited 22 female participants, with a mean (SD) age of 51.62 (8.79) years, none of whom was excluded, and one withdrew from the study for reasons unrelated to the study (see Annex 1, Supplementary Figure 1). Ninety per cent (n=20) of the 22 participants were not regularly using any dietary supplements, 36% (n=8) were occasional users, and 54% (n=12) did not use any dietary supplements. Additionally, 10% (n=2) used dietary supplements regularly.
Figure S1: Participants’ flowchart.
Tolerability
After 56 days of continuous use of the nutraceutical, none of the participants presented erythema, xerosis/desquamation, edema, exudation, comedogenicity, or alterations in skin pigmentation. One participant reported a mildly increased frequency of bowel movements, possibly related to use of the product.
Biometric Assessment of Anti-aging Efficacy
The efficacy of the synergistic formula with both structural and functional active ingredients as an anti-aging product was evaluated by assessing changes in biometric measurements throughout the study. Hydration was quantified in the forehead using microtopography (See Figure 4A showing the same participant's microtopography at D0, D28 and D56, and its GrayIndex values). Hydration levels increased from mean (SD) 81.30 AU (30.22) at D0 to 114.44 AU (41.55) at D56 (p=0.01) (Figures 4A and B). Global wrinkle density (entire face) and forehead wrinkle number did not show significant changes throughout the study (Table 3, p>0.5 and Supplementary Figure 2, Annex 1). Likewise, facial definition and sagging, both measured as positive volume, were not statistically different before and after treatment administration (Table 3, p>0.5).
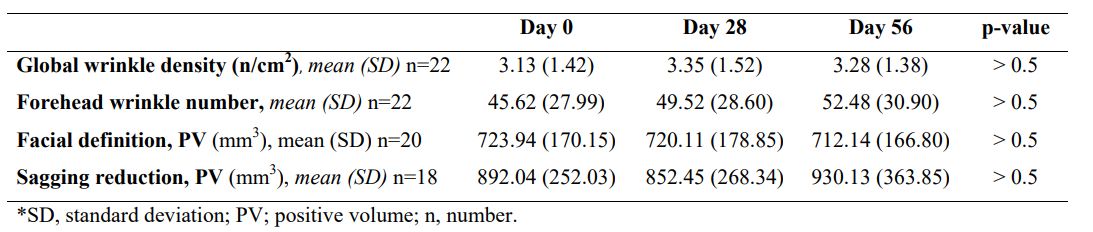
Table 3: Nutraceutical (INDIBA Sicily Collagen) AEVA3D-HE2 VisioHOP Biometric evaluation.
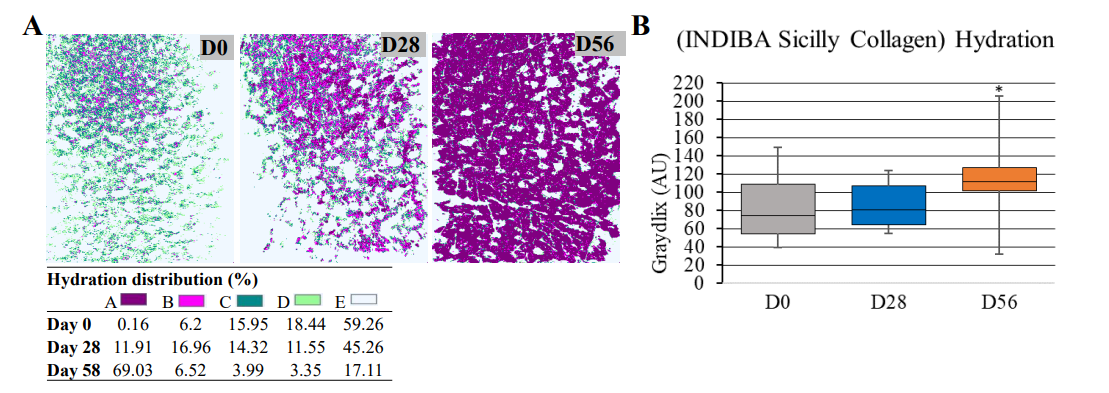
Figure 4: Hydration levels (GrayIndex). (A), representative images showing the forehead microtopography at and quantified hydration levels (GrayIndex scale) at D0, D28, and D56. (B), Box plot showing the nutraceutical formula (INDIBA Sicily Collagen) effect on the forehead hydration at D0, D28, and D56. Horizontal lines within the boxes denote median values; boxes extend from the 25th to the 75th percentile of each group's distribution of values; whiskers denote adjacent values (i.e., the most extreme values within 1.5 interquartile range of the 25th and 75th percentile of each group); *, p=0.01.
Figure S2: Global wrinkle density at days 0, 28, and 56.
Dermatological Evaluation of Anti-aging Efficacy
The external dermatologist classified wrinkles in the forehead, frown, crow’s feet, and the area around the nasolabial fold as shallow to moderately deep wrinkles, with mean (SD) scores ranging from 2.81 points (1.29) to 3.67 (1.11). No significant changes were observed during the study period (p>0.5) (Table 4).
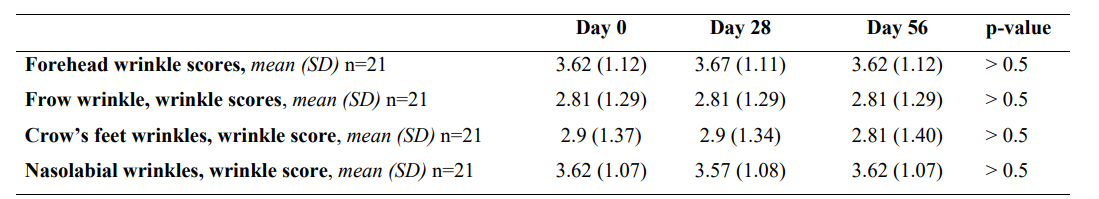
*SD, standard deviation; n, number
Table 4: Nutraceutical (INDIBA Sicily Collagen) dermatological evaluation .
Subjective Evaluation
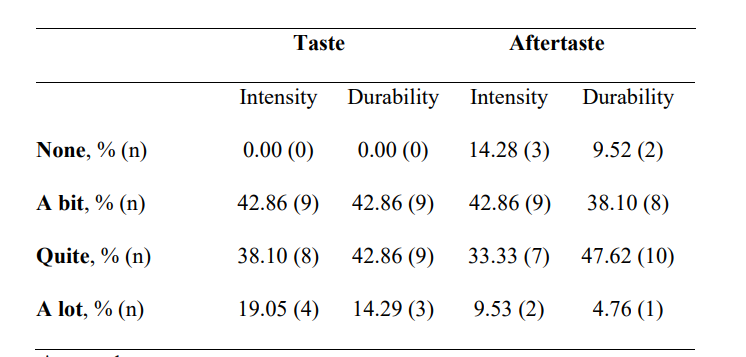
The questionnaire's average scores for organoleptic characteristics of the formula are presented in Table 5. Less than half of the participants found a bit of taste durability and intensity (42.86% and 42.86%, respectively). Aftertaste intensity was defined as “a bit” by 42.86% of patients and durability as “quite” by 47.62%.
Regarding global organoleptic characteristics, after 56 days of continuous use of the nutraceutical, 80.95% (n=17) of the participants liked the product a lot and moderately (Table 6). Aroma and color were liked a lot and moderately by 69.91% (n=13) and 47.62% (n=10) of participants, respectively. Texture and flavor were found satisfactory by 42.86% (n=9) and 71.43% (n=15). Regarding perception of efficacy, after using the product, 76.19% of participants found the area more nourished (See Supplementary Table 3, Annex I, totally agree and agree, n=16), the skin soft (76.19%, n=16), more moisturized (80.95%, n=17) and less tight (71.43%, n=15). The nutraceutical improved the self-perceived appearance of the skin in 71.43% of participants (n=15), skin texture in 76.19% (n=16), and firmness and elasticity in 61.91% (n=13) and 57.15% (n=12), respectively. Packing subjective evaluation is presented in Supplementary Table 4 (Annex 1).
*n, number.
Table 5: Subjective evaluation of the nutraceutical (INDIBA Sicily Collagen) taste and aftertaste
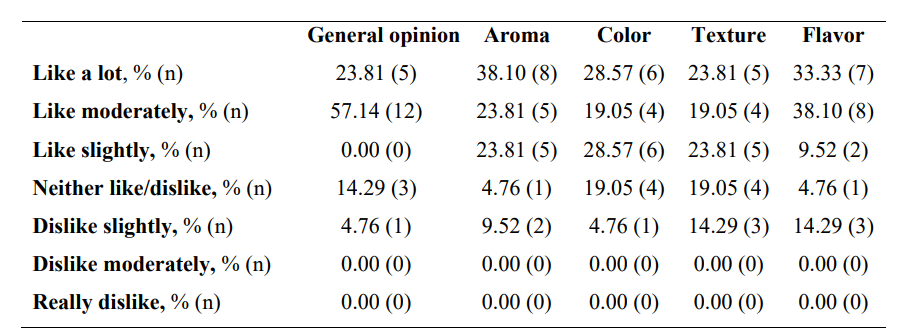
Table 6: Subjective opinion on global assessment and organoleptic characteristics of the synergistic formula (INDIBA Sicily Collagen).
Discussion
Our findings show that the synergistic formula (INDIBA Sicily Collagen) stimulated the proliferative capacity of both HDFs and aged HDFs in vitro. Additionally, it enhanced ECM accumulation of collagen Types I and III, along with upregulation of COL1A2, HAS2, and MMP1 expression. Clinically, the nutraceutical showed good tolerability, increased skin hydration levels, and no measurable changes in wrinkle density or number.
The synergistic nutraceutical induced a notable stimulation in the proliferative capacity of HDFs and aged (senescent) HDFs. At concentrations below 0.32 mg/mL, this increase was accompanied by enhanced accumulation of Collagen Type I in the ECM, without altering COL1A2 expression. This suggests that the formula may regulate collagen turnover rather than promote increased production through gene upregulation. Importantly, the increased proliferative capacity did not alter SIRT1 expression, a marker associated with cellular stress.17 Changes in SIRT1 activity are linked to increased oxidative stress via a bidirectional crosstalk mechanism. Since collagen synthesis process is often associated with oxidative stress, the unaltered SIRT1 expression levels might indicate a positive synergistic effect of the formula’s antioxidant ingredients (HA, vitamin C, and Coenzyme Q10 among the most efficient antioxidants in the nutraceutical formula) during the endogenous synthesis of new collagen.
Interestingly, Type I collagen accumulation in the ECM induced by 0.32 mg/mL of the synergistic formula was similar to the levels triggered by TGF-1, a potent inducer of tissue collagen deposition. At higher concentrations (0.62 mg/mL), the formula widely surpassed the effect of TGF-1, potentially indicating an equal or superior capacity of the synergistic formula to induce ECM accumulation of collagen I. At this higher concentration, the nutraceutical also induced the accumulation of Type III collagen. The nutraceutical did not significantly stimulate aged fibroblast proliferation at this 0.62 mg/mL concentration. Still, this concentration kept the cell proliferation rate slightly above controls (mean [SD] increase 6.32% [4.17]) and produced a slight but yet not significant increase in MMP1 expression (34% [13]). Both trends were similar at higher concentrations of the formula (1.25 mg/mL), with sustained proliferation of aged HDFs, increased expression of MMP1 (76% [11]), and production of Type I and III collagen well above the levels induced by TGF-1. Together, these results might suggest that the synergistic formula enabled a physiological skin repair mechanism. Remarkably, the increase in collagen III/collagen I ratio, accompanied by MMP1 induction, is characteristic of the wound healing process [18].
In this scenario, collagen I deposition is followed by collagen III accumulation, while MMP1 cleaves undamaged collagens as part of the restorative tissue remodeling and to facilitate proliferating fibroblast migration [18,19]. In this context, enhanced collagen fragmentation by MMP1 might keep the already enhanced collagen production in balance, allowing the migration of proliferating fibroblasts. Interestingly, micro-needling therapy, initially introduced for skin rejuvenation, seems to follow a similar sequence: increased deposition of collagen I and III that will be later remodeled. Moreover, in the wound healing process, several MMP1-stimulating factors have been reported to physiologically modulate collagen turnover, resulting in an anti-fibrotic (scar-free) effect [19,20].
On the other hand, despite the increased in vitro fibroblast proliferation and collagen accumulation on the ECM, no significant impact on the biometric analysis or wrinkle quantification was observed in clinical evaluation, as reported for other hydrolyzed fish collagen formulations [21,22]. In this regard, collagen turnover in the skin is estimated at around 74 days, indicating that longer treatment periods may be required to observe visible effects [23].
Additionally, our results showed a sustained fold increase in the expression of HAS2, the gene encoding hyaluronan synthetase 2, associated with the aging-dependent reduction in HA. The increased expression of HAS2 in vitro correlates well with the enhanced skin hydration levels quantified in vivo and the participants' self-perception of increased hydration after 56 days of treatment with the synergistic formula. Remarkably, elevated levels of HA are synthesized during scar-free fetal tissue repair, and the prolonged presence of HA ensures such scarless tissue repair.3 Indeed, HA plays a key role in the wound healing process, as well as in maintaining an optimal skin barrier, with HA alterations being found in skin barrier-related pathologies, including atopic dermatitis (AD) [24]. In this context, it is worth mentioning that topically applied HA has a limited restoring capacity because it is cleared rapidly from the dermis and degraded [25]. In this regard, and since the nutraceutical was well tolerated, further research is needed to determine the potential of the formula as an inside-out approach to alleviating barrier-related disorders. Likewise, this synergistic formula (INDIBA Sicily Collagen) has the potential to stimulate key factors in the wound-healing process, including fibroblast proliferation, promotion of endogenous HA and collagen production, and MMP1-mediated reduction of fibrosis.
Limitations of this study include the study size, the short time frame of the clinical study, which could preclude the macroscopic detection of enhanced fibroblast proliferation and collagen turnover, and the focus on wrinkle number and density alone. Parameters such as skin thickness or biometric wrinkle depth were reported for other fish collagen formulations to be significant after no less than 90 days of treatment.23 Longer study periods may be required to observe significant changes in skin parameters, such as wrinkle density and number, aligning with the estimated collagen turnover timeframe in the skin. Additionally, variables such as HA content, oxidative stress levels, or HA epidermal-dermal distribution in the context of wound-healing and/or barrier-related alterations might offer insights into yet unexplored potential uses of the product.
Conclusion
The synergistic formula with structural and functional active ingredients, including fish collagen, HA, antioxidants, and vitamins (INDIBA Sicily Collagen), may positively impact skin health by enhancing fibroblast proliferation and increasing the accumulation of key ECM proteins, particularly Type I and III collagen. Additionally, the synergistic formula upregulated the expression of HAS2, an enzyme involved in the endogenous synthesis of HA, a key molecule for water retention and skin hydration. This molecular effect supports the clinical findings, where volunteers showed significant improvement in skin hydration levels after treatment with the nutraceutical, reinforcing its role as a skin hydration booster. Although the in vivo evaluation did not reflect changes in wrinkle density or number, the tolerability, improved skin hydration, enhanced self-perceived hydration and texture, and subjective satisfaction were good after 56 days of daily intake. Overall, our findings provide a promising foundation for further exploring the potential benefits of this synergistic formula as a wound healing adjuvant and its role in addressing skin barrier alterations, where promotion of endogenous HA production could alleviate the associated clinical signs and symptoms.
Conflict of interest
Aleix Cuenca is employed by INDIBA as a Medical & Educational Manager but has not been involved nor has he had access to the treatment of the experimental data. Aleix Cuenca declares that he has not received any pressure or influence from INDIBA in the writing of this article. The remaining authors declare that they have no conflicts of interest. All authors ensure the independence and integrity of the data and the analysis thereof.
References
- Quan, T., & Fisher, G. J. (2015). Role of age-associated alterations of the dermal extracellular matrix microenvironment in human skin aging: a mini-review. Gerontology, 61(5), 427- 434.
- Shin, S. H., Lee, Y. H., Rho, N. K., & Park, K. Y. (2023). Skinaging from mechanisms to interventions: focusing on dermal aging. Frontiers in Physiology, 14, 1195272.
- Depalma, R. L., Krummel, T. M., Durham III, L. A., Michna,B. A., Thomas, B. L., Nelson, J. M., & Diegelmann, R. F. (1989). Characterization and quantitation of wound matrix in the fetal rabbit. Matrix, 9(3), 224-231.
- Fisher, G. J., Varani, J., & Voorhees, J. J. (2008). Looking older: fibroblast collapse and therapeutic implications. Archives of dermatology, 144(5), 666-672.
- Walker, M. (2022). Human skin through the ages. Internationaljournal of pharmaceutics, 622, 121850.
- Asserin, J., Lati, E., Shioya, T., & Prawitt, J. (2015). The effect of oral collagen peptide supplementation on skin moisture and the dermal collagen network: evidence from an ex vivo model and randomized, placebo-controlled clinical trials. Journal of cosmetic dermatology, 14(4), 291-301.
- Boo, Y. C. (2022). Ascorbic acid (vitamin C) as a cosmeceutical to increase dermal collagen for skin antiaging purposes: emerging combination therapies. Antioxidants, 11(9), 1663.
- De Luca, C., Mikhal’chik, E. V., Suprun, M. V., Papacharalambous, M., Truhanov, A. I., & Korkina, L.G. (2016). Skin antiageing and systemic redox effects of supplementation with marine collagen peptides and plant-derived antioxidants: a single-blind case-control clinical study. Oxidative medicine and cellular longevity, 2016(1), 4389410
- Kim, K. K., Sheppard, D., & Chapman, H. A. (2018). TGF-β1 signaling and tissue fibrosis. Cold Spring Harbor perspectives in biology, 10(4), a022293.
- Galvez-Martin, P., Soto-Fernandez, C., Romero-Rueda, J., Cabañas, J., Torrent, A., Castells, G., & Martinez- Puig, D. (2023). A novel hyaluronic acid matrix ingredient with regenerative, anti-aging and antioxidant capacity. International journal of molecular sciences, 24(5), 4774.
- Sohn, E. J., Kim, J. M., Kang, S. H., Kwon, J., An, H. J.,Sung, J. S., ... & Choi, J. S. (2018). Restoring effects of natural anti-oxidant quercetin on cellular senescent human dermal fibroblasts. The American journal of Chinese medicine, 46(04), 853-873.
- Potekaev, N. N., Borzykh, O. B., Medvedev, G. V., Petrova,M. M., Gavrilyuk, O. A., Karpova, E. I., ... & Shnayder, N. A. (2021). Genetic and epigenetic aspects of skin collagen fiber turnover and functioning. Cosmetics, 8(4), 92.
- Terazawa, S., Nakajima, H., Tobita, K., & Imokawa, G.(2015). The decreased secretion of hyaluronan by older human fibroblasts under physiological conditions is mainly associated with the down-regulated expression of hyaluronan synthases but not with the expression levels of hyaluronidases. Cytotechnology, 67(4), 609-620.
- Fisher, G. J., Quan, T., Purohit, T., Shao, Y., Cho, M. K., He, T.,... & Voorhees, J. J. (2009). Collagen fragmentation promotes oxidative stress and elevates matrix metalloproteinase-1 in fibroblasts in aged human skin. The American journal of pathology, 174(1), 101-114.
- Bielach-Bazyluk, A., Zbroch, E., Mysliwiec, H., Rydzewska- Rosolowska, A., Kakareko, K., Flisiak, I., & Hryszko, T. (2021). Sirtuin 1 and skin: implications in intrinsic and extrinsic aging—a systematic review. Cells, 10(4), 813.
- Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. methods, 25(4), 402-408.
- Benslimane, Y., Bertomeu, T., Coulombe-Huntington, J., McQuaid, M., Sánchez-Osuna, M., Papadopoli, D., ... & Harrington, L. (2020). Genome-wide screens reveal that resveratrol induces replicative stress in human cells. Molecular Cell, 79(5), 846-856.
- Mathew-Steiner, S. S., Roy, S., & Sen, C. K. (2021). Collagen in wound healing. Bioengineering, 8(5), 63.
- Kandhwal, M., Behl, T., Singh, S., Sharma, N., Arora, S., Bhatia, S., ... & Bungau, S. (2022). Role of matrix metalloproteinase in wound healing. American journal of translational research, 14(7), 4391.
- Li, Y., Kilani, R. T., Rahmani-Neishaboor, E., Jalili, R. B., & Ghahary, A. (2014). Kynurenine increases matrix metalloproteinase-1 and-3 expression in cultured dermal fibroblasts and improves scarring in vivo. Journal of Investigative Dermatology, 134(3), 643-650.
- Evans, M., Lewis, E. D., Zakaria, N., Pelipyagina, T., & Guthrie, N. (2021). A randomized, tripleâ?ÂÂblind, placeboâ? controlled, parallel study to evaluate the efficacy of a freshwater marine collagen on skin wrinkles and elasticity. Journal of cosmetic dermatology, 20(3), 825-834.
- Maia Campos, P. M. B. G., Franco, R. S. B., Kakuda, L.,Cadioli, G. F., Costa, G. M. D. A., & Bouvret, E. (2021). Oral supplementation with hydrolyzed fish cartilage improves the morphological and structural characteristics of the skin: a double-blind, placebo-controlled clinical study. Molecules, 26(16), 4880.
- Rucklidge, G. J., Milne, G., McGaw, B. A., Milne, E., & Robins, S. P. (1992). Turnover rates of different collagen types measured by isotope ratio mass spectrometry. Biochimica et Biophysica Acta (BBA)-General Subjects, 1156(1), 57-61.
- Neuman, M. G., Nanau, R. M., Oruña-Sanchez, L., & Coto,G. (2015). Hyaluronic acid and wound healing. Journal ofpharmacy & pharmaceutical sciences, 18(1), 53-60.
- Reed, R. K., Laurent, U. B., Fraser, J. R., & Laurent,T. C. (1990). Removal rate of [3H] hyaluronan injected subcutaneously in rabbits. American Journal of Physiology- Heart and Circulatory Physiology, 259(2), H532-H535.
Supplementary Tables
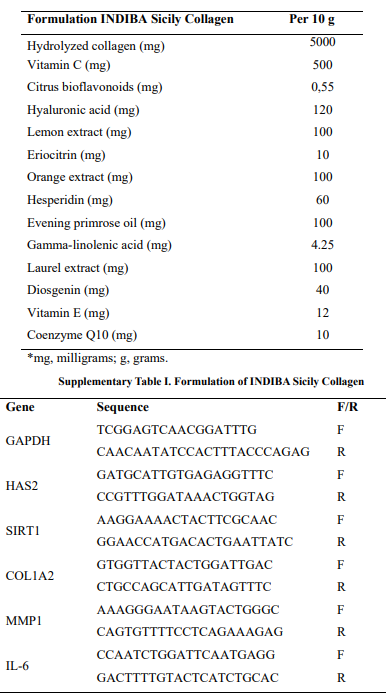
*PCR, Polymerase chain reaction: F, forward; R, reverse.
Supplementary Table II. Primers used in real-time quantitative PCR
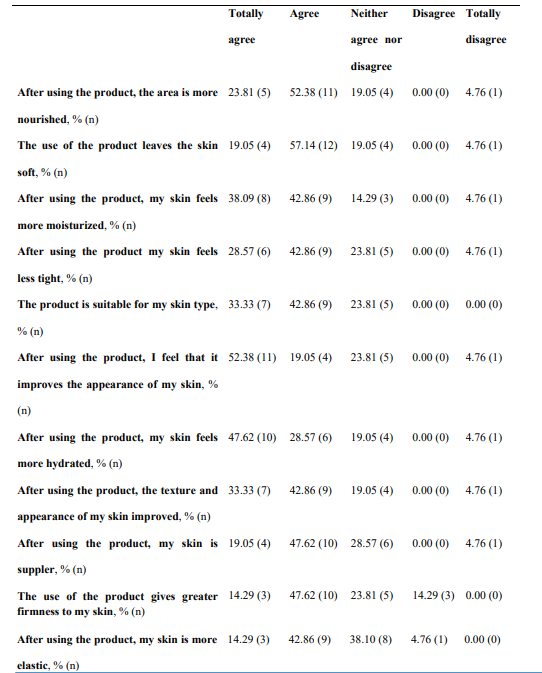
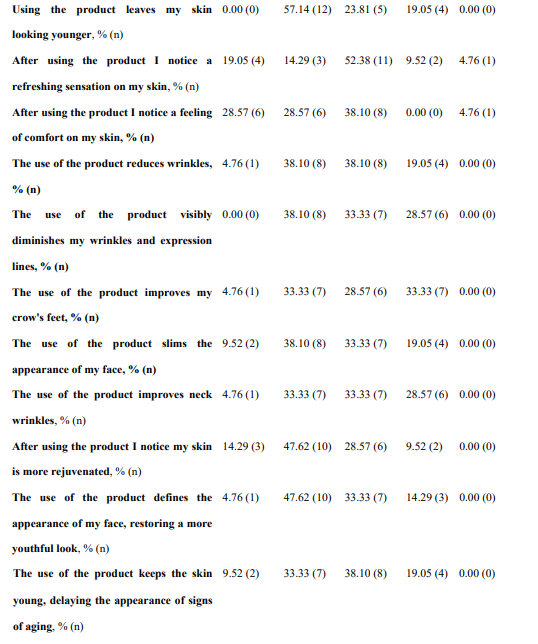
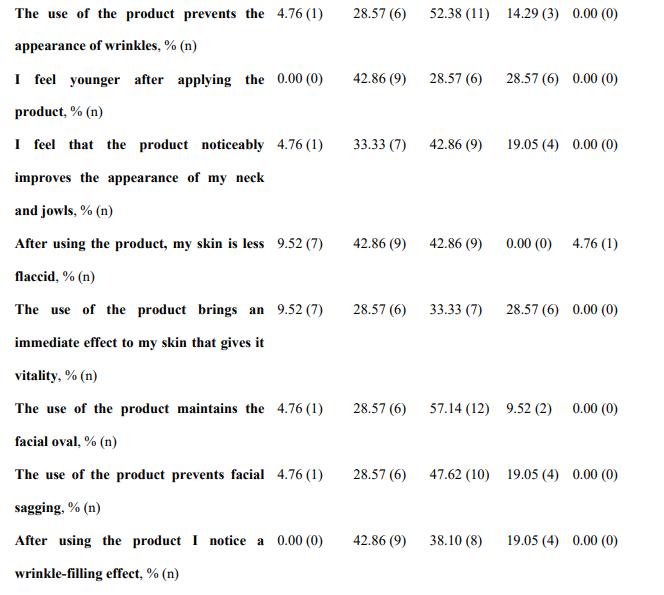
*n, number.
Supplementary Table III. Subjective evaluation of the nutraceutical (INDIBA Sicily Collagen)
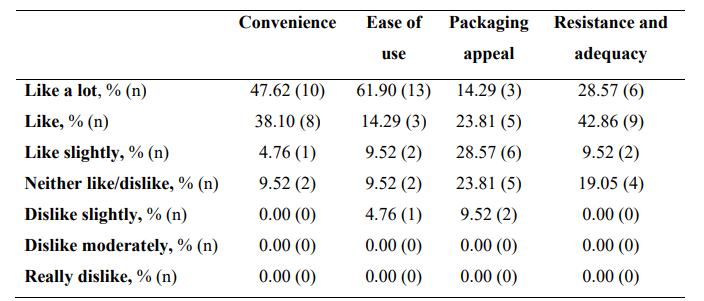
*n, number.
Supplementary Table IV. Subjective opinion on INDIBA Sicily Collagen packing


