Research Article - (2023) Volume 1, Issue 2
Specific Immunotherapy Can Be A Useful Treatment in Seasonal Pollen Induced Esophagitis
2Department of Pediatrics, Hospital Virgen de la Concha, Zamora, Spain
3Molecular Biology Department, Cellular Biology Área. León University. Leon, Spain
4Histology Department. Valladolid University, Spain
5Gastroenterology Service. Hospital Rio Carrión. Palencia, Spain
6Biodiversity and Environmental Management, University of Leon, Leon. Spain & Institute of Atmospheric Sciences and Climate. National Research Council. Bologna, Italy
Received Date: Nov 10, 2023 / Accepted Date: Dec 02, 2023 / Published Date: Dec 30, 2023
Copyright: ©2023 Alicia Armentia, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Armentia, A., Mart
Abstract
Although pollen allergy is very common in patients with eosinophilic esophagitis (EoE), it is doubtful that pollen is an etiological agent to consider.
We selected 255 patients suffered from esophagitis with seasonal exacerbation, and performed a real life study on the efficacy of immunotherapy with the detected pollen and avoidance of food, if was also detected. Allergens involved in EoE were identified by prick, specific IgE and molecular analysis: component resolved diagnosis (CRD) by microarrays.
Microscopic examination of esophageal biopsies of patients with EoE were made to verify the presence of callose (polysaccharide abundant in the polinic tubes during germination, but absent in animal tissues) in the esophagus. Endoscopy and biopsy were performed ever six months of treatment. Esophageal mucosal sections were analyzed by scanning electron microscope Immunotherapy guided by molecular analysis and biopsy study was useful to decide the treatment (avoidance or targeted immunotherapy). This treatment allowed us a more reasonable restriction of food in the diet and specific immunotherapy aimed at the suspected allergens responsible for the disease. After immunotherapy, 188 (74%) patients were discharged whit negative biopsy, no symptoms, no medication, without relapse. Specific molecular guided immunotherapy can be a useful treatment in seasonal pollen induced esophagitis.
Keywords
Pollen Tubes, Eosinophilic Esophagitis, Seasonal Esophagitis, Callose, Germination, Biopsies, Specific Immunother- apy.
Introduction
Eosinophilic esophagitis (EoE) is associated with atopic diseas- es including asthma, allergic rhinitis, and atopic dermatitis and is considered as an atopic disease of unclear etiology [1]. As in allergic asthma, mucosal barrier dysfunction has been reported [2]. Many EoE patients present dysphagia and food impaction. EoE exacerbations are often seasonal and recently has been de- scribed as a late manifestation of the allergic march [3,4].
In a previous study we hypothesized that the inflammatory re- sponse of the esophageal mucosa in patients with high levels of antibodies to pollen allergens and worsened seasonal EoE might be due to swallowing and germination of airborne pollen in esophagus mucosa [5].
We thought it might be possible the intrusion into the esophageal mucosa of pollen allergens through the tubes emitted after pollen germination, which encounter a pH and humidity resembling the stigma of spermatophytes during the pollination.
Microscopic examination of esophageal biopsies of 129 adult patients with EoE, 82 of them with seasonal exacerbation, and 100 controls with gastroesophageal reflux without eosinophilic infiltrate, were made to verify the presence of callose (polysac- charide abundant in pollen tubes but absent in animal tissues) in the esophagus. Callose was detected using histological sections stained with sirofluor fluorochrome. Esophageal mucosal sec- tions were analyzed by scanning electron microscope to detect pollen or spores. Allergens involved in EoE were identified by prick, specific IgE and component resolved diagnosis by mi- croarrays.
Callosa was detected in 82 (67%) EoE patients with positive CRD to group 1 grass pollen. All these patients suffered clini- cal exacerbation in pollen season. Exacerbation was observed at the same time only in 4 control patients. Clinical evaluations and esophageal biopsies were made every six months. Gastroin- testinal (dysphagia, heartburn, stomach upset, vomiting, consti- pation, diarrhea, failure to thrive) and allergic (rhinitis, asthma, dermatitis, anaphylaxis) histories were reviewed.
Pollen allergens were detected in 87.6% of patients with EoE. The predominant allergens were group 1 grass (55%), Art v 3 (11.3%) and lipid transfer proteins (LTPs) (19.4%) of common Mediterranean foods such as peach, hazelnuts, walnuts and wheat.
This finding aimed us 3 years ago to study more patients suffered from esophagitis and seasonal exacerbation, and to carried out a treatment with specific immunotherapy with the detected pollen and/or avoidance of the detected food guided by CRD.
Materials and Methods
We performed a real life study in all EoE patients referred to our Allergy Clinic. Informed consent and the approval of the Rio Hortega Hospital Research Ethics Committee were obtained (Ref. CEIm:2011/PI02). A total of 327 patients met clinical and biopsy criteria of EoE with seasonal exacerbations. In all these patients conventional EoE treatment (PPi , diet or steroides) pro- duce little improvement.
Allergens involved in EoE were identified by prick, specific IgE and component resolved diagnosis (CRD) by microarrays (Thermofisher diagnostic, Sweden). We began allergen specific immunotherapy (AIT) with a commercial polymerized pollen extract in 291 patients that met criteria of pollen hypersensitivity (Positive clinical data, Prick, specific IgE, and CRD) and callosa in diagnostic biopsy, who suffered from esophagitis with sea- sonal exacerbation. The mean age was 36± 18.34 years and 72% were males. Thirty-six patients reused this immunotherapy or not finished 2 years of treatment due to different reasons.
Clinical evaluations and esophageal biopsies were made every six months and we finished this therapy after a clear clinical and biopsy improvement. The immunotherapy had been applied for two years at the time of analysis of results. Patiens treated with immunotherapy and patients that denied this treatment were in- cluded in a statistical analysis.
Results
In our patients, CRD detected allergen hypersensitivity in more than 80% of patients with EoE. The predominant allergens were grass group 1 (65%) and lipid transfer proteins (LTP) of peach, hazelnuts and walnuts. Callose from pollen tubes was found in 86% of their biopsies and in all the selected patients for specific pollen immunotherapy.
After two years of CRD-guided elimination diet and/or AIT, EoE patients showed significant clinical improvement (p<0.017) and 188 (74%) patients were discharged whit negative biopsy, no symptoms, no medication, without relapse. Evolution of EoE patients is show in table 1.
|
Intervention 255 patients with EoE |
No AIT/ no avoidance |
AIT only |
Avoidance only |
AIT+ avoid- ance |
Pollen/pollen tubes |
Callose |
|
AIT |
36 |
204 |
19 |
68 |
238 |
240 |
|
Group 1 grasses pollen |
|
182 |
|
68 |
207 |
182 |
|
Other pollen mixtures |
|
25 |
|
1 |
25 |
21 |
|
Avoidance |
|
|
|
|
|
|
|
Hazelnut |
|
|
1 |
|
|
|
|
Hazelnut+walnut |
|
|
2 |
|
|
|
|
Peach/fruits |
|
|
16 |
|
|
|
|
AIT/avoidance |
|
|
|
|
|
|
|
Hazelnut |
|
|
|
23 |
|
|
|
rCor a8/hazelnut |
|
|
|
22 |
|
|
|
Peach/fruits |
|
|
|
15 |
|
|
|
Sea food |
|
|
|
8 |
|
|
|
Significant biopsy im- provement at 2 years |
1 (0.3%) |
188 (74%) |
14 (73.7%) |
64 (94.1%) |
|
|
|
Symptom free at 2 years |
1 (0.3%) |
188 (74%) |
11(57.9%) |
64 (94.1%) |
|
|
Table S1: Evolution of EoE patients after two years of specific allergen immunotherapy (AIT) and/or elimination diet.
CRD-directed AIT and/or elimination diet was efficient in treat- ing EoE patients and was well tolerated. AIT-treated patients had also better outcomes (odds ratio 177.3, 95% CI 16.2-1939.0).
We frequently observed pollen tubes surrounded by eosinophils in the proximal and middle esophageal mucosa, (Figure 1) re- sulting in micro-abscesses. Eosinophils seemed to act as if they were responding to parasitic infections. In figures 1,2 and 3 we add images from biopsies obtained during pollen season before Immunotherapy. Figure 4 shows histological changes after im- munotherapy.
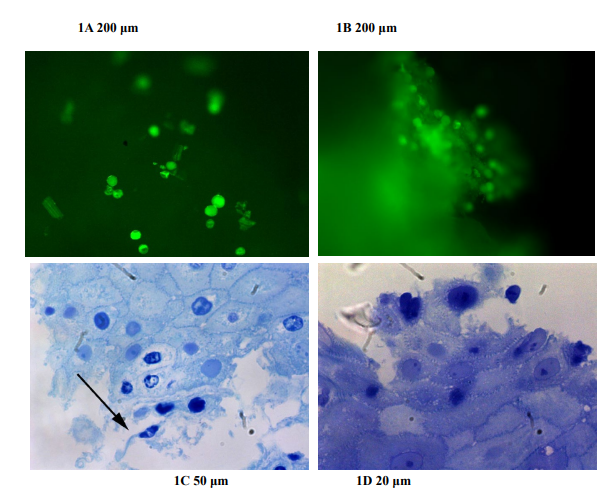
Figure S1: Epifluorescence (A and B) shows pollen, spores and other plant elements on the surface of esophageal biopsies before histological fixation. (C and D): Plant impactions and esophageal mucosae showing damaged epithelial spinous cells. Semi-thin sections with toluidine blue stain. Arrow shows pollen tubes.
Figure S2: A: damaged epithelial cells and intercellular spaces. Arrow shows elongated pollen tube

Figure S3: B: Micro-impaction mainly composed of pollen grains of the Poaceae family infiltrating intercellular spaces. Arrow shows characteristic annulus of grass pollen.
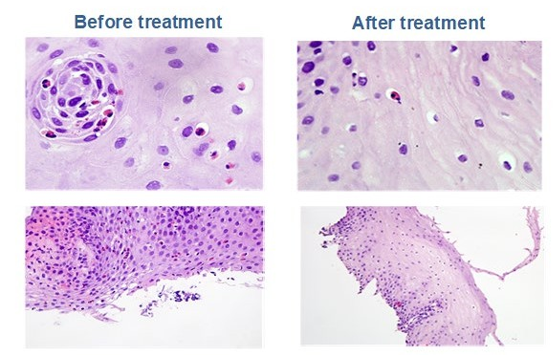
Figure S4: Human histology showed eosinophilic infiltration before AIT and elimination diet with significant decrease of eosino- phil infiltrate at two years. EoE biopsies showed eosinophilic infiltration gradually lessened after etiological treatment with diet and specific AIT. (Before AIT H/E 100x, > 15 Eo/CGA: After AIT (H/E 40x). H/E: Hematosiline-eosine stain.
Discussion
Identification of environmental and food allergens in patients suffered from EoE is important and can guide therapy. Evidence supports a link between EoE, gut inflammation and environmen- tal aeroallergens [6-10]. In a study by Ram et al including 160 pediatrics patients with EoE, 20% had biopsy examination-con- firmed variation of EoE triggered by aeroallergens and 84% of them had allergic rhinitis and asthma. Components of pollen grains are suspected to trigger changes in gut functions, some- thing leading to inflammatory conditions. Buhner et al investi- gated the effect of aqueous pollen extracts on enteric and spinal sensory neurons from humans and guinea-pig and concluded that altered nerve signaling as a result of severe pollen exposure may be a pathophysiological feature to allergic and non-allergic challenges.
Encouraged by our previous findings we treat patients with pol- len germinated in their esophagus and with positivity in CRDs with pollen specific immunotherapy. Those who also had an awareness of related foods, often described were selectively eliminated from their diet. After two years of specific immuno- therapy achieved objective improvement by clinic and negative biopsy in 74% of patients.
Recent update suggests disease exacerbations owing to the in- crease in aeroallergens to which patients are sensitized. It was also recently postulated that treatment of allergic rhino-conjunc- tivitis with allergen specific immunotherapy can improve the symptoms of EoE [11].
Our study aims to consider swallowed pollen in the triggering and exacerbation of EoE, since in addition to being airborne and inhalant, can also be ingested as food.
Currently, biomarkers and biologic therapies are not helpful for diagnosis or inducing clinical and histological remission of the disease [12]. So, we must try to identify the cause of the disease to ensure an etiological treatment of EoE.
As of yet, no single agent has been approved by the US Food and Drug Administration to treat eosinophilic esophagitis [13]. Successful therapies exist, including different medica- tions (budesonide effervescent tablet, biologics used for other Th2 mediated diseases) and dietary modifications, but disease typically recurs when the intervention is discontinued [14-16]. Specific Immunotherapy can avoid disease recurrence and other treatment complications. We have not found relapse after one year of finishing the AIT.
The efficacy of the treatment must be demonstrated. Endoscopy and biopsy is essential for diagnosis, assessment of response to therapy and ongoing monitoring of patients in histological re- mission [17].
We have been able to found in numerous patients germinated pollen in their esophagus, thanks to using a specific stain for plant cells that are different to the stains (hematoxilyn-eosin), normally used in human tissue.
Alteration of the mucosal barrier might cause the penetration of pollen grains tubes into the esophageal tissues. The subsequent germination of pollen and the release of highly-allergenic di- gestion-resistant molecules, such as group 1 grass (β-expansins) and LTPs, might be responsible for the increase in symptoms in these patients during periods of greater pollination. Stacks of rough endoplasmic reticulum are largely dissociated in heat- stressed pollen, consequently affecting protein processing and secretion.
In EoE patients, endoscopic and biopsy studies searching for intrusion to plant foods and pollen, and specific-guided diet and immunotherapy after plant structures detection in biopsies, might be effective. Emerging immunologic treatment with spe- cific molecular targets are likely to change EoE management paradigms in the next years. A multidisciplinary approach (aller- gist, gastroenterologist, plant biologist, immunologist, dietitians and pathologist) is recommended, given the complexity of this disease
Data Availability Statement
All dataset necessary to interpret, verify and extend the research it is available to readers who require it, as well as biopsy images and microarrays reports.
Acknowledgements
We sincerely thank our patients for their courage and interest. They deserve our best efforts in the fight to overcome their dis- ease. They were very receptive to any positive health interven- tion on their behalf. The authors thank David Buss for his edito- rial assistance
Author Contributions
The authors Alicia Armentia, Javier Santos and Sara Martín have participated in the conception, design of the clinical study, analysis and interpretation of the data. Javier Santos made the endoscopies and biopsies. Blanca Martin carried out all labora- tory analyses and Sara Martin-Armentia the study of pediatric patients. Rafael Alvarez-Nogal and Delia Fernández made the pollen histological study and they verify the presence of callose (polysaccharide abundant in the polinic tubes during germina- tion) in the esophagus. Sara Gayoso carried out the scanning electron microscope study. All authors have contributed to the generation, analysis and interpretation of biopsy data with the remaining authors.
Declaration of Interest
We don’t have any financial conflicts of interest
References
- Furuta, G. T., Katzka, D.A. (2015). Eosinophilic Esophagi- tis. N Eng J Med. 373:1640-8.
- Wu, L., Oshima, T., Li, M., Tomita, T., Fukui, H., Watari, J., & Miwa, H. (2018). Filaggrin and tight junction proteins are crucial for IL-13-mediated esophageal barrier dysfunction. American Journal of Physiology-Gastrointestinal and Liver Physiology, 315(3), G341-G350.
- Ram, G., Lee, J., Ott, M., Brown-Whitehorn, T. F., Cian- feroni, A., Shuker, M., & Spergel, J. M. (2015). Seasonal exacerbation of esophageal eosinophilia in children with eosinophilic esophagitis and allergic rhinitis. Annals of Al- lergy, Asthma & Immunology, 115(3), 224-228.
- Hill, D. A., Grundmeier, R. W., Ramos, M., & Spergel, J.M. (2018). Eosinophilic esophagitis is a late manifestation of the allergic march. The Journal of Allergy and ClinicalImmunology: In Practice, 6(5), 1528-1533.
- Armentia, A., Martín-Armentia, S., Álvarez-Nogal, R., Ar- mentia, B. M., Gayoso, M. J., & Fernández-González, D(2019). Germination of pollen grains in the oesophagus of individuals with eosinophilic oesophagitis. Clinical & Ex- perimental Allergy, 49(4), 471-473.
- Schneider, R., Hanak, T., Persson, S., & Voigt, C. A. (2016). Cellulose and callose synthesis and organization in focus, what's new?. Current Opinion in Plant Biology, 34, 9-16.
- Letner, D., Farris, A., Khalili, H., & Garber, J. (2018). Pol- len-food allergy syndrome is a common allergic comorbid- ity in adults with eosinophilic esophagitis. Diseases of the Esophagus, 31(2), dox122.
- Armentia, A., Martín-Armentia, S., Martín-Armentia, B., Santos-Fernández, J., Álvarez, R., Madrigal, B., ... & Gay- oso, M. J. (2018). Is eosinophilic esophagitis an equivalent of pollen allergic asthma? Analysis of biopsies and therapy guided by component resolved diagnosis. Allergologia et Immunopathologia, 46(2), 181-189.
- Ram, G., Lee, J., Ott, M., Brown-Whitehorn, T. F., Cian- feroni, A., Shuker, M., & Spergel, J. M. (2015). Seasonal exacerbation of esophageal eosinophilia in children with eosinophilic esophagitis and allergic rhinitis. Annals of Al- lergy, Asthma & Immunology, 115(3), 224-228.
- Buhner, S., Schäuffele, S., Giesbertz, P., Demir, I. E., Zeller, F., Traidl-Hoffmann, C., & Gilles, S. (2023). Allergen-free extracts from birch, ragweed, and hazel pollen activate hu- man and guinea-pig submucous and spinal sensory neurons. Neurogastroenterology & Motility, 35(7), e14559.
- Gomez, G. A., & Torrijos, E. (2022). Eosinophilic esopha- gitis due to aeroallergens: a systematic review and update. Journal of Investigational Allergology and Clinical Immu- nology, 32(6), 438-450.
- Armentia, A., San, M. A., Martin-Armentia, B., Martín, S., Gonzalez Sagrado, M., San Miguel-Hernandez, A. (2021). Usefulness of Measurement of soluble Interleukin-2 recep- tor (sIL-2R) and Interleukin 6 to monitor efficacy of specif- ic Immunotherapy in eosinophilic esophagitis. International Journal of Medical Research& Health Sciences.10:173-179.
- Barni, S., Arasi, S., Mastrorilli, C., Pecoraro, L., Giovanni- ni, M., Mori, F., & Novembre, E. (2021). Pediatric eosino- philic esophagitis: a review for the clinician. Italian Journal of Pediatrics, 47(1), 1-14.
- Gonsalves, N. P., & Aceves, S. S. (2020). Diagnosis and treatment of eosinophilic esophagitis. Journal of Allergy and Clinical Immunology, 145(1), 1-7.
- Greuter, T., Hirano, I., & Dellon, E. S. (2020). Emerging therapies for eosinophilic esophagitis. Journal of Allergy and Clinical Immunology, 145(1), 38-45.
- Lehman H, Lam W. (2019). Eosinophilic esophagitis. Pedi- atri Clin North Am. 955-965.
- Aceves, S. S., Alexander, J. A., Baron, T. H., Bredenoord,A. J., Day, L., Dellon, E. S., & Katzka, D. A. (2022). En- doscopic approach to eosinophilic esophagitis: American Society for Gastrointestinal Endoscopy Consensus Confer- ence. Gastrointestinal endoscopy, 96(4), 576-592.