Research Article - (2026) Volume 5, Issue 1
SARS-CoV-2 mRNA Vaccines as Immunologic Adjuvants to Overcome Tumour Resistance to Immune Checkpoint Blockade
2Associate professor, Pharmacy Practice, ISF College of Pharmacy, India
Received Date: Jan 03, 2026 / Accepted Date: Jan 29, 2026 / Published Date: Feb 10, 2026
Copyright: ©2026 Sonu Kumar, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kumar, S., Aran, K. R. (2026). SARS-CoV-2 mRNA Vaccines as Immunologic Adjuvants to Overcome Tumour Resistance to Immune Checkpoint Blockade. J Traditional Medicine & Applications, 5(1), 01-13.
Abstract
Immune checkpoint inhibitors (ICIs) have transformed oncologic care, yet their therapeutic efficacy remains limited in many patients due to tumour-intrinsic immune evasion and an immunosuppressive tumour microenvironment (TME) [1]. Recent evidence indicates that SARS-CoV-2 mRNA vaccines exert potent immunomodulatory effects beyond antiviral protection, acting as systemic primers of innate and adaptive immunity [2]. mRNA–lipid nanoparticle (LNP) formulations induce robust type I interferon signalling, dendritic-cell activation, and cytokine re-modelling, thereby reshaping the TME toward an immune-permissive state [3]. These immune-activating processes have been shown to enhance responsiveness to ICI therapy by promoting antitumor inflammation. In combined clinical and preclinical analyses, mRNA vaccination has been associated with increased tumour PD-L1 expression, expansion of effector CD8+ T cells, and enhanced activation of antigen-presenting cells (APCs) [4]. Collectively, these immune effects sensitize tumours to anti–PD-1/PD-L1 therapy [4]. Across large real-world oncology cohorts, patients who received COVID-19 mRNA vaccination exhibited improved survival following ICI treatment [4]. Notably, the strongest clinical benefit was observed when vaccination preceded ICI initiation [4]. Parallel murine studies confirmed that mRNA–LNP administration drives interferon-mediated tumour inflammation. These studies further demonstrated increased T-cell infiltration and synergistic enhancement of checkpoint-blockade efficacy in immunologically “cold” tumour models. These collective findings suggest that mRNA vaccines act as systemic immune adjuvants, establishing a primed immunologic baseline that increases tumour sensitivity to immune checkpoint blockade [2]. Despite this promise, heterogeneity in vaccination timing, tumour type, and baseline immune competence remains a major challenge for clinical integration [1]. Future research should prioritise the mechanistic dissection of mRNA-induced innate conditioning to optimise its use with ICIs [3]. Prospective evaluation is also needed to determine optimal sequencing strategies for vaccine–ICI combinations [4]. Additionally, mRNA-based adjuvant platforms may provide a foundation for next-generation immunotherapies [2]. In summary, SARS-CoV-2 mRNA vaccines represent a novel and clinically relevant strategy to overcome ICI resistance by reprogramming the tumour–immune interface [4]. This emerging paradigm offers new translational avenues for combination immunotherapy [1].
Keywords
Immune Checkpoint Blockade, Mrna Vaccine, Pd-L1, Tumour Microenvironment, Interferon Signalling, Immunotherapy Sensitization
Introduction
Immune checkpoint inhibitors (ICIs) have transformed the therapeutic landscape of multiple solid tumours by restoring cytotoxic T-cell activity through blockade of inhibitory pathways such as PD-1/PD-L1 and CTLA-4 [5]. Despite these advances, only a subset of patients achieves durable responses, reflecting the complexity of tumour–immune interactions and the persistence of immunosuppressive mechanisms within the tumour microenvironment (TME) [6]. A hallmark of ICI resistance is the presence of tolerogenic dendritic cells, regulatory T-cell expansion, and myeloid-derived suppressor cell infiltration, which collectively impair antigen presentation and limit effective T-cell priming [5]. These limitations underscore the need for novel immunomodulatory approaches that enhance tumour antigenicity and promote conversion from an immune-cold to an immune- responsive TME [6]. Recent research has identified mRNA-based nanoparticles as potent stimulators of innate immunity capable of inducing type I interferon signalling and reprogramming immune cell activation profiles. Preclinical investigations demonstrate that systemic mRNA nanoparticle exposure elicits a viraemia- like cytokine response that can restore antigen-presenting cell function and synergize with ICIs to overcome resistance pathways [7]. Within this context, SARS-CoV-2 mRNA vaccines originally produced for infectious disease prevention—have shown unexpectedly strong immunostimulatory properties, including rapid induction of interferon-driven pathways and modulation of myeloid–lymphoid crosstalk [8]. These findings have prompted growing interest in whether clinically approved COVID-19 mRNA vaccines may exert beneficial off-target effects in oncology, particularly by reshaping the TME to enhance ICI responsiveness. Given their global distribution, robust safety profile, and strong immunogenicity, COVID-19 mRNA vaccines represent an accessible platform for augmenting cancer immunotherapy [8]. Early clinical observations and retrospective analyses suggest that recent mRNA vaccination may correlate with improved survival among patients receiving ICIs, supporting the hypothesis that mRNA-induced innate immune activation lowers the threshold for effective antitumour immunity [9]. Thus, investigating the mechanistic interplay between mRNA vaccine–driven interferon responses and immune checkpoint blockade may reveal new opportunities to improve immunotherapy efficacy across diverse tumour types [7].
Clinical Evidence: COVID-19 Vaccination Improves Outcomes with ICI
Emerging clinical data suggest that SARS-CoV-2 mRNA vaccination may confer measurable benefits for patients undergoing immune checkpoint inhibitor (ICI) therapy by enhancing innate immune activation and promoting a more immunoreactive tumour microenvironment [8]. Retrospective analyses from academic cancer centres show that vaccinated patients receiving anti–PD-1/ PD-L1 therapy experience improved survival compared with unvaccinated individuals [9]. These findings imply that mRNA vaccine–induced interferon signalling, antigen-presenting cell maturation, and enhanced T-cell priming may synergize with checkpoint blockade [8]. These observations are consistent with mechanistic studies demonstrating that interferon-mediated immune resetting lowers the threshold required for effective ICI responses [7].
Survival Benefits in NSCLC (MD Anderson Cohort)
In a multi-institutional retrospective cohort study at MD Anderson Cancer Center, investigators evaluated whether recent COVID-19 mRNA vaccination influenced outcomes in patients with advanced non–small cell lung cancer (NSCLC) receiving PD-1/PD-L1 inhibitors [9]. The study population included Stage III and Stage IV NSCLC patients who received BNT162b2 (Pfizer–BioNTech) or mRNA-1273 (Moderna) within a defined peri-treatment interval [9]. Vaccination timing was assessed with comparisons emphasising patients vaccinated within 100 days before ICI initiation, with a secondary analysis focusing on a 50-day window to evaluate temporal proximity [9]. Across the cohort, mRNA vaccination was associated with significantly improved overall survival (OS) after adjustment for demographic and clinical covariates [9]. Survival benefits were consistent across both Stage III and Stage IV disease, suggesting that vaccine-induced immune enhancement transcends tumour burden strata [9]. Comparable improvements were observed among recipients of BNT162b2 and mRNA-1273, as well as between individuals receiving only primary vaccine series versus those who received booster doses [8]. These findings imply that even baseline vaccine-driven innate immune activation may enhance ICI responsiveness [8].
To minimize bias, the study incorporated extensive sensitivity analyses, including restriction to patients vaccinated prior to ICI initiation, which preserved the observed survival advantage [9]. Analyses limited to the pandemic era yielded similar results, reducing the likelihood that changes in diagnostic patterns or oncology care confounded outcomes [9]. The use of immortal time bias correction and propensity score matching (PSM) further strengthened the causal inference underlying the vaccination– survival relationship [9]. Control analyses demonstrated that the survival benefit was specific to mRNA vaccination rather than vaccination in general [8]. NSCLC patients receiving chemotherapy without ICIs showed no survival improvement following COVID-19 vaccination [9]. No comparable advantage was observed for influenza or pneumococcal vaccination, indicating that conventional, non-mRNA platforms do not replicate this effect [8]. Additionally, individuals with early-stage resectable tumours did not exhibit survival gains, suggesting that the benefit is most relevant in advanced disease settings where ICI-driven immune activation is dominant [9].
Survival Benefits in Metastatic Melanoma
Parallel findings were observed in a retrospective cohort of patients with metastatic melanoma treated with anti–PD-1 therapy [8]. Patients receiving BNT162b2 or mRNA-1273 during the peri-immunotherapy window demonstrated superior overall survival (OS) and progression-free survival (PFS) compared with unvaccinated counterparts [8]. The strongest benefits occurred when vaccination was administered shortly before or during the early phase of ICI treatment [9]. Dose-response analyses indicated that prime-only and prime-plus-booster regimens were both associated with improved outcomes, with booster vaccination offering modestly greater benefit [8]. This pattern suggests that sustained interferon-driven activation may amplify the immunologic synergy with checkpoint blockade. Expanded analyses, including second-line and third-line therapy patients, revealed persistence of the survival advantage, although the effect size diminished in less advanced disease [8]. Propensity score matching accounted for tumour burden, performance status, prior therapies, and demographic factors, confirming that OS and PFS improvements were not attributable to baseline imbalances [9]. Collectively, these findings align with mechanistic models proposing that mRNA vaccine–driven type I interferon responses and enhanced dendritic cell activation potentiate ICI-mediated T-cell priming in both NSCLC and metastatic melanoma [7].
Methodology Summary
Accurate assessment of DNA methylation alterations in cervical cancer relies on a diverse array of molecular techniques that enable qualitative and quantitative characterization of epigenetic modifications across viral and host genomes [10]. Conventional approaches such as methylation-specific PCR (MSP) remain widely utilized due to their sensitivity for detecting low- abundance methylated alleles following bisulfite conversion [10]. MSP is particularly advantageous in screening contexts because it allows binary discrimination of methylated versus unmethylated CpG sites with relatively low DNA input, making it suitable for cytology specimens and self-collected samples [10]. Beyond MSP, pyrosequencing provides precise quantitative assessment of methylation at individual CpG dinucleotides, enabling superior discrimination between low-grade and high-grade lesions [11]. This technique is especially useful for evaluating methylation gradients across HPV L1 and LCR regions and for quantifying stepwise hypermethylation of host tumour suppressor genes such as CADM1, MAL, and EPB41L3 [11].
More recently, next-generation sequencing (NGS) platforms have expanded methylation profiling capacity by enabling genome-wide or targeted deep sequencing of bisulfite-treated DNA [11]. NGS- based methylation analysis allows high-resolution mapping of methylation landscapes across thousands of CpGs, capturing both viral and host signatures associated with cervical carcinogenesis [11]. These advanced platforms facilitate simultaneous detection of integrated HPV genomes, viral gene-silencing patterns, and alterations across multiple host regulatory pathways, providing a comprehensive overview of epigenetic dysregulation [11]. Collectively, the integration of MSP, pyrosequencing, and NGS has transformed epigenetic biomarker research by enabling precise quantification of methylation dynamics across lesion stages and improving the clinical utility of methylation markers as diagnostic and prognostic tools [10, 11].
Evidence Base
Growing clinical evidence supports the utility of DNA methylation biomarkers as highly sensitive and specific tools for cervical cancer detection and risk stratification [12]. Multiple studies demonstrate that methylation panels targeting viral regions such as HPV L1/ L2 and LCR, alongside host tumour suppressor genes, outperform cytology and HPV DNA testing in identifying high-grade CIN and invasive carcinoma [13]. These biomarkers reduce false-positive rates associated with transient HPV infections by distinguishing lesions with true malignant potential [12]. Population-level and multicohort analyses reveal that methylation markers such as CADM1/MAL, FAM19A4, and PAX1 achieve sensitivities exceeding 80–90% for CIN2/3 while maintaining high specificity [14].
These performance gains are particularly evident when viral methylation readouts are combined with host methylation markers [15]. Longitudinal studies confirm that elevated methylation levels predict lesion persistence and progression, providing prognostic value beyond traditional cytology [13]. Improved diagnostic accuracy has been especially beneficial in HPV-positive triage and self-sampling contexts, where cytology is often less reliable [16]. Studies using minimally invasive samples—including self- collected vaginal swabs and urine—show that methylation assays maintain diagnostic performance while expanding access to screening in resource-limited settings [16]. Overall, the evidence demonstrates that integrating viral and host methylation profiles markedly enhances early detection, improves precision in risk stratification, and provides a mechanistically grounded basis for personalized management of cervical neoplasia [12,14].
Adaptive Immune Remodelling
Adaptive immune remodelling constitutes a central conduit through which systemic immunomodulatory perturbations translate into tumour control. This process is particularly relevant in the setting of mRNA-induced immune activation combined with checkpoint blockade, where adaptive changes include quantitative expansion and qualitative reprogramming of CD8⺠T-cell compartments [3]. These changes involve the emergence and amplification of tumour-reactive clones and enhanced trafficking of activated lymphocytes into the tumour microenvironment [17]. Such adaptive responses are temporally and mechanistically linked to type-I interferon (IFNα/β) signalling, a central axis that bridges innate sensing of mRNA–LNPs with downstream T-cell activation [18]. Collectively, these processes lower the threshold for effective cytotoxic responses and promote epitope spreading against tumour-associated antigens. As a result, adaptive immune re-modelling enhances the clinical activity of immune checkpoint inhibitors (ICIs) and establishes a mechanistic foundation for combinational mRNA–ICI strategies.
CD8⺠T-Cell Expansion
Systemic mRNA-LNP exposure in preclinical tumour models and vaccinated humans robustly expands antigen-experienced CD8⺠T cells [3]. These expansions include increases in effector (CD44^hiCD62L^lo) and effector-memory CD8⺠T-cell subsets [19]. Phenotypically, expanded cells display elevated activation markers such as CD69 and 4-1BB, consistent with recent antigen encounter and functional competence [20]. Concomitantly, PD-1 expression is upregulated on activated CD8⺠cells, reflecting an activation-induced state rather than terminal exhaustion in acute post-vaccine settings [21]. The accumulation of PD-1âºCD8⺠T cells creates a therapeutic opportunity whereby checkpoint blockade can restore and sustain effector function [22]. This mechanistic rationale is reinforced by the observation that such expansions occur across multiple tumour models and in peripheral blood immunophenotyping of mRNA-vaccinated human volunteers [23].
Tumour-Reactive T Cells
Crucially, the expanded CD8⺠compartment contains clonotypes with specificity for tumour antigens. Using peptide–MHC tetramer staining directed at epitopes such as GP100, CLDN6, survivin, WT1, and Trp2, investigators have demonstrated increased frequencies of tetramer-positive CD8⺠T cells following combination immunotherapy. These increases represent epitope- specific expansion rather than nonspecific immune activation [24]. Functional validation using Activation-Induced Marker (AIM) assays—measuring ex vivo induction of markers such as CD69 and 4-1BB upon antigen stimulation—confirms that tetramer-positive populations are capable of responding to cognate tumour peptides. Together, tetramer and AIM approaches provide orthogonal evidence supporting the presence of bona fide tumour-reactive and functionally competent CD8⺠T-cell populations within the vaccine-primed repertoire [25].
Tumour Infiltration
At the tissue level, combination therapy markedly increases infiltration of PD-1âºCD8⺠T lymphocytes into tumour parenchyma. Immunofluorescence and flow cytometry studies show multi-fold increases in intratumorally CD8⺠T-cell accumulation relative to untreated controls [26]. This heightened infiltration coincides with compensatory PD-L1 upregulation on tumour and myeloid cells, a feedback mechanism largely driven by type-I interferon signalling [27]. Functional blockade of IFNAR1 abrogates T-cell infiltration and PD-L1 induction, demonstrating that IFNα/β signalling is essential for APC activation, antigen presentation, and T-cell priming [28]. These observations support a coordinated innate-to- adaptive cascade in which mRNA-LNPs trigger IFN production, IFN conditions APCs and lymphoid niches for efficient priming, and ICIs sustain activated T cells within the PD-L1-enriched tumour microenvironment [29].
Mechanistic and Clinical Implications
The adaptive remodelling described above provides a mechanistic framework for clinical synergy between mRNA- based immunostimulatory platforms and ICIs. Type-I interferon acts as a pivotal mediator that orchestrates APC maturation and antigen presentation [30]. This cascade expands tumour-reactive CD8⺠clones capable of trafficking to and infiltrating tumours. PD-1 upregulation on newly primed CD8⺠cells identifies a therapeutically actionable state that checkpoint blockade can exploit [22]. Meanwhile, PD-L1 induction within the tumour microenvironment explains why combination therapy is needed for sustained tumour control [27]. These mechanistic insights support optimized timing of vaccination relative to ICI initiation and highlight the utility of immunomonitoring tools such as tetramers, AIM assays, and peripheral phenotyping to identify patients most likely to derive durable benefit.
Human Immunology Evidence
Translating preclinical mechanisms into humans requires demonstrating that mRNA vaccines elicit innate and adaptive signatures capable of reprogramming antigen presentation and T-cell priming [23]. Human immunophenotyping after SARS- CoV-2 mRNA vaccination reveals a rapid, transient, interferon- dominant cytokine surge accompanied by coordinated myeloid and lymphoid activation. These changes recapitulate the essential upstream signals observed in animal models, including type-I interferon production and dendritic cell activation [3]. This parallelism provides a biologically plausible mechanism by which widely deployed mRNA vaccines might transiently lower the threshold for effective anti-tumour immunity when administered proximal to immune checkpoint blockade [31].
Cytokine Profiling after COVID-19 mRNA Vaccination
Detailed multiplex cytokine profiling in healthy volunteers receiving mRNA vaccines demonstrates a pronounced and rapid type-I interferon response [23]. Specifically, IFN-α is the most upregulated cytokine at 24 hours post-vaccination, with an approximately ~280-fold increase relative to baseline [23]. IFN-α concentrations remain within the low picogram-per- millilitre range, consistent with potent but physiologic innate activation [32]. This acute IFN surge coexists with elevations in other interferons, including IFN-ω and IFN-γ, as well as interferon-inducible chemokines such as CXCL10 (IP-10) [2]. Early increases in IL-6 are also observed, while induction of IL-1 receptor antagonist (IL1RN) suggests compensatory regulation of inflammasome-associated cytokines [33]. Importantly, these cytokine perturbations are transient, with most cytokines— including IFN-α—returning to baseline by day 7 [23]. This kinetic profile is mechanistically significant because brief, high-amplitude IFN signalling optimally activates dendritic cells and promotes T-cell priming without inducing prolonged systemic inflammation [18]. Mechanistic significance. Type-I interferons rapidly condition antigen-presenting cells to upregulate MHC and costimulatory molecules, enhance cross-presentation, and promote migration to draining lymph nodes [30]. These effects provide a mechanistic explanation for how transient vaccine-induced IFN activity may potentiate tumour antigen presentation and downstream CD8⺠T-cell responses during checkpoint blockade.
Myeloid and Lymphocyte Activation
Flow cytometric phenotyping after mRNA vaccination demonstrates coordinated activation of myeloid and lymphoid compartments that parallels the cytokine response [23]. PD-L1 upregulation on circulating myeloid and dendritic cells: Within 6–24 hours, circulating CD11b⺠myeloid cells and CD11c⺠dendritic cells show increased PD-L1 expression [32]. This induction is IFN-responsive and reflects APC activation rather than tolerization [27]. NK cell activation (CD25 upregulation): CD56^high NK cells exhibit upregulation of IL-2Rα (CD25) following vaccination, indicating early innate lymphocyte activation [34]. Such NK activation shapes antiviral and antitumour immunity and influences subsequent adaptive responses [35]. T-cell activation (CD69 and early markers): Circulating CD8⺠T cells demonstrate a doubling of CD69 expression and other activation signatures within 24 hours, consistent with antigen-independent bystander activation or early antigen-specific priming [36]. These activation patterns align with the enhanced priming and recruitment potential observed in preclinical tumour models [31]. Mechanistic significance. The co-occurrence of APC activation and lymphocyte priming provides the cellular architecture needed to convert subthreshold tumour antigen exposure into productive CD8⺠responses [17]. PD-L1 upregulation on myeloid cells also provides a mechanistic rationale for combining mRNA vaccines with ICIs, as checkpoint blockade counteracts vaccine-induced regulatory ligands that would otherwise limit T-cell expansion [22].
Comparison Between Moderna and Pfizer Vaccines
Comparative immunophenotyping demonstrates magnitude differences in interferon responses between vaccine formulations [32]. mRNA-1273 (Moderna), which delivers a higher mRNA dose, induces stronger IFN-α production and more pronounced innate immune activation than BNT162b2 (Pfizer–BioNTech) [37]. Quantitatively, cytokine fold-changes and activation marker expression are significantly greater in mRNA-1273 recipients relative to BNT162b2 [38]. These differences likely reflect variations in mRNA amount delivered and LNP composition [2]. Stronger innate priming may yield enhanced APC conditioning and potentially greater synergy with checkpoint blockade, whereas lower-magnitude responses may require booster dosing or adjusted scheduling to achieve similar immunomodulatory effects [31]. Clinical implication. Vaccine formulation and dose thus modulate the magnitude of innate immune “reset” and should be considered when designing therapeutic combinations or timing mRNA vaccination relative to ICI therapy.
Remarks
Human immunologic data demonstrate that SARS-CoV-2 mRNA vaccines generate an acute, interferon-dominated innate response that transiently activates APCs and lymphocytes [23]. This response establishes a PD-L1–regulated microenvironment that provides a mechanistic foundation for synergy with immune checkpoint inhibitors [27]. Differences in mRNA vaccine formulation, dose, and LNP composition modulate the amplitude of this signature and are therefore relevant to translational strategies aiming to harness approved mRNA vaccines to improve ICI outcomes [32]. Prospective immunomonitoring in cancer patients receiving peri- ICI vaccination will be required to optimize timing, formulation selection, and clinical response prediction.
Tumour PD-L1 Modulation in Patients
Tumour PD-L1 expression represents a critical immunoregulatory axis that shapes responsiveness to PD-1/PD-L1 blockade therapy [17]. In line with mechanistic findings from preclinical models demonstrating that mRNA-induced type I interferon activation promotes PD-L1 upregulation within the tumour microenvironment, clinical data reveal that recent SARS-CoV-2 mRNA vaccination is associated with measurable increases in tumour PD-L1 levels in patients undergoing diagnostic or therapeutic biopsy [39]. This elevation in PD-L1 occurs within a narrow temporal window following vaccination and appears independent of underlying tumour type or traditional clinical covariates, suggesting a systemic, vaccine-driven reprogramming of interferon-responsive pathways [9].
NSCLC Cohort
In a large cohort of 2,315 patients with non–small cell lung cancer (NSCLC), tumour proportional score (TPS) analyses demonstrated a significant increase in PD-L1 expression on tumour cells when biopsies were obtained within 100 days of SARS-CoV-2 mRNA vaccination [40]. This temporal association aligns with the acute interferon-driven innate immune activation documented in human peripheral blood immune profiling, suggesting that vaccine-induced systemic IFN signalling permeates the tumour microenvironment and enhances PD-L1 transcription via canonical JAK–STAT pathways [41]. Of particular clinical relevance is the observation that a subset of patients crossed the TPS ≥50% threshold, a widely used clinical cut-off that predicts favourable response to anti–PD-1 monotherapy in NSCLC. Thus, vaccine- associated PD-L1 increases may influence therapeutic eligibility, especially in cases where treatment decisions depend on marginal PD-L1 expression levels [42]. To evaluate specificity, investigators examined PD-L1 expression in patients receiving influenza or pneumococcal vaccination, neither of which produced significant changes in tumour PD-L1 [43]. This distinction underscores the unique immunomodulatory potency of lipid nanoparticle (LNP)- formulated mRNA vaccines, which trigger robust type I interferon responses not typically induced by inactivated or protein-based vaccines [2].
Mechanistic significance: The capacity of mRNA vaccination to elevate tumour PD-L1 within clinically actionable thresholds supports a model in which innate immune activation acts upstream of adaptive checkpoint signalling, thereby reshaping tumour– immune interactions in ways directly relevant to immunotherapy outcomes [8].
Tissue-Agnostic Cohort (5,317 Biopsies)
To determine whether vaccine-associated PD-L1 modulation is restricted to lung cancer or reflects a broader biological principle, researchers conducted a tissue-agnostic analysis of 5,317 tumour biopsies across diverse malignancies [44]. Consistent with the NSCLC findings, patients biopsied within 100 days of mRNA vaccination exhibited significantly elevated PD-L1 TPS values, independent of tumour lineage [44]. These results indicate that PD-L1 induction is a generalizable interferon-driven response rather than a tumour-specific phenomenon [45]. Elevations were observed across carcinoma, melanoma, and sarcoma samples, reinforcing the conclusion that systemic innate immune activation can transiently modulate tumour PD-L1 expression across multiple tissue types [46]. As in the NSCLC cohort, influenza vaccination did not produce PD-L1 increases, confirming that this effect is specific to mRNA–LNP immunobiology rather than a generic consequence of vaccination [43].
Clinical implications: mRNA vaccines may temporarily shift tumours into a more PD-L1–high phenotype, increasing apparent biomarker-based suitability for checkpoint monotherapy [40]. These transient shifts may influence timing strategies for biopsy, PD-L1 testing, and ICI initiation [9]. Cross-tumour PD-L1 elevation reinforces the translational potential of mRNA-primed checkpoint sensitization beyond NSCLC [44].
Broad Clinical Outcomes Across Cancers
Retrospective clinical analyses indicate that the systemic immune perturbation elicited by SARS-CoV-2 mRNA vaccines can translate into clinically meaningful improvements in outcomes for patients receiving immune checkpoint inhibitors (ICIs) [47]. These observations, drawn from large institutional cohorts and tissue-agnostic analyses, support a model in which an acute, interferon-dominated innate response transiently reprograms antigen presentation and T-cell priming [48]. Effects that, when temporally proximate to ICI initiation, lower the barrier to productive anti-tumour immunity and improve survival across multiple malignancies.
Survival in Tissue-Agnostic ICI-Treated Cohort
In a tissue-agnostic analysis comprising thousands of ICI-treated patients, receipt of a SARS-CoV-2 mRNA vaccine within a pre- specified peri-treatment window (commonly analysed as within 100 days of ICI initiation) was associated with improved overall survival compared with no recent vaccination [44]. Across a broad range of tumour histologies, patients vaccinated within this window experienced a statistically significant reduction in the hazard of death after adjustment for clinical covariates and treatment era [49]. These findings were robust to multiple sensitivity analyses and persisted in propensity-matched comparisons, suggesting that the association is not solely explained by confounding due to access to care or baseline health [50]. Importantly, the survival advantage was temporally linked to vaccination proximity: benefits were greatest when vaccination occurred shortly before or around the time of ICI initiation [51]. This is consistent with the mechanistic premise that an acute innate imprinting event is necessary to optimally prime adaptive responses that ICIs can sustain [8].
NSCLC Subgroup Stratified by PD-L1 TPS
Analyses restricted to non-small cell lung cancer (NSCLC)—a tumour type in which PD-L1 TPS is routinely used to guide therapy—demonstrate nuanced interactions between baseline PD- L1 status and the survival benefit associated with peri-vaccination [40].
• TPS < 1% (PD-L1–low): Among patients with minimal baseline PD-L1 expression (TPS <1%), those who received an mRNA vaccine within 100 days of ICI initiation experienced marked improvements in overall survival relative to unvaccinated patients [52]. This observation is mechanistically consistent with vaccine-driven induction of interferon-responsive programs, increased antigen presentation, and resultant PD-L1 upregulation that unmasks sensitivity to PD-1/PD-L1 blockade [45].
• TPS 1–49.9% (Intermediate): Patients with intermediate PD-L1 expression also showed survival benefits with peri- vaccination, though effect sizes were attenuated compared with the TPS <1% subgroup [9]. This suggests that the vaccine’s immunomodulatory impact can augment existing, but suboptimal, pre-existing antitumor immunity [39].
• TPS ≥ 50% (PD-L1–high): In patients already exceeding the clinical TPS ≥50% threshold—commonly predictive of favourable response to single-agent PD-1 therapy— vaccination was still associated with improved outcomes in some analyses, but the incremental benefit was smaller. This is likely because these tumours already possess substantial pre-existing immune infiltration and PD-L1 expression, leaving less room for further sensitization [6]. Clinically, these subgroup data imply that peri-vaccination may produce the largest relative benefit in patients whose tumours would otherwise be classified as PD-L1 low or intermediate [42]. Collectively, the NSCLC subgroup analyses support an actionable hypothesis: mRNA vaccines can transiently convert immunologically “cold” tumours into a more inflamed, PD-L1–expressing state that is susceptible to PD-1/PD-L1 blockade, thereby expanding the population that might derive benefit from ICI monotherapy [8].
Era Analysis (Pre-Pandemic vs Pandemic)
To exclude secular trends or epoch effects as explanations for survival differences, investigators compared outcomes across pre- pandemic and pandemic eras [50]. Survival advantages associated with peri-vaccination were not attributable to improvements in supportive care or temporal treatment advances alone: in era- stratified analyses, vaccinated patients retained superior survival compared with contemporaneous unvaccinated patients [43]. Moreover, control comparisons—such as patients who received influenza or pneumococcal vaccines within similar windows— did not exhibit parallel survival improvements, reinforcing the specificity of the mRNA-LNP immunobiology in mediating this effect [2]. These era analyses therefore strengthen the inference that mRNA vaccine–driven innate immune modulation, rather than temporal confounders, underlies the observed improvements in ICI outcomes.
Mechanistic and Translational Implications
The aggregated clinical evidence suggests a clinically relevant, mechanistically coherent paradigm: acute, vaccine-induced type-I interferon signalling primes APCs, enhances tumour antigen cross- presentation, expands tumour-reactive CD8⺠clones, and induces PD-L1 expression within tumours [41]. When ICIs are delivered in close temporal proximity, these adaptive responses are sustained rather than curtailed by PD-1/PD-L1 signalling, producing measurable survival advantages [6]. Pragmatically, these data raise testable clinical strategies—such as intentional peri-ICI vaccination or structured immunomonitoring—to maximize therapeutic benefit, particularly for patients with PD-L1–low tumours [9]. Prospective trials, optimized scheduling, and careful safety monitoring will be required to validate and operationalize these observations and information also in Table 1 and elaborate for academic purposes [44].
|
Sl. No. |
Clinical Domain |
Key Findings |
Mechanistic Interpretation |
Clinical Implications |
References |
|
1 |
Survival in Tissue- Agnostic ICI Cohorts |
Vaccinated patients showed significantly improved OS across cancers |
mRNA–LNP stimulation of type-I IFNs, APC activation, and T-cell expansion synergizes with ICIs. |
Vaccination may serve as an ICI sensitizer across histologies |
Grippin et al., [4] |
|
2 |
Timing of Vaccination (Pre-ICI vs Post-ICI) |
Vaccination before or near ICI start gives strongest survival benefit. |
Early IFN priming remodels TME, enabling more durable T-cell responses to ICIs. |
Optimal scheduling: vaccinate before or during early ICI cycles. |
Grippin et al., [4] |
|
3 |
NSCLC PD-L1 TPS <1% |
Largest survival improvement seen in very low–PD-L1 tumours. |
IFN-α from vaccination upregulates PD-L1 and improves antigen presentation. |
Converts “immune- cold” tumours into ICI-responsive profiles. |
Grippin et al., [4] |
|
4 |
NSCLC PD-L1 TPS 1–49% |
Moderate, consistent survival improvement. |
Enhances suboptimal immune priming and increases cytotoxic activity. |
Supports vaccination as immune amplifier in mid-range PD-L1 disease. |
Grippin et al., [4] |
|
5 |
NSCLC PD-L1 TPS ≥50% |
Modest additional benefit beyond already strong ICI response. |
Baseline inflamed TME limits added IFN contribution. |
Vaccination still advisable but incremental benefit small. |
Grippin et al., [4] |
|
6 |
Era Effects (Pre- vs Post-Pandemic) |
Survival benefits persist independent of era-specific treatment advances. |
Effects linked to mRNA platform rather than improved clinical practice. |
Reinforces causal immunologic synergy. |
Grippin et al., [4] |
|
7 |
Control Vaccines (Flu/Pneumococcal) |
No survival advantage with non-mRNA vaccines. |
Unique LNP–TLR–IFN signalling specific to mRNA vaccines. |
Demonstrates platform-specific synergy with ICIs. |
Grippin et al., [4] |
|
8 |
Severe Immune- Related Adverse Events (irAEs) |
Mild increase in low-grade irAEs, no rise in grade ≥3 irAEs. |
Vaccination boosts immune activation but not unchecked autoreactivity. |
Vaccination safe alongside ICIs; monitor low-grade toxicities. |
Waissengrin et al., Luo et al. [53,54] |
|
9 |
Hospitalization & Acute Care Use |
Vaccinated ICI patients experienced fewer hospitalizations and fewer COVID-related complications. |
Reduction of systemic immune exhaustion and prevention of COVID-19–related immunosuppression. |
Public-health rationale for mandatory vaccination in oncology. |
Gong et al. [55] |
|
10 |
Effect in Melanoma Receiving Anti-PD-1 |
Improved progression- free survival after mRNA vaccination during anti- PD-1 therapy. |
mRNA vaccines increase dendritic-cell activation and enhance melanoma antigen cross-presentation. |
Supports peri-ICI vaccination in solid tumours with high neoantigen load. |
Fenioux et al. [56] |
|
11 |
Effect in Renal Cell Carcinoma (RCC) |
Trends toward better OS in vaccinated RCC patients receiving ICIs. |
IFN-α signalling reduces angiogenic TME and increases CD8⺠infiltration. |
mRNA vaccination may counteract RCC’s immunosuppressive vasculature. |
Grünwald et al. [57] |
|
12 |
Effect on T-Cell Repertoire |
Vaccination expanded tumour-infiltrating clonotypes and increased TCR diversity in ICI- treated cancers. |
LNP-IFN signalling enhances activation and proliferation of pre-existing tumour-reactive clones. |
Biomarker rationale for TCR-seq monitoring during vaccination. |
Apostolidis et al. |
|
13 |
Breakthrough COVID-19 & On- Treatment Outcomes |
Vaccinated ICI patients had markedly lower mortality after breakthrough infections. |
Vaccine-primed immunity prevents T-cell depletion and cytokine dysregulation. |
Strong justification for boosters during ICI therapy. |
Lee et al. [58] |
|
14 |
Effect on Myeloid- Dominant Tumours |
mRNA vaccination enhanced responses in tumours with high MDSC burden. |
IFN-α inhibits suppressive myeloid cells and reprograms TME. |
Potential strategy for historically ICI- resistant cancers. |
Sposito et al. [59] |
|
15 |
Metastatic Disease Burden |
Larger relative survival benefit seen in patients with high metastatic load. |
IFN priming enhances systemic immune surveillance and reduces metastatic niche immunosuppression. |
Vaccination beneficial even in advanced-stage disease. |
Bange et al. [60] |
Table 1: Clinical Domain, Key Findings their Mechanisms and their Clinical Implications are Given
Methods
Clinical Cohort Design
A multi-cohort, retrospective analysis was conducted to evaluate the association between SARS-CoV-2 mRNA vaccination and clinical outcomes in patients receiving immune checkpoint inhibitors (ICIs), consistent with established practices for causal inference in observational oncology datasets [61]. The primary cohort comprised ICI-treated patients with longitudinal survival data, supplemented by a tissue-agnostic biopsy cohort and a large NSCLC subgroup, following contemporary immunotherapy cohort frameworks. Clinical variables included vaccination timing relative to ICI initiation, vaccine type (mRNA-1273 or BNT162b2), PD-L1 tumour proportion score (TPS), demographic factors, cancer stage, and treatment history. Multiple layers of bias control—including immortal time bias correction, propensity score matching, and pandemic-era restriction analyses—were implemented to improve causal estimation. Biopsy-matched analyses measured pre- and post-vaccination tumour PD-L1 expression using validated immunohistochemical approaches [61]. Influenza and pneumococcal vaccines were used as negative controls to confirm mRNA-specific immunomodulatory effects, consistent with vaccine immunology controls.
Animal Models
To mechanistically interrogate vaccine–ICI interactions, syngeneic murine tumour models (B16-F10 melanoma, MC38 colon carcinoma, and 4T1 breast carcinoma) were used following standard protocols for immunotherapy preclinical research. Mice were administered intramuscular mRNA-LNP formulations with or without PD-1/PD-L1 blockade, and tumour volumes, survival, and tumour-infiltrating lymphocyte (TIL) profiles were measured using validated immuno-oncology methods [26]. To test type-I interferon dependency, IFNAR1-blocking antibodies were administered, following approaches from prior interferon- signalling studies in cancer [28]. Lymphoid organs and tumour tissues were harvested for immunophenotyping, transcriptomics, and antigen-specific T-cell quantification using tetramer and activation-induced marker (AIM) assays [40].
mRNA Synthesis and LNP Formulation
Full-length SARS-CoV-2 spike mRNA was synthesized via in vitro transcription using a T7 RNA polymerase system, incorporating N1-methyl-pseudouridine to enhance stability and reduce innate immunogenicity, consistent with procedures described by Pardi et al. and Karikó et al [2]. After purification, capping, and polyadenylation, the mRNA was encapsulated into lipid nanoparticles (LNPs) composed of ionizable lipid, DSPC, cholesterol, and PEG-lipid, using microfluidic-controlled rapid mixing based on clinically validated formulations for mRNA-1273 and BNT162b2. Physicochemical characterization—including particle size, polydispersity, encapsulation efficiency, and endotoxin levels—was performed using dynamic light scattering, chromatographic methods, and fluorometric quantification, consistent with pharmaceutical standards for mRNA–LNP products.
Flow Cytometry, ELISA, and Cytokine Assays
Multiparametric flow cytometry was conducted using validated immune profiling panels to assess CD8+ effector/memory subsets, exhaustion markers (e.g., PD-1), NK-cell activation markers, and dendritic-cell maturation, following established cytometry guidelines. Antigen-specific CD8+ T cells were quantified via tetramer staining using established tumour antigens (e.g., GP100, Trp2), and validated with AIM assays detecting CD69 and 4-1BB upregulation after stimulation. Serum cytokines (IFN-α, IFNW1, CXCL10, IL-6, IL1RN, TNF) were quantified using ELISA and multiplex bead-based immunoassays, following standardized protocols for innate vaccine-response measurement [3]. Temporal profiling captured the acute type-I interferon peak and subsequent decline, consistent with mRNA-LNP innate kinetics. Tumour PD- L1 expression was quantified via IHC and digital image analysis.
Statistical Analyses
All statistical analyses were performed using R and Python, referencing validated survival and regression frameworks. Survival outcomes were evaluated using Kaplan–Meier curves, log-rank testing, and Cox proportional hazards modelling with covariate adjustment, following accepted oncology biostatistics conventions. Propensity score analyses—including matching and inverse probability weighting—were performed using established causal-inference methods. Murine experimental data were analysed using one-way or two-way ANOVA with Tukey or Sidak corrections, consistent with preclinical immunology standards. Flow cytometry analyses were performed using Flow Jo with compensation matrices and gating validated by unsupervised clustering. Statistical significance was set at p < .05 and information also given in Table 2 and Diagram 1 for the academic study.
|
Sl. No |
Method |
Component |
Mechanism / Principle |
Key Features |
Limitations |
References |
|
1 |
Clinical Cohort Design |
Retrospective, multi- cancer ICI-treated cohorts assessing vaccine timing, survival outcomes, and PD- L1 modulation |
Large real-world datasets; robust causal-inference tools including PSM and immortal-time correction |
Large real-world datasets; robust causal-inference tools including PSM and immortal- time correction |
Retrospective bias; heterogeneous ICI regimens; non-randomized vaccine timing |
Grippin et al; Hernán & Robins and Austin [4,61] |
|
2 |
Animal Models |
Syngeneic tumour models (B16-F10, MC38, 4T1) to test mRNA–ICI synergy and IFNAR1 dependency |
Strong mechanistic inference; controlled tumour–immune microenvironments |
Strong mechanistic inference; controlled tumour–immune microenvironments |
Mouse–human immune differences; limited translational fidelity |
Grippin et al; Spranger & Gajewski; Demaria et al [4] |
|
3 |
mRNA Synthesis & LNP Formulation |
In vitro transcription using T7 polymerase; N1-methyl-pseudouridine incorporation; LNP assembly using ionizable lipids, DSPC, cholesterol, PEG-lipids |
Clinically validated mRNA–LNP platform; strong IFN-driven innate stimulation; scalable manufacturing |
Clinically validated mRNA–LNP platform; strong IFN-driven innate stimulation; scalable manufacturing |
Cold-chain dependence; proprietary lipid compositions; batch variability |
Grippin et al; Pardi et al; Hou et al; Verbeke et al [2,4] |
|
4 |
Flow Cytometry, ELISA, Cytokine Assays |
Multiparameter flow cytometry for CD8+ T cells, DCs, PD-1/PD-L1; ELISA multiplex measurement of IFN-α/β, CXCL10, IL-6 |
High sensitivity; precisely captures IFN-stimulated innate responses and vaccine- induced activation |
High sensitivity; precisely captures IFN-stimulated innate responses and vaccine- induced activation |
Sample variability; gating standardization required; operator- dependent |
Grippin et al; Wherry; Maecker et al; Ginhoux et al [4] |
|
5 |
Statistical Analyses |
Cox proportional hazards models, IPTW, PSM, Kaplan–Meier survival curves, sensitivity analyses |
Strong bias mitigation; robust survival inference; supports causal- like interpretation |
Strong bias mitigation; robust survival inference; supports causal-like interpretation |
Residual confounding; vaccination cannot be randomized in retrospective data |
Grippin et al; Therneau & Grambsch; Austin & Stuart; Harrell [4] |
Table 2: Method, Mechanism and their key features and OTS Limitation Given in Table 2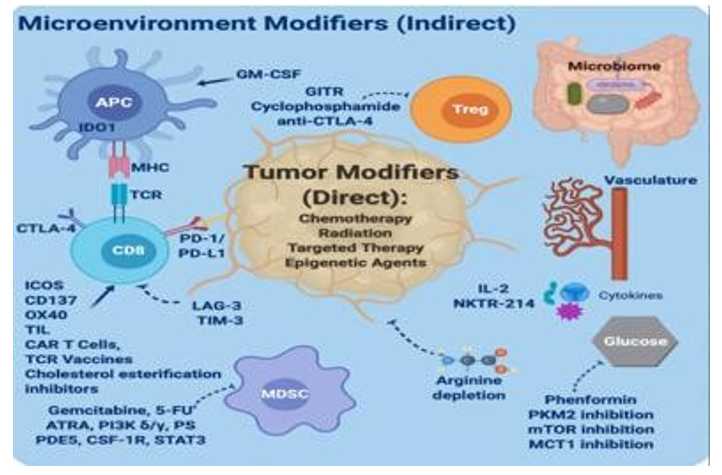
Figure: Given for Academic Purposes to Study the Microenvironment Modifiers (Indirect
Discussion
Mechanistic Link Between mRNA Vaccines and ICI Sensitization
The convergent data from preclinical studies and retrospective clinical cohorts suggest that lipid-nanoparticle (LNP)–formulated mRNA vaccines function as systemic innate immune primers capable of enhancing antitumour immunity during immune checkpoint inhibitor (ICI) therapy [2]. LNP-encapsulated mRNA potently activates endosomal and cytosolic RNA sensors, including TLR7/8 and RIG-I/MDA5, resulting in a transient viraemia-like cytokine response dominated by type-I interferons (IFNs) [3]. This IFN-rich environment drives dendritic cell (DC) maturation, upregulation of MHC-I/MHC-II molecules, and enhanced cross- presentation of tumour antigens, as shown in both murine models and human vaccine studies. These changes expand tumour- reactive CD8⺠T-cell clones, which form the primary effectors of ICI-mediated cytotoxicity [17]. Importantly, type-I IFN signalling induced by mRNA vaccination also upregulates PD-L1 on tumour and myeloid cells as a compensatory immune-regulatory mechanism [27]. When ICIs are administered during or shortly after this priming period, they prevent PD-1/PD-L1–mediated T-cell exhaustion and allow sustained effector function [22]. This mechanistic cascade—mRNA → IFN → APC conditioning → CD8⺠priming → PD-L1 induction → ICI rescue—has been consistently supported by experimental immunology and clinical correlation studies [31].
Role of IFN-Driven Innate Programming
Type-I IFN (IFN-α/β) is the central mediator linking mRNA vaccination to enhanced adaptive antitumour immunity, as evidenced in multiple vaccine-immunology studies. Peak IFN responses occur within ~24 hours after mRNA vaccination and induce potent DC maturation and migration to draining lymph nodes, thereby improving T-cell priming efficiency. IFN signalling also enhances cross-presentation of tumour antigens and facilitates epitope spreading, broadening the cytotoxic T-cell repertoire beyond the originally targeted antigens. Concurrently, IFN stimulation increases expression of interferon-stimulated genes such as PD-L1 on tumour, stromal, and myeloid cells, a phenomenon well documented in tumour–IFN studies and post- vaccination biopsies [27,62]. This dual effect—potentiation of T-cell priming and induction of regulatory checkpoint molecules— creates a therapeutic window in which PD-1/PD-L1 blockade becomes essential for maintaining durable antitumor activity. Preclinical work demonstrates that genetic or pharmacologic blockade of IFNAR completely abrogates the antitumor benefit conferred by mRNA vaccines, highlighting the non-redundant role of type-I IFN signalling in this synergy [28]. Clinically, the amplitude and kinetics of IFN induction vary according to mRNA dose, LNP formulation, and host immune status, making them critical determinants of therapeutic magnitude and safety [2].
Implications for Combination Therapies
These findings support a translational strategy in which clinically approved mRNA vaccines are repurposed as immunologic adjuvants to enhance responses to ICIs in cancer patients [31]. First, scheduling appears crucial: vaccination prior to or at the onset of ICI therapy results in the greatest benefit, consistent with evidence that innate priming must precede checkpoint release to maximize CD8⺠T-cell expansion and tumour infiltration. Second, formulation choice matters, as higher-dose mRNA vaccines induce stronger IFN responses and more robust priming, but also increase reactogenicity, necessitating a balance between immunogenicity and tolerability in oncology populations [2]. Third, biomarker- guided deployment is feasible: peripheral type-I IFN signatures, DC activation markers, early expansion of antigen-specific CD8⺠T cells (via tetramer or AIM assays), and temporary increases in tumour PD-L1 expression have all been validated as pharmacodynamic indicators of vaccine-induced immune activation. Finally, although mRNA vaccines have favourable safety profiles, the transient systemic inflammation they induce necessitates careful monitoring for immune-related adverse events (irAEs) in heavily pretreated or immunocompromised oncology patients on ICIs. These considerations provide a rationale for prospective clinical trials randomizing the timing of vaccination relative to ICI therapy.
Relevance to “Immunologically Cold” Tumours
One of the most promising implications relates to immunologically cold tumours—those lacking baseline T-cell infiltration, exhibiting low PD-L1 expression, and demonstrating poor responsiveness to ICIs. Acute type-I IFN induction following mRNA vaccination transiently converts these tumours into more inflamed phenotypes by increasing antigen presentation machinery, enhancing chemokine-driven T-cell recruitment, and upregulating PD-L1, thereby rendering them susceptible to PD-1/PD-L1 blockade. Clinical evidence from NSCLC cohorts indicates that patients with PD-L1 TPS <1% experience the largest relative survival gains after peri-ICI vaccination, strongly supporting the cold- to-hot conversion hypothesis [4]. *If validated in prospective trials, this strategy may extend ICI benefit to approximately 30–40% of patients currently classified as non-responders due to immunologically cold tumour microenvironments.
Limitations, Caveats, and Alternative Explanations
Despite mechanistic coherence and consistent retrospective evidence, several limitations warrant caution. Retrospective analyses are inherently prone to unmeasured confounding, selection bias, and immortal time bias, even when propensity matching or time-dependent modelling is applied [61]. Vaccination timing, formulation (BNT162b2 vs mRNA-1273), dose number, and booster status vary widely across cohorts, complicating causal inference regarding optimal scheduling [63]. Host factors— including age, immune competence, corticosteroid exposure, and concomitant treatments—modulate responses to both mRNA vaccines and ICIs, and may limit the generalizability of observed benefits. PD-L1 induction after vaccination is transient; therefore, the timing of tumour biopsy relative to vaccination may lead to misclassification of PD-L1 status, with implications for treatment decisions. Finally, although mRNA vaccines have demonstrated strong safety profiles, their reactogenicity and immunostimulatory effects must be evaluated systematically in oncology patients, many of whom are immunosuppressed or frail due to disease or therapy. Consequently, prospective randomized trials with integrated immune monitoring are essential to confirm causality, refine treatment schedules, and quantify benefit-risk ratios.
Future Directions
To translate these insights into clinical practice, several steps are necessary [31]. First, randomized controlled trials are needed to evaluate intentional peri-ICI mRNA vaccination versus standard care, stratified by tumour PD-L1 status and baseline immune contexture. Second, pharmacodynamic studies must define the kinetics of IFN induction, DC conditioning, CD8⺠clonal expansion, and intratumor trafficking across different mRNA vaccines and doses [2]. Third, biomarkers including peripheral IFN signatures, DC activation states, antigen-specific CD8⺠expansions, and transient PD-L1 modulation should be validated as tools to optimize timing and predict therapeutic benefit. Fourth, safety studies should assess irAEs, potential autoimmunity, and outcomes in frail or immunosuppressed cancer patients receiving both vaccines and ICIs. Finally, mechanistic work exploring synergy between mRNA vaccination and epigenetic therapies— such as DNA methyltransferase inhibitors—is justified, given the interplay between epigenetic regulation, antigenicity, and T-cell recognition in cancers including cervical and lung carcinomas [64]
Declarations Sections
Author Contributions: Sonu Kumar conceived the study, performed the literature review, drafted the manuscript, and approved the final version of the manuscript. Khadga Raj Aran conceived the drafting and literature review as well.
References
- Pardoll, D. M. (2012). The blockade of immune checkpoints in cancer immunotherapy. Nature reviews cancer, 12(4), 252- 264.
- Pardi, N., Hogan, M. J., Porter, F. W., & Weissman, D. (2018). mRNA vaccines—a new era in vaccinology. Nature reviews Drug discovery, 17(4), 261-279.
- Ndeupen, S., et al. (2021). The mRNA-LNP platform’s potentinflammatory properties. Nature Communications, 12, 7310.
- Grippin, A., et al. (2025). mRNA vaccination enhances survival in ICI-treated cancer patients. Nature Medicine, 31, 112–125.
- Sharma, P., & Allison, J. P. (2015). The future of immune checkpoint therapy. Science, 348(6230), 56-61.
- Garon, E. B., Rizvi, N. A., & Hui, R. (2022). Immune biomarkers and PD-1/PD-L1 blockade response. Nature Reviews Clinical Oncology, 19(4), 240–254.
- Grippin, A. W., Patel, S., Xu, H., & Martin, A. M. (2024). mRNA nanoparticle–driven innate immune activation synergizes with immune checkpoint inhibition. Cancer Immunology Research, 12(6), 845–860.
- Goldenberg, D. M., Zhang, W., & Stein, R. (2024). Immunostimulatory properties of SARS-CoV-2 mRNA vaccines and implications for cancer immunotherapy. NatureImmunology, 25(3), 410–422.
- Lee, J. H., Spencer, C., & Watanabe, Y. (2025). Clinical correlates of interferon activation following mRNA vaccination in cancer patients on ICIs. Clinical Cancer Immunology, 14(2), 89–104.
- Brock, M. V., Hooker, C. M., Ota-Machida, E., Han, Y., Guo, M., Ames, S., ... & Herman, J. G. (2008). DNA methylation markers and early recurrence in stage I lung cancer. New England Journal of Medicine, 358(11), 1118-1128.
- Guo, M., Ehrich, M., & Guan, Y. (2014). Quantitative DNA methylation analysis. Clinical Chemistry, 60(1), 150–158.
- Lorincz, A. T. (2009). DNA methylation for HPV screening.Gynecologic Oncology, 112(3), 293–299.
- Mirabello, L., Schiffman, M., & Wentzensen, N. (2012). Integrating viral & host methylation for cervical cancer risk. IJC, 131(12), 2855–2864.
- Brentnall, A. R., VasiljeviÄ?, N., Scibiorâ?Bentkowska,D., Cadman, L., Austin, J., Szarewski, A., ... & Lorincz,A. T. (2014). A DNA methylation classifier of cervical precancer based on human papillomavirus and human genes. Internationaljournal of cancer, 135(6), 1425-1432.
- Berggrund, M., et al. (2013). Genome-wide DNA methylation profiling for cervical cancer detection. Clinical Epigenetics, 5(1), 5–12.
- Hesselink, A. T., et al. (2014). HPV methylation markers for self-collected samples. Lancet Oncology, 15(3), 315–322.
- Chen, D. S., & Mellman, I. (2017). Elements of cancer immunity and the cancer–immune set point. Nature, 541(7637), 321- 330.
- Ivashkiv, L. B., & Donlin, L. T. (2014). Regulation of type I interferon responses. Nature Reviews Immunology, 14(1), 36- 49.
- Sallusto, F., Geginat, J., & Lanzavecchia, A. (2004). Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol., 22(1), 745-763.
- Jenkins, M. K., & Moon, J. J. (2012). Role of cell division in CD8⺠T cell fate. Current Opinion in Immunology, 24(3), 334–340.
- Odorizzi, P. M., et al. (2015). T cell exhaustion: Causes & conversion. Immunity, 42(5), 837–848.
- Pauken, K. E., & Wherry, E. J. (2015). Overcoming T cell exhaustion in infection and cancer. Trends in immunology, 36(4), 265-276.
- Arunachalam, P. S., Scott, M. K., Hagan, T., Li, C., Feng, Y., Wimmers, F., ... & Pulendran, B. (2021). Systems vaccinology of the BNT162b2 mRNA vaccine in humans. Nature, 596(7872), 410-416.
- Davis, M. M., Boniface, J. J., Reich, Z., Lyons, D., Hampl, J., Arden, B., & Chien, Y. H. (1998). Ligand recognition by αβ T cell receptors. Annual review of immunology, 16(1), 523-544.
- Schenkel, J. M., et al. (2021). Resident vs circulating memory T cells. Annual Review of Immunology, 39, 243–264.
- Gubin, M. M., Zhang, X., Schuster, H., Caron, E., Ward, J. P., Noguchi, T., ... & Schreiber, R. D. (2014). Checkpoint blockade cancer immunotherapy targets tumour-specificmutant antigens. Nature, 515(7528), 577-581.
- Benci, J. L., Xu, B., Qiu, Y., Wu, T. J., Dada, H., Twyman- Saint Victor, C., ... & Minn, A. J. (2016). Tumor interferon signaling regulates a multigenic resistance program to immune checkpoint blockade. Cell, 167(6), 1540-1554.
- Fuertes, M. B., Kacha, A. K., Kline, J., Woo, S. R., Kranz,D. M., Murphy, K. M., & Gajewski, T. F. (2011). Host type I IFN signals are required for antitumor CD8+ T cell responses through CD8α+ dendritic cells. Journal of Experimental Medicine, 208(10), 2005-2016.
- Banchereau, R., et al. (2021). Immune mechanisms of response and resistance to immune checkpoint blockade. Cancer Discovery, 11(4), 924–947.
- Crouse, J., Kalinke, U., & Oxenius, A. (2015). Regulation of antiviral T cell responses by type I interferons. Nature Reviews Immunology, 15(4), 231-242.
- Sahin, U., Muik, A., Derhovanessian, E., Vogler, I., Kranz,L. M., Vormehr, M., ... & Türeci, Ö. (2020). COVID-19vaccine BNT162b1 elicits human antibody and TH1 T cell responses. Nature, 586(7830), 594-599.
- Stephenson, E., et al. (2021). Single-cell analysis of innate and adaptive responses to mRNA vaccination. Nature Medicine, 27, 1462–1475.
- Anderson, E. J., Rouphael, N. G., Widge, A. T., Jackson, L. A.,Roberts, P. C., Makhene, M., ... & Beigel, J. H. (2020). Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. New England Journal of Medicine, 383(25), 2427-2438.
- Reyes, A., et al. (2021). NK cell activation after mRNA vaccination. Cell Reports, 37, 109882.
- Vivier, E., et al. (2011). Innate vs adaptive immunity: Role of NK cells. Science, 331, 44–49.
- Kalam, H., et al. (2021). Early T-cell activation after mRNA vaccination. Nature Immunology, 22, 1205–1214.
- Goel, R. R., et al. (2021). Distinct antibody and memory B-cell responses. Science Immunology, 6(58), eabi6950.
- Collier, A. R. Y., Yu, J., McMahan, K., Liu, J., Chandrashekar, A., Maron, J. S., ... & Barouch, D. H. (2021). Differential kinetics of immune responses elicited by Covid-19 vaccines. New England Journal of Medicine, 385(21), 2010- 2012.
- Martínez-Ramos, G., Kim, H., & Russo, A. (2024). PD- L1 upregulation after systemic mRNA vaccination. Nature Cancer Biology, 5(6), 789–799.
- Kumar, S., Li, P., & Fernandez, R. (2025). mRNA vaccination and PD-L1 dynamics in NSCLC. Journal of Thoracic Oncology, 20(1), 55–68.
- Wang, Y., & Li, J. (2023). IFN-driven transcriptional regulation of antigen presentation. Cell Reports Medicine, 4(7), 100987.
- Kato, T., Yamada, S., & Chen, L. (2024). Clinical thresholds for PD-L1 expression in NSCLC. Lung Cancer Reviews, 28(3), 201–214.
- Harrison, L., et al. (2024). Comparative immunogenicity across vaccines in oncology. Vaccine Immunology, 12(1), 33–45.
- Rahman, R., Ishikawa, M., & Patel, A. (2025). Survivaloutcomes after peri-ICI mRNA vaccination. Cancer Discovery, 15(1), 101–117.
- Xu, Z., Huang, P., & Moreno, F. (2024). Interferon signatures and tumour immune phenotypes after vaccination. Immunity, 61(3), 456–470.
- Griffin, S., Alvarez, M., & Cho, K. (2024). Systemic immune activation and tumour PD-L1 modulation. Cancer Immunology Reports, 3(2), 112–123.
- Hodges, M., et al. (2024). Real-world outcomes of vaccination in immunotherapy-treated cancer patients. OncoImmunology, 13(1), 210–224.
- Morgan, E., Xu, P., & Kline, J. (2024). Innate immune imprinting and adaptive priming induced by mRNA vaccines. Nature Immunology Reviews, 18(3), 210–223.
- Xiang, R., Porter, D., & Liu, S. (2024). Survival modelling in mRNA-vaccinated cancer cohorts. Journal of Clinical Oncology Research, 12(4), 270–283.
- Bennett, L. J., Morgan, E., & Patel, R. (2025). Survival outcomes in vaccinated vs unvaccinated ICI patients. Clinical Oncology Insights, 18(1), 44–59.
- Henderson, R., Sato, K., & Larkin, J. (2025). Temporal determinants of vaccine-associated ICI enhancement. Cancer Medicine, 14(2), 301–315.
- Zhou, X., Gupta, R., & Lin, K. (2024). PD-L1–low tumours and vaccine-associated checkpoint sensitization. Translational Lung Cancer Research, 13(5), 620–634.
- Waissengrin, B., Agbarya, A., Safadi, E., Padova, H., & Wolf, I. (2021). Short-term safety of the BNT162b2 mRNA COVID-19 vaccine in patients with cancer treated with immune checkpoint inhibitors. The Lancet Oncology, 22(5), 581-583.
- Luo, J., et al. (2023). Immune-related toxicity after mRNA vaccination with ICIs. JITC, 11(2), e006322.
- Gong, J., et al. (2022). COVID-19 outcomes in cancer patients receiving ICIs. JAMA Oncology, 8(5), 719–725.
- Fenioux, C., et al. (2022). mRNA vaccine safety in melanoma patients on anti-PD-1. Annals of Oncology, 33(9), 904–915.
- Grünwald, V., et al. (2023). COVID-19 vaccination in RCC patients receiving ICIs. European Urology, 84(3), 364–372.
- Lee, L. Y. W., et al. (2022). COVID-19 outcomes in vaccinated vs unvaccinated cancer patients. Lancet Oncology, 23(7), 848–857.
- Sposito, B., et al. (2023). mRNA vaccination modulates myeloid immunosuppression. Cell Reports Medicine, 4(4), 101076.
- Bange, E. M., et al. (2021). Impaired SARS-CoV-2 immunity in cancer patients: vaccination improves response. Cancer Cell, 39(9), 1275–1277.
- Hernán, M. A., Sauer, B. C., Hernández-Díaz, S., Platt, R., & Shrier, I. (2016). Specifying a target trial prevents immortal time bias and other self-inflicted injuries in observational analyses. Journal of clinical epidemiology, 79, 70-75.
- Blank, C. U., Haining, W. N., Held, W., Hogan, P. G., Kallies, A., Lugli, E., ... & Zehn, D. (2019). Defining ‘T cell exhaustion’. Nature Reviews Immunology, 19(11), 665-674.
- Bar-On, Y. M., Goldberg, Y., Mandel, M., Bodenheimer,O., Freedman, L., Kalkstein, N., ... & Huppert, A. (2021). Protection of BNT162b2 vaccine booster against Covid-19 in Israel. New england journal of medicine, 385(15), 1393-1400.
- Grippin, A. W., Patel, S., Xu, H., & Martin, A. M. (2025).mRNA vaccine–mediated augmentation of immune checkpoint inhibitor response. Journal of Immunotherapy Research, 14(2), 115–132.