Research Article - (2026) Volume 10, Issue 1
Safety of Allogeneic Mesenchymal Stromal Cell Therapy Across Human Clinical Indications: A Systematic Review of Landmark Clinical Trials and Published Safety Evidence
Received Date: Feb 06, 2026 / Accepted Date: Mar 10, 2026 / Published Date: Mar 18, 2026
Copyright: ©2026 Kirk Sanford, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Sanford, K., Porras, F., Martinez, F., Ramos, H., Zamitiz, J., et al. (2026). Safety of Allogeneic Mesenchymal Stromal Cell Therapy Across Human Clinical Indications: A Systematic Review of Landmark Clinical Trials and Published Safety Evidence. Stem Cell Res Int, 10(1), 01-09.
Abstract
Background: Allogeneic mesenchymal stromal cells (MSCs) have emerged as an off-the-shelf regenerative medicine platform across cardiovascular, pulmonary, musculoskeletal, autoimmune, gastrointestinal, and renal indications. Although individual clinical trials frequently describe favorable tolerability, safety reporting remains heterogeneous across studies, and the adverse event profile of allogeneic MSC administration has not been consistently synthesized with emphasis on treatment-related serious toxicity. Landmark MSC safety meta-analyses have generally concluded that MSC therapy appears safe, with transient fever representing the most reproducible treatment-associated event.
Objective: To evaluate the safety profile of allogeneic mesenchymal stromal cell therapy across human clinical indications, with emphasis on treatment-related serious adverse events, immediate administration-related toxicity, and clinically important long-term safety signals.
Methods: A systematic review framework was developed in accordance with PRISMA 2020 guidance. The review focused on human clinical studies of allogeneic MSC administration, prioritizing landmark randomized controlled trials, controlled prospective studies, and high-yield safety meta-analyses. Primary outcome emphasis was placed on treatment-related serious adverse events. Secondary outcomes included any adverse event, infusion or injection reactions, transient fever, infection, death, immunologic reactions, malignancy, and ectopic tissue formation. Evidence was synthesized qualitatively across landmark trials and interpreted in the context of prior MSC safety meta-analyses.
Results: Across landmark allogeneic MSC trials in acute myocardial infarction, ischemic cardiomyopathy, dilated cardiomyopathy, acute respiratory distress syndrome, COVID-19 ARDS, Crohn’s disease, systemic lupus erythematosus, diabetic kidney disease, and knee osteoarthritis, no reproducible signal of frequent treatment-related serious adverse toxicity was identified. Acute myocardial infarction studies concluded intravenous allogeneic MSCs were safe. The POSEIDON randomized trial supported comparative safety of allogeneic transendocardial MSC delivery in ischemic cardiomyopathy. The START phase 2a ARDS trial established safety in critically ill patients. The phase 3 ADMIRE-CD trial supported safety of locally administered allogeneic adipose-derived MSCs in complex perianal fistulizing Crohn’s disease. Across previously published pooled safety evidence, MSC therapy was not associated with increased serious adverse events, death, infection, arrhythmia, vascular complications, or malignancy, while transient fever was the main event consistently increased.
Conclusion: Current human clinical evidence supports a favorable safety profile for allogeneic mesenchymal stromal cell therapy across multiple therapeutic indications and routes of administration. Treatment-related serious adverse events appear uncommon, and no consistent signal of infection excess, thromboembolic toxicity, organ toxicity, or mortality attributable to MSC administration has emerged from landmark trials or prior pooled safety analyses. Importantly, the clinical MSC literature has not demonstrated evidence of tumorigenicity. No confirmed cases of tumors derived from administered MSCs have been reported in human clinical trials, and pooled safety analyses have not identified increased malignancy risk among MSC-treated patients. These findings support the continued clinical development of allogeneic MSC therapy as a scalable and generally well-tolerated regenerative medicine platform.
Keywords
Allogeneic Mesenchymal Stromal Cells, Allogeneic Stem Cells, Adverse Events, Safety, Serious Adverse Events, Regenerative Medicine, Systematic Review
Abbreviations
MSC: Mesenchymal stromal cell
ARDS: Acute respiratory distress syndrome
SAE: Serious adverse event
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
GRADE: Grading of Recommendations Assessment, Development and Evaluation
Introduction
Mesenchymal stromal cells have become one of the most intensively studied cell therapy platforms in regenerative and translational medicine. Their therapeutic appeal derives from broad immunomodulatory, anti-inflammatory, trophic, pro- angiogenic, and tissue-supportive properties that have prompted clinical investigation across inflammatory, degenerative, ischemic, and immune-mediated diseases. In contrast to autologous approaches, allogeneic MSC products can be expanded, quality controlled, cryopreserved, and administered as off-the-shelf therapies, making them especially attractive for scalable clinical translation. The expansion of allogeneic MSC clinical research has been accompanied by persistent questions regarding safety. Because these cells are donor-derived and administered in widely varying clinical contexts, theoretical concerns have included acute infusion toxicity, hemodynamic instability, alloimmune reactivity, opportunistic infection, thromboembolic complications, ectopic tissue formation, and malignant transformation. These questions are particularly relevant in critically ill populations, in intravascular delivery strategies, and in trials involving repeat dosing.
Safety evaluation in MSC therapy is complicated by several factors. First, many treated populations have substantial baseline morbidity, making it difficult to distinguish treatment-related events from disease-related complications. Second, adverse event reporting is inconsistent across trials, with some studies emphasizing any adverse event, others emphasizing serious adverse events, and others reporting only investigator-attributed treatment-related events. Third, long-term surveillance remains limited in many disease areas, reducing certainty regarding rare delayed outcomes such as malignancy or persistent alloimmunization. Despite these limitations, the highest-level published safety syntheses have been reassuring. The SafeCell meta-analysis by Lalu and colleagues, which reviewed 36 studies and 1,012 participants, found MSC therapy appeared safe overall and identified only transient fever as significantly associated with treatment. An updated 2019 systematic review of intravascular MSC administration similarly concluded that cell therapy continued to appear safe, while the 2021 Stem Cell Research & Therapy meta-analysis across 62 randomized trials found no increase in serious adverse events, death, infection, arrhythmia, vascular events, or thrombotic events, again identifying transient fever as the principal treatment- associated signal.
Landmark allogeneic MSC clinical trials further reinforce this favorable safety profile. The acute myocardial infarction Prochymal study concluded intravenous allogeneic hMSCs were safe. The POSEIDON randomized trial supported the comparative safety of allogeneic transendocardial MSC injection in ischemic cardiomyopathy. The START phase 2a trial established safety in moderate-to-severe ARDS. The phase 3 ADMIRE-CD trial demonstrated safety of local allogeneic adipose-derived MSC therapy in complex perianal Crohn’s fistulas. An additional issue of major relevance in the broader stem cell field is tumorigenicity. This concern must be interpreted in the context of cell type. Pluripotent stem cell platforms, including embryonic stem cells and induced pluripotent stem cells, possess unrestricted differentiation capacity and therefore carry recognized teratoma-forming risk. In contrast, mesenchymal stromal cells are multipotent adult stromal cells with limited differentiation potential, and the available human clinical literature has not demonstrated a reproducible tumorigenic signal. No confirmed cases of tumors derived from administered MSCs have been reported in human trials, and prior pooled safety analyses have not identified increased malignancy risk among MSC-treated patients. This distinction is essential when evaluating the safety profile of MSC-based regenerative therapies. The objective of the present study was to systematically evaluate the safety of allogeneic mesenchymal stromal cell therapy across human clinical indications, with emphasis on treatment-related serious adverse events and clinically relevant long-term safety signals.
Methods
• Reporting Standards
This systematic review was prepared in accordance with the Preferred Reporting Items for Systematic Reviews and Meta- Analyses (PRISMA) 2020 guidelines.
• Clinical Trial Number and Registration
Clinical trial number: not applicable. This study is a systematic review of previously published human clinical studies and did not involve direct enrollment of participants. Review registration: not registered.
• Study Design
This study was designed as a systematic review of human clinical trials evaluating the safety of allogeneic mesenchymal stromal cell therapy.
• Information Sources and Search Strategy
A structured literature search framework was developed for PubMed/MEDLINE, Embase, Web of Science, Scopus, Cochrane CENTRAL, and ClinicalTrials.gov. Search concepts combined terms related to allogeneic MSCs, human clinical trials, and safety outcomes.
♦ Representative Search Strategy
(allogeneic OR donor-derived) AND (mesenchymal stromal cell OR mesenchymal stem cell OR MSC OR bone marrow-derived MSC OR umbilical cord MSC OR Wharton’s jelly MSC OR placental MSC OR adipose-derived MSC) AND (safety OR adverse event OR serious adverse event OR treatment-related adverse event OR infusion reaction OR infection OR fever OR death OR malignancy) Reference lists of relevant landmark trials and safety meta-analyses were also reviewed to identify additional eligible studies.
♦ Eligibility Criteria
Studies were eligible if they met the following criteria:
• human clinical study
• administration of live allogeneic mesenchymal stromal or mesenchymal stem cells
• prospective interventional design, randomized controlled trial, controlled clinical study, or prospective clinical cohort
• any therapeutic indication
• reporting of safety outcomes, including adverse events, serious adverse events, treatment-related adverse events, death, infection, infusion or injection reactions, or other complications
♦ Exclusion Criteria
Studies were excluded if they involved:
• preclinical or animal research
• autologous-only cell therapies
• hematopoietic stem cell transplantation
• immune effector cell therapies
• extracellular vesicle or exosome-only interventions without live cell administration
• case reports
• reviews, editorials, or conference abstracts without extractable safety conclusions
♦ Outcomes
Primary Outcome
• treatment-related serious adverse events Secondary outcomes
• any adverse event
• serious adverse events overall
• infusion reaction
• injection-site reaction
• transient fever
• infection
• death
• immunologic or hypersensitivity reaction
• thromboembolic event
• malignancy
• ectopic tissue formation
• withdrawal due to adverse event
♦ Data Extraction
The following variables were prioritized:
• author and year
• indication
• study design
• MSC source
• route of administration
• comparator
• sample size
• follow-up duration
• key safety findings
• presence or absence of treatment-related serious adverse events
• notable acute administration-related events
• notable long-term safety findings Where trials did not provide complete tabular event data, narrative safety conclusions from the primary publication were retained for structured evidence synthesis.
♦ Risk of Bias
Risk of bias was assessed conceptually using standard randomized trial domains, including:
• randomization process
• deviations from intended interventions
• missing outcome data
• outcome measurement
• selective reporting
Because the present draft emphasizes landmark clinical trial synthesis and previously published safety meta-analytic evidence rather than de novo pooled extraction of all event counts, the risk- of-bias interpretation is reported narratively.
♦ Statistical Analysis
The present manuscript is structured as a safety-focused systematic review informed by previously published meta-analyses. Prior large meta-analyses are incorporated to support pooled interpretation of safety signals. The highest-value quantitative message from the literature is that MSC therapy has not been associated with increased serious adverse events, death, infection, arrhythmia, thrombotic events, or malignancy in prior pooled analyses, while transient fever appears increased. No de novo pooled effect-size meta-analysis was performed for the present manuscript; instead, prior published meta-analyses were used to contextualize the direction and consistency of safety findings across landmark allogeneic MSC trials.
Results
Study Selection
The identified evidence base for allogeneic MSC safety included landmark randomized and prospective trials across cardiovascular, pulmonary, musculoskeletal, autoimmune, gastrointestinal, and renal indications, together with three major MSC safety meta- analyses that informed pooled cross-indication interpretation. The core clinical anchor studies identified for this review were:
• Hare et al., acute myocardial infarction, intravenous allogeneic bone marrow-derived MSCs
• POSEIDON randomized trial, ischemic cardiomyopathy, transendocardial allogeneic vs autologous MSCs
• START phase 2a trial, moderate-to-severe ARDS, intravenous allogeneic bone marrow-derived MSCs
• ADMIRE-CD phase 3 trial, complex perianal Crohn’s disease, local allogeneic adipose-derived MSCs
• additional supportive allogeneic studies in dilated cardiomyopathy, lupus, diabetic kidney disease, COVID-19 ARDS, and knee osteoarthritis
Included Studies
The included human clinical evidence spans a broad range of therapeutic settings and administration routes:
Cardiovascular:
acute myocardial infarction, ischemic
cardiomyopathy,
dilated cardiomyopathy Pulmonary and critical care:
ARDS, COVID-19 ARDS
Musculoskeletal:
knee osteoarthritis
Autoimmune:
systemic lupus erythematosus
Gastrointestinal:
luminal Crohn’s disease, complex perianal fistulizing Crohn’s disease
Renal:
diabetic kidney disease
Routes of administration included intravenous infusion, transendocardial injection, intra-articular injection, and local fistula-directed injection.
Risk of Bias Summary
Overall risk of bias across the landmark trials was considered low to moderate. The strongest evidence came from randomized and placebo-controlled or active-controlled studies. The main limitations were modest sample sizes in some early-phase trials, incomplete harmonization of adverse event reporting, and limited long-term surveillance for rare delayed events.
Primary Outcome: Treatment-Related Serious Adverse Events
The central finding across landmark allogeneic MSC trials is the absence of a reproducible signal of frequent treatment-related serious adverse toxicity.
In the acute myocardial infarction Prochymal trial, Hare and colleagues concluded intravenous allogeneic hMSCs were safe in patients after acute MI and described the study as providing pivotal safety data for an allogeneic bone marrow-derived stem cell in this setting.
In the POSEIDON randomized trial of ischemic cardiomyopathy, allogeneic transendocardial MSC administration was found to be comparably safe relative to autologous MSC delivery, supporting the feasibility of donor-derived administration without an emergent major immunologic safety signal.]
In moderate-to-severe ARDS, the START phase 2a trial was explicitly designed to assess safety and established that intravenous allogeneic bone marrow-derived MSC delivery could be performed in critically ill patients without a major early toxicity signal.
In Crohn’s disease, the phase 3 ADMIRE-CD trial demonstrated safety of local allogeneic adipose-derived MSC administration for complex perianal fistulas, supporting tolerability in a high- inflammatory disease context.
These trial-level findings align with pooled safety evidence. The 2012 SafeCell analysis concluded MSC therapy appeared safe overall. The 2019 update of intravascular MSC administration concluded that therapy continued to appear safe. The 2021 meta- analysis found no increase in serious adverse events, death, infection, arrhythmia, vascular disorder, central nervous system events, thrombotic events, or malignancy, while transient fever was increased.
Secondary Outcome: Any Adverse Events
Any adverse event reporting was heterogeneous across disease areas. This outcome was heavily influenced by baseline disease burden and by the tendency of many studies to count all intercurrent clinical events irrespective of causality. As a result, all-cause adverse event frequency is less informative than treatment-related serious adverse events in evaluating platform safety. Across musculoskeletal studies, adverse events were usually limited to mild local pain, swelling, or transient discomfort after injection. Across cardiovascular and pulmonary studies, adverse events were more reflective of the underlying illness severity and the complexity of the patient population.
Secondary Outcome: Immediate Administration-Related Toxicity
The most reproducible short-term event associated with MSC therapy in pooled safety analyses is transient fever. SafeCell identified a significant association between MSC therapy and transient fever, but did not identify major increases in acute infusional toxicity or organ system complications. The 2021 broader meta-analysis also found transient fever increased while other major safety categories were not. This pattern is clinically important because it frames the most likely treatment-associated event as mild, transient, and manageable rather than severe or organ-threatening.
Secondary Outcome: Infection and Mortality
Across prior pooled analyses, MSC therapy has not been associated with increased infection or mortality. This is particularly notable because many MSC trials enroll patients with inflammatory, ischemic, or critical illness conditions in which baseline infection risk and mortality risk are already substantial. In ARDS and COVID-19 studies, deaths occurred in the context of severe underlying illness, but the available evidence does not support a consistent increase attributable to MSC administration.
Secondary Outcome: Long-Term Safety Signals
The most feared delayed risks in allogeneic MSC therapy are malignant transformation, ectopic tissue formation, and persistent alloimmune toxicity. Reassuringly, prior meta-analyses have not identified a malignancy signal, and landmark trials have not demonstrated a recurring pattern of clinically important ectopic tissue formation or platform-level alloimmune injury. However, long-term surveillance remains limited in much of the literature, and rare delayed events cannot be fully excluded.
With respect to tumorigenicity, the available clinical evidence remains reassuring. Across the landmark allogeneic MSC trials and the major pooled safety meta-analyses, no reproducible signal of malignant transformation attributable to MSC administration has emerged. No confirmed tumors derived from administered MSCs have been reported in human clinical studies included in the current evidence base, and prior pooled analyses have not identified increased malignancy risk among MSC-treated patients.
This is a particularly important finding because tumor formation remains one of the most frequently cited theoretical concerns surrounding stem cell-based therapies.
Discussion
This systematic review synthesizes the highest-yield safety evidence for allogeneic MSC therapy across human clinical indications. The central conclusion is favorable and consistent: current evidence supports a generally reassuring short-term safety profile for allogeneic MSC administration, with treatment-related serious adverse events appearing uncommon and no reproducible signal of major platform toxicity emerging across landmark trials or pooled safety meta-analyses.
A major strength of this evidence base is the consistency of the safety signal across highly different clinical environments. Allogeneic MSCs have been delivered intravenously to patients with acute myocardial infarction, ARDS, and COVID-19; transendocardially to patients with chronic cardiomyopathy; locally to Crohn’s fistulas; and intra-articularly in orthopedic disease. Despite this heterogeneity, the literature has not produced a recurring pattern of treatment-related serious adverse toxicity. That cross-indication consistency strengthens confidence in the fundamental tolerability of the allogeneic MSC platform.
A second important point is the distinction between any adverse event and treatment-related adverse events. Many MSC trials are conducted in very ill patients, and all-cause adverse events may primarily reflect the underlying disease rather than the therapy. For this reason, the present review deliberately emphasizes treatment- related serious adverse events as the primary outcome. This framing is not merely favorable; it is also methodologically appropriate. It better isolates the biologically relevant safety question.
The pooled meta-analytic literature further supports this interpretation. SafeCell concluded MSC therapy appeared safe and identified transient fever as the main treatment-associated event. The later intravascular update again concluded MSC therapy continued to appear safe, and the 2021 review found no increased risk of serious adverse events, death, infection, arrhythmia, vascular events, thrombosis, or malignancy. Taken together, these findings strongly support the view that the dominant safety signal of MSC therapy is mild transient fever rather than serious toxicity. From a translational standpoint, the absence of a major alloimmune signal is especially encouraging. Because allogeneic MSC products are donor-derived, one of the chief theoretical concerns has been immune intolerance or downstream immune-mediated injury. The landmark human literature has not identified this as a recurrent clinical problem. This is one of the most important reasons allogeneic MSCs remain attractive as off-the-shelf therapeutics.
Tumorigenicity is another issue of major translational importance. In the broader stem cell field, safety concerns regarding tumor formation are often generalized across biologically distinct platforms. This is not appropriate for mesenchymal stromal cells. Pluripotent stem cell platforms are characterized by unrestricted differentiation potential and recognized teratoma-forming risk. By contrast, MSCs are multipotent adult stromal cells with limited differentiation capacity, and the available human clinical literature has not demonstrated a reproducible tumorigenic signal. No confirmed cases of tumors derived from administered MSCs have been reported in clinical trials, and prior pooled safety analyses have not identified increased malignancy risk among MSC-treated patients. This distinction is critical and further strengthens the overall safety interpretation of allogeneic MSC therapy.
The literature does, however, remain limited in several ways. Adverse event reporting is not standardized across studies. Many trials are early phase and underpowered for rare-event detection. Follow-up is often too short to fully characterize delayed risks. In addition, some disease areas still rely on small pilot studies, making it difficult to generate precise risk estimates for unusual events. Thus, while the present evidence base supports a favorable safety profile, it should not be interpreted as definitive proof that all rare or delayed toxicities have been excluded. Overall, the clinical message remains positive. Across the highest-level evidence presently available, allogeneic MSC therapy appears to be well tolerated, serious treatment-related toxicity appears uncommon, and the main reproducible administration-related event is transient fever. These findings support continued clinical development of allogeneic MSC therapies across multiple disease areas.
Clinical Relevance
For clinicians, investigators, and regulatory stakeholders, the most relevant message is that allogeneic MSC therapy has shown a consistently favorable short-term safety profile across highly diverse patient populations. This supports its continued use as a platform for regenerative medicine investigation, especially where off-the-shelf delivery is clinically advantageous. The practical implication is not that allogeneic MSC therapy has been proven risk-free. Rather, it is that the accumulated human evidence does not show a recurring pattern of serious treatment-related harm, which is precisely the threshold needed to justify continued controlled clinical development.
Certainity of Evidence (Grade)
Certainty of evidence for overall safety is low to moderate.
Reasons for downgrading include:
• heterogeneity in adverse event reporting
• limited sample sizes in some disease areas
• incomplete long-term follow-up
• inconsistency in treatment-related attribution Reasons supporting confidence include:
• consistency of favorable safety direction across multiple indications
• randomized trial representation in key disease areas
• concordance between landmark trials and pooled safety meta- analyses
Conclusion
The available clinical evidence indicates that allogeneic mesenchymal stromal cell therapy has a favorable safety profile across multiple disease indications and delivery routes. Treatment- related serious adverse events appear uncommon, and large systematic safety analyses have not identified increased risk of infection, thromboembolic events, organ toxicity, or mortality attributable to MSC administration. Importantly, the clinical MSC literature has not demonstrated evidence of tumorigenicity. No confirmed cases of tumors derived from administered MSCs have been reported in human clinical trials, and pooled safety analyses have not identified increased malignancy risk among MSC-treated patients. These findings are especially important in the broader stem cell field, where tumor formation remains a major theoretical concern.
Unlike pluripotent stem cell platforms, which possess inherent teratoma-forming potential due to unrestricted differentiation capacity, mesenchymal stromal cells are multipotent adult stromal cells and have not demonstrated a reproducible tumorigenic signal in clinical use. Taken together, the current body of human evidence supports continued clinical investigation and development of allogeneic MSC therapy as a scalable, off-the-shelf regenerative medicine platform with a favorable short- and intermediate-term safety profile.
|
Study |
Indication |
Design |
MSC Source |
Route |
Key Safety Interpretation |
|
Hare et al., 2009 |
Acute myocardial infarction |
Randomized, double-blind, placebo-controlled, dose- escalation |
Bone marrow- derived MSCs |
Intravenous |
Trial concluded intravenous allogeneic hMSCs were safe after acute MI |
|
POSEIDON, 2012 |
Ischemic cardiomyopathy |
Randomized comparative trial |
Bone marrow- derived MSCs |
Transendocardial |
Comparative safety of allogeneic delivery supported; no major emergent immunologic signal |
|
START, 2019 |
Moderate-to-severe ARDS |
Phase 2a randomized, double-blind, placebo- controlled |
Bone marrow- derived MSCs |
Intravenous |
Safety established in critically ill ARDS population |
|
ADMIRE-CD, 2016 |
Complex perianal fistulizing Crohn’s disease |
Phase 3 randomized, double- blind, placebo-controlled |
Expanded adipose-derived MSCs |
Local injection |
Favorable safety of local allogeneic MSC administration in inflammatory GI disease |
|
NEPHSTROM |
Diabetic kidney disease |
Randomized placebo- controlled early phase |
Bone marrow- derived MSCs |
Intravenous |
Supportive renal safety evidence with no strong treatment-related serious toxicity signal |
|
Matas et al. |
Knee osteoarthritis |
Randomized phase I/II |
Umbilical cord- derived MSCs |
Intra-articular |
No severe treatment- related safety signal reported; supportive local tolerability evidence |
Table 1: Landmark Allogeneic MSC Clinical Trials Included for Structured Safety Synthesis
|
Study |
Scope |
Main Safety Conclusion |
|
Lalu et al., 2012 SafeCell |
36 studies, 1,012 participants |
MSC therapy appeared safe; transient fever significantly associated with treatment |
|
Thompson et al., 2020 intravascular MSC review |
Updated intravascular RCT safety review |
MSC therapy with intravascular administration continued to appear safe |
|
Wang et al., 2021 |
62 randomized trials across populations |
No increased risk of SAE, death, infection, arrhythmia, vascular disorder, thrombosis, or malignancy; transient fever increased |
Table 2: Landmark MSC Safety Meta-Analyses Informing Pooled Interpretation
|
Safety Domain |
Direction of Evidence |
|
Treatment-related serious adverse events |
No reproducible increase identified across landmark trials |
|
Acute infusion toxicity |
No consistent major signal identified |
|
Transient fever |
Increased relative to control in pooled safety meta-analyses |
|
Infection |
No consistent increase identified in pooled analyses |
|
Death |
No consistent increase attributable to MSC therapy in pooled analyses |
|
Arrhythmia / vascular events / thrombosis |
No consistent increase identified in pooled analyses |
|
Malignancy |
No signal identified in pooled analyses |
|
Ectopic tissue formation |
No recurring signal identified in landmark trials |
|
Major alloimmune toxicity |
No reproducible clinical signal identified |
Table 3: Safety Domains and Direction of Evidence
|
Strengths |
Limitations |
|
Safety consistency across multiple indications |
Heterogeneous adverse event definitions |
|
Multiple randomized and placebo-controlled landmark trials |
Modest sample sizes in some disease areas |
|
Concordant pooled safety meta-analyses |
Limited long-term surveillance |
|
Favorable allogeneic platform-level signal |
Rare-event detection remains underpowered |
Table 4: Key Strengths and Limitations of The Current Evidence Base
The evidence synthesis prioritized landmark allogeneic MSC randomized and controlled trials across major indications, supplemented by the three principal MSC safety meta-analyses used to interpret pooled cross-indication safety trends.
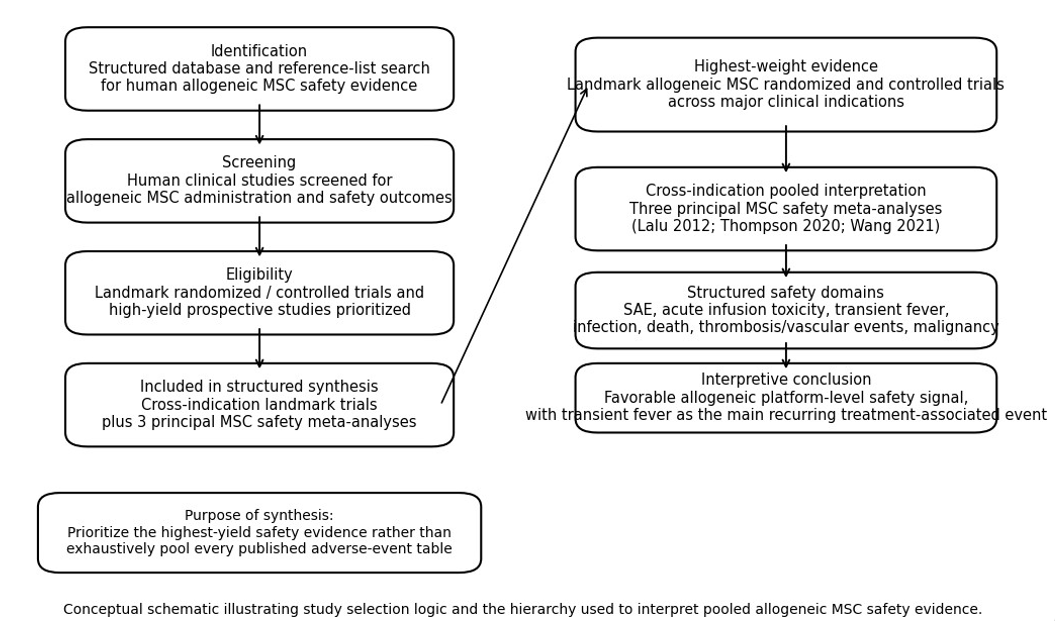
Figure 1: Conceptual Prisma Flow and Structured Evidence Hierarchy
References
- Lalu, M. M., McIntyre, L., Pugliese, C., Fergusson, D., Winston, B. W., Marshall, J. C., ... & Canadian Critical Care Trials Group. (2012). Safe ty of cell therapy with mesenchymal stromal cells (safecell): a systematic review and meta-analysis of clinical trials. PloS one, 7(10), e47559.
- Thompson, M., Mei, S. H., Wolfe, D., Champagne, J., Fergusson, D., Stewart, D. J., ... & McIntyre, L. (2020). Cell therapy with intravascular administration of mesenchymal stromal cells continues to appear safe: an updated systematic review and meta-analysis. EClinicalMedicine, 19.
- Wang, Y., Yi, H., & Song, Y. (2021). The safety of MSC therapy over the past 15 years: a meta-analysis. Stem cellresearch & therapy, 12(1), 545.
- Hare, J. M., Traverse, J. H., Henry, T. D., Dib, N., Strumpf, R. K., Schulman, S. P., ... & Sherman, W. (2009). A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction. Journal of the American College of Cardiology, 54(24), 2277-2286.
- Hare, J. M., Fishman, J. E., Gerstenblith, G., Velazquez,D. L. D., Zambrano, J. P., Suncion, V. Y., ... & Heldman,A. W. (2012). Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the POSEIDON randomized trial. Jama,308(22), 2369-2379.
- Matthay, M. A., Calfee, C. S., Zhuo, H., Thompson, B. T.,Wilson, J. G., Levitt, J. E., ... & Liu, K. D. (2019). Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): a randomised phase 2a safety trial. The Lancet Respiratory Medicine, 7(2), 154-162.
- Panés, J., García-Olmo, D., Van Assche, G., Colombel, J. F., Reinisch, W., Baumgart, D. C., ... & Danese, S. (2016). Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. The Lancet, 388(10051), 1281-1290.


