Review Article - (2018) Volume 3, Issue 2
Role of P-53 Gene in Preventing Breast Cancer
2Professor of Physiology, Titu Maiorescu University, Bucharest, Romania
Received Date: Jun 22, 2018 / Accepted Date: Jun 27, 2018 / Published Date: Jul 04, 2018
Copyright: ©Aurelian Udristioiu. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Breast cancer affects more than one million patients annually in the world and is a leading cause of mortality. Histological type, grade, tumor size, lymph node involvement, and estrogen receptor and HER-2 receptor status, all influence prognosis and the probability of response to systemic therapies. P53 gene is mutated in about 30% of breast cancers/.The possible links between alterations of p53 and clinical or pathological features of breast tumors have been widely investigated. The first study to examine gene-expression patterns of breast cancer suggested that at least four major molecular classes of breast cancer exist: luminal-like, basal-like, normal like, and HER-2 positive.
Basal-like breast cancer account for 15% of breast cancers and are often described as triple negative breast cancers (TNBCs). In fact, TNBCs, defined by lack of expression of estrogen receptor, progesterone receptor, and HER2, probably include both basal-like breast cancers and some poorly differentiated luminal breast cancers. They are also associated with a younger age and a poor prognosis. TNBCs also have an increased frequency of TP53 mutations. Recently, it was shown that mutant p53 status was a strongly unfavorable prognostic factor for relapse-free survival and overall survival only in a triple negative group in patients treated with adjuvant anthracycline-containing chemotherapy.
Inflammatory breast cancer (IBC) is a clinical diagnosis known as the T4d category in the TNM classification. It is a distinct clinical subtype of locally advanced breast cancer (LABC), with a particularly aggressive behavior and poor prognosis. TP53 mutations are more frequent in inflammatory breast cancer (50%) than in non-inflammatory breast cancer (20–30%).
The results from these studies served as the justification for attempts to vaccinate patients using p53-derived peptides, and a number of clinical trials are in progress. The most advanced work used a long synthetic peptide mixture derived from p53 (p53-SLP; Netherlands).The vaccine is delivered in the adjuvant setting and induces T helper type I response in the majority of patients. However, the response may not be potent enough to result in clinical benefit as a mono-therapy because most patients had T-helper cells that failed to produce key cytokines, indicating that this treatment should also be associated with another type of conventional or immunotherapy therapy.
Keywords
Inflammatory Breast Cancer, Triple Negative Breast Cancers, P53 gene, MDM2–p53 Complex, Nutlin
Introduction
Breast cancer affects more than one million patients annually in the world. The prognosis of patients with localized breast cancer is determined by clinical and biological factors such as age at diagnosis, tumor size, nodal status, tumor histological, grading, expression of estrogen/progesterone receptors. And Her 2 status. Treatments combine surgery on the primary tumor, radiation therapy of the breast, chemotherapy, and hormone therapy. Breast cancer is a heterogeneous disease. Histological type, grade, tumor size, lymph node involvement, and estrogen receptor and HER-2 receptor status, all influence prognosis and the probability of response to systemic therapies [1].
Body review
P53 gene is mutated in about 30% of breast cancers/.The possible links between alterations of p53 and clinical or pathological features of breast tumors have been widely investigated. The first study to examine gene-expression patterns of breast cancer suggested that at least four major molecular classes of breast cancer exist: luminal-like, basal-like, normal like, and HER-2 positive [2].
Table 1: Summary of the breast tumor molecular subtypes, intrinsic subtype, IHC status, grade prognosis
|
Intrinsic subtype |
IHC status |
Grade prognosis |
|
-Luminal A |
[ER+|PR+] HER2-KI67-, -1|2 |
Good prognosis |
|
-Luminal B |
[ER+|PR+] HER2-KI67, +2|3 |
Intermediate prognosis |
|
-HER2 over-expression |
[ER-PR-] HER2+2|3 |
Poor prognosis |
|
-Basal* |
ER-PR-] HER-, basal marker |
3Poor prognosis |
|
Normal-like |
[ER+|PR+] HER2-KI67-1| |
3Intermediate7.8% prognosis. |
Basal-like breast cancers account for 15% of breast cancers and are often described as triple negative breast cancers (TNBCs). In fact, TNBCs, defined by lack of estrogen receptor, progesterone receptor, and HER2, probably include both basal-like breast cancers and some poorly differentiated luminal breast cancers. They are also associated with a younger age and a poor prognosis TNBCs also have an increased frequency of TP53 mutations. Recently, it was shown that p53 status was a strongly unfavorable prognostic factor for relapse-free survival and overall survival only in a triple negative group in patients treated with adjuvant anthracycline-containing chemotherapy [3].
Inflammatory breast cancer (IBC) is a clinical diagnosis known as the T4d category in the TNM classification. It is a distinct clinical subtype of locally advanced breast cancer (LABC), with a particularly aggressive behavior and poor prognosis. TP53 mutations are more frequent in inflammatory breast cancer (50%) than non-inflammatory breast cancer (20–30%).
Interpretation of prognostic data is complicated by the fact that earlier studies only used immunohistochemistry to detect the accumulation of p53. Breast tumors with positive immunostaining for p53 are usually ER and PR negative. This is often associated with a high rate of proliferation, a high histological grade, aneuploidy, and a poor prognosis [4].
Specific germline mutations in the hereditary breast-ovarian cancer susceptibility (HBC/HBOC) genes, BRCA1, BRCA2 and PALB2, have been shown to recur in French Canadians of Quebec, Canada. Germline TP53 mutation carriers are known to segregate in Li-Fraumeni syndrome families, which feature young age of onset breast cancer.P53 germline mutations found in French Canadian cancer families provided from hereditary cancer clinics and investigate 37 new BRCA1 and BRCA2 mutation-negative HBC/HBOC families for the TP53 mutations not selected for family history of cancer [5].
Level of TP53 among the sera of the studied group’s level of TP53 has been measured in U/ml using ELISA technique. The patients’ age range was 22-84 years (mean 51.29) most of them were in the fourth decade. 21 patients (42%) were premenopausal and (58%) were pos-menopausal. The study showed the demographical features which indicated that the mean of age of the majority of patients were within or above the menopause duration (51.29 ± 12.18 years) with highly significant difference in comparison with control (29.42±10.21years), this result is comparable to some extent with the previous study (50.9+11.8) (Haider, 92010). The mean of TP53 of malignant cases was 47±33.5 U/ml in comparison with 27.9±12.7 U/ml for healthy control [6]. The function of p53 is altered in nearly 50% of cancers, p53 being inactivated by mutations in the DNA binding domain or deletion of the carboxy-terminal domain. In other cases, loss of p53 function is related to other mechanisms such as interaction with a viral protein (inlymphoma hepatocellular carcinoma), multiplication of the MDM2 gene (in sarcomas), and deletion of p14ARF gene (in breast or lung tumors). P-53 transcriptional activity is based on the formation of tetramers (dimers of dimers). Mutant proteins may interfere with wild-type p53 by forming hetero-oligomers that are less competent for specific DNA binding. It has been shown that some missense mutations gain oncogenic properties (Figure 1) [7].
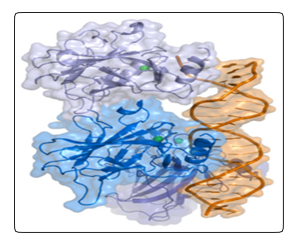
Figure 1: P-53 protein in tetrameric form.
The best method for to test the normal functionality of p-53 protein is the Fasay test. Detection of p53 by immune-hysto-chemistry cannot make the determination of TP53 function status but only the presence or absence of p-53 in normal or malignant cells (Figure 2).
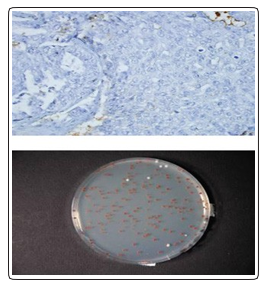
Figure 2: Detection of p53 by immunohystochemistry and determination of TP53. [8]. (Varna M, Bousquet G, Plassa FL, Bertheau P, Janin A. TP53 Status and Response to Treatment in Breast Cancers. J Biomed Biotechnol. 2011; 2011: 284584)
Similarly, methylation at the CDKN2A (INK4a/ARF) locus can epigenetically silence the expression of the p14ARF protein, and block the ability of activated oncogenes to stabilize the p53 response. In experimental models, disrupting the MDM2–p53 interaction restored p53 function and sensitized tumors to chemotherapy or radiotherapy. This strategy could be particularly beneficial in treating cancers that harbor TP53 mutations. In these tumor types, the induction of p53—using a small-molecule inhibitor of MDM2, nutlin-3—can induce apoptosis in malignant cells. On the other hand, nonmalignant cells seem to tolerate MDM2 inhibition and no high-grade toxic effects have been observed in normal tissues. With the recent discovery of specific MDM2 inhibitors that activate p53 it is now possible to probe the p53–MDM2 response in different tumor settings [9].
The most well studied small-molecule inhibitor of the MDM2–p53 complex is nutlin. Nutlins are a group of cis-imidazoline analogs, first identified by Vassilev and colleagues that have a high binding potency and selectivity for MDM2. Nutlin induces cell-cycle arrest in normal cells and is associated with minimal toxic effects in xenograft mouse models. However, intrinsic DNA damage in tumor cells may result in elevated levels of post-translationally modified p53 (for example phosphorylation at Ser46 and Ser392). Further stabilization of modified forms of p53 by nutlin-3, which inhibits the negative auto regulatory loop between MDM2 and p53, results in the activation of other p53-dependent proapoptotic pathways. By enhancing the apoptotic signals through exposure to radiation or other exogenous cytotoxic drugs that induce DNA damage, synergism between nutlin-3 and other therapeutics towards apoptosis in tumor cells can be achieved (Figure 3) [10].
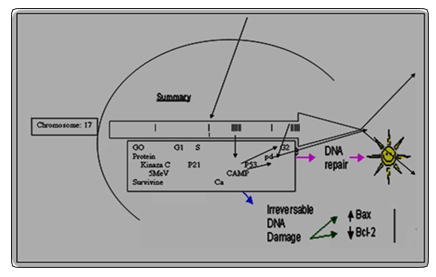
Schedule 1: P-53 protein in interaction by enhancing of the apoptotic signals for DNA repair.[10].(Udristioiu A, Florescu C, Popescu MA, Cojocaru M. High Concentration of anaerobic ATP implicated in aborted apoptosis from CLL. LabMed. 2010; 41: 203-208).
Breast cancer (BC) is the foremost cause of cancer-related mortality in women worldwide. An increasing number of studies has confirmed that micro RNAs (mi RNAs or mi Rs) play an important role in the development and progression of BC. Nucleic acid microRNA-214 (miR-214), a member of the mi RNA family, has been demonstrated to function as both a tumor suppressor and oncogene in various types of human cancer. Furthermore, p53, which has been reported to be down regulated in BC, was predicted to be the target gene of miR-214 using bioinformatics software programs. Moreover, luciferase reporter vectors were constructed and it was confirmed that p53 is a target of miR-214. Following the transfection of miR-214 into BC cells, we found that the over expression of miR-214 markedly enhanced cell invasion through the down regulation of p53 expression. By contrast, the over expression of p53 abrogated the effects of miR-214. In conclusion, this study demonstrates that miR-214 functions as an oncogene in BC, at least partly by promoting cell invasion through the down regulation of p53 [11].
Antibodies specific for total p53 and for p53 phosphorilated at three different sites within the activation domain were used in parallel analyses. This has led to a number of active clinical trials using with large peptides derived from p53 and a search for evidence of a natural immune response to p53 in cancer patients. While it has been established for some time that cancer patients make antibodies to p53 the frequency and magnitude of this response is still debated. However, a large number of patients with cancer produce p53-reactive T cells; short-term in-vitro stimulation with p53-transfected dendritic cells revealed that more than 40% of patients with breast cancer have p53-reactive CD4 and CD8 T cells in their peripheral blood. These responses occur most frequently in patients with high p53 protein expression in their tumors, although they are also found rarely in patients with low p53 expression or in healthy individuals [12, 13].
The results from these studies served as the justification for attempts to vaccinate patients using p53-derived peptides, and a number of clinical trials are in progress. The most advanced work used a long synthetic peptide mixture derived from p53 (p53-SLP; Netherlands). This vaccine consists of 10 overlapping peptides (0.3 mg per peptide), representing the p53 protein from amino acids 70 to 248. This region is recognized by the T cells of all colorectal patients displaying p53 immunity and it harbors most of the known MHC class I and II epitopes. The vaccine is delivered in the adjuvant setting and induces T helper type I response in the majority of patients. However, the response may not be potent enough to result in clinical benefit as a mono therapy because most patients had T-helper cells that failed to produce key cytokines indicating that these p53-specific T-helper responses are not polarized. Therefore, approaches are being investigated to promote a stronger and more correctly polarized response using both DNA-based and dendritic cell-delivered p53 vaccines [14].
Some studies suggested that p53 may influence response to anti hormonal treatments. TP53 mutations are less frequent in patients with ER-positive breast cancers, but they are associated with a poorer prognostic in these patients. In vitro studies on human breast cancer cell lines, MN1 (p53WT) and MDD2 (p53MUT) derived from MCF-7, it was shown that p53 mutated cells were more resistant to cytotoxic effects of 4-hydroxy-tamoxifen compared to p53wild-type cells. Clinical studies on patients with locally advanced breast cancer treated with tamoxifen or primary chemotherapy showed that mutations in the TP53 gene are associated with a poor survival. Temporal activation of p53 by a specific MDM2 inhibitor is selectively toxic to tumors and leads to complete tumor growth inhibition [15].
TP53 status shows a strong prognosis impact and this could be useful in the choosing the best treatment for breast cancer. Generally, TP53 mutated is associated with a poor response to chemotherapy, hormone therapy or radiotherapy. Discordant studies concerning its predictive value exist, and this is linked to method of detection of TP53 status. We show that FASAY test and sequencing of TP53 are better than immunohistochemistry to determine if TP53 is mutated or not mutant [Table 2].
Table 2: PD-1 and PD-L1 Antibodies in Clinical Development (Abbreviations: Ab, antibody; IgG, immunoglobulin G; PD-1, programmed cell death protein 1; PD-L1, programmed cell death protein 1 ligand
|
Target |
Agent |
Class |
|
PD-1 |
Nivolumab (MDX1106, BMS-936558) |
IgG4 fully human Ab |
|
Pembrolizumab (MK-3475) |
IgG4 engineered humanized Ab |
|
|
Pidilizumab (CT-011) |
IgG1 humanized Ab |
|
|
PD-L1 |
BMS935559 (MDX-1105) |
IgG4 fully human Ab |
|
MPDL3280A |
IgG1 engineered fully human Ab |
|
|
MEDI4736 |
IgG1 engineered fully human Ab |
|
|
MSB0010718C |
IgG1 fully human Ab |
|
|
PD-1–positive T cells |
AMP-224 |
Fc of human IgG–PD-L2 fusion |
Immune therapeutic success
After chemotherapy treatment, tumor antigens are taken up by cells presenting antigen (APC) and are presented in the context of the co-stimulatory molecules B7 from dendritic cells. T cells recognize antigens to become activated. T-cells may differentiate into memory T cells that can turn into tumor recurring presence not only through induction of genetic programs, which leads to a proliferation and differentiation, but also to induce receptor inhibitor mediated by CTLA-4 program, which ultimately is going to stop proliferation. As T-cell receptor CTLA-4, T-cell receptors, PD-1 is expressed only in activated T cells to stop. In many laboratory studies, today are ongoing the clinical trials with anti-CTLA-4 and immunological control points, i.e. PD-1 / PDL1 can improve the prospects of patients with various malignancies (Figure 3) [16].
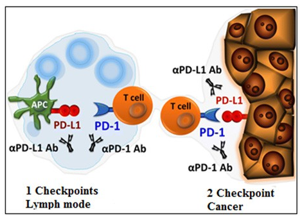
Figure 3: PD-1 inhibits T-cell responses by interfering with the T cell receptor signaling unlike competing out-CD28 [16]. (Postow AM, Kallahan K, Wolchok JD. Immune Checkpoint Blockade in Cancer Therapy. J Clin Oncol. 2015; 33: 1974-1982].
Figure 3: PD-1 inhibits T-cell responses by interfering with the T cell receptor signaling unlike competing out-CD28 [16]. (Postow AM, Kallahan K, Wolchok JD. Immune Checkpoint Blockade in Cancer Therapy. J Clin Oncol. 2015; 33: 1974-1982].
Interactions between PD-1 and its ligands, PD-L1 and PD-L2, are complex and occur in several stages of an immune response (Figure 2). According to Postov and collaborators, there is an activation mechanism in the lymph node where PD-L1 / PD-L2 on an antigen presenting cell (dendritic cell) negatively regulates T-cell activity by PD-1 and an interaction between B7 and PD- L1. The PD-1 pathway is also likely to be important in the tumor micromedium where PD-L1 expressed by tumors interact with PD-1 on T cells to suppress effector function of T. MHC cells, a major histocompatibility complex.It is important to remember that the breast cancer survival rates is, in reality, going to be higher.This is because the breast cancer survival rates data is gathered from a large number of people with the disease over a 5 year period. Thus, with the ongoing improvements and advancements in breast cancer screening, research, early detection and advanced tailored treatment, the outcomes at present will be even better than in past years. Also, it is important to bear in mind other factors discussed in this post, such as stage, grade and hormone receptor status play an important role in prognosis.
Conclusion
The special laboratory techniques used to identify the genetic make-up of cancers, this genetic information may become a better predictor of cancer aggressiveness and outcome than stage, which has been the diagnostic indicator of choice in the past. Additionally, this genetic information will likely play an increasing role in directing treatment. Specifically, the genes involved in each cancer may indicate more aggressive treatment for some cancers and less aggressive treatment for other forms.Prospective studies using these two methods could better determine its predictive value according to response to treatments
References
- Beije N, Onstenk W, Kraan, J, Sieuwerts MA, Hamberg P (2016) Prognostic Impact of HER2 and ER Status of Circulating Tumor Cells in Metastatic Breast Cancer Patients with a HER2-Negative Primary Tumor. Neoplasia 18: 647-653.
- Dai X, Li T, Bai Z, Yang Y, Liu X (2015) Breast cancer intrinsic subtype classification, clinical use and future trends. Am J Cancer Res 5: 2929-2943.
- Esfahani DS, Denkert C, Stenzinger A, Salat C, Sinn. B (2016) Role of TP53 mutations in triple negative and HER2-positive breast cancer treated with neoadjuvant anthracycline/taxane-based chemotherapy. Oncotarget 187: 67686-67698.
- Anderson FW, Schairer C, Bingshu E. Kenneth W, Hance C, et al. (2005) Epidemiology of Inflammatory Breast Cancer (IBC). Breast Dis 22: 9-23.
- Arcand LS, Akbari RM, Mes-Masson MA, Provencher D, Foulkes DW (2017) Germline TP53 mutational spectrum in French Canadians with breast cancer. BMC Med Genet 16: 24.
- Jabir AF, Hoidy HW (2017) No Evaluation of Serum P53 Levels in Iraqi Female Breast Cancer Patients. Asian Pac J Cancer Prev 18: 2551-2553.
- Eymin E, Gazzeri S, Brambilla C, Brambilla C, E. Brambilla (2002) Mdm2 overexpression and p14ARF inactivation are two mutually exclusive events in primary human lung tumors. Oncogene 21: 2750-2761.
- Varna M, Bousquet G, Plassa FL, Bertheau P, Janin A (2011) TP53 Status and Response to Treatment in Breast Cancers. J Biomed Biotechnol 2011: 284584.
- Deben C, Wouters A, Beeck OK, Bossche DJ, Jacobs J (2005) The MDM2-inhibitor Nutlin-3 synergizes with cisplatin to induce p53 dependent tumor cell apoptosis in non-small cell lung cancer. Oncotarget 6: 22666-22679.
- Udristioiu A, Florescu C, Popescu MA, Cojocaru M (2010) High Concentration of anaerobic ATP implicated in aborted apoptosis from CLL. LabMed 41: 203-208.
- Wang F1, Lv P1, Liu X1, Zhu M1, Qiu X2 (2015) microRNA-214 enhances the invasion ability of breast cancer cells by targeting p53. Int J Mol Med 35: 1395-1402.
- Shangary S, Qin D, Mc Eachern D, Liu M (2008) Temporal activation of p53 by a specific MDM2 inhibitor is selectively toxic to tumors and leads to complete tumor growth inhibition. Proc Natl Acad Sci 105: 3933-3938.
- Van der Burg SH, Cock K, Menon AG, Franken KL (2001) Long lasting p53-specific T cell memory responses in the absence of anti-p53 antibodies in patients with respected primary colorectal cancer. Eur J Immunol 31: 146-155.
- Speetjens F, Kuppen P, Welters M, Essahsah F (2009) Induction of p53-specific immunity by a p53 synthetic long peptide vaccine in patients treated for metastatic colorectal cancer. Clin Cancer Res 15: 1086-1095.
- Fernandez-Cuesta L, Anaganti S, Hainaut P, Olivier M (2011) p53 status influences response to tamoxifen but not to fulvestrant in breast cancer cell lines. Int J Cancer 128: 1813-1821.
- Postow AM, Kallahan K, Wolchok JD (2015) Immune Checkpoint Blockade in Cancer Therapy. J Clin Oncol 33: 1974-1982


