Research Article - (2026) Volume 1, Issue 1
Role of Functional Groups in H2 Production from Small Organic Molecules Over Noble-Metal-Free Tio2 Under Photohydrothermal Activation
Received Date: Jan 23, 2026 / Accepted Date: Feb 25, 2026 / Published Date: Mar 06, 2026
Copyright: ©2026 Chantal Guillard, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Mediouni, N., Dappozze, F., Berhault, G., Guillard, C. (2026). Role of Functional Groups in H2Production from Small Organic Molecules Over Noble-Metal-Free Tio2 Under Photohydrothermal Activation. J Archit Eng Built Environ, 1(1), 01-16.
Abstract
This study aims to investigate the transformation mechanisms of simple organic molecules containing one or two carbon atoms and functional groups such as alcohols, aldehydes, or carboxylic acids, under photohydrothermal treatment (TPUV) using TiO2 UV100 as a photocatalyst in an inert atmosphere under UV irradiation. The main objective is to better understand the processes involved in TPUV by monitoring both liquid and gaseous products formed during the reaction. A comparative approach was adopted, evaluating the behavior of each molecule under individual thermal catalysis (TP) and photocatalytic (PUV) treatments, as well as under the combined TPUV process, all in the presence of TiO2 and inert gas. The impact of temperature and pressure was studied on only one compound, glyoxylic acid in order to assess its influence on the transformation and product formation. The study markedly reveals different behaviors depending on the nature of the functional group, particularly for molecules containing aldehyde groups. Moreover, with the exception of those molecules containing an aldehyde group, the coupled TPUV process led to higher H2 production compared to individual processes. This difference is discussed in terms of reduction of aldehyde function in competition with H2 formation. These findings provide new insights into the reactivity of small organic compounds under combined photonic and hydrothermal activation of TiO2 and highlighting its potential as an efficient noble-metal-free strategy for photocatalytic applications.
Keywords
TiO2, Photohydrothermal, Temperature, Pressure, UV, H2, Mechanism
Introduction
The energy crisis has driven researchers to investigate new hybrid processes. Although still relatively unexplored, the coupling of hydrothermal process and photocatalysis has shown promising results, especially in key areas such as CO2 reduction, hydrogen (H2) production, and biomass valorization. These early findings highlight the potential of this method as a sustainable and energy-efficient alternative for chemical transformations. Most publications investigating this hybrid process directed toward CO2 photoreduction using TiO2 based materials with noble metal (Au) at temperatures up to 95°C and pressures reaching 20 bars, or Ag/g-C3N4 catalysts conducted at 190°C under autogenous P and visible light irradiation [1-4]. Beyond CO2 photoreduction, photohydrothermal approaches have also been applied to hydrogen production from biomass-derived substrates, where the synergy of light and heat enhances both rates and yields.
For example, reported that photo-assisted hydrothermal gasification of biomass-derived substrates, such as glucose, can significantly enhance H2 and CH4 generation at high pressures (up to 30 MPa) and temperatures exceeding 350°C [5]. further explored photohydrothermal hydrogen production from various biomass-derived substrates, including levulinic acid, formic acid, and glucose, using noble-metal-decorated TiO2 catalysts (Pt, Pd, Ag, Au) [6]. Among these, glucose exhibited the highest H2 yield under relatively mild conditions (80°C, 4 bars), highlighting both its superior reactivity and the strong synergistic effect between light and heat. More recently, our work on glucose valorization shown that pristine TiO2 (without noble metal) allowed to improve the selectivity to high valueadded products which are attributed to the increase of the acidity [7].
Hypotheses to explain this behavior include the in-situ generation of Bronsted acidity during photocatalysis, and/or the enhanced Lewis acidity of TiO2 under UV irradiation which may facilitate hydrothermal transformations (improvement of reaction driven by acidity of solution and/or materials). Two years later, using dihydroxyacetone (DHA) as a model substrate, and TiO2 nanoparticles with varying acid-base properties under UV irradiation at 120°C and 5 bars, we demonstrated that the acidity of the catalyst did not significantly influence the observed synergistic effect. However, we confirmed the participation of Bronsted acidity to the formation of a new product [8].
In this case, this new product corresponds to the dehydration of an intermediate generated during the hydrothermal process, and promoted by H+ generation under photocatalytic conditions. Additionally, we highlighted the enhancement of H2 and CO2 production as well as the appearance of a reduction reaction attributed to the action of photogenerated electrons on a compound generated during the hydrothermal process. Altogether, while the synergistic effects of light, heat, and pressure have shown promise, particularly for energy-oriented applications, the fundamental mechanisms governing photocatalytic hydrothermal processes remain unclear, calling for deeper investigation.
Building on our previous DHA study, we aim here to broaden the scope to other molecules to better understand functional group behavior under such conditions [9]. This study aims to gain a deeper understanding of the transformation mechanisms and chemical pathways involved in photohydrothermal catalysis (TPUV) in the presence of TiO2 and an inert gas. To achieve this, we will use simple molecules containing one or two carbon atoms and bearing various functional groups (carboxylic, hydroxyl, and aldehyde). The TPUV process will be compared with the individual processes—hydrothermal treatment (TP) and photocatalysis (PUV)—by analyzing both the transformation of the molecules and the products formed in the liquid and gas phases. Lastly, the influence of temperature on this coupled process will be investigated.
Experimental Section
Materials
All compounds used in the photocatalytic tests, with or without hydrothermal contribution were: formic acid (Sigma Aldrich 98%), methanol (Alfa Aesar 99.9%), acetic acid (Sigma Aldrich 99%), ethanol (Carlo Erba), glycolic acid (Sigma Aldrich), ethylene glycol (Sigma Aldrich 99.8%), oxalic acid (sigma Aldrich), glyoxylic acid (Alfa Aesar 97%), and glyoxal (Sigma Aldrich 40% wt / water solution). A commercial titanium dioxide, pure TiO2 anatase Hombikat UV100, was purchased from Sachtleben Chemie and used as the photocatalyst; it has a specific surface area of 300 m²/g. Ultrapure water (18 MΩ.cm) was used for all experiments.
Photocatalytic, Hydrothermal and Combined Processes
The experiments were performed in a 100 mL stainless-steel autoclave (Figure 1), allowing experiments under controlled temperature and pressure. The reactor was equipped with a sapphire window at the bottom, permitting UV light irradiation using a PLL lamp (18 W, 365 nm, 6.2 mW.cm-2).
Figure 1: Scheme Representing the Photocatalytic Reactor Used Under Pressure and Temperature
All reactions were conducted using 50 mL of aqueous solutions of the different selected molecules at 0.5 g.L-1, with 0.5 g. L-1 of TiO2 UV100 used as the photocatalyst. The reaction mixtures were continuously stirred at 500 rpm, and the pressure was maintained at 5 bars under argon. Three types of experiments were performed, where only temperature and UV irradiation were varied:
• Hydrothermal reactions (TP): conducted at 90°C, 120°C, and 150°C in the dark, without UV irradiation.
• Photocatalytic reactions (PUV): performed at ambient temperature under UV light.
• Photohydrothermal reactions (TPUV): carried out at 90°C, 120°C, and 150°C under the same UV irradiation as PUV.
To prevent undesired transformations during thermal treatments, TiO2 was first dispersed in water in the autoclave which was then heated to the target temperature. Once the desired temperature was reached, the substrate was added, and UV irradiation applied if required, defining in this way the start of the reaction. At predetermined time points, 1000 µL samples were collected, filtered through a 0.45 µm hydrophilic MILLEX HVLP membrane (Millipore), and analyzed by HPLC.
Analytical Procedure
High-Performance Liquid Chromatography (HPLC)
The different molecules and their products formed in the aqueous phase were quantified using a Shimadzu Prominence HPLC system equipped with both a refractive index detector (RID) and a photodiode array detector (PDAD). Separation was performed on a Transgenomic ICSep ICE COREGEL-87H3 organic acid column, using 277 µL. L-1 H2SO4 as the mobile phase at a flow rate of 0.7 mL.min-1, with the column oven maintained at 40 °C. Detection was carried out using the RID and the PDAD at 210 nm.
The conversion of the starting molecule (denoted as X) and the yield of the product formed from its transformation (denoted as Y) were calculated using the following equations:
Conversion of X % x 100
![]()
where [X]i is the initial concentration of the starting molecule, [X] t is its concentration at a given reaction time.
Gas Phase Analysis
Gas-phase products were analyzed directly from the reactor using a Perkin Elmer Clarus 590 gas chromatograph. The gas flow was split between two detection pathways. Light gases (H2, Ar, O2, N2, CH4, CO) were separated on an RT Msieve5A molecular sieve column (30 m × 0.32 mm × 30 µm df, Restek) and detected using a pulsed discharge helium ionization detector (PDHID). Heavier organic compounds were directed through a PoraPlot Q column (25 m × 0.53 mm × 20 µm df, Agilent J&W Technologies) and passed through a Polyarc (R) system (Activated Research Company) consisting of two catalytic beds that first oxidize organics to CO2 and then reduce them to CH4, with detection by a flame ionization detector (FID). The GC oven was programmed from 50 °C, ramping at 20 °C.min-1 to 150 °C, which was then held until the end of the analysis.
Results and Discussion
Influence of Carbon Chain Length and Functional Groups on Their Transformation Under TPUV
A preliminary screening of the transformation of a series of small organic compounds containing either one or two carbon atoms was performed under photohydrothermal catalysis conditions (TPUV). The molecules included a variety of functional groups, such as carboxylic acids, aldehydes, and alcohols, allowing for the evaluation of both carbon chain length (C1 vs C2) and the influence of functional group on reactivity under photohydrothermal catalysis conditions (TPUV). All reactions were carried out under an inert atmosphere and in the presence of TiO2 UV100.
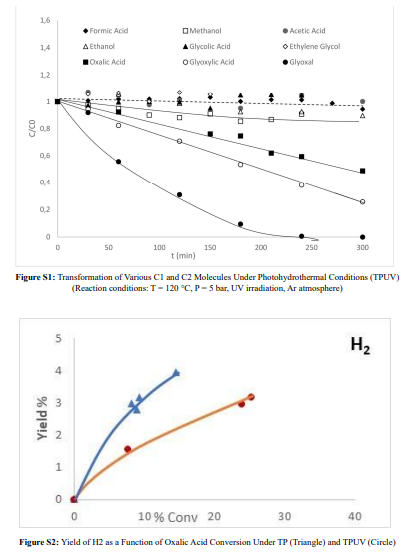
The results are presented in Figure S1. All compounds studied contain one or two carbon atoms with an alcohol or carboxylic acid group, or two alcohol groups, or one alcohol group and one carboxylic acid group, and show negligible transformation under photohydrothermal (TPUV) catalytic conditions. However, three two-carbon compounds stand out: one contains two carboxylic acid groups (oxalic acid), and the other two contain an aldehyde group (one with two aldehyde groups (glyoxal) and the other with one aldehyde group and one carboxylic acid group (glyoxylic acid)). These compounds undergo significant transformation under TPUV catalytic conditions, with the two aldehyde-containing compounds showing notably higher efficiency.
Considering these first results, the next step has been to compare their transformation in regards to their transformation under individual processes, hydrothermal process (TP) and photocatalysis (PUV). Given the negligible efficiency of formic acid, methanol, acetic acid, ethanol, ethylene glycol, and glycolic acid under TPUV, the comparison of these compounds will be addressed collectively while a separate section will focus on the three other compounds: oxalic acid, glyoxal, and glyoxylic acid.
Comparison of Thermal, Photocatalytic and Coupling Processes in The Transformation of The Different Model Molecules
The transformation of each model molecule was investigated under combined hydrothermal and photocatalytic processes (TPUV), hydrothermal-only treatment (TP) and pure photocatalytic reaction at ambient temperature (PUV). In all cases, commercial TiO2 UV100, a pure anatase phase with a BET surface area of 300 m2/g was used. Degradation rates and formation of liquid and gas products were systematically analyzed to better understand the degradation pathways and the role of each process.
Formic Acid, Methanol, Acetic Acid, Ethanol, Ethylene Glycol, and Glycolic Acid
For formic acid, methanol, acetic acid, ethanol, ethylene glycol, and glycolic acid, differences in disappearance rates under the three processes cannot be reliably estimated due to their very low overall degradation. Regarding the products formed in the liquid phase, it was observed that for all alcohols (methanol, ethanol, and ethylene glycol), aldehydes were detected under TPUV at concentrations close to those observed under PUV, indicating that photocatalysis is the dominant reaction mechanism in this case (Equations 1 and 2).
Equation 1: R-OH à R’-CHO + H2 (With R= CH3 and CH3-CH2 and R’ = H and CH3 respectively)
Equation 2: HO-CH2-CH2-OH à HOCH2–CHO + H2
However, in the case of ethanol and ethylene glycol, new compounds appeared under TPUV corresponding to dehydration reactions, leading to the formation of ethylene (CH2=CH2) and acetaldehyde (CH3CHO), respectively (Equations 3 and 4)
Equation 3: HO-CH2-CH2-OH à CH3-CHO + H2O
Equation 4: CH3-CH2OH à CH3-CHO +H2O
When a carboxylic group is present, decarboxylation was significantly more pronounced under TPUV compared to PUV and TP, with the latter exhibiting negligible decarboxylation (Figure 2).
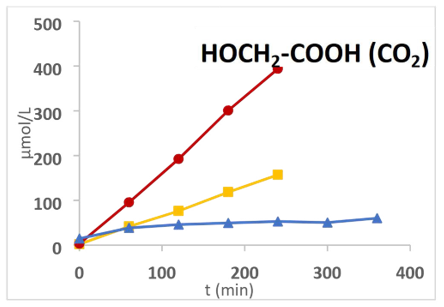
Figure 2: CO2 Formation During the Transformation of Formic Acid, Acetic Acid and Glycolic Acid Under TP (Triangle), PUV (Square) and TPUV (Circle)
It is also interesting to note that, for all these compounds, although the transformation under TPUV was very low (less than 15%), making it difficult to observe any significant difference compared to PUV or TP, the formation of H2 was consistently higher under TPUV (Figure 3), the hydrogen yields nevertheless remaining below 2% of the initial product concentration.
Figure 3: H2 Formation During the Transformation of Formic Acid, Methanol, Acetic Acid, Ethanol, Ethylene Glycol and Glycolic Acid Under TP (Triangle), PUV (Square) and TPUV(Circle)
The higher production of H2 may be attributed to the generation of H+ ions during the hydrothermal process due to protolysis of H O. In presence of irradiation and TiO2, these ions help separate charges by attracting electrons and favoring oxidation reactions, which improve the transformation of organic molecules. However, this increase of transformation under TPUV is not clearly observed because the conversion rates are low under all processes. Nevertheless, this hypothesis is consistent with the significant decarboxylation observed.
Oxalic Acid (OXA)
As shown in Figure 4, a moderate oxalic acid transformation occurred under TP while no significant transformation was observed under PUV. Degradation reached approximately 50% after 6 hours under TPUV, showing that the combination of temperature and UV light enhances reactivity.
Under TP and TPUV, traces of some products, which could be glycolic acid, acetic acid, formic acid, were detected in the liquid phase whereas no product was detected under the PUV process.
This result shows that the catalytic reduction of oxalic acid occurs under medium temperature and pressure [9] and can explain the detection of traces of methane and CO in the gas phase (Figure 4).
Figure 4: Disappearance of Oxalic Acid as A Function of Time Under Different Processes, And Gas Phase Products Formed During Degradation: Hydrogen (H2), Carbon Monoxide (Co), Carbon Dioxide (Co2) And Methane (Ch4) Under TP (Triangle), PUV (Square) and TPUV (Circle)
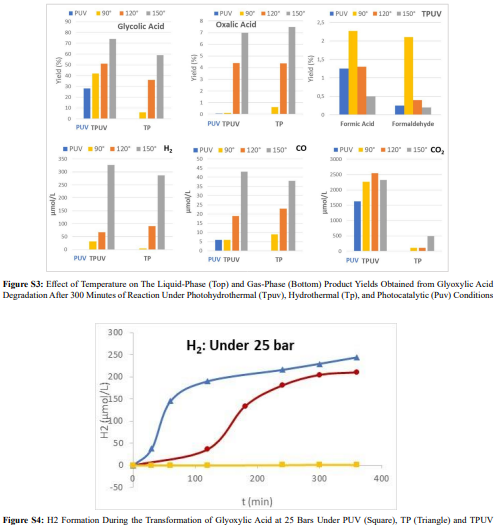
In addition to CH4 and CO, which were observed at similar concentrations under both TP and TPUV conditions, H2 and CO2 were detected across all processes (Figure 4). Their production, however, was enhanced under TPUV, consistent with previous observations for CO2 in the case of carboxylic compounds (Figure 2) and for H2 with all molecules previously studied (Figure 3). For oxalic acid, since its transformation under TP and TPUV is significant, we were able to plot H2 formation as a function of oxalic acid conversion (Figure S2). This clearly shows that the higher H2 production under TPUV results from increased oxalic acid conversion, supporting our earlier hypothesis that H+ generation via the hydrothermal process (through water autoprotolysis) promotes charge separation.
Glyoxal (GOX)
Glyoxal transformation under TP, PUV, and TPUV conditions are shown in Figure 5. As shown, GOX exhibits complete degradation under both TP and TPUV within 300 minutes, while under PUV, the conversion remained limited, reaching only ~13% after 6 hours and up to 46% after 27 hours. This result shows that glyoxal degradation is primarily temperaturedriven while UV irradiation alone only results in slow transformation.
Figure 5: Glyoxal Degradation Under TP (Triangle), PUV (Square) and TPUV (Circle) Conditions
The liquid and gas products formed during glyoxal transformation under these three processes (Figure 6) show that under TPUV, no new product was observed but favors greatly the formation of some of these products.
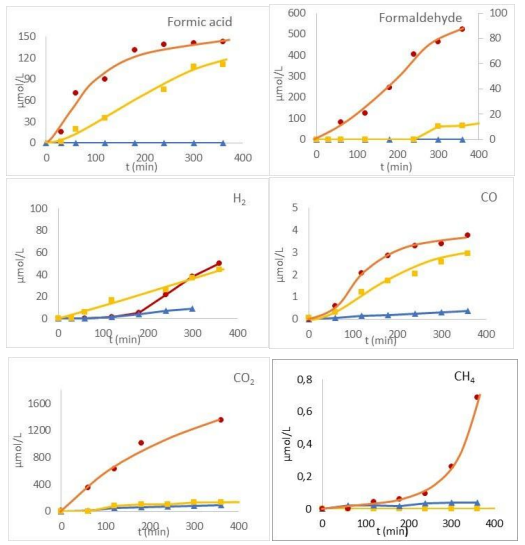
Comparison of The Products Formed By Individual Processes During Glyoxal Transformation
Under hydrothermal (TP) conditions, the main product of the glyoxal transformation was glycolic acid. Its formation can be attributed to a redox (dismutation) reaction in which one aldehyde group is reduced to an alcohol (—CH2OH) while the other aldehyde is oxidized to a carboxylic acid (—COOH). This process is typical of a degradation process occurring in the presence of a base such as OH- corresponding to a Cannizzaro-type mechanism. The formation of OH- by water protolysis is probably at the origin of this reaction. Its formation can also be explained by hydration of glyoxal in presence of H+ also formed by water protolysis. Both mechanisms are given in Equation 5 and 6 respectively:
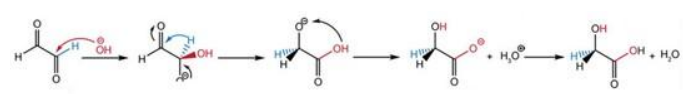
Equation 6: Hydration mechanism for glyoxal transformation
Gaseous products such as H2, CO, CH4 and CO2 were detected, but only in trace amounts.
Under UV-based (photocatalytic) conditions, the main products identified were formic acid and glycolaldehyde, both of which being absent under purely hydrothermal conditions. Glycolaldehyde and formic acid result from the photocatalytic transformation of glyoxal, 2 photogenerated electrons allowing the reduction of one glyoxal molecule into one molecule of glycolaldehyde whereas 2 OH radicals formed in parallel react with one molecule of glyoxal to generate 2 molecules of formic acid. Equations 7 to 11 describe the mechanism for glycolaldehyde and formic acid formations under photocatalysis.
Equation 7: 2H2O + 2h+ à 2H+ + 2OH°
Equation 8: 2H+ + 2e- à 2H°
Equation 9: HCO-COH + 2HO° à 2 HCOOH
Equation 10: HCO-COH + H° à HCO-CH2O°
Equation 11: HCO-CH2 O° + H° à HCO-CH2OH
A low concentration of formaldehyde was detected after 300 minutes of irradiation, suggesting its formation as a secondary product from glycolaldehyde. Regarding gaseous products, UV activation of TiO2 led to a higher formation of H2 and CO compared to TP while CO2 was produced only in slightly higher quantities under UV.
Comparison of The Products Formed by TPUV in Regards to Individual Processes
Glycolaldehyde, formaldehyde, formic acid and CO2 are strongly favored by coupling the TiO2 activation under temperature and UV light. It is also important to note the increase of the CH4 formation after about 2h30 of reaction indicating the increase of reduction reaction processes under TPUV. Another interesting point is also the transformation under UV light of glycolic acid formed by hydrothermal process. Individual process experiments of glycolic acid showed that no transformation occurred under TP suggesting that while thermal energy favors the formation of glycolic acid, photocatalyis may decompose it into formaldehyde and CO2 in presence of h+ (Equations 12 to 14) favoring the glyoxal reduction reaction to generate glycolaldehyde (Equations 15 to 17).
Equation 12: H+, -OOC-CH2OH + h+ à °OOC-CH2OH +H+
Equation 13: °OOC-CH2OH à CO2 + °CH2OH
Equation 14: °CH2OH à CH2O + H°
Equation 15: HCO-COH + H° à HCO-CH2O°
Equation 16: H+ + e- à H°
Equation 17: HCO-CH2O° + H° à HCO-CH2OH
The increase of CH4 formation by coupling activation of TiO2 under temperature and UV under pressure could be related to the reduction of glycolaldehyde. This point could be verified by investigating the transformation of glycolaldehyde. However, this compound is commercialized under dimer form and cannot be found as monomer. In summary, thermal conditions (TP) promote a Cannizzaro-type reaction, leading to stable liquid-phase intermediates (glycolic acid) and minimal gas formation. In contrast, UV based processes generate glycolaldehyde thanks to a direct reduction with electrons, this step being strongly favored under TPUV.
This is due to the increase of charge separation by decarboxylation of glycolic acid generated by Cannizzaro reaction under hydrothermal process leading in addition to an increase of glyoxal transformation. It is also important to note that, unlike all other previously tested molecules, H2 formation is not enhanced under TPUV conditions. This behavior may be attributed to the reduction of glyoxal in the presence of H° radicals, which is then unavailable for H2? production. This in-depth understanding of glyoxal transformation behavior now provides a solid foundation for investigating another molecule, glyoxylic acid (GA), which contains both aldehyde and carboxylic acid groups and for determining whether a similar mechanism can explain its transformation.
Glyoxylic Acid
Glyoxylic acid exhibits a distinct transformation behavior under the three processes, in contrast to the one observed for glyoxal. As shown in Figure 7, glyoxylic acid (GA) exhibits a low reactivity under thermal treatment alone (TP) while photocatalytic conditions (PUV) led to moderate conversion. The application of combined thermal and photocatalytic conditions (TPUV) significantly increased the degradation rate.
Figure 7: Glyoxylic Acid Degradation Under TP (Triangle), PUV (Square) and TPUV (Circle) in Presence of Tio2 Uv100
Beyond degradation rates, liquid phase analysis revealed the formation of four main degradation products in liquid phase: glycolic acid, oxalic acid, formic acid, and formaldehyde, and in the gas phase, H2, CO, CO2 and CH4, each showing distinct formation behaviors depending on the process used (Figure 8).
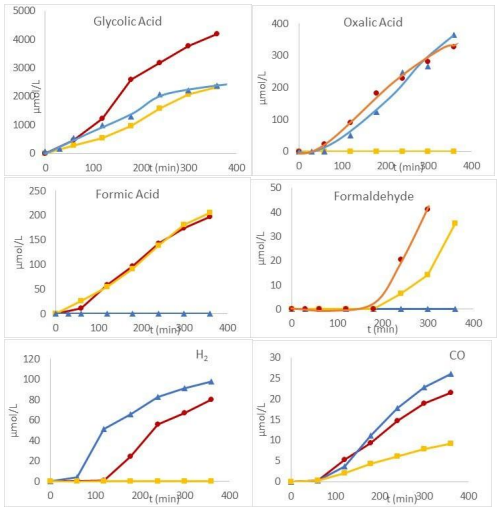
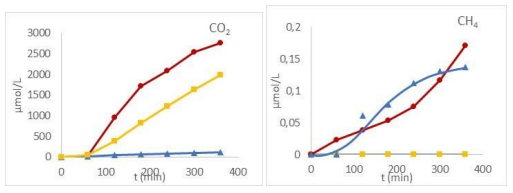
Figure 8: Liquid and Gas-Phase Degradation Products of Glyoxylic Acid as A Function of Time Under TP (Triangle), PUV (Square) and TPUV (Circle) Processes
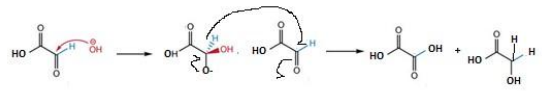
Oxalic acid, CO, H2 and CH4 are specifically formed by the hydrothermal process and are not improved by TPUV conditions. In contrast, formic acid and formaldehyde are exclusively produced through the photocatalytic process, with formaldehyde showing a slight increase under TPUV conditions. Glycolic acid is the predominant product under all conditions with the highest concentration observed under the photohydrothermal catalytic process (TPUV). The formation of glycolic acid and oxalic acid via the hydrothermal process (PT) suggests a Cannizzaro-like mechanism where two glyoxylic acid (GA) molecules disproportionate in the presence of hydroxide ions (OH–): one molecule is reduced to glycolic acid (CH2OH–COOH), and the other is oxidized to oxalic acid (COOH–COOH) (Equation 18).
Equation 18:
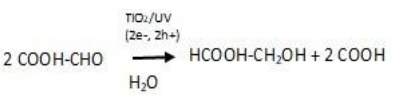
The generation of H2 and CH4 is consistent with the reductive decomposition pathways of oxalic acid under thermochemical processing, suggesting the occurrence of reduction reactions. Under PUV, the main product formed corresponds to glycolic acid. Its formation is explained by glyoxylic reduction with two electrons generated during photocatalysis while the two asformed h+ participate in the formic acid generation. Equation 19 summarizes the reduction of glyoxylic under photocatalysis conditions.
Equation 19:
This observation highlights that in photocatalytic systems, reduction and oxidation processes are intrinsically coupled due to the simultaneous generation of photogenerated electrons nd holes. The formation of formaldehyde, generated under UV activation of TiO2 and appearing only after about 180 min, is explained by the decomposition of glycolic acid formed by glyoxylic acid degradation as explained previously in the case of glyoxal transformation.
Under TPUV, an improved formation of glycolic acid, CO2 and in a lesser extent formaldehyde was observed. The higher concentration of glycolic acid observed under TPUV conditions, compared to the individual processes, is likely due to the combined effects of both individual processes. The slight improvement of formaldehyde generation after about 180 min and the improvement of the formation of CO2 can be explained by the photocatalytic degradation of glycolic acid following the equations 8-10, but also the decarboxylation of oxalic acid.
During the transformation of glyoxylic acid, the lowest H2 formation under TPUV compared to individual processes could be explained by the significant participation of photoelectrons in the reduction of glyoxylic acid to glycolic acid leading consequently to a partial decrease of their participation in the H2 formation. Similar phenomenon was also observed with glyoxal. But in this case, no inhibition of H2 production was noted. This behavior could be explained considering the small reduction observed since only small amount of glycolaldehyde was formed.
In summary, the transformation of glyoxylic acid under TPUV conditions confirms the phenomena previously observed during the conversion of glyoxal under the same conditions. First, the products resulting from the Cannizzaro reaction are formed under hydrothermal conditions (TP), then transformed under photocatalytic irradiation (PUV). Secondly, glyoxylic acid and glyoxal undergo reduction by photogenerated electrons during photocatalysis. Indeed, under TPUV, these electrons are no longer fully available to improve the production of H2, because the protons (H+) resulting from the protolysis of water under hydrothermal conditions lead to the formation of hydrogen radical (H°) under photocatalysis, reducing the aldehyde function instead of forming H2.
Effect of Process Parameters on The Glyoxylic Acid Transformation Under Photohydrothermal Conditions
Effect of Temperature
Temperature is a critical parameter in the photohydrothermal catalytic process since it influences both reaction kinetics and the intermediate species. To evaluate its effect, glyoxylic acid (GA) degradation was studied under TP and TPUV conditions at three different temperatures: 90°C, 120°C, and 150°C, while maintaining constant pressure (5 bars), UV irradiation, and an inert argon atmosphere.
As shown in figure 9, increasing the temperature leads to a clear enhancement in GA disappearance rate confirming that the thermal energy plays a crucial role in accelerating the reaction. Figure 9 also indicates that below 120°C, photocatalysis is the dominant reaction, whereas at 150°C, only the hydrothermal process takes place likely due to the reduced efficiency of the photocatalyst at elevated temperatures via charge carrier recombination.
Figure 9: Effect of Temperature on The Degradation Rate of Glyoxylic Acid Under TP (Triangle), PUV (Square) and TPUV (Circle) Conditions
The influence of temperature on the distribution of products is detailed in supplementary Figure S3, which presents the liquid-and gas-phase yields after 5 hours of reaction under TPUV and TP at 90°C, 120°C, and 150°C, compared to a reference PUV condition at 30°C.
• In the liquid phase, glycolic acid is consistently the main product under TPUV, with its yield increasing significantly with the temperature. Oxalic acid formation is strongly temperature dependent, occurring predominantly at high temperatures under TP and TPUV, and remaining negligible at 90°C and below. This behavior confirms that oxalic acids and a part of glycolic acid originate from the base-induced Cannizzaro reaction, which is thermally activated.
• In contrast, formic acid and formaldehyde, absent under thermal processing, accumulate mainly at lower temperatures (notably at 90°C under TPUV), indicating their photocatalytic origin and subsequent thermal decomposition at temperatures above 90°C.
• In the gas phase, the production of H2 and CO increases with temperature under both TPUV and TP, highlighting the thermal nature of these reductive pathways. In contrast, CO2 formation reaches a maximum under TPUV at moderate temperature (120°C), but decreases at 150°C. This result suggests that at moderate temperature, UV-driven oxidative decarboxylation of intermediates (formic acid, formaldehyde, oxalic acid) is favored. However, at higher temperature, enhanced charge recombination reduces photocatalytic efficiency and limits mineralization.
Altogether, these observations reveal a temperature-dependent balance between oxidative and reductive routes under TPUV conditions. While moderate temperatures promote oxidative decarboxylation and CO2 formation, higher temperatures favor reductive pathways, enhancing H2 and CO production but limiting full mineralization.
Effect of Pressure
The influence of pressure was investigated using glyoxylic acid at 120°C in the presence of TiO2 UV100 under a total pressure of 25 bars and compared with experiments conducted at 5 bars. Under purely thermal conditions (TP), an increase in oxalic acid concentration was observed whereas under combined thermophotocatalytic conditions (TPUV), no such increase was detected (Figure 10 a, b). Instead, a slight decrease in oxalic acid content was observed after 200 min, which can likely be attributed to its photodegradation under PUV irradiation. This hypothesis is consistent with the concomitant rise in CO2 formation (Figure 10c), as previously reported at 5 bars.
In addition, as also observed at 5 bars, hydrogen evolution at 25 bars was less significant under TPUV than under TP conditions (Figure S4). This behavior is explained by the competitive reduction of glyoxylic acid, which occurs at the expense of H2 production. Overall, these results indicate that increasing the pressure from 5 to 25 bars slightly enhances the Cannizzaro reaction under thermal conditions and promotes decarboxylation processes under TPUV. This effect can be ascribed to water protolysis, generating OH– and H+ species. The hydroxide ions favor the Cannizzaro reaction while protons facilitate charge separation through interaction with electrons (e–) and simultaneously promote decarboxylation via their reaction with photogenerated holes (h+).
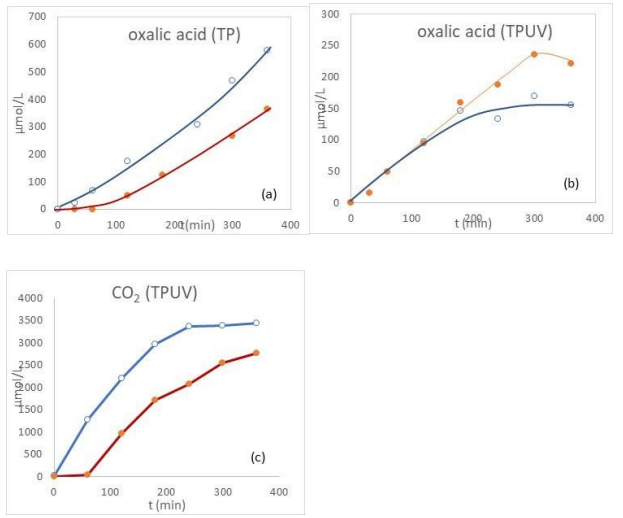
Figure 10: Oxalic Acid Formation Under 5 And 25 Bars Under TP (A) and TPUV (B) And Co2 Formation Under TPUV During the Transformation of Glyoxylic Acid in The Presence of Tio2 Uv100, 120°C and Inert Gas: Open Circles (25 Bars), Full Circles (5 Bars)
Conclusion
The comparison between the photohydrothermal process (TPUV) and the individual processes (thermal – TP, and photonic – PUV) in the presence of TiO2 under an inert atmosphere using simple C1–C2 organic molecules (carboxylic acids, alcohols, and aldehydes) demonstrates a significant enhancement in H2 production under TPUV, except for aldehyde-containing compounds. A marked improvement in the decarboxylation reaction is also observed. The increase in hydrogen evolution is attributed to water protolysis under hydrothermal conditions, which increases proton (H+) availability and thus enhances photocatalytic charge separation, decarboxylation reactions and H2 formation. In contrast, for aldehydefunctionalized molecules, H2 production is not improved because the preferential reduction of the aldehyde group competes with H2 formation. Under hydrothermal conditions, the Cannizzaro reaction occurs for aldehyde-containing compounds due to hydroxide ions (OH–) formed from water protolysis.
The resulting products undergo further transformation during photocatalysis. For example, in glyoxal conversion, TPUV promotes the formation of formaldehyde and CO2 through photocatalytic degradation of glyoxylic acid formed via the Cannizzaro reaction under TP. Simultaneously, TPUV significantly enhances glycolaldehyde formation through glyoxal reduction by hydrogen radicals (H°), enabled by the increased generation of H+. This process therefore appears promising for the selective valorization of organic compounds via reductive pathways, as evidenced by the improved glycolaldehyde yield from glyoxal. Moreover, pressure enhances the Cannizzaro reaction through intensified water protolysis, while temperature studies reveal a clear transition: TPUV boosts H2 production between 30 °C and 150 °C, but above 150 °C, the reaction is primarily driven by hydrothermal catalysis with minimal photonic contribution.
Overall, these findings demonstrate that photocatalysis assisted by hydrothermal conditions can significantly enhance H2 generation by increasing H+ availability, without requiring noble metal co-catalysts such as platinum. This highlights the potential of TPUV as a noble-metal-free alternative, warranting further comparison with conventional Pt/TiO2 photocatalysis. Finally, while TPUV shows improved H2 formation, it is important to note that, as in conventional photocatalysis, the conversion rates of organic substrates remain low (<10%) under the tested conditions (e.g., 500 mg/L), which currently limits the industrial viability of the process. Further optimization is therefore required to improve substrate conversion while maintaining high H2 yields.
Acknowledgment
The authors acknowledge the financial support of the ECOCHEM project within the PEPR SPLEEN research program. We also thank OpenAI for using ChatGPT to improve the English in this manuscript.
References
- Rossetti, A. Villa, C. Pirola, L. Prati, G. Ramis. (2014). A novel high-pressure photoreactor for CO2 photoconversion to fuels, RSC Adv. 4, 28883–28885.
- E. Bahadori, A. Tripodi, A. Villa, C. Pirola, L. Prati, G. Ramis,I. Rossetti. (2018). High Pressure Photoreduction of CO2:Effect of Catalyst Formulation, Hole Scavenger Addition and Operating Conditions, Catalysts 8, 430.
- I. Rossetti, A. Villa, M. Compagnoni, L. Prati, G. Ramis,C. Pirola, C.L. Bianchi, W. Wang, D. Wang. (2015). CO2 photoconversion to fuels under high pressure: effect of TiO2 phase and of unconventional reaction conditions, Catal. Sci. Technol. 5, 4481–4487.
- X. Han, M. Li, Y. Ma, Y. Li, H. Ma, C. Wang, Thermal coupled photocatalysis to enhance CO2 reduction activities on Ag loaded g-C3N4 catalysts, Surf. Interf. 23 (2021) 101006.
- A. Nakatani, N. Kometani. (2010). Photocatalytic effect of TiO2 on hydrothermal gasification of glucose, J. Phys. Conf. Ser. 215, 012091.
- G. Ramis, E. Bahadori, I. Rossetti. (2021). Design of efficient photocatalytic processes for the production of hydrogen from biomass derived substrates, Int. J. Hydrogen Energy 46, 12105–12116.
- I. Abdouli, F. Dappozze, M. Eternot, N. Essayem, C. Guillard. (2022). Hydrothermal process assisted by photocatalysis: Towards a novel hybrid mechanism driven glucose valorization to levulinic acid, ethylene and hydrogen, Appl. Catal. B: Environ. 305, 121051.
- A. Flory, F. Dappozze, C. Guillard. (2024). Dehydration and reduction reactions favored under medium pressure and temperature assisted by photocatalysis, Catal. Today 432, 114625.
- E. Schuler, L. Grooten, P. Oulego, N. R. Shiju, G.-J. M. Gruter. (2023) Selective reduction of oxalic acid to glycolic acid at low temperature in a continuous flow process, RSC Sustainability, 1, 2072-2080.
Supplementary Document
Role of Functional Groups in H2 Production from Small Organic Molecules Over Noble-Metal-Free Tioâ?? Under Photohydrothermal Activation