Review Article - (2025) Volume 4, Issue 2
Review on Novel Antibiotic Development: Highlighting Strength and Weakness
Received Date: Jun 06, 2025 / Accepted Date: Aug 11, 2025 / Published Date: Aug 20, 2025
Copyright: ©Â©2025 Anina Qureshi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Qureshi, A., Usman, S., Komish, H., Khan, A. N., Jameel, M. H. (2025). Prevalence and Predictors of Post-Partum Depression in Pakistan. Archives Clin Med Microbiol, 4(2), 01-06.
Abstract
Antimicrobial resistance (AMR) is a growing global health threat, making the need for new antibiotics more urgent than ever. This review explores recent advancements in antibiotic development while addressing the scientific, economic, and regulatory barriers that slow progress. It emphasizes the importance of innovation, policy changes, and responsible antibiotic use in the fight against resistance. A well-coordinated, multifaceted approach is essential to ensure that effective treatments remain available in the future.
However, significant challenges still stand in the way. Many new antibiotics are simply modifications of existing ones, limiting their long-term impact. High research costs, slow approval processes, and a lack of financial incentives discourage pharmaceutical companies from investing in new drug development. At the same time, poor global access and weak antibiotic stewardship programs continue to fuel resistance. Addressing these issues requires fresh approaches, better policies, and stronger global cooperation to secure the future of infectious disease treatment.
Keywords
Nandrolone decanoate, Osteomyelitis, Orthopedic rehabilitation, Anabolic steroids, Postoperative recovery
Introduction
Novel antibiotic drug development is a crucial area of research. The fight against antimicrobial resistance (AMR), which is becoming the greater danger to global health as germs acquire resistance to current medications, depends heavily on developing the new antibiotics. Older, less toxic antibiotics can be repurposed for use against resistant strains of bacteria. In contrast, new antibiotics can target can target mechanism of bacteria that have not yet been discovered like it techniques used any technological developments such as microbial genomics an X-ray crystallography has a speed up the search for novel antibiotics possibilities by exposing genetic pathways and bacterial structures that can be used to create new drugs nevertheless the fields have several obstacles to overcome such at the high expense and long development. It frequently takes more than 10 years and billions of dollars to bring new antibiotics to market. Additionally, economic dissent impedes progress because antibiotics are often utilized, traditional drug discovery strategies involve new chemical entities having a novel mechanism of action, involve various challenges, weaknesses and limitations [1].
Antibiotic resistance is a persistent worldwide health concern that is discussed in this article along with efforts to address this complicated issue. To slow the rising rate of antibiotic resistance, political goals, laws, scientific research and educational programs all are crucial. In an era with few new medicines for a bacterial illness, the difficult task of managing antibiotic resistance falls to researchers, politicians, and prescribers. The authors suggest that policy, practice, surveillance, monitoring and innovative treatment can address antibiotic resistance in both the human and agricultural domains. The intricacy of antibiotic resistance is emphasized in the essay as it is the necessity of the multimodal strategy to enhance the health outcome [2].
The rate at which multidrug-resistant bacteria are emerging and significant bacteria pathogens becoming resistant to standard antibiotic treatments is worrying. Only a few new antibiotics are in the clinical pipeline, and the current lack of effective medications and effective preventative strategies make it difficult to fight bacterial infections and the disease they cause. Instead, new treatment options and alternative antimicrobial therapies will need to be developed.
Strengths in Novel Antibiotics
The development pipeline for antibiotics exploded between 1940 and the 1960s, during the "golden age of antibiotics."Twenty therapeutically significant classes of antibiotics were found and developed during the fast-paced period between 1940 and 1962 [3,4]. The market has seen fewer new medications over the last 20 years, which has raised resistance to bacterial diseases, especially Gram-negative antibiotics such as MRSA, S. pneumoniae, and Pseudomonas aeruginosa. Only two significant novel classes, the cyclic lipopeptides and the oxazolidinones, have entered the market, both since 2000 (A. R. M. Coates & Hu, 2007) [5]. Additionally, novel substances were created using non-natural chemical means, such as metronidazole, isoniazid, oxazolidinones, and prontosil (the forerunner to sulpha medicines) (A. R. M. Coates & Hu, 2007) [5]. As molecular biology, medicinal chemistry, and advanced genomic techniques, the pharmaceutical industry appeared to be more capable than ever to find the next generation of innovative antibiotic drugs [3]. Historically, the primary targets of antibiotics have been bacterial DNA replication, protein synthesis, or peptidoglycan production. Table: 1 provides an overview of the new targets that researchers are looking for to combat bacterial infections as a result of widespread resistance [6].
|
Novel Targets Identified with References |
Discovery Approaches |
Sources |
|
Essential amino acid biosynthesis |
Informatics-based mining |
Medicinal plants |
|
Cell wall lipid biosynthesis |
Cryptic Biosynthetic Gene Clusters (BGC) activation |
Marine invertebrates |
|
Lipid insertion enzymes |
|
Insect and vertebrate symbionts |
|
Metal chelator biosynthesis |
Microbial co-cultures |
|
|
Quorum sensing metabolism |
BGC engineering |
Endophytic fungi and bacteria |
|
Clp proteases |
|
Uncultured microbes |
Table 1: Emerging Targets for Antibiotic Research and Development, Current Discovery Approaches, and Potential Sources forNovel Antibiotics
Among the latest remarkable discoveries is teixobactin, a depsipeptide that effectively combats Gram-positive bacteria by targeting the formation of their cell walls. Salinamide F is a substance that inhibits bacterial RNA polymerase and has strong antibacterial activity against both Gram-positive and Gram- negative bacteria. Gram-positive bacteria are effectively inhibited by copsin, a peptide with six cysteine links that prevents the development of cell walls. One polyaromatic oligopeptide that effectively inhibits bacterial DNA gyrase is albicidin. Moreover, cystobactamids block DNA gyrase [7]. The global public health community faces a serious threat from bacteria that are resistant to therapeutically useful drugs. The absence of research and development of new antibiotics is one of the factors that has been analyzed as contributing to this catastrophe [6].
The Pew Trust talks about how to better focus antibiotic discovery efforts and shows its progress in trying to promote the sharing of antibiotic discovery data and knowledge. It highlights initiatives to boost antibiotic development by the National Institute of Allergy and Infectious Diseases (NIAID) and the Global Antibiotic Research and Development Partnership (GARDP) and calls for more coordinated action and ongoing financing for early- stage research. Additionally, these two groups have offered their perspectives. Although the NIAID describes the free services it offers through the Division of Microbiology and Infectious Diseases, GARDP explains its founding and goals. Antimicrobial researchers, who might not be aware of its availability, should be incredibly interested in this help [8].
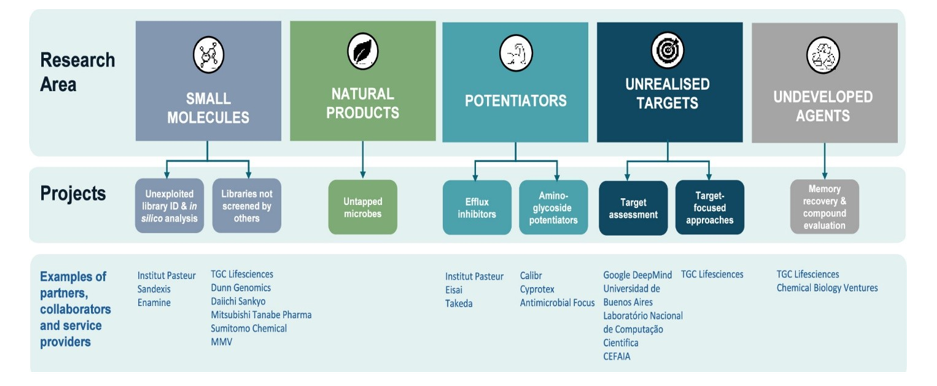
Figure 1: GARDP Discovery & Exploratory Research program’s research areas, projects, and examples of partners, collaborators, and service providers.
As the most effective method for creating novel antibiotics over the previous forty years, several publications concentrate on enhancing conventional antibiotic classes. As evidenced by the quantity of research articles, novel β-lactamase inhibitor (BLI)/β-lactam combos make up a significant portion of the clinical antibiotic pipeline Large pharmaceutical companies are controlling the production and research of antibiotics, according to WHO data, which is causing a gap in NDM-1 action and resistance to broad-spectrum antibiotics [8]. Antimicrobial resistance (AMR) is a serious issue that cannot be solved by new therapies. The development of about 41 medicines for bacterial illnesses in 2019 underscores the need for more research funding [9].
Challenges
The rapid rise in antimicrobial resistance due to overuse and misuse of antibiotics in human medicine and agriculture is one of the biggest challenges in developing novel antibiotics. Pathogens are evolving resistance mechanisms, making existing antibiotics ineffective. The process of discovering and developing a novel antibiotic is extremely costly, with estimates reaching billions of dollars. Pharmaceutical companies may not find it economically viable to invest in antibiotics, given the limited profit margins compared to drugs for chronic conditions.
Identifying novel antibiotic candidates is challenging due to the complexity of microbial interactions and the difficulty of targeting bacterial pathogens without harming human cells. Moreover, drug discovery can be hindered by the lack of robust screening methods and the failure of many compounds in clinical trials.
The regulatory process for approving new antibiotics is lengthy and complicated. Although there are incentives like the GAIN Act (Generating Antibiotic Incentives Now), navigating regulatory hurdles and ensuring that new antibiotics are safe, effective, and properly tested is difficult. The overuse and inappropriate prescribing of antibiotics in both healthcare and agriculture contribute to the rapid development of resistance. Balancing the necessity for new antibiotics with effective stewardship programs remains a critical issue. Even newly developed antibiotics can quickly lose their efficacy as bacteria evolve resistance mechanisms. This presents a continuous challenge in maintaining the usefulness of new antibiotics in clinical practice.
While novel antibiotics are being developed, ensuring global access, particularly in low- and middle-income countries (LMICs), remains a challenge. Lack of infrastructure, regulatory barriers, and cost can limit the availability and appropriate use of new antibiotics
Novel Antibiotic Development
• Enhancing Established Antimicrobial Substances
There are two ways to increase a known antimicrobial agent's activity. Firstly, altering an antimicrobial agent's fundamental chemical makeup to get beyond resistance to antibacterial mechanisms, such as tigecycline. Secondly, substances that block the processes of resistance to an antibiotic drug; novel β-lactamase inhibitors are one method utilized in this area [10].
• Peptide Imitation and Peptides with Antimicrobial Activity
In 2018, De Breij et al. substituted an amino acid from the LL-37 chain's ending carbon to create an LL-37 derivative (SAAP-148) with strong antibacterial properties. When tested against a variety of ESKAPE pathogenic bacteria, including E. fecium, S. aureus, K. pneumoniae, A. baumannii, P. aeruginosa, and Enterobacter species, this LL-37 derivative showed a minimum inhibitory concentration [MIC] ranging from 0.4 to 12.8 µM without selecting for resistance. Additionally, this AMP variant demonstrated anti- biofilm action against P. aeruginosa, A. baumannii,andS.aureus.
Another significant peptide antibiotic, colistin, is generated by Bacillus polymyxa var. colistinus and is used as a last option for combating multidrug-resistant infections. It has become a crucial treatment for Gram-negative bacterial infections, particularly those in hospitalized individuals brought on by MDR pathogens [11].
• Virulence-Blocking Agents
Targeting particular virulence factors associated with this process can influence pathogenesis as an alternative to the traditional drug development approach. By preventing the bacterium from becoming resistant, this tactic seeks to stop its propagation [10].
By injecting virulence proteins known as effectors that rewire the host cell machinery and enable immune response evasion, the majority of pathogenic Gram-negative bacteria use the bacterial type III secretion system (T3SS) as a virulence factor to trigger illness. Commensal bacteria do not have the T3SS; hence treatments that target it should not impact commensal bacteria. The T3SS is one of the virulence factors that are controlled by quorum sensing (QS), a collection of mechanisms that control the activity of genes in a concentration-dependent fashion. Both beneficial and harmful bacteria use QS. By decreasing the pathogens' infectivity, compounds that block QS may not be fatal to bacteria but could stop growth and disease [12].
• Metal Nanoparticles
Metal Nanoparticles (MNPs) are materials or particles that are smaller than a nanometer. Although these particles' exact mechanisms of action are unknown, three steps are thought to happen simultaneously [10]:
- By disrupting metabolic processes and causing modifications to the form and function of membranes, MNPs impair the permeability of cell membranes.
- Metal ions are discharged into the surrounding environment when MNPs are in solution. Metal ions cause oxidative stress in bacteria and produce active oxygen species, such as hydroxyl radicals and oxygen ions. Cell membrane damage can result from oxidative stress, which plays a major role in changing the porousness of bacterial membranes. Additionally, abnormal enzyme activity and alterations in cell structure brought on by metal ions might interfere with regular physiological functions.
- Interaction with substances like DNA that include phosphorus and sulfur, which inhibit transcription and DNA unfolding [11].
• Phage Treatment Combined with Enzybiotics
With their unique anti-pathogen action mechanisms, enzybiotics are a very promising substitute for conventional antibiotics. The words "enzyme" and "antibiotic" merge to form the phrase "enzybiotics.". Enzybiotics fall into six major types. These include the following phage-derived enzymes: Endolysins and virion- associated peptidoglycan hydrolases are examples of peptidoglycan hydrolases (PGHs),while polysaccharide depolymerases, or PSDs, are enzymes that destroy biofilms. Furthermore, among enzybiotics are enzymes generated from bacteria: enzymes that disrupt biofilms, bacterial enzymes called autolysins that degrade peptidoglycan to allow dividing cells to separate after splitting, and bacteriocins, which are antibacterial proteins or peptides made by bacteria that prevent the growth of similar bacteria. Furthermore, animal-derived enzymes, such as the lysozymes present in bodily fluids like tears, saliva, milk, and mucus, are included in the category of enzymes [1-6].
• Antisense Technologies
Antisense technologies disrupt biological processes by using oligonucleotides or analogs, frequently preventing the expression of certain genes. They may be used to treat several illnesses, such as cancer, genetic abnormalities, and bacterial and viral infections. Antisense technologies are not yet widely used in prokaryotes, though. Antisense chemicals used as adjuvants with antibiotics are the subject of some research because they can diminish or quiet the genes that cause antibiotic resistance. Steric hindrance and target mRNA cleavage are two important inhibition methods [13].
Future Directions
Antibiotic development in the future necessitates a thorough and multidimensional strategy that tackles the financial, legal, and scientific obstacles while encouraging creativity and cooperation. Drug development can be completely transformed by investing in cutting-edge technology like:
Phage Therapy and Microbiome Research
Offering an alternative to traditional antibiotics, bacteriophages (viruses that attack bacteria) and microbiome-based treatments allow us to fight dangerous infections more safely.
Precision Medicine
Using tailored treatments derived from molecular and genetic data makes it possible to target infections specifically while reducing unnecessary antibiotic usage.
Nanotechnology-Based Therapies
The usage of antimicrobial peptides and nanoparticles may prove to be an effective way to kill resistant bacteria without harming healthy cells.
CRISPR and Gene Editing
Restoring effectiveness of antibiotics can be achieved by neutralizing the resistance mechanisms in bacteria through modification of their genes with CRISPR technology.
Improved Surveillance and Quick Diagnostics
Faster and more precise diagnostics paired with AI tools will detect and target resistant infections in real time, allowing faster analysis and treatment selection.
Antibiotics and Alternatives
AI will assist in the creation of new antibiotics and alternative therapies by analyzing large datasets to accurately predict drug candidates that are likely to be successful.
Global Governance Control and Stewardship
Prohibition, enhanced governance and collaboration around AMR will prove useful in controlling resistant antibiotics alongside efforts in fostering responsible antibiotic usage.
A long-term strategic perspective is necessary to guarantee sustainable innovation in the development of antibiotics. Important initiatives include creating sustainable business models that separate profit from sales volume, promoting fair access to antibiotics, and giving antimicrobial resistance top priority in global health agendas. The healthcare community can secure a strong pipeline of innovative antibiotics and lessen the growing threat of antimicrobial resistance for future generations by coordinating scientific achievements with legislative reforms and international cooperation.
Conclusion
Beyond drug development, the responsible use of antibiotics in healthcare and agriculture is crucial to slowing the spread of resistance. Strengthening global surveillance systems, improving infection prevention measures, and enhancing public awareness campaigns are all necessary steps in preserving the efficacy of existing antibiotics. International collaboration will be key, as AMR is a cross-border issue requiring coordinated action among nations.
Ultimately, ensuring a sustainable pipeline of effective antibiotics will require a long-term commitment from governments, the pharmaceutical industry, and the scientific community. By fostering innovation, revising economic models, and implementing robust stewardship programs, we can mitigate the growing threat of antimicrobial resistance and safeguard the future of infectious disease treatment. The fight against AMR is complex, but with sustained global efforts, a balance between innovation and responsible antibiotic use can be achieved, securing the health and well-being of future generations.
The development of new antibiotics remains an urgent global priority, particularly in the face of rising antimicrobial resistance (AMR), which threatens to undermine decades of progress in modern medicine. Even with all the progress in developing new antibiotics, there are still some big challenges that need to be addressed. One major issue is that most new antibiotics are just improved versions of existing ones, rather than completely new types of drugs. Since bacteria are constantly evolving resistance, we need antibiotics that work in entirely different ways, but research in this area is still limited. More focus should be placed on discovering new ways to target bacteria so that we can stay ahead in the fight against resistance.
Another big challenge is the high cost and long time it takes to develop new antibiotics. It often takes over a decade and billions of dollars to bring a new antibiotic to market, and because antibiotics are typically used for short-term treatments rather than long-term conditions, pharmaceutical companies don’t see them as profitable. This lack of financial incentive slows down innovation. At the same time, the process of getting new antibiotics approved is complicated and time-consuming, even though some policies, like the GAIN Act, aim to help. There needs to be a better system to encourage companies and researchers to invest in antibiotic development.
Alternative treatments, like bacteriophage therapy, antimicrobial peptides, and metal nanoparticles, have shown a lot of promise, but they haven’t been widely adopted. The main reasons are a lack of large-scale clinical trials, strict regulations, and challenges in manufacturing these treatments. If more research and funding were directed toward these alternatives, they could provide new ways to treat infections, especially those caused by drug-resistant bacteria.
Access to antibiotics is another major concern. Even when new antibiotics are developed, many people around the world, particularly in low- and middle-income countries, can’t get them due to high costs, lack of proper distribution, and regulatory barriers. This is a serious issue because antibiotic resistance is a global problem, and if some countries can’t access effective treatments, resistant bacteria will continue to spread worldwide. More international collaboration is needed to ensure that life-saving antibiotics are available to everyone, not just in wealthier nations. Finally, we need better programs to track antibiotic resistance and promote responsible antibiotic use. Overuse and misuse of antibiotics both in healthcare and agriculture are major drivers of resistance. While some antibiotic stewardship programs exist, they are not always effective, especially in areas with fewer resources. Strengthening these programs, improving public awareness, and promoting smarter antibiotic use are crucial steps in making sure new antibiotics remain effective for as long as possible.
Solving these challenges will require a mix of scientific breakthroughs, better financial and regulatory policies, and global cooperation. If we don’t take action now, the growing problem of antibiotic resistance could make even common infections much harder to treat. Investing in research, improving access, and ensuring responsible antibiotic use will be key to protecting future generations from untreatable infections [14-25].
References
- Yoneyama, H., & Katsumata, R. (2006). Antibiotic resistance in bacteria and its future for novel antibiotic development. Bioscience, biotechnology, and biochemistry, 70(5), 1060-1075.
- Frieri, M., Kumar, K., & Boutin, A. (2017). Antibiotic resistance. Journal of infection and public health, 10(4), 369- 378.
- Boyd, N. K., Teng, C., & Frei, C. R. (2021). Brief overview of approaches and challenges in new antibiotic development: a focus on drug repurposing. Frontiers in cellular and infection microbiology, 11, 684515.
- Coates, A., Hu, Y., Bax, R., & Page, C. (2002). The future challenges facing the development of new antimicrobial drugs.Nature reviews Drug discovery, 1(11), 895-910.
- Coates, A. R. M., & Hu, Y. (2007). Novel approaches to developing new antibiotics for bacterial infections. British journal of pharmacology, 152(8), 1147-1154.
- Mantravadi, P. K., Kalesh, K. A., Dobson, R. C., Hudson, A. O., & Parthasarathy, A. (2019). The quest for novel antimicrobial compounds: emerging trends in research, development, and technologies. Antibiotics, 8(1), 8.
- Moloney, M. G. (2016). Natural products as a source for novel antibiotics. Trends in pharmacological sciences, 37(8), 689- 701.
- Blaskovich, M. A. (Ed.). (2020). Antibiotics special issue: challenges and opportunities in antibiotic discovery and development. ACS Infectious Diseases, 6(6), 1286-1288.
- Verma, T., Aggarwal, A., Singh, S., Sharma, S., & Sarma,S. J. (2022). Current challenges and advancements towards discovery and resistance of antibiotics. Journal of Molecular Structure, 1248, 131380.
- Vila, Moreno-Morales, J., & Ballesté-Delpierre, C. (2020). Current landscape in the discovery of novel antibacterial agents. Clinical Microbiology and Infection, 26(5), 596-603.
- León-Buitimea, A., Garza-Cárdenas, C. R., Garza-Cervantes,J. A., Lerma-Escalera, J. A., & Morones-Ramírez, J. R. (2020). The demand for new antibiotics: antimicrobial peptides, nanoparticles, and combinatorial therapies as future strategies in antibacterial agent design. Frontiers in microbiology, 11, 1669.
- Hotinger, J. A., Morris, S. T., & May, A. E. (2021). The case against antibiotics and for anti-virulence therapeutics. Microorganisms, 9(10), 2049.
- Jani, S., Ramirez, M. S., & Tolmasky, M. E. (2021). Silencing antibiotic resistance with antisense oligonucleotides. Biomed- icines, 9(4), 416.
- Davies, J., & Davies, D. (2010). Origins and evolution of antibiotic resistance. Microbiology and molecular biology reviews, 74(3), 417-433.
- World Health Organization (WHO). (2019). Antimicrobial resistance: Global report on surveillance 2014.
- Outterson, K., Powers, J. H., & Rex, J. H. (2015). "A New Business Model for Antibiotics." Science Translational Medicine, 7(317), 317ps318.
- Laxminarayan, R., Duse, A., Wattal, C., Zaidi, A. K., Wertheim, H. F., Sumpradit, N., ... & Cars, O. (2013). Antibiotic resistance—the need for global solutions. The Lancet infectious diseases, 13(12), 1057-1098.
- Boucher, H. W., & Talbot, G. H. (2009). "The global challenge of antibiotic resistance." Infectious Disease Clinics of North America, 23(4), 1-20.
- Zhang, L., & Fong, S. K. (2017). Novel Antibacterial Agents: Challenges and Opportunities in Drug Discovery. Frontiers in Microbiology, 8, 1310.
- Shlaes, D. M., & Gibbons, J. P. (2010). "Regulatory Issues in the Development of Antibiotics." Clinical Infectious Diseases, 50(7), 920-926.
- Founou, L. L., Founou, R. C., & Ndip, R. N. (2018). Antibiotic resistance in human pathogens: A review. Journal of Infection and Public Health, 11(2), 220-230.
- World Health Organization (WHO). (2017). Antimicrobial Stewardship Programmes in Health Care Facilities in Low- and Middle-Income Countries: A WHO Practical Toolkit.
- O'Neill, J. (2016). Tackling drug-resistant infections globally:final report and recommendations.
- Hay, S. I., Abajobir, A. A., Abate, K. H., et al. (2017). "Global and national morbidity and mortality for 332 diseases and injuries in 195 countries and territories, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015." The Lancet, 388(10053), 1545-1602.
- Anis-Wlodarczyk, K. M., Wozniak, D. J., & Abedon, S. T. (2021). Treating bacterial infections with bacteriophage- based enzybiotics: in vitro, in vivo and clinical application. Antibiotics, 10(12), 1497.



