Research Article - (2024) Volume 4, Issue 1
Research on Complex Processing of Ash and Slag at Angren Tpp
Received Date: Jun 22, 2024 / Accepted Date: Jul 31, 2024 / Published Date: Aug 07, 2024
Copyright: ©©2024 Asadjon Kambarov, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Kambarov, A., Sevara, K., Rano, T., Nilufar, A., Nigora, A., et al. (2024). Research on Complex Processing of Ash and Slag at Angren Tpp. J Oil Gas Res Rev, 4(1), 01-08.
Abstract
All over the world, at this stage, the development of technology and technology requires more and more metal-containing products. The depletion of land resources today forces scientists around the world to look for other, alternative sources for extracting metals. Technogenic waste from metallurgical and chemical enterprises is of considerable interest. Recently, the most pressing issues related to improving the environmental situation not only in Uzbekistan, but also beyond its borders, are of a technogenic nature. Over the many years of operation of the Angren and Novo-Angren thermal power plants, a huge amount of ash and slag waste has been generated, which requires close attention for the purpose of disposal or processing to improve the environmental situation in the country and solve production problems with the production of a certain type of raw materials.
Keywords
Ash and Slag, Ammonium Fluoride, Silicon Dioxide, Iron
Thermal power plants do not lag behind other technogenic waste in terms of their chemical composition, since they contain both non-ferrous and rare metals. Research carried out in some foreign countries makes it possible to completely recycle ASW, but in our country the problem of their recycling has not yet been solved. The main objective of this work is to begin the comprehensive processing of ammonia waste from the Angren thermal power plant using halogen-ammonium technology. This technology is used for the first time in the processing of ASW. This article presents the results of research on the extraction of silicon and iron from ash and slag waste from the Angren thermal power plant. The authors have developed a technology for producing silicon dioxide from the ashes of the Angren thermal power plant using ammonium fluoride, which, in terms of its technical characteristics, is the best analogue in foreign countries. The granulometric characteristics of the resulting product were studied using modern instruments.
Introduction
Ash and slag waste can be classified as technogenic mineral raw materials, generated in large quantities at thermal power plants, which accumulate over time, but are not depleted. Among industrial wastes, ash and slag wastes occupy one of the first places in terms of volume from the combustion of solid fuels (various types of coal, oil shale, peat) at thermal power plants (TPPs). The annual production volume of ash and slag at many thermal power plants exceeds 1 million tons, and at stations the combustion of ash fuel reaches 5 million cubic meters. m. Waste adversely affects the environment due to the composition of heavy metals and toxic substances in it, and also occupy large areas, generate dust, and pollute groundwater. Recently, industrial emissions of industrial waste have become more of an economic problem; its comprehensive processing is advisable. The development of technologies for the use of industrial ash waste in order to obtain valuable components makes it possible to solve the problems of economic and environmental problems of many industries [1,2].
The use of ash and slag waste for industrial purposes in developed countries is more than 60%. According to research by a number of scientists, ash and slag contains about 50 types of metals. This figure shows the production of ASW in the world in 2012.
Figure 1: Production of Ash and Slag Waste in The World in Million Tons For 2012
In foreign countries, much attention is paid to the problem of processing ash and slag waste. For example in Germany and France this figure is 70%, and in Finland - about 90%, which allows most thermal power plants to utilize ASW [3]. Special associations such as the American Coil Council (USA), Asian Coal Ash Association (China, Indonesia, Australia), European Coal Combustion Products Association (it includes 28 energy companies from 15 countries).
In the USA, 38% of the ash produced by power plants is used only in the production of mortars and concrete, in the Czech Republic and Slovakia 75% Products made from cellular concrete are made from ash, in Poland - more than 50%. Ashes from thermal power plants are widely used abroad in the production of fired ceramic bricks and artificial porous aggregates. One of the promising areas for processing ash and slag waste is the extraction of useful metals from them. Today, this method of producing aluminum from ash is actively used in China.
The annual increase in ash waste in ash dumps in the Primorsky Territory is 2.5-3.0 million tons per year, in the Khabarovsk Territory - up to 1.0 million tons. In the vicinity of Khabarovsk, about 16 million tons of ash and cinders have been accumulated [4]. In Uzbekistan, more than 13 million tons of ash waste, which needs disposal, have currently been accumulated in the dumps of the Angren thermal power plant. In the future, extracting useful components from ashes can provide economic and environmental benefits and reduce the negative impact on the environment.
Currently, there are two main directions for processing ash and slag waste, the first of which is the extraction of metals, the other is the recycling of waste for its further use. Technologies for processing ash and slag waste are constantly developing, new ways of using them are being explored, which makes this task increasingly relevant in the modern world. The main objective of this work is to begin the comprehensive processing of ammonia waste from the Angren thermal power plant using halogen-ammonium technology. This technology is used for the first time in the processing of ASW.
Description of the Object and Research Methods
We used the ashes of the Angren thermal power plant as source materials. From thermal coal combustion wastes such as ash and slag waste, they are a valuable technogenic raw material not only for the construction industry, but can also serve as a source for extracting valuable impurity elements from coal. A spectral analysis of the ASW of the Angren thermal power plant was carried out. The results are shown in Table 1 and Fig. 1.
The results showed the presence of 34 elements. Predominantly contains silicon, aluminum and iron. Non-ferrous metals, copper, zinc, cobalt, cadmium, rare elements such as molybdenum, tungsten, yttrium, titanium, etc. are present.
|
Component |
Result |
Unit |
Stat.Err |
LLD |
LLQ |
|
|
1 |
Cl |
0.0131 |
mass % |
0.0002 |
0.0002 |
0.0006 |
|
2 |
Ra |
(0.0007) |
mass % |
0.0002 |
0.0005 |
0.0014 |
|
3 |
Mg |
1.75 |
mass % |
0.0170 |
0.0238 |
0.0714 |
|
4 |
Al |
11.8 |
mass % |
0.0179 |
0.0066 |
0.0199 |
|
5 |
Si |
48 |
mass % |
0.0154 |
0.0043 |
0.0128 |
|
6 |
S |
0.480 |
mass % |
0.0013 |
0.0012 |
0.0037 |
|
7 |
K |
1.03 |
mass % |
0.0072 |
0.0055 |
0.0164 |
|
8 |
Ca |
8.35 |
mass % |
0.0166 |
0.0054 |
0.0163 |
|
9 |
Ti |
0.506 |
mass % |
0.0029 |
0.0033 |
0.0100 |
|
10 |
V |
0.0207 |
mass % |
0.0010 |
0.0027 |
0.0081 |
|
11 |
Cr |
(0.0015) |
mass % |
0.0003 |
0.0009 |
0.0027 |
|
12 |
Mn |
0.164 |
mass % |
0.0013 |
0.0023 |
0.0069 |
|
13 |
Fe |
4.32 |
mass % |
0.0023 |
0.0028 |
0.0083 |
|
14 |
Co |
(0.0112) |
mass % |
0.0012 |
0.0041 |
0.0122 |
|
15 |
Cu |
0.0064 |
mass % |
0.0002 |
0.0003 |
0.0008 |
|
16 |
Zn |
0.305 |
mass % |
0.0010 |
0.0003 |
0.0008 |
|
17 |
Ga |
0.0028 |
mass % |
0.0001 |
0.0003 |
0.0009 |
|
18 |
Ge |
0.0027 |
mass % |
0.0001 |
0.0002 |
0.0007 |
|
19 |
As |
0.0214 |
mass % |
0.0003 |
0.0007 |
0.0022 |
|
20 |
Rb |
0.0115 |
mass % |
0.0001 |
<0.0001 |
0.0002 |
|
21 |
Sr |
0.119 |
mass % |
0.0003 |
0.0002 |
0.0006 |
|
22 |
Y |
0.0052 |
mass % |
<0.0001 |
0.0002 |
0.0005 |
|
23 |
Zr |
0.165 |
mass % |
0.0010 |
0.0005 |
0.0016 |
|
24 |
Nb |
0.0020 |
mass % |
0.0002 |
0.0004 |
0.0012 |
|
25 |
Mo |
0.0058 |
mass % |
0.0007 |
0.0011 |
0.0033 |
|
26 |
Ag |
0.0008 |
mass % |
<0.0001 |
0.0002 |
0.0005 |
|
27 |
Cd |
0.0012 |
mass % |
0.0001 |
0.0002 |
0.0006 |
|
28 |
Sn |
0.0014 |
mass % |
0.0001 |
0.0003 |
0.0006 |
|
29 |
Ba |
0.162 |
mass % |
0.0015 |
0.0009 |
0.0028 |
|
30 |
W |
0.0265 |
mass % |
0.0006 |
0.0013 |
0.0040 |
|
31 |
Ir |
0.0033 |
mass % |
0.0004 |
0.0010 |
0.0029 |
|
32 |
Pt |
0.0019 |
mass % |
0.0002 |
0.0005 |
0.0016 |
|
33 |
Pb |
0.0773 |
mass % |
0.0004 |
0.0003 |
0.0010 |
|
34 |
Eu |
0.0869 |
mass % |
0.0038 |
0.0122 |
0.0367 |
Table 1: Results of the Analysis of the Ashes of the Angren TPP
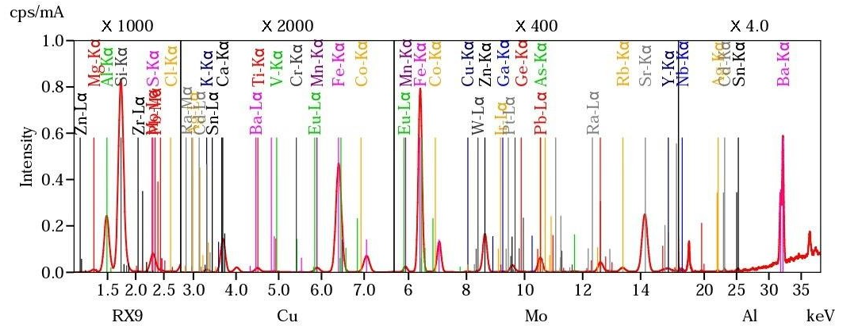
Figure 2: Spectral Analysis of the Ashes of the Angren Thermal Power Plant
Research Methods
Transmission (TEM) and scanning electron microscopes (SEM) were used to study the morphology and size of particles. Electron microscopic studies of the ashes were carried out using a scanning electron microscope (SEM). The information obtained is presented in Figures 1 and 2 demonstrating the morphological features of the sample. In the overview drawings of the ash, we see that in the structure of the sample there is a wide fraction of spherical melted particles with a size of 10 - 20 microns or less. The surface relief of the particles has a wide degree of roughness and porous structure
Figure 3: Overview photos of ash

Figure 4: Overview photos of ash
Since highly dispersed particles tend to stick to each other and form agglomerates, to accurately determine the particle sizes of the synthesized SiO2 powders and their size distribution, a technique based on the creation of an aqueous nanosuspension and its subsequent transillumination using a laser analyzer was used.
To do this, first, agglomerates of synthesized silica particles were dispersed by ultrasonic waves with a frequency of 40 kHz in an aqueous medium, and then the sizes of individual particles were measured.
A Nano Sight LM10 laser analyzer (Malvern Analytical) was used to study the distribution of SiO2 particles in an aqueous nanosuspension. The temperature of the aqueous solution was +30 °C, and the concentration of particles in the solution was 1.27•109 particles/ml. The qualitative and quantitative composition of ash components depends on the coal deposit. The composition of coal components can be considered relatively constant for macro components, while for micro components it is not constant even within the same seam. The chemical properties of ash components, as well as their ability to concentrate during coal combustion, are determined by their phase composition. The phase composition of compounds depends primarily on the temperature conditions of coal combustion and the composition of the gas phase of the atmosphere in which the compounds are formed.
Research Methodology
Silicon Dioxide Separation
Progress of Work
We propose to develop a technology for processing ash and slag waste from the Angren thermal power plant using fluoride (NH4F) or bifluoride ammonium (NH4F*HF) salt for desiliconization of cakes, which occurs according to the following reaction: When processed in the regeneration of ammonium fluoride, it turns into ammonium bifluoride. Ammonium bifluoride reacts with silicon dioxide according to the reaction:
SiO2+6NH4F=(NH4)2+SiF6+4NH3+2H2O (1)
The ammonium hexafluorosilicate formed as a result of the reaction transforms into a gaseous state when heated. Gaseous ammonium hexafluorosilicate sublimates at a temperature of 340-370°C and is subject to ammonia precipitation according to the reaction:
SiO2+6NH4F=(NH4)2+SiF6+4NH3+2H2O (1)
The ammonium hexafluorosilicate formed as a result of the reaction transforms into a gaseous state when heated. Gaseous ammonium hexafluorosilicate sublimates at a temperature of 340-370°C and is subject to ammonia precipitation according to the reaction:
(NH4)2SiF6+4NH4OH= SiO2+6NH4F+2H2O (3)
The ammonium fluoride formed as a result of the reaction is evaporated and crystallizes in the form of ammonium bifluoride. Silicon dioxide is released in finely dispersed form. The theoretical foundations, experimental results and some proposals for the possibility of implementing ammonium fluoride technology for the production of silicon dioxide are examined below.
A stoichiometric sample of ammonium fluoride and tailings weighing 30 g was placed in a furnace, the installation was sealed and, after checking for leaks, it was heated in the furnace to a certain temperature and held for a specified time. After removal from the oven, the glass with the remaining product was weighed. The degree of desublimation was determined by the mass of the residue (Table 1).
|
Name |
EXPERIMENTS |
||||
|
1 |
2 |
3 |
4 |
5 |
|
|
(NH4)2SiF6, g |
48 |
47 |
47 |
48 |
48 |
|
Remaining, g |
14,5 |
14 |
14.5 |
14 |
14 |
Table 2: Results of Separation of Ammonium Hexaphtrosilicate
Studies of the desublimation process show that 95-96% of the evaporated product condensed in the cooled zone of the apparatus. Thus, there is no loss of substance during the process of sublimation-desublimation. This confirms the data of works (2,3,4) that ammonium hexafluorosilicate (AFSA) does not decompose into gas components during sublimation, since, otherwise, the decomposition of HFSA would irreversibly lead to the loss of part of the substance.
From the given experimental Data, it is clear that the temperature of 3700 C is the most favorable for the formation of a volatile product - within 45 minutes the yield is more than 98%. Determination of the degree of ammonium fluoride regeneration as stated above, ammonium fluoride is regenerated according to reaction (2). The data obtained during the experiments are given in Table 2.
|
Indicator name |
Values for brands |
Received sample |
||
|
Newsil 115 |
News il 125 |
Newsil 175 |
||
|
1.Silicon oxide content in in terms of dry matter, % |
≥ 98 |
≥ 98 |
≥ 98 |
99.6 |
|
2. Appearance |
White powder |
|||
|
3.Screening on a 45 micron sieve, % |
≤ 0,5 |
|
|
≤ 0,5 |
|
4.Drying loss (within 2 hours at 105°C), % |
4,0-8,0 |
|
|
4,0-8,0 |
|
5.loss on ignition (in for 2 hours at 1000 °C),% |
≤ 7,0 |
|
|
≤ 7,0 |
|
6. pH |
6,0-7,5 |
|
|
6,0-7,5 |
|
7. Absorption value DBP, |
|
|
|
|
|
cm3/100g |
200-350 |
|
|
200-350 |
|
|
|
|
|
|
|
8.Specific surface area BET, m2/g |
100-120 |
100-140 |
165-185 |
100-120 |
|
9.Na2SO4, % |
≤ 1,60 |
|
|
not detect. |
|
10.Cu, mg/kg |
≤30 |
|
|
not detect. |
|
11.Fe, mg/kg |
≤ 1000 |
|
|
80 |
|
12.Mn, mg/kg |
≤ 50 |
|
|
not detect. |
|
13.Ni,mg/kg≥ |
|
|
|
20 |
|
14. Density, g/l |
≤ 200 |
|
|
≥ 200 |
Table 3: Comparative Data of the Obtained Silicon Oxide with Industrial Samples
According to the data in Table 3, the silicon dioxide we obtained is purer and lower in impurity content. In all other respects it is better.
Nanopowder Research SiO2
In Fig. Figure 2 shows an SEM image of the synthesized SiO2 powders. It can be seen that the majority of silica particles form agglomerates by binding several individual particles. Particle agglomeration can occur due to both van der Waals attractions and Coulomb or other relatively weak interactions. In this case, the original particles retain their shape and size.
Figure 6: SEM image of synthesized SiO2 powders.
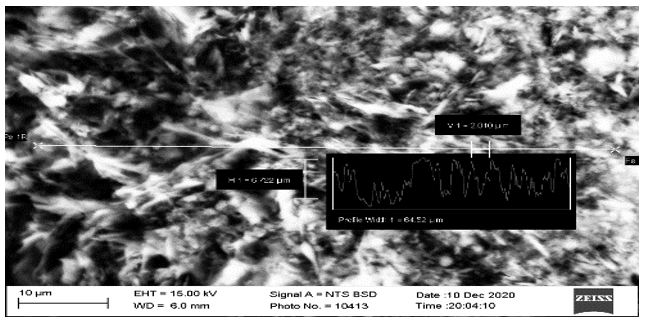
Figure 5: Powder Surface Morphology Showing the Porous-Fibrous Structure of the Sio2 Particle Agglomerate.
In Figure 5, in an area measuring 64.5 x 6.7 µm, you can see the morphology of the surface of the powders in the form of a developed porous-fibrous structure and notice a change in the contrast of the surface relief due to the heterogeneity of the agglomerate structure. Therefore, we measured the sizes of individual particles after dispersing the agglomerates with ultrasonic waves.
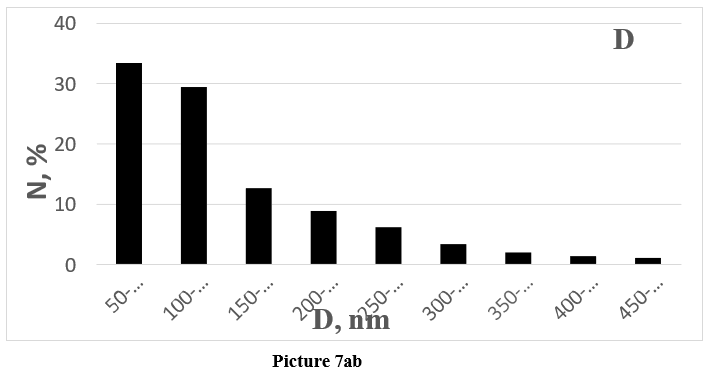
Picture 7: Size distribution of synthesized amorphous SiO2 nanoparticles and corresponding histograms of the distribution of the number (N) of synthesized particles by size.
Separation of Iron from Ash and Slag After Separation of Silicon Oxide Progress
After separating silicon oxide from ash and slag using the halogen-ammonium method, iron was separated by magnetic separation. It should be noted that the iron content in the non-magnetic part is significant. To increase the extraction of iron from the non-magnetic part of the collective concentrate, a technique has been developed for converting non-magnetic iron oxide into a magnetic form using radiation treatment of the material with a pulsed infrared emitter based on functional ceramics, synthesized at the Institute of Materials Science on a large Solar Furnace (Parkent). In this case, in total, more than 88% of the iron turns into magnetic iron oxide. Table 3 shows the results of the experiments.
|
Name |
Magnetic part, g |
Non-magnetic part. G |
Total |
Exit, % |
|
|
1 |
Direct magnetic separation |
7 |
_ |
7 |
58 |
|
2 |
Processed IIG |
_ |
3.6 |
3,6 |
30 |
|
3 |
Sum |
7 |
3,6 |
10,6 |
88 |
Table 4: Results of Magnetic Separation of Ash and Slag
Submitted for analysis. Table 4 shows the content of elements in the magnetic fraction.
|
Name |
Si |
Fe |
Al |
Cu |
Zn |
Ni |
Sn |
Na |
Mo |
W |
|
Original |
48,8 |
4,32 |
11,8 |
0,0064 |
0,305 |
- |
0,0014 |
- |
0,0058 |
0,0265 |
|
Magn. tailcoat |
12 |
53 |
6,9 |
4 |
4 |
- |
0,3 |
- |
3,2 |
0,3 |
Table 5: Content of Elements in the Magnetic Fraction of Asw %
To obtain purer iron, the magnetic fraction was dissolved in an 8 N solution of nitric acid. Iron was precipitated with a 12% am- monia solution. The precipitate was filtered, dried and calcined at a temperature of 900-1100 °C.
|
Component name |
Content, % |
Content, g/kg (magnetic concentrate) |
|
|
1 |
Fe3O4 |
97,16 |
971,6 |
|
2 |
Cr2O3 |
0,17 |
1,70 |
|
3 |
MnO |
0,34 |
3,40 |
|
4 |
VO2 |
0,33 |
3,30 |
Table 6: Lcontent Of Some Elements in Purified Magnetic Concentrate
Conclusion
It was shown for the first time that silicon dioxide was produced from the ashes of the Angren thermal power plant using ammo¬nium fluoride, which, in terms of its technical characteristics, is the best analogue in foreign countries. Its granulometric charac¬teristics were studied using modern instruments [5-9]. The pro¬posed technology for processing ash and slag from the Angren TPP is as follows: at the first stage, silicon dioxide is removed, which includes:
• opening of ash and slag at 170 °C with ammonium fluoride with the formation of fluoroammonium compounds and impurity metals.
• sublimation separation of silicon hexafluorosildicate (HFSA) at 350-370 °C.
• hydrolysis of HFSA with ammonia water to obtain silicon di-oxide.
• filtering SiO2 from the pulp, drying and calcining it to obtain the finished product in the form of powder.
• distillation of ammonia mother liquors, crystallization of ammonium fluoride. The second stage of processing enriched cakes, separating iron by magnetic separation.
• A laboratory technological scheme has been developed for pro-cessing ashes from the Angren thermal power plant to obtain valuable components such as iron and silicon.
References
- Sheludyakov, L. N., Kosyanov, E. A., & Yu., A. (1985). Complex processing of silicate waste Markonrenkov. Alma-Ata: Nauka, 123.
- Abdulloev, M. M. (2000). Physicochemical studies of aluminum production waste and development of their processing technology: author's abstract. diss. candidate of technical sciences, Dushanbe, 22.
- Leonov, S. B., Fedotov, K. V., & Senchenko, A. E. (1998). Industrial gold mining from ash and slag dumps of thermal power plants. Mining magazine, 5, 67-68.
- Arbuzov, S. I., Ershov, Rare, V. V., & Potseluev, A. L. (1999). Elements in coals of the Kuznetsk basin. Kemero-vo, 248.
- Kenzhaeva, S. A., Toshkodirova R. E. Study on the extraction of valuable components from zinc production clinker.
- Ernazarov, M., Askarova, N., Kenjayeva, S., Toshkodirova, R., & Toshpulatov, A., et al. (2024). Study of the complex processing of ash and slag at Angren Thermal Power Plant. E3S Web Conf., 498, 01010.
- Toshkodirova, R. E., & Kenzhaeva, S. A. (2023). STUDY AND RESEARCH OF ZINC PRODUCTION CLINKER PROCESSING TECHNOLOGIES. Journal of Advances in Engineering Technology, (2), 87-91.
- Kenzhaeva, S. A., & Toshkodirova, R. E. (2022). A Study on The Extraction of Valuable Components From Zinc Production Clinker. Oriental renaissance: Innovative, educational, natural and social sciences, 2 (5), 616-622.
- Muhammad-Tohir, E., & Sevara Absamatovna, K. (2024). A study on the processing of ash and slag of the angren tpp. Sanoatda raqamli texnologiyalar Digital technologies in industry, 2 (2), 25-30.



