Research Article - (2022) Volume 7, Issue 1
Relatedness of California Mastitis Test Scores and Bacteriological Plate Counts in the diagnosis of Subclinical Mastitis in white Fulani cattle from selected dairy herds
Received Date: Feb 19, 2022 / Accepted Date: Feb 26, 2022 / Published Date: Mar 14, 2022
Copyright: ©Hussaini I M. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Suleiman, A.B., Hussaini, I.M., Anchau, Z.G. (2022). Relatedness of California Mastitis Test Scores and Bacteriological Plate Counts in the diagnosis of Subclinical Mastitis in white Fulani cattle from selected dairy herds. Adv Nutr Food Sci, 7(1), 182-188.
Abstract
Background: Subclinical mastitis remains a major challenge in the dairy industries as it affects nutritional content of milk and milk products, volume of milk production and also leads to economic loss. This study was carried out to determine the relationship between California Mastitis Test (CMT) scores and bacteria counts in the diagnosis of subclinical mastitis. A total of 339 white Fulani cattle milk samples were aseptically collected from six geographical regions of Plateau State and screened for mastitis and bacteriological analysis using the California Mastitis Test (CMT), Total Bacterial Count (TBC) and Total Coliform Count (TCC) methods respectively.
Results: The result of the study reveals that 31.0% of the milk samples were positive for mastitis. Based on the CMT scores, 61.9% of the samples were CMT negative, 7.1% were CMT trace while 6.5%, 11.5% and 13.0% were CMT +, CMT ++ and CMT +++ respectively. Most of the CMT negative samples (55.71%) were culture negative while almost all the CMT +++, were positive (90.91%) culture positive. A positive correlation was observed between CMT scores and plate counts (Total Bacteria count and Total Coliform Count). The Total Bacteria count ranged from 5.7x101 – 5.9x106 CFU/mL while the Total Coliform Count ranged from 0.6x101 – 3.0x105.CMT scores correlates well with bacteriological plate count of the mastitis milk samples.
Conclusion: There is a need for the improvement in quality and safety of dairy product because of public health risk associated with it.
Keywords
Milk, Mastitis, Bacteriological, Count, CMT Scores
Introduction
Mastitis is an important disease of public health importance due to the consumption of raw or improperly pasteurized milk. Mastitis is an important disease of great economic importance in diary in-dustry due to intramammary infection (IMI) which results into re¬duced secretory capacity of the mammary gland [1, 2]. In general, mastitis is caused by bacteria pathogens such as Staphylococcus aureus, Streptococcus agalactiae, environmental streptococci and coliforms leading to direct milk contamination. Cows suffering from systemic disease could also secrete pathogenic bacteria such as Mycobacterium sp., Brucella sp. and Bacillus sp. Majority of the cattle herds are not under veterinary supervision without ad¬equate disease and health programs especially without mastistis screening. Both clinical and subclinical mastistis in dairy cows in Nigeria have been reported by several authors [3-5]. Also, unregu¬lated administration of antibiotics to cattle by the herdsmen could result in their residues that is also detrimental to milk quality and dairy technology [2, 6].
There is rising demand for milk and other dairy products in de-veloping countries including Nigeria. Milk is an excellent source of high quality protein diet and also containing a lot of essential vitamins and minerals which can be easily digested and absorbed by all mammals especially the infants. Milk and milk products are major global diets providing about 5-10% of the total calo¬ries of the daily human diet in several countries and are the best natural sources of essential amino acids, calcium and phosphorus. These nutrients are essential role in body building, especially the bones and teeth development [7]. These nutritional attributes have made milk an acceptable and staple global diet of both infants, and adults [2].
The largest proportion of milk produced for human consumption is obtained from cattle. Dairy cows efficiently convert human-in-edible food and by-products into nutrient-rich milk [8]. The recent dairy farming improvement efforts in Nigeria through public-pri-vate partnership scheme comprising collection of milk from differ-ent cattle herds owned by nomadic and sedentary pastoralists. This Nigerian Dairy Development Program (NDDP) is geared towards improving the livelihoods of smallholder dairy farmers and im¬proving cattle productivity by integration traditional milk source into the formal dairy value chain in Nigeria [2].
Subclinical mastitis is a major disease affecting the dairy industry [9-10]. It causes economic losses by decreasing milk production, costs of therapy, and unused milk during the withdrawal period. Clinical mastitis can be detected by the farmer, but subclinical mastitis can only be detected by the measurement of inflammatory components and pathogens in the milk [11]. Since somatic cell count (SCC) in milk has been shown to be an excellent marker for subclinical mastitis, the use of SCC is inevitable for specifying the reduction of the milk yield [10]. SCC is directly related to the CMT scores as follows: Somatic cell ranges of 0 – 200,000 cell/ mL, 200,000 – 400,000 cell/mL, 400,000 – 1,200,000 cell/mL, 1,200,000 – 5,000,000 cell/mL and Over 5,000,000 cell/mL are equivalent to CMT scores Negative, Trace, CMT +, CMT ++ and CMT +++ respectively [12].
The contents and nutritional values of milk, which are important for human nutrition, may be changed with a systemic or mamma¬ry gland infection of host animals. High bacterial contamination in milk reduces the quality of both milk and dairy products, af¬fects milk shelf life and flavor, and deteriorates the physicochem-ical properties of milk. Mastitic milk contains both pathogens and bacterial toxins and its consumption may directly or indirectly in¬crease the risk of foodborne illnesses [10].
Different methods have been suggested for detection of subclin-ical mastitis, such as the California mastitis test (CMT), SCC, some biochemical methods, the presence of pathogens in the milk, and electrical conductivity (EC) [10]. The husbandry and milk¬ing practice by the local dairy producers could result in poor milk quality, since milk is an excellent growth medium for both endog-enous and environmental pathogenic and non-pathogenic bacte-ria [13]. Microbial growth and multiplication in milk result from pre-harvest intramammary infections as well as milking and post milking environmental [12] contamination with deleterious effects on quality, wholesomeness and safety of milk and milk products. Safe and high quality milk can only be obtained from mastitis free and healthy animals. Also, herd hygiene, management and milk¬ing processes influence the microbial quality of cow manure milk while cows suffering from a disease could excrete the pathogenic bacteria in the udder with the milk, thereby predisposing consum¬ers to food/milk borne disease. Milk borne outbreaks represent 2-6% of bacterial food-borne outbreaks reported by surveillance systems from several countries [2].
The unhygienic hand milking in the open filed with the milk pooled from unscreened cows collected and stored in local cala-bash guards commonly practiced by herdsmen in Nigeria portent food safety risks from such products. Private-Public- Partnership efforts of Nigeria government on dairy development aim at im¬provement of yield, safety and quality milk produced by indige¬nous cows. Improvement of traditional and nomadic milk produc¬tion through dairy development programs in Nigeria will therefore require routine quality and safety monitoring of milk parameters such as CMT, SCC, total aerobic plate and coliform counts both at herd level and milk collection centers [2]. Therefore, this study was aimed at elucidating the relatedness of CMT scores and bac¬terial presence in mastitis milk for safety of consumption of milk and the overall health of the animal.
Introduction
Mastitis is an important disease of public health importance due to the consumption of raw or improperly pasteurized milk. Mastitis is an important disease of great economic importance in diary in-dustry due to intramammary infection (IMI) which results into re¬duced secretory capacity of the mammary gland [1, 2]. In general, mastitis is caused by bacteria pathogens such as Staphylococcus aureus, Streptococcus agalactiae, environmental streptococci and coliforms leading to direct milk contamination. Cows suffering from systemic disease could also secrete pathogenic bacteria such as Mycobacterium sp., Brucella sp. and Bacillus sp. Majority of the cattle herds are not under veterinary supervision without ad¬equate disease and health programs especially without mastistis screening. Both clinical and subclinical mastistis in dairy cows in Nigeria have been reported by several authors [3-5]. Also, unregu¬lated administration of antibiotics to cattle by the herdsmen could result in their residues that is also detrimental to milk quality and dairy technology [2, 6].
There is rising demand for milk and other dairy products in de-veloping countries including Nigeria. Milk is an excellent source of high quality protein diet and also containing a lot of essential vitamins and minerals which can be easily digested and absorbed by all mammals especially the infants. Milk and milk products are major global diets providing about 5-10% of the total calo¬ries of the daily human diet in several countries and are the best natural sources of essential amino acids, calcium and phosphorus. These nutrients are essential role in body building, especially the bones and teeth development [7]. These nutritional attributes have made milk an acceptable and staple global diet of both infants, and adults [2].
The largest proportion of milk produced for human consumption is obtained from cattle. Dairy cows efficiently convert human-in-edible food and by-products into nutrient-rich milk [8]. The recent dairy farming improvement efforts in Nigeria through public-pri-vate partnership scheme comprising collection of milk from differ-ent cattle herds owned by nomadic and sedentary pastoralists. This Nigerian Dairy Development Program (NDDP) is geared towards improving the livelihoods of smallholder dairy farmers and im¬proving cattle productivity by integration traditional milk source into the formal dairy value chain in Nigeria [2].
Subclinical mastitis is a major disease affecting the dairy industry [9-10]. It causes economic losses by decreasing milk production, costs of therapy, and unused milk during the withdrawal period. Clinical mastitis can be detected by the farmer, but subclinical mastitis can only be detected by the measurement of inflammatory components and pathogens in the milk [11]. Since somatic cell count (SCC) in milk has been shown to be an excellent marker for subclinical mastitis, the use of SCC is inevitable for specifying the reduction of the milk yield [10]. SCC is directly related to the CMT scores as follows: Somatic cell ranges of 0 – 200,000 cell/ mL, 200,000 – 400,000 cell/mL, 400,000 – 1,200,000 cell/mL, 1,200,000 – 5,000,000 cell/mL and Over 5,000,000 cell/mL are equivalent to CMT scores Negative, Trace, CMT +, CMT ++ and CMT +++ respectively [12].
The contents and nutritional values of milk, which are important for human nutrition, may be changed with a systemic or mamma¬ry gland infection of host animals. High bacterial contamination in milk reduces the quality of both milk and dairy products, af¬fects milk shelf life and flavor, and deteriorates the physicochem-ical properties of milk. Mastitic milk contains both pathogens and bacterial toxins and its consumption may directly or indirectly in¬crease the risk of foodborne illnesses [10].
Different methods have been suggested for detection of subclin-ical mastitis, such as the California mastitis test (CMT), SCC, some biochemical methods, the presence of pathogens in the milk, and electrical conductivity (EC) [10]. The husbandry and milk¬ing practice by the local dairy producers could result in poor milk quality, since milk is an excellent growth medium for both endog-enous and environmental pathogenic and non-pathogenic bacte-ria [13]. Microbial growth and multiplication in milk result from pre-harvest intramammary infections as well as milking and post milking environmental [12] contamination with deleterious effects on quality, wholesomeness and safety of milk and milk products. Safe and high quality milk can only be obtained from mastitis free and healthy animals. Also, herd hygiene, management and milk¬ing processes influence the microbial quality of cow manure milk while cows suffering from a disease could excrete the pathogenic bacteria in the udder with the milk, thereby predisposing consum¬ers to food/milk borne disease. Milk borne outbreaks represent 2-6% of bacterial food-borne outbreaks reported by surveillance systems from several countries [2].
The unhygienic hand milking in the open filed with the milk pooled from unscreened cows collected and stored in local cala-bash guards commonly practiced by herdsmen in Nigeria portent food safety risks from such products. Private-Public- Partnership efforts of Nigeria government on dairy development aim at im¬provement of yield, safety and quality milk produced by indige¬nous cows. Improvement of traditional and nomadic milk produc¬tion through dairy development programs in Nigeria will therefore require routine quality and safety monitoring of milk parameters such as CMT, SCC, total aerobic plate and coliform counts both at herd level and milk collection centers [2]. Therefore, this study was aimed at elucidating the relatedness of CMT scores and bac¬terial presence in mastitis milk for safety of consumption of milk and the overall health of the animal.
Materials and Methods
Study area
This study was conducted in Plateau State, located at the middle belt of Nigeria. Six geographic regions of the state (Jos South, Bar¬kin Ladi, Bassa, Riyon, Jos East and Jos North) were selected for the sampling for this study The area is characterized with heavy cattle rearing and milk production for the region.
Sample collection and analysis
A total of 339 milk samples comprising of (61, 61, 47, 66, 34 and 70 from Jos South, Barkin Ladi, Bassa, Riyon, Jos East and Jos North respectively) were collected aseptically from different dairy herds cattle from the selected regions. Each sample was examined physically for colour, odour, pH reaction and consistency.
California Mastitis Test
The CMT was performed and interpreted as described by Kandeel et al. (2018). Briefly, 2 mL of fresh foremilk sample from each quarter was placed in the appropriate chamber of the CMT plastic paddle and mixed with 2 mL of CMT reagent at ambient tempera¬ture by gently moving the paddle in a circular motion. A change in viscosity indicated an increase in quarter SCC, with the CMT re¬action being visually scored at 45 seconds after adding the reagent. A 5-point scale was used to measure the score of viscosity as fol¬lows: negative, mixture remains liquid with no evidence of forma¬tion of precipitate; trace, a slight precipitate evident which tends to disappear with continued movement of the paddle; CMT +, a distinct precipitate but no tendency toward gel formation; CMT ++, the mixture thickens immediately with some gel formation, and with motion, the mixtures tend to move in toward the center leaving the bottom of the outer edge of the cup exposed, and out again covering the bottom of the cup if the motion stopped; CMT +++, a distinct gel forms which tends to adhere to the bottom of the paddle and a distinct central peak forms during swirling.
Total bacterial count (TBC) and Total coliform count (TCC)
Total bacteria count and Coliform count were determined as pre¬viously described with slight modifications. Briefly, a 1mL milk sample was serially up to 10-5. From this dilution, a 0.1 mL of the diluents was transferred onto Nutrient agar and MacConkey agar for determination of TBC and TCC respectively and was incubated at 30 °C for 48 h [10].
Statistical analysis
The mean TBC and TCC in samples from different regions results were compared using ANOVA.
Results
All the milk samples collected were creamy or whitish in colour, odourless with pH range of between 6.6 and 6.7 and are consis-tency in viscosity. Out of the 339 cow milk samples screened, 105 were found to be positive for mastitis using the CMT screening method, hence giving an overall prevalence of 31.0% (Table 1). The distribution of CMT scores of the cow milk samples is pre¬sented in Table 2. Out of the 339 samples screened, a total of 234 (69.0%) were of presumably good quality. This consist of 210 (61.9%) samples with negative CMT scores and 24 (7.1%) sam¬ples with trace CMT scores. The remaining 105 (31.0%) samples had weak to strong positive CMT scores. Samples collected from dairy farms in Jos North had the highest CMT negative scores while samples from Jos East had the highest CMT positive scores.
Table 3 presents the distribution of CMT scores of bulk cow milk samples collected from dairy farms in Plateau State. For the bulk milk, 56.1% had negative to trace CMT score (presumably good quality). While the remaining 43.9% accounts for the samples with weak to strong positive milk samples.
Correlation between CMT scores and culture results of cow milk samples collected is presented in Table 4 and Figure 1 respectively. Most of the milk samples with negative to trace CMT scores had no growth while most of those with weak to strong positive CMT scores had bacterial growth.
All milk samples with Negative CMT scores and most milk sam¬ples with Trace CMT scores had TBC less than the acceptable limit of 1.0x105 CFU/mL and TCC less than the acceptable limit of 1.0x102 CFU/mL by PMO, 2009. The TCC of samples with weak to strong positive CMT scores were higher than the acceptable lim¬it of 1.0x102 while some samples with weak positive CMT scores had TBC lower than the acceptable limit of 1.0x105. Almost all the samples with strong CMT scores had TBC and TCC higher than the acceptable limits.
Table 1: Prevalence of mastitis in cow by California Mastitis Test (CMT)
|
Location |
Number of samples screened |
Number positive |
Prevalence (%) |
|
Jos South |
61 |
17 |
27.9 |
|
Barkin Ladi |
61 |
18 |
29.5 |
|
Bassa |
47 |
17 |
36.2 |
|
Riyon |
66 |
22 |
33.3 |
|
Jos East |
34 |
13 |
38.2 |
|
Jos North |
70 |
18 |
25.7 |
|
Total |
339 |
105 |
31.0 |
Table 2: Distribution of CMT scores of cow milk samples collected from dairy farms in Plateau State
|
Location |
Negative (%) |
Trace (%) |
CMT + (%) |
CMT ++ (%) |
CMT +++ (%) |
|
Jos South (61) |
33 (54.1) |
11 (18.0) |
4 (6.6) |
10 (16.4) |
3 (4.9) |
|
Barkin Ladi (61) |
39 (63.9) |
4 (6.6) |
5 (8.2) |
10 (16.4) |
3 (4.9) |
|
Bassa (47) |
27 (57.4) |
3 (6.4) |
7 (14.9) |
3 (6.4) |
7 (14.9) |
|
Riyon (66) |
42 (63.6) |
2 (3.0) |
3 (4.6) |
6 (9.1) |
13 (19.7) |
|
Jos East (34) |
18 (52.9) |
3 (8.8) |
0 (0.0) |
6 (17.7) |
7 (20.6) |
|
Jos North (70) |
51 (72.9) |
1 (1.4) |
3 (4.3) |
4 (5.7) |
11 (15.7) |
|
Total (339) |
210 (61.9) |
24 (7.1) |
22 (6.5) |
39 (11.5) |
44 (13.0) |
Table 3: Distribution of CMT scores of bulk cow milk samples collected from dairy farms in Plateau State
|
Location |
Negative (%) |
Trace (%) |
CMT + (%) |
CMT ++ (%) |
CMT +++ (%) |
|
Jos South (6) |
2 (33.3) |
2 (33.3) |
1 (16.7) |
1 (16.7) |
0 (0.0) |
|
Barkin Ladi (9) |
5 (55.6) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
4 (44.4) |
|
Bassa (6) |
2 (33.3) |
0 (0.0) |
2 (33.3) |
2 (33.3) |
0 (33.3) |
|
Riyon (6) |
1 (16.7) |
1 (16.7) |
1 (16.7) |
2 (33.3) |
1 (16.7) |
|
Jos East (4) |
3 (75.0) |
1 (25.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
Jos North (10) |
6 (60.0) |
0 (0.0) |
0 (0.0) |
1 (10.0) |
3 (30.0) |
|
Total (41) |
19 (46.3) |
4 (9.8) |
4 (9.8) |
6 (14.6) |
8 (19.5) |
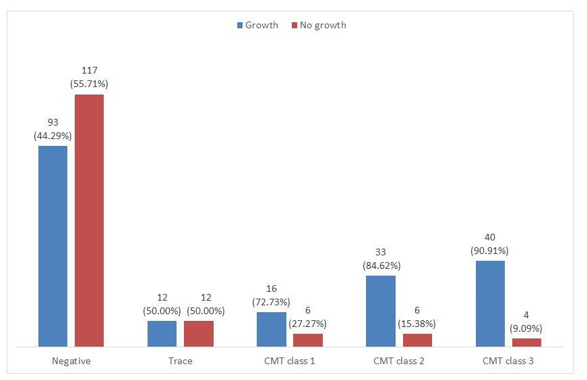
Figure 1: Correlation between CMT scores and culture results of cow milk samples collected from dairy farms in Plateau State
Table 4: Distribution of the correlation between CMT scores and culture results of cow milk samples collected from dairy farms in Plateau State
|
Location |
Negative (%) |
Trace (%) |
CMT + (%) |
CMT ++ (%) |
CMT +++ (%) |
|
Jos South |
|||||
|
Growth |
17 (51.5) |
3 (27.3) |
4 (100.0) |
8 (80.0) |
3 (100.0) |
|
No growth |
16 (48.5) |
8 (72.7) |
0 (0.0) |
2 (20.0) |
0 (0.00) |
|
Barkin Ladi |
|||||
|
Growth |
18 (46.2) |
3 (75.0) |
5 (100.0) |
10 (100.0) |
3 (100.0) |
|
No growth |
21 (53.8) |
1 (25.0) |
0 (0.0) |
0 (0.0) |
0 (0.0) |
|
Bassa |
|||||
|
Growth |
15 (55.5) |
1 (33.3) |
4 (57.1) |
3 (100.0) |
6 (85.7) |
|
No growth |
12 (44.4) |
2 (66.7) |
3 (42.9) |
0 (0.0) |
1 (14.3) |
|
Riyon |
|||||
|
Growth |
14 (33.3) |
2 (100.0) |
1 (33.3) |
4 (80.0) |
12 (92.3) |
|
No growth |
28 (66.7) |
0 (0.0) |
2 (66.7) |
1 (20.0) |
1 (7.7) |
|
Jos East |
|||||
|
Growth |
12 (66.7) |
3 (100.0) |
0 (0.0) |
5 (71.4) |
7 (100.0) |
|
No growth |
6 (33.3) |
0 (0.0) |
0 (0.0) |
2 (28.6) |
0 (0.0) |
|
Jos North |
|||||
|
Growth |
17 (33.3) |
0 (0.0) |
2 (66.7) |
3 (75.0) |
9 (81.8) |
|
No growth |
34 (66.7) |
1 (100.0) |
1 (33.3) |
1 (25.0) |
2 (18.2) |
Table 5: Mean Total Bacteria Count and mean Total Coliform Count of cow milk samples collected from dairy farms in Plateau State
|
Location |
CMT scores |
Mean TBC ± SE (CFU/mL) |
Mean TCC ± SE (CFU/mL) |
|
Jos South |
Negative |
1.2x102±2.3 |
1.7x101±1.2 |
|
Trace |
7.9x102±5.8 |
2.4x101±2.3 |
|
|
CMT + |
4.7x103±115.5 |
3.0x102±1.15 |
|
|
CMT ++ |
1.4x104±577.4 |
5.8x102±3.5 |
|
|
CMT +++ |
2.8x105±377.7 |
3.0x103±71.0 |
|
|
Barkin Ladi |
Negative |
5.7x101±1.2 |
0.6 x101±0.6 |
|
Trace |
6.8 x101±0.6 |
1.8 x101±0.6 |
|
|
CMT + |
2.9 x102±5.8 |
0.8 x102±1.7 |
|
|
CMT ++ |
8.7 x102±4.0 |
1.3 x102±2.3 |
|
|
CMT +++ |
6.2x104±836.7 |
0.4x103±57.7 |
|
|
Bassa |
Negative |
9.8 x101±0.6 |
1.0 x101±0.7 |
|
Trace |
2.8 x102±2.3 |
4.3 x101±0.6 |
|
|
CMT + |
4.9 x104±71.0 |
0.9 x102±1.0 |
|
|
CMT ++ |
3.1 x105±712.5 |
9.3 x102±1.2 |
|
|
CMT +++ |
5.9x106±57735.0 |
6.7x103±125.3 |
|
|
Riyon |
Negative |
2.5 x102±2.9 |
1.9 x101±0.6 |
|
Trace |
8.3 x102±1.2 |
2.7 x102±1.0 |
|
|
CMT + |
3.5 x103±39.3 |
9.7 x102±3.5 |
|
|
CMT ++ |
2.4 x104±51.4 |
5.3 x103±1.2 |
|
|
CMT +++ |
1.1x105±3179.8 |
3.7x104±101.3 |
|
|
Jos East |
Negative |
3.0 x102±6.9 |
1.3 x101±1.1 |
|
Trace |
9.2 x102±4.6 |
2.5 x102±4.0 |
|
|
CMT + |
4.3 x103±6.9 |
8.8 x102±4.6 |
|
|
CMT ++ |
3.9 x104±71.0 |
4.7 x103±13.3 |
|
|
CMT +++ |
8.5x104±132.7 |
1.3x104±213.7 |
|
|
Jos North |
Negative |
7.8 x101±0.6 |
8.5 x101±0.6 |
|
Trace |
3.0 x102±1.2 |
2.7 x102±4.6 |
|
|
CMT + |
4.9 x104±70.4 |
3.9 x103±16.2 |
|
|
CMT ++ |
2.4 x105±5214.6 |
7.1 x104±71.0 |
|
|
CMT +++ |
1.1x106±4308.4 |
3.0x105±7273.4 |
Discussion
In order to obtain good quality milk and prevent economic losses, early screening and prevention of subclinical mastitis have to be prioritized during collection, handling and transportation of milk. One of the rapid and inexpensive test that can be used to determine the somatic cell concentration in milk indirectly is CMT. CMT is a practical and easy technique used in demonstrating intramammary infection [10]. Mastitis in cow causes high economic loss to the milk industry hence it is regarded as the most complicated and costly disease [14, 15].
In this study, 69.0% of the milk samples were CMT negative hence had good quality while 31.0% of the milk samples were CMT pos¬itive suggesting that 31.0% of the milk samples were from cows with subclinical mastitis. This poor milk quality could be due to hand milking process especially in the open and transportation of milk under unrefrigerated condition.
Higher prevalence of mastitis observed in the bulk samples (43.9%) is an indication that the many cows from the herds had subclinical mastitis. Olatoye et al. [2] reported a lower prevalence of mastitis (26%) in bulk milk collected from dairy herds in Oyo. Increase in the percentage of milk samples with bacterial growth was observed with an increase in CMT scores of the milk samples.
Out of the 210 milk samples with negative CMT results, micro-bial growth was observed in 44.3% of the milk samples. So also growth was observed in 90.91% of the 44 milk samples with CMT +++ positive result. This implies that CMT is a good technique used in the diagnosis of subclinical mastitis. Similar trend of in-crease in number of culture positive samples with increase in CMT scores was previously reported by Kandeel et al. [16].
A positive correlation was observed between the bacteriologi¬cal plate counts (TBC and TCC) and CMT scores. The range of TBC for milk samples with negative CMT scores was 5.7x101 to 3.0x102 CFU/mL while the range of TBC for samples with CMT +++ scores was 6.2x104 to 5.9x106 CFU/mL. So also the range of TCC was higher in milk samples with CMT +++ scores (0.4x103 to 3.0x105 CFU/mL) compared to the range of milk samples with negative CMT scores (0.6x101 to 8.5 x101 CFU/mL).
The range of TBC and TCC reported by Olatoye et al. [2] were in contrast with the range of TBC and TCC observed in this study. They reported TBC range of 10x103 to 12x103 CFU/mL and 203x103 to 249x103 CFU/mL for CMT negative and CMT +++ samples respectively. While the TCC range were 83 to 103 CFU/ mL and 24107.8 to 36307.8 CFU/mL for CMT negative and CMT +++ samples respectively. This differences might be due to differ-ence in cattle breed, hygienic practices observed in the selected farms and season during which the studies were conducted.
The TCC of most milk samples with weak (CMT +) to strong (CMT +++) positive CMT scores were high, this suggests the presence of fecal contaminants either from the udder of the cow, handlers or containers used in milking and transportation.
Conclusion
The overall prevalence of mastitis in cow by California Masti¬tis Test was 31.0%. Out of the 339 milk samples screened, 210 (61.9%), 24 (7.1%), 22 (6.5%), 39 (11.5%) and 44 (13.0%) were CMT negative, CMT trace, CMT +, CMT ++ and CMT +++ re-spectively. Samples with CMT +++ positive results had the highest number of culture positive and bacteria counts.
Abbreviations
CMT: California Mastitis Test
TBC: Bacterial Count
TCC: Total Coliform Count
NDDP: Nigerian Dairy Development Program
SCC: Somatic cell count
Acknowledgements
We hereby thank the Technical Staff of Department of Microbiolo¬gy, Ahmadu Bello University for their help in data process
Authors Contributions
Conceived and designed the study: SAB. Collection of data and Experiment: SAB, HIM. Analysis and interpretation of data: SAB HIM AZG. Drafting of initial manuscipt and revision of draft: SAB, AZG. All authors read and approved the final manuscript
Funding
No grant or external funding was received for this work
Availability of Data and Materials
The dataset generated and analyzed during the current that is not shown in the manuscript is from the corresponding author on rea¬sonable request.
Ethics Approval and Consent to Participate
Exemption was granted for this research because the milk samples were purchased at the point of sales to consumers, hence ethics approval and consent for participation is not applicable
Consent for Publication
Not applicable
Competing Interest
The authors declare that they have no competing interest
References
- Sulieman AB, Kwaga JK, Umoh VJ, Okolocha EC, Mu-hammed M, Lammler C, Shaibu SJ, Akineden O, Weiss R. Macro-restriction analysis of Staphylococcus aureus isolated from subclinical bovine mastitis in Nigeria. Afr J Microbiol Res 2012; 33:6270–4
- Olatoye O, Amosun A, Ogbu U, Okunlade Y. Bulk tank somatic cell count and associated microbial quality of milk from selected dairy cattle herds in Oyo State, Nigeria. Italian J Food Saf. 2018;3;7– 10.
- Amosun EA, Ajuwape ATP Adetosoye AI. Bovine Strepto-coccal mastitis in Southwest and Northern States of Nigeria. Afri J Biomedl Res., 2010;13:33-37
- Junaidu AU, Salihu MD, Tambuwal FM, Magaji AA, JaafaruS. Prevalence of mastitis in lactating cows in some selected commercial dairy farms in Sokoto metropolis. Adv Appl Sci Res. 2011; 2:290-4.
- Shittu A, Abdullahi J, Jibril A, Mohammed AA, Fasina FO. Sub-clinical mastitis and associated risk factors on lactating cows in the Savannah Region of Nigeria. BMC Vet Res. 2012; 1:8:134 – 9.
- Gonzalo C, Carriedo JA, García-Jimeno MC, Pérez-Bilbao M, De la Fuente LF. Factors influencing variation of bulk milk antibiotic residue occurrence, somatic cell count, and total bacterial count in dairy sheep flocks. J Dairy Sci. 2010; 93:1587-95.
- Pandey GS, Voskuil GC. Manual on milk safety, quality and hygiene. Golden Valley Agricultural Research Trust, Zambia. 2011 pg;52.
- van Hooijdonk T, Hettinga K. Dairy in a sustainable diet: a question of balance. Nutr Rev. 2015; 73:48-54.
- Durr JW, Cue RI, Monardes HG, Moro-Mendez J, Wade KM Milk losses associated with somatic cell counts per breed, parity and stage of lactation in Canadian dairy cattle. Livestock Science, 2008;117: 225-232.
- KaÅ?ikci G, Cetin Ö, Bingöl EB, Gündüz MC. Relations between electrical conductivity, somatic cell count, California mastitis test and some quality parameters in the diagnosis of subclinical mastitis in dairy cows. Turkish J Vet Anim Sci. 2012; 30:36:49-55.
- Atasever S, Erdem H. Association between subclinical mastitis markers and body condition scores of Holstein cows in the Black Sea region, Turkey. J. Anim. Vet. Adv. 2009; 8:476-80.
- Ruegg PL, Pantoja JC. Understanding and using somatic cell counts to improve milk quality. Irish J Agric Food Res. 2013;101-17.
- Olatunji EA, Jubril AE, Okpu EO, Olafadehan OA, Ijah UJ,Njidda AA. Bacterial assessment and quality analysis of raw milk sold in gwagwalada area Council of the Federal Capital Territory (FCT) Abuja, Nigeria. Food Sci Qual Manag. 2012; 7:1-4.
- Barkema HW, Green MJ, Bradley AJ, Zadoks RN. Invited review: The role of contagious disease in udder health. J dairy Sci. 2009; 92:4717-29.
- Olde-Riekerink RGM, Barkema HW, Scholl DT, Poole DE, Kelton DF. Management practices associated with the bulk-milk prevalence of Staphylococcus aureus in Canadian dairy farms. Prev Vet Med, 2010;97:20-28
- Kandeel SA, Morin DE, Calloway CD, Constable PD. Association of California mastitis test scores with intramammary infection status in lactating dairy cows admitted to a veterinary teaching hospital. J Vet Inter Med. 2018; 32:497-505.



