Research Article - (2024) Volume 8, Issue 2
RAGE Expression and Invasiveness of Oral Squamous Cell Carcinoma Cells Via Differential Expression of Matrix Remodeling Enzymes
2College of Dental Medicine, Roseman University of Health Sciences, South Jordan, UT, USA
Received Date: May 08, 2024 / Accepted Date: Jun 03, 2024 / Published Date: Jun 07, 2024
Copyright: ©©2024 Juan Arroyo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Clarke, D., Yeates, E., Davis, G., Harward, K., Robertson, P., et al. (2024). RAGE Expression and Invasiveness of Oral Squamous Cell Carcinoma Cells Via Differential Expression of Matrix Remodeling Enzymes. J Oral Dent Health, 8(2), 01-06.
Abstract
Oral squamous cell carcinoma is an aggressive cancer that affects approximately 30,000 people annually. Tobacco use is known to induce diseases characterized by abnormal oral squamous cell carcinoma cells. The receptor for advanced glycation end-products is a surface receptor that is up-regulated by cigarette smoke in diverse tissue types, including oral squamous cell carcinoma cells. Our objective was to determine the receptor for advanced glycation end-products message and protein expression, cell invasiveness, and potential regulatory enzymes implicated in matrix remodeling during cigarette smoke extract exposure.
Gingiva-derived Ca9-22 cells and tongue-derived Cal27 cells were cultured and exposed to 0.05% cigarette smoke extractfor 24 hours. Cell invasion was determined and cells were lysed for either quantitative polymerase chain reaction or western blot analysis. The receptor for advanced glycation end-products (mRNA and protein) was determined as well as protein expression for metalloprotease 2, metalloprotease 13 and the tissue inhibitor of metalloproteinases.
Treatment of these two cell lines with cigarette smoke extract resulted in: (i) increased receptor for advanced glycation end- products mRNA and protein expression; (ii) increased Ca9-22 and Cal27 cell invasion; (iii) increased metalloprotease 2, metalloprotease 13 and the tissue inhibitor of metalloproteinases in Ca9-22 cells; (iii) decreased metalloprotease 13 and the tissue inhibitor of metalloproteinases in Cal27 cells.
We conclude that cigarette smoke extract increases invasion of oral squamous cell carcinoma cells in a cell dependent fashion. These results can provideinsights into the understanding of oral squamous cell carcinoma cellular behavior in smoke environments.
Keywords
Oral Squamous Cell Carcinoma, Receptor for Advanced Glycation End-Products, Metalloproteinase, Cell Invasion
Introduction
Oral squamous cell carcinoma (OSCC) is an aggressive cancer and one of the most frequently occurring cancers in developing countries [1]. OSCC involves cellular compromise characterized by early metastatic migration with a recurrence rate that surpasses 50% and a 5-year survival rate of 50–60% of all cases [1-6]. Tobacco exposure alone is recognized as a major cause of this group of cancers with higher numbers of OSCC identified in smokers when compared to no-smoker counterparts [1]. Although cigarette smoke has been implicated in altered OSSC cell behavior, reports detailing precise regulation of OSCC dysfunction caused by cigarette smoke remains to be fully elucidated.
The receptor for advanced glycation end products (RAGE) is a multiligand cell surface receptor expressed by numerous cell types including OSCC cells [1]. This receptor is implicated in the pathogenesis of many inflammatory diseases, such as diabetes, atherosclerosis, and diverse rheumatological disorders [1,7]. RAGE expression is detected in gingival tissues from humans with chronic periodontitis; therefore, suggesting a likely role in tissue inflammation during disease progression [8]. Recently, RAGE expression has also been linked to the initiation and progression of various cancers [9]. Specifically, RAGE has been linked to OSCC angiogenesis and the recurrence of this type of cancer over time [10,11]. Subsequent to RAGE augmentation, pro-inflammatory response in local cell types is perpetuated, adding to the severity of gingival tissue pathologies observed in smokers [1]. Importantly, previous studies in our laboratory demonstrated that cigarette smoke regulates OSCC cells invasionin a RAGE dependent manner but molecules associated with this regulation remained to be clarified [1].
To better understand OSCC invasiveness during CSE induced RAGE expression, we decided to investigate several key molecules involved in targeting matrix during invasion. One common group of enzymatic factors involved in matrix targeting are matrix metalloproteinases (MMPs). MMPs are highly involved in tissue remodeling, angiogenesis, embryogenesis, altered migration, and cell invasion [12,13]. Specifically, we sought to investigate the expression of MMP-2 due to its central role in enhanced invasive potential of oral cancer cells [14,15]. Invasive metrics were also evaluated in the context of MMP-13, a factor centrally implicated in the cellular migration of oral cancer cells [16,17]. Lastly, inhibitors of MMPs (TIMP) were quantified due to their role in managing overall tissue destabilization during oral cell cancer progression [18].
Methods
Cell Culture
Ca9-22 human gingiva squamous carcinoma cells and Cal27 human tongue squamous carcinoma cells were used in these experiments (both from ATCC, Manassas, VA). Cells were maintained in RPMI medium plus 10% fetal bovine serum (FBS) (Invitrogen, Carlsbad, CA, USA) and 1% penicillin.
Cigarette smoke extract (CSE)
Cigarette smoke extract (CSE) was generated as previously described [1]. Briefly, one research cigarette (University of Kentucky, Lexington, KY) was continuously smoked by connecting the filtered end of the cigarette to a vacuum pump, pulling the particles into 5 mL of media and the resulting medium was defined as 100% CSE, which was diluted with culture medium to generate 0.05% CSE used in the cell culture experiments.
Cell Treatment
Cells were grown to 70-80% confluency before treatment with 1 mL fresh media or 0.05% CSE continuously for 24 hours.
RNA Isolation and Analysis
cDNAamplification was performed using Bio-Rad iTaq Universal SYBR® Green One-Step Kit. Data analysis was performed using a Bio-Rad Single Color Real Time PCR detection system (Bio-Rad Laboratories, Hercules, CA). The following primers were synthesized by Invitrogen Life Technologies (Grand Island, NY): RAGE (For-CCC TTA GCT GGC ACT TAG ATG G and Rev-TGA CCG CAG TGT AAA GAG TCC C) and β-actin (For-ACA GGA TGC AGA AGG AGA TTA C and Rev- CAC AGA GTA CTT GCG CTC AGG A).
Immunoblotting
Western Blots were performed as outlined previously [1]. Briefly, cells were lysed using a protein lysis buffer (RIPA; Thermofisher, Rochester, NY). Protein lysates (50 mg) were separated using electrophoresis on mini-Protean TGX Precast gels (Bio-Rad Laboratories, Hercules, CA) and then transferred onto a nitrocellulose membrane (Bio-Rad Laboratories). Membranes were incubated overnight with the antibodies against tissue inhibitor of metalloproteinases 1 (TIMP-1; 1:200 Cell Signaling, Danvers, MA), MMP-2 (1:500; Cell Signaling), MMP-9 (1:200; Cell Signaling), RAGE (1:500 (Abcam, Waltham, MA), or beta Actin (1:2500 Cell Signaling). For protein detection, membranes were incubated with the following secondary antibodies: 680RD Donkey anti-Mouse, 800CW Donkey anti-Mouse, 680RD Donkey anti-Rabbit, or 800CW Donkey anti-Rabbit (all at 1:2,500; from LI-COR; Lincoln, NE) at room temperature for 1 hour. Membranes were scanned using a LI-COR Odyssey DLx. All results were normalized to beta Actin. Fluorescent densities were measured via image J and comparisons were made between control and experimental groups.
Real-Time Cell Invasion
Cell invasion was measured using the xCELLigence RTCA cell monitoring system following the manufacturer’s protocol (ACEA Biosciences, Blue Spring, MO). Real-time cell invasion was analyzed following the treatments noted above. Measurements were performed in a 16 well-Cim-Plate (ACEA Biosciences, Blue Spring, MO, USA). The plates contained an upper chamber coated with type IV collagen (1:40; Corning, Corning, NY) and a lower chamber consisting of 10% FBS RPMI. Cells were plated in the top chamber at a concentration of 50,000 cells per 100 μl per well of serum free media to reach a total volume of 100 μl in each well. The plates were then placed into the RTCA instrument and invasion readings were taken every 15 min for 24 hours.
Statistical Analysis
Results were checked for normality and data were presented as mean ± SE. Differences in cell invasion, RAGE expression, MMP-2, MMP-9, and TIMP proteins were measured between control and treated cells using the Mann–Whitney U-test. Significant differences between groups were noted at P < 0.05.
Results
RAGE Expression and Cell InvasionRAGE activation is increased in cigarette smoke environments [1]. First, we sought to determine if RAGE expression is differentially induced in CSE-treated OSCC cells. Treatment with CSE led to increased expression of RAGE mRNA (1.5-fold; p<0.03) in the Ca9-22 cells (Figure 1A). In contrast, RAGE mRNA was decreased (1.5-fold; p<0.03) in Cal27 cells treated with CSE (Figure 1B). We observed a 1.8-fold induction (p<0.03) of RAGE protein in the CSE treated Ca9-22 cells (Figure 1C). Similarly, RAGE protein was increased (2.4-fold; p<0.03) Cal27 cells treated with CSE (Figure 1D). Increased cellular invasion is a cellular hallmark of OSSC cells. We therefore sought to quantitatively determine invasive effects of cultured OSCC cells treated with CSE. CSE treatment increased Ca9-22 (1.9-fold; p<0.0002) and Cal27 (1.5-fold; p<0.0003) invasion in culture (Figure 2A and B)
Figure 1: Expression of RAGE following exposure CSE. qRT-PCR showed increased RAGE mRNA in Ca9-22 cells with CSE treatment (A) and decreased RAGE mRNA was detected in Cal27 cells treated with CSE (B). Western blot analysis revealed increased expression of RAGE (1.8-fold induction; P<0.03) with CSE treatment in Ca9-22 cells (2.4-fold; p<0.03) when compared with controls (C). Similarly, increased RAGE protein expression was detected in Cal27 cells treated with CSE (D).*Statistically different from control (P < 0.05).
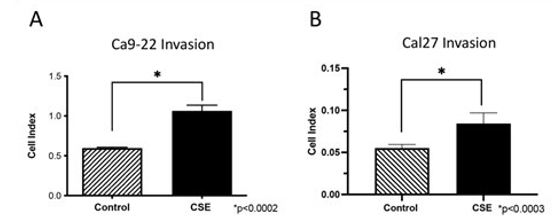
Figure 2: Invasiveness of Ca9-22 cells and Cal27 following exposure to CSE. Real-time invasion of Ca9-22 (A) and Cal27 (B) cells was significantly increased following exposure to CSE when compared to controls. *Statistically different from control (P < 0.05).
MMP-2, MMP-9 and TIMP
In order to understand the expression profile of molecules associated with the OSCC invasion, we decided to quantify matrix metalloproteinases (MMPs) following CSE exposure. MMPs are enzymes responsible for the degradation extracellular matrixes necessary for efficient cellular invasion. We specifically quantified MMP-2 and MMP-13 because they are known to be involved in the migration of oral cancers [1,15,19]. CSE treatment increased MMP-2 in Ca9-22 (2.1-fold; p<0.03) treated cells (Figure 3A). However, MMP-2 protein was unaffected by CSE in Cal27 OSCC cells (Figure 3B). An increase in MMP-13 was observed by Ca9-22 OSCC cells (2.9-fold; p<0.02) treated with CSE; however, MMP-13 expression was diminished (1.5-fold; p<0.02) following CSE treatment of Cal27 cells (Figure 4A and B). Tissue inhibitor of metalloproteases (TIMP) are proteins that inhibit MMPs [13]. We therefore investigated expression of TIMP to determine a potential regulatory correlation between inhibitory TIMP and the two MMPs. CSE treatment increased expression of TIMP (1.5-fold; p<0.03) in the Ca9-22 OSCC cells (Figure 5A); however, TIMP was decreased (1.2-fold; p<0.04) in the Cal27 OSCC cells when treated with CSE (Figure 5B).
Figure 3: Expression of matrix metalloproteinase (MMP)-2 following CSE treatment. Western blot analysis revealed increased expression of MMP-2 (2.1-fold; p<0.03) by Ca9-22 cells treated with CSE when compared with controls (A). CSE treatment did not affect MMP-2 expression in Cal27 cells compared to controls (B). *Statistically different from control (P < 0.05).
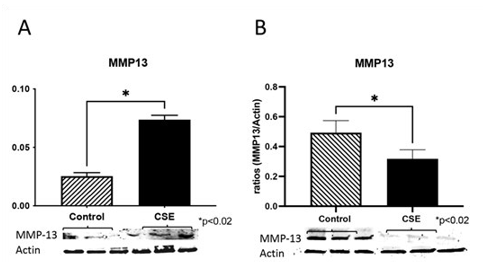
Figure 4: Expression of matrix metalloproteinase (MMP)-13 following CSE treatment. Western blot analysis revealed increased expression of MMP-13 (2.9-fold; p<0.02) by Ca9-22 cells treated with CSE when compared to controls (A). CSE treatment decreased MMP-13 expression(1.5-fold; p<0.02) in Cal27 cells compared to controls (B). *Statistically different from control (P < 0.05).
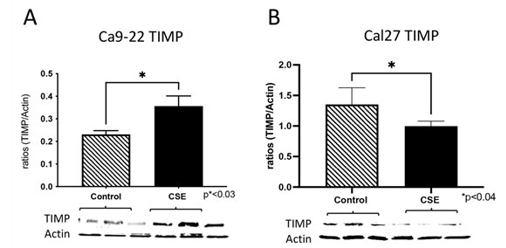
Figure 5: Expression of tissue inhibitor of metalloproteinases (TIMP) during CSE treatment. Immunoblotting revealed increased expression of TIMP (1.5-fold; p<0.03) by Ca9-22 cells treated with CSE when compared to controls (A). CSE treatment decreased TIMP expression (1.2-fold; p<0.04) in Cal27 cells compared to controls (B). *Statistically different from control (P < 0.05).
Discussion
Despite the many efforts presented to date seeking cigarette smoke cessation, cigarette smoke remains on the list of top ten contributors of preventable diseases and its use is suspected to continue to rise [20,21]. OSSC is an aggressive invasive cancer and tobacco is one of the major risk factors for the development of this deleterious set of diseases [1]. Moreover, studies have shown that tobacco may have a mechanistic role in the regulation of OSCC cellular invasion [1]. In fact, our laboratory previously demonstrated a RAGE-dependent increase in Ca9-22 OSCC cell invasion during cigarette smoke extract (CSE) exposure [1]. This discovery buttressed the research of others that revealed the RAGE pathway as a means of alleviating OSCC severity [22]. RAGE is a cell-surface receptor expressed in many cells including OSCC cells [1,23]. In this last case, RAGE was screened in 38 OSCC cases and found to be highly correlative to well-differentiated cases [23]. Although RAGE is mostly considered a perpetuator of inflammation, recent studies have also shown parallel roles for this receptor in cases of DNA repair, regulation of cellular invasion, and other processes [1,7,9,24].
We first wanted to determine RAGE expression in the OSCC cells. In our studies we tested two different OSCC cell lines: Ca9-22 derived from gingiva OSCC and Cal27 obtained from lingual OSCC. Our experiments confirmed increased RAGE mRNA and protein expression in the Ca9-22 cells exposed to CSE while only RAGE protein was elevated in Cal27 cells. Although both cell types are considered OSCC cells, lingual Cal27 cells have often returned different behaviors making it important to differentiate the connectiveness of cell responses between both our experimental conditions. For instance, a retrospective study that included over 5,000 patients revealed that progression to malignancy, or risk of cancer, was highest in the tongue (11.4%) vs. those centered in the gingiva (6.5%) [25].
Cell invasion is the process in which cells dissociate from a source tissue and migrate into neighboring organs often culminating in the development of cancers [1]. Our investigation revealed that the addition of CSE was sufficient to increase the invasiveness of both Ca9-22 and Cal27 OSCC cells. While not specifically evaluated in the current investigation, concomitant RAGE upregulation suggests a potential role for the receptor in mechanisms of invasion that must be clarified.
To better understand the regulation of invasion in these cell populations, we next studied MMP-2 and MMP-13 as well as a key inibitor, TIMP. These and other family members are important enzymatic proteins known to be involved in the destabilization of matrix required for efficient cellular invasion. MMPs are endopeptidases that degrade extracellular matrix in order to facilitate invasion of migratory cells. They perform these functions via the degradation of extracellular matrix (ECM) components. MMP-2 degrades collagen, elastin, fibronectin, gelatin, and laminin and is known to have pro-inflammatory and anti-inflammatory properties [26]. In addition, increased expression of MMP-2 is associated with enhanced metastatic invasion of OSCC cells [14]. MMP-13 degrades extracellular matrix components such as collagens, gelatin, aggrecan, and fibronectin [27]. During OSCC progression, we show that MMP-13 is elevated by tobacco smoke in ways that mirror its upregulation by e-cigarette vapor in the presence of nicotine [28]. In our studies, we observed increased MMP-2 and MMP-13 following exposure to CSE in Ca9-22 OSCC cells when compared to controls. This observation suggests that these MMPs could be associated with the increased invasion observed when these cells are exposed to CSE. In addition, we observed increased expression of TIMP proteins. Perhaps the increased expression of TIMP could be a means of mechanistically adjusting invasiveness of these cells when exposed to CSE. Although this inhibitory function of TIMP is well known, it is important to mention that TIMP also has cytokine-like functions that can influence processes including cellular growth, apoptosis and oncogenesis [29].
In relation of the Cal27 OSCC cells, we observed that MMP-2 protein expression was not affected when cells were treated with CSE when compared to controls. further, we observed decreased MMP-13 and TIMP expression. These results were unexpected, particularly in the context of increased invasion caused by CSE in these cells. Collectively, these results support the idea that a different mechanism potentially regulates Cal27 OSCC cells invasion that may be independent of MMP-2 and MMP-13. Future confirmatory studies are necessary in order to settle these assumptions.
Conclusion
In Conclusion we demonstrated that CSE is capable of eliciting increased invasion of OSCC cells and that the regulation of invasion is potentially specific to the source of the OSCC cell population exposed to CSE. Further research is necessary to better understand the plausible roles for these and other MMPs and targeting studies are essential in order to discern reasons for differences in RAGE expression during CSE exposure. The current studies set the foundation for additional experimental insights into the cell specific behaviors of OSCC cells exposed to smoke environments.
Acknowledgements:
Much appreciation is extended to a team of exceptional undergraduate students in the Lung and Placenta Laboratory at Brigham Young University for vital assistance with various experiments.
Declarations
Availability of data and materials:
All data are presented within the article. Data and other materials are available from the corresponding author on reasonable request.
Funding Sources:
This work was supported by grants National Institutes of Health (1R15HL152257 and 1R15HD108743-01; JAA and PRR).
Authors’ Contributions:
JAA and PRR assisted in experimental design. JAA, DC, KC, EY, GD, KH, and PR assisted in experimental procedures. PRR and JAA conceived of the study and supervised in its implementation and interpretation. The manuscript was written by JAA. Manuscript review by PRR, FWL, and DRW. All authors assisted in manuscript preparation and approved of the final submitted version.
References
- Chapman, S., Mick, M., Hall, P., Mejia, C., & Sue, S., et al. (2018). Cigarette smoke extract induces oral squamous cell carcinoma cell invasion in a receptor for advanced glycation end-products-dependent manner. European Journal of Oral Sciences, 126(1), 33-40.
- Aberle, D. R., Abtin, F., & Brown, K. (2013). Computed tomography screening for lung cancer: has it finally arrived? Implications of the national lung screening trial. Journal of Clinical Oncology, 31(8), 1002.
- He, S., Zhang, W., Li, X., Wang, J., & Chen, X., et al. (2021). Oral squamous cell carcinoma (OSCC)-derived exosomal MiR-221 targets and regulates phosphoinositide-3-kinase regulatory subunit 1 (PIK3R1) to promote human umbilical vein endothelial cells migration and tube formation. Bioengineered, 12(1), 2164-2174.
- Kaminagakura, E., Tango, R. N., Cruz-Perez, D., Bonan, R., & Yamamoto de Almeida, L., et al. (2022). Oral squamous cell carcinoma outcome in adolescent/young adult: Systematic review and m eta-analysis. Head & Neck, 44(2), 548-561
- Johnson, N. W., Jayasekara, P., Amarasinghe, A. H. K.,& Hemantha, K. (2011). Squamous cell carcinoma and precursor lesions of the oral cavity: epidemiology and aetiology. Periodontology, 2011, 57(1), 19.
- Jiang, X., Wu, J., Wang, J., & Huang, R. (2019). Tobacco and oral squamous cell carcinoma: A review of carcinogenic pathways. Tobacco induced diseases, 17, 29.
- Tsai, K. Y., Tullis, B., Breithaupt, K. L., Fowers, R., & Jones, N., et al. (2021). A role for RAGE in DNA double strand breaks (DSBs) detected in pathological placentas and trophoblast cells. Cells, 10(4), 857.
- Katz, J., Bhattacharyya, I., Farkhondeh-Kish, F., Perez, F. M., & Caudle, R. M., et al. (2005). Expression of the receptor of advanced glycation end products in gingival tissues of type 2 diabetes patients with chronic periodontal disease: a study utilizing immunohistochemistry and RT-PCR.
- Muthyalaiah, Y. S., Jonnalagadda, B., John, C. M., & Arockiasamy, S. (2021). Impact of Advanced Glycation End products (AGEs) and its receptor (RAGE) on cancer metabolic signaling pathways and its progression. Glycoconjugate journal, 38(6), 717-734.
- Sasahira, T., Kirita, T., Bhawal, U. K., Ikeda, M., & Nagasawa, A., et al. (2007). The expression of receptor for advanced glycation end products is associated with angiogenesis in human oral squamous cell carcinoma. Virchows Archiv, 450, 287-295.
- Sasahira, T., Kirita, T., Bhawal, U. K., Yamamoto, K., & Ohmori, H., et al. (2007). Receptor for advanced glycation end products (RAGE) is important in the prediction of recurrence in human oral squamous cell carcinoma. Histopathology, 51(2), 166-172.
- Novikov, N. M., Zolotaryova, S. Y., Gautreau, A. M., & Denisov, E. V. (2021). Mutational drivers of cancer cell migration and invasion. British journal of cancer, 124(1), 102-114.
- Cabral-Pacheco, G. A., Garza-Veloz, I., Castruita-De la Rosa, C., Ramirez-Acuña, J. M., & Perez-Romero, B. A., et al. (2020). The roles of matrix metalloproteinases and their inhibitors in human diseases. International journal of molecular sciences, 21(24), 9739.
- Yamada, S. I., Yanamoto, S., Naruse, T., Matsushita, Y., & Takahashi, H., et al. (2016). Skp2 regulates the expression of MMP-2 and MMP-9, and enhances the invasion potential of oral squamous cell carcinoma. Pathology & Oncology Research, 22, 625-632.
- Lawal, A. O., Adisa, A. O., Kolude, B., & Adeyemi, B. F. (2015). Immunohistochemical expression of MMP-2 and MMP-8 in oral squamous cell carcinoma. Journal of clinical and experimental dentistry, 7(2), e203-207.
- Wu, C. Y., Liu, J. F., Tsai, H. C., Tzeng, H. E., & Hsieh, T. H., et al. (2022). Interleukin-11/gp130 upregulates MMP-13 expression and cell migration in OSCC by activating PI3K/ Akt and AP-1 signaling. Journal of Cellular Physiology, 237(12), 4551-4562.
- Huang, S. H., Law, C. H., Kuo, P. H., Hu, R. Y., & Yang,C. C., et al. (2016). MMP-13 is involved in oral cancer cell metastasis. Oncotarget, 7(13), 17144-17161.
- de Vicente, J. C., Fresno, M. F., Villalain, L., Vega, J. A., & Arranz, J. S. L. (2005). Immunoexpression and prognostic significance of TIMP-1 and-2 in oral squamous cell carcinoma. Oral oncology, 41(6), 568-579.
- Vilen, S. T., Salo, T., Sorsa, T., & Nyberg, P. (2013). Fluctuating roles of matrix metalloproteinase-9 in oral squamous cell carcinoma. The Scientific World Journal, 2013: 920595.
- Sanders, N. T., Dutson, D. J., Durrant, J. W., Lewis, J. B., & Wilcox, S. H., et al. (2017). Cigarette smoke extract (CSE) induces RAGE-mediated inflammation in the Ca9-22 gingival carcinoma epithelial cell line. Archives of Oral Biology, 80, 95-100.
- Deitel, M. (2003). Overweight and obesity worldwide now estimated to involve 1.7 billion people. Obesity surgery, 13(3), 329-330.
- Ren, L., Lou, Y., & Sun, M. (2021). The anti-tumor effects of evodiamine on oral squamous cell carcinoma (OSCC) through regulating advanced glycation end products (AGE)/ receptor for advanced glycation end products (RAGE) pathway. Bioengineered, 12(1), 5985-5995.
- Landesberg, R., Woo, V., Huang, L., Cozin, M., & Lu, Y., et al. (2008). The expression of the receptor for glycation endproducts (RAGE) in oral squamous cell carcinomas. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology, 105(5), 617-624.
- Kumar, V., Fleming, T., Terjung, S., Gorzelanny, C., & Gebhardt, C., et al. (2017). Homeostatic nuclear RAGE–ATM interaction is essential for efficient DNA repair. Nucleic acids research, 45(18), 10595-10613.
- McCord, C., Achita, P., Kiss, A., Magalhaes, M. A., & Darling, M., et al. (2023). Progression to malignancy in oral potentially malignant disorders: a retrospective study of 5,036 patients in Ontario, Canada. Oral Surgery, Oral Medicine, Oral Pathology and Oral Radiology, 136(4), 466-477.
- Cancemi, P., Aiello, A., Accardi, G., Caldarella, R., & Candore, G., et al. (2020). The role of matrix metalloproteinases (MMP-2 and MMP-9) in ageing and longevity: focus on sicilian long-living individuals (LLIs). Mediators of Inflammation, 2020.
- Berkey, D. B. (1988). Clinical decision-making for the geriatric dental patient. Gerodontics, 4(6), 321-326.
- Robin, H. P., Trudeau, C. N., Robbins, A. J., Chung, E. J., & Rahman, E., et al. (2022). Inflammation and invasion in oral squamous cell carcinoma cells exposed to electronic cigarette vapor extract. Frontiers in oncology, 12, 917862.
- Ries, C. (2014). Cytokine functions of TIMP-1. Cellular and molecular life sciences, 71, 659-672.


