Research Article - (2023) Volume 1, Issue 2
Pre-Clinical and Randomized Clinical Trial with Bromhexine and N-Acetylcysteine for COVID-19
2INCT-Biomedicina no Semiárido Brasileiro (INCT- Biomedicina), Núcleo de Biomedicina (NUBIMED), Facul, Brazil
3Faculdade de Medicina, USP-RP, Ribeirao Preto, Sao Paulo, Brazil
4Secretaria de Vigilância em Saúde (SVS) e Laboratórios Central de Saúde Pública (LACEN), Brazil
5Departamento de Bioquímica, UNIFESP, São Paulo, SP, Belize
6Rede Vírus, Ministério da Ciência, Tecnologia, Inovações e Comunicações (MCTIC), Brasília-DF, Brazil
Received Date: Sep 24, 2023 / Accepted Date: Oct 15, 2023 / Published Date: Oct 25, 2023
Copyright: ©Ã?©2023 Aldo AM Lima, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Pires-Neto, R. J., Clementino, M., Mesquita, R. F., Lima, M. S., Lima, A. A. M., et al. (2023). Pre-Clinical and Randomized Clinical Trial with Bromhexine and N-Acetylcysteine for COVID-19. Transl Med OA, 1(2), 68-75.
Abstract
Treatment options for mild to moderate COVID-19 is limited. N-acetylcysteine and bromhexine have antiviral activity and show potential as treatment options against SARS-CoV-2 infections. This study evaluates the in vitro antiviral effect of bromhexine (BMX) for SARS-CoV-2 and determines the efficacy of treatment with BMX in combination with N-acetylcysteine (NAC) to reduce clinical scores in patients with mild to moderate COVID-19. Upon evidence from pre- clinical studies, a single center randomized trial of BMX + NAC (ClinicalTrials.gov Identifier: NCT04928495) with 420 participants in total took place in Fortaleza, CE, Brazil. Out of the 420 participants 140 received placebo, 140 received NAC alone, and 140 received NAC + BMX. Patients were monitored for 10-14 days, where physicians recorded all signs and symptoms reported. Nasopharyngeal swabs and blood samples were collected for SARS-CoV-2 RNA testing during the first visit, as well as 3 and 10 days after. Blood samples were collected at first visit and 10 days after for immuno-inflammatory biomarkers measurements. Treatment with NAC+BMX reduced clinical scores and symptoms when compared to placebo group (2/26; 8% vs 7/18; 39%; p < 0.05). Fever (≥37.8°C) was reduced by NAC + BMX treatment when compared to treatment with NAC alone and placebo. This study was limited by a largely vaccinated population. Our analysis showed that BMX reduces SARS-CoV-2 infection in vitro. Clinical trial results suggested that combinatory treatment with NAC + BMX is beneficial in mild to moderate COVID-19.
Introduction
There is an urgent need for antiviral drugs to treat or ameliorate mild and severe cases of COVID-19. Among several studies that have been published on the repurposing of drugs to treat COVID-19 [1,2], the most promising report was the use of remdesivir, originally developed to treat hepatitis C and Ebola fever [3]. Remdesivir was superior to placebo in reducing recovery time in adults hospitalized with COVID -19 and lower respiratory tract infection. RNA viruses such as SARS-CoV-2 use the NF-κB pathway within host cells in their viral replication process. In experiments with human coronaviruses (HCoV- 229E), suppression of the NF-κB pathway was associated with reduction in the rate of virus replication in vitro [4,5]. NAC inhibits the NF-κB pathway and replication of human influenza virus (H5N1, Vietnam / strain VN1203) in human lung epithelial cells [5]. Considering the lower cost of NAC we investigated its potential as an inhibitor of SARS-CoV-2 replication, due to its ability to downregulate the NF-κB pathway. NAC also reduces the production of pro-inflammatory cytokines (IL-8, CXCL10, CCL5, and IL-6), and the chemotaxis of monocyte migration [6]. NAC has been shown to inhibit the replication of other viruses such as human immunodeficiency virus (HIV) [7] and respiratory syncytial virus (RSV) [8]. In SARS-CoV-2, the Mpro protease is required for virus replication [9].
NAC can bind to Cys-145, an active site of Mpro, which can inhibit its protease activity and consequently inhibit viral replication. The canonical entry into cells of the SARS-CoV-2 virus depends on the binding of the viral spike protein (S) to the angiotensin-converting enzyme 2 (ACE-2) cell receptor, a fact preceded by the initiation of the S protein through the action of the host cell membrane-associated type II transmembrane serine protease (TMPRSS2) [9]. Several research groups have proposed reusing drugs capable of interfering with the action of TMPRSS2, such as bromhexine hydrochloride, for the treatment of patients with COVID-19 [10-12]. Bromhexine selectively inhibits TMPRSS2 activity [13] and, consequently, could suppress protein cleavage SARS-CoV-2 S, thus limiting the reproduction of the new coronavirus. This study evaluated in vitro BMX inhibition of SARS-CoV-2 replication, as well as the effectiveness of NAC alone or combined with BMX in reducing the clinical score of COVID-19 through a randomized, double- blind, placebo-controlled clinical trial. Our results showed that BMX treatment efficiently disrupts of SARS-CoV-2 infection in vitro and benefit of NAC + BMX treatment by reducing ordinal clinical scores, specifically by its reduction of fever in patients with mild and moderate respiratory syndrome with SARS- CoV-2 infection.
Methods
Molecular Docking
The transmembrane serine protease TMPRSS2 (accession number: O15393) was obtained from the Uniprot database (Nucleic Acids Res, 2019). The TMPRSS2 extracellular domain was used to build a homology model using the SWISS- MODEL web server. Bromhexine hydrochloride (CID 5702220) was obtained from the PubChem Database (https://pubchem. ncbi.nlm.nih.gov/) in sdf format and then converted to mol2 format. The molecular docking of the protein TMPRSS2 and the bromhexine hydrochloride molecule was performed using the Achilles blind docking server (https://bio-hpc.ucam.edu/ achilles/).
Antiviral Activity of Bromhexine in Vitro
All in vitro protocols were places in a BSL-3 laboratory following all biosafety regulations of the University of São Paulo. Cell lineages Caco-2 (human colon, ATCC TB-37) HUH7 (human liver, JCRB0403) and Vero (monkey kidney, ATCC CCL-81) were cultured at 37Ë?C in 5% CO2 as recommend by ATCC. Bromhexine hydrochloride (BMX) (Shanghai Shengxin Medicine Chemical Co., Ltd.) was kindly donated by Ourofino. Camostat mesylate were purchased from Sigma Aldrich (Sigma-Aldrich, Inc., Missouri, USA). Antiviral properties of BMX in Caco-2 cells (Human, colon - ATCC HTB-37) was determined by the neutral red (NR) and Deoxyribonucleic Acid (DNA) Content assays. Briefly, 24-well plates treated with the compounds were infected in triplicate with SARS-CoV-2 (MOI=1) and incubated for adsorption for 2 hours at 37°C in 5% CO2. The inoculum was then removed, cultures were washed with PBS, and then treated with medium containing 750 nM BMX or vehicle (0.004% DMSO). The positive control consisted of 100 mM camostat mesylate (Sigma Aldrich, Inc., Missouri, USA), a known TMPRSS2 inhibitor. After 48 h, 250 μL of supernatant was collected for RNA extraction and viral load quantification by real-time RT-PCR using a standard curve. Next an in vitro dose-response experiment was done with Caco- 2 cells pretreated with BMX ranging from 250 to 1500 nM for 2 hours, followed by inoculation with SARS-CoV-2 (MOI=1) for 2 h. The monolayers were then washed with PBS and replenished with fresh medium containing BMX at the same concentrations used in the pretreatment. Supernatants were collected for viral genome quantification 48 hours postinfection. The virucidal activity of bromhexine on SARS-CoV-2 was tested using 5x106 TCID50 inoculum and incubated with the drug diluted in serum free DMEM at a final concentration of 1500 nM. Two hours later, virus titers were measured by endpoint titration by TCID50 in 96-wel plates.
Single Centre Clinical Trial
Study Design, Site, and Population
A randomized, double-blind, placebo-controlled clinical trial was performed to test the efficacy of NAC+BMX treatment for mild or moderate COVID-19. This trial was approved by the Brazilian National Research Ethics Commission (no. 47715321.9.0000.5054), and carried out in the city of Fortaleza, Northeastern Brazil. Patients with mild to moderate respiratory infection and clinical suspicion of COVID-19 were invited to participate in the study after signing a consent form.
Selection, Inclusion, and Exclusion Criteria
Adult patients of both sexes were invited to participate in the study. Inclusion criteria were: (a) patients with clinical signs and symptoms of mild or moderate respiratory infection defined by the absence of dyspnea, respiratory distress and an O2 saturation <95%; and (b) patients between ≥18 and ≤60 years of age. Exclusion criteria were: (a) patients participating in another clinical intervention study; (b) patients having an illness or other medical condition that precludes the use of the drugs in this intervention; (c) patients unable to ingest, retain, and absorb intervention medications; and (d) mentally incapacitated patients. The study team recruited the patients from November 29, 2021, to October 24, 2022. A total of 486 patients were selected to enter the study protocol and 66 of them were excluded based on before mentioned exclusion criteria. Therefore, 420 patients were randomly distributed into the three treatment groups. We used a computer random list in blocks of three sequentially permuted numbers to assign the treatment groups. Thus, 140 patients received placebo control (Vitamin C – 500 mg/day for 10 days), 140 patients received Nacetylcysteine (NAC; 1800 mg/ day for 10 days), and 140 patients received NAC (1800 mg/day, for 10 days) + Bromhexine Hydrochloride (BMX; 32 mg/day, for 10 days). Treatments were prepared with similar formulation vehicle to keep treatments blind.
Protocol Study Duration and Treatments Evaluation
Patients enrolled were evaluated by the study medical staff and health professionals during three visits: day 1, day 3 (±2 days) and day 10 (±3 days), at an outpatient primary care clinic. On day 1 of recruitment, demographic, epidemiologic and clinical data, and nasopharyngeal swab, and blood samples were collected, as well as started registration of adverse events (AEs) and serious adverse events (SAEs) was collected. On day 3, clinical sign and symptoms information and AEs and SAEs, and nasopharyngeal swab were collected. On day 10, we collected clinical information, nasopharyngeal swab, and blood samples.
Diagnosis of SARS-CoV-2
Molecular diagnostic analyzes was performed by Real Time PCR (qPCR) according to official guidelines determined by the American CDC (CDC-006-00019, Revision 03). Samples of nasopharyngeal swabs followed nucleic acid isolation (AcNu). All qPCR reactions were by uniplex diagnostics as described before [14].
Immune-Inflammatory Biomarkers
Immune-inflammatory biomarkerd were evaluated by Luminex XMAP Technology, as described before [14]. This technique performs a simultaneous analysis, in the same plasma sample (25 uL volume), of a panel of protein markers of pro and anti- inflammatory activity (IL-1β, IL-6, MCP-3, IL-10, G-CSF, TNF-α). Glutathione (GSH), superoxide dismutase (SOD) and catalase (CAT) were measured in plasma sample using a standard kit and performed as recommended (Thermofisher, Frederick, MD, EUA).
Adverse Events and Serious Adverse Events Data
Adverse events (AEs) and serious adverse events (SAEs) were collected using a specific form and reported within 14 days do Data Safety Managing Board (DSMB). All SAEs recorded were reviewed and reported according to good clinical practice procedures (Code of Federal Regulations & ICH Guidelines, 2019).
Sample Size and Statistical Analysis
For the samples size calculation, we used the post-hoc power calculator for two independent treatment groups (https:// clincalc.com/Stats/SampleSize.aspx). We used the percentages of the clinical score’s incidence of the NAC + BMX group of 8% compared with 38% incidence clinical score of the control group (Vit. C). Considering an alpha value of 0.05 (type-1 error), we obtained a post-hoc power value of 70% (1 – Beta; type-II errors). The randomized trial data were entered twice into spreadsheets and verified by one independent researcher to ensure accuracy. Data were de-identified and statistical analysis was performed using Statistical Package for the Social Sciences (SPSS v.20.0; IBM Corporation, https://www.ibm.com\).
We used descriptive analysis and checked the variables for normal distribution using the Kolmogorov–Smirnov test. Mann- Whitney U test and Kruskal-Walli’s test were used for independent nonparametric samples or Wilcoxon signed rank test was used for paired samples and for parametric variables. The chi-square test or Fisher´s exact test was used for assessing qualitative variables. All statistical tests were two-sided with a significant level of p < 0.05. For the pre-clinical study in vitro data, we used ordinary one-way ANOVA, followed by Bonferroni’s post hoc test. Differences were considered statistically significant when p < 0.05. Statistical analyses and graph plots were carried out using the GraphPad® Prism 8.4.2 software.
Results
Molecular docking showed that bromhexine binds to TMPRSS2. A blind docking server generated a three-dimensional structure representing the interaction between TMPRSS2 and BMX, with 15 possible clusters of binding poses. The selected bromhexine pose in the active site of TMPRSS2 exhibited a -5.7 kcal/mole binding energy, with hydrogen bonds with Ser460 (Figure 1A). A doseresponse effect of bromhexine on SARS-CoV-2 replication in vitro was observed in the Caco-2 cells infected with both parental and P1 variant strains. The estimated inhibitory concentration (IC50) of bromhexine was around 1μM (Figure 1B). When we tested the antiviral activity of bromhexine using a non-cytotoxic concentration (0.75 μM), we verified that the drug strongly inhibited SARS-CoV-2 infection in Caco- 2 cells (p < 0.001) (Figure 1C). Remarkably, bromhexine was more effective in reducing the SARS-CoV-2 replication than the positive control camostat mesylate (p < 0,0001), with 1.5-fold- higher inhibition efficiency than camostat mesylate (Figure 1C). Moreover, antiviral effects of bromhexine over SARS- CoV-2 infection was not reproduced when we use HUH7 and Vero CCL81 cells lines (Figure 1D). Infectious virus titration experiments determined by TCID50 revealed that the drug did not exhibit direct virucidal activity at 1500 nM after 2 hours of incubation at room temperature (Figure 1E).
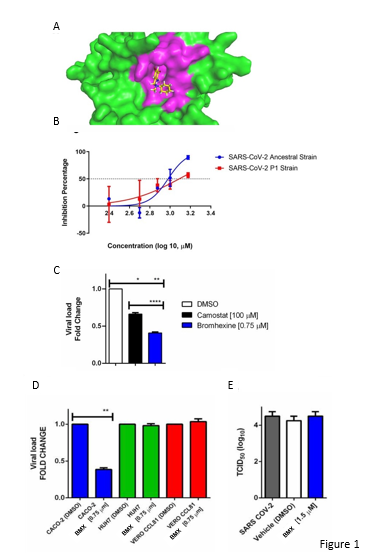
Figure 1. Bromhexing (BMX) shows potential to inhibit SARS-CoV-2 proliferation in vitro. A. Docking model of the 3D structuresTMPRSS2 protein and BMX using the Achiles blind docking server. Green and pink colors indicate the TMPRSS2 protein and its active site residue, respectively. The chemical structure represents the BMX. B. Dose-response effect of BMX concentrations in the SARS-CoV-2 progeny RNA production in parental lineage (blue line) and P.1 lineage (red line). C. Absolute quantitation of viral load of SARS-CoV-2 in Caco-2 cells treated for 48h with BMX at 0.75 μM, Camostat and mock (DMSO 0.004%). D. BMX inhibition of SARS-CoV2 replication in Caco-2, HUH7, and Vero cell lineages. E. Virucidal activity of BMX treatment represented by SARS-CoV-2 titers with TCID50 assay. (* P < 0.05; ** P < 0.01; **** P < 0.001)
A randomized, Double-Blind, Placebo-Controlled Clinical Trial Tested the Efficacy of NAC+BMX Treatment for Mild or Moderate COVID-19
Considering the in vitro results, we proposed a clinical trial to test the efficacy of treating mild respiratory infections caused by SARS-CoV-2 with N-acetyl cysteine (NAC) and NAC combined with bromhexine (NAC + BMX). Figure 2 shows a flowchart of selection, randomization, and follow-up of patients in the study protocol. After signing the consent form, a total of 486 patients with mild and moderate respiratory syndrome were evaluated for eligibility for entry into the study. Of these, 420 (86%) were enrolled and randomized to treatment with placebo (Control), N-acetyl cysteine alone (NAC), and NAC combined with Bromhexine (NAC + BMX). During the first visit, 420 (100%) patients with mild and moderate respiratory syndrome were evaluated and allocated to one of the experimental groups. During the second visit (3 + 2 days) we evaluated 313 (74%) patients. During the third and final visit (10 + 3 days) of the study, we evaluated 261 (62%) patients. Supplementary figure 1 shows a heat map showing the dispersion of confirmed COVID-19 cases (2,406 georeferenced cases) at the peak of the third wave of COVID-19 in Fortaleza, between January and February 2022. According to the Municipal Health department (February 21, 2022), this period coincides with the period of greatest recruitment in the clinical trial, under the predominance of the Omicron variant of SARS-CoV-2.
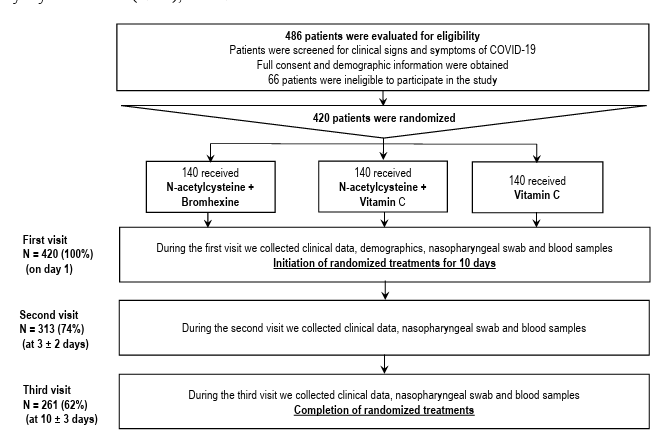
Figure 2. Flowchart representing the selection, randomization, and follow-up of patients during the single center clinical trial to test the efficacy of treating mild respiratory infections caused by SARS-CoV-2 with N-acetyl cysteine (NAC) and NAC combined with bromhexine (NAC + BMX).
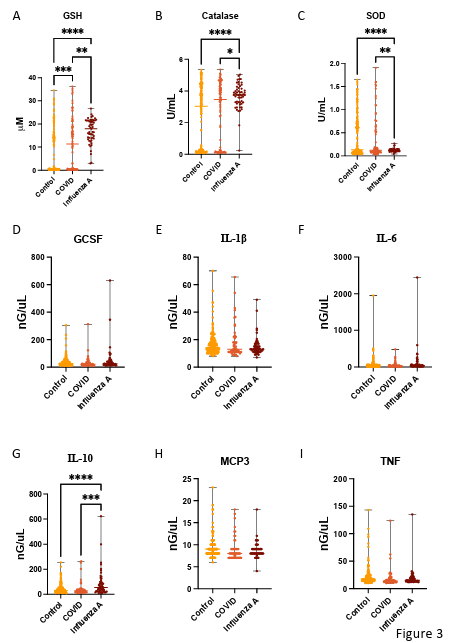
Figure 3. Serum Antioxidant activity and cytokine levels of patients with acute respiratory syndrome without diagnosis (Control), with SARS-CoV-2 infection (COVID), and with Influenza A virus infection (Influenza A). A. Concentration of Glutatione (GSH) in serum of patients with acute respiratory syndrome. B. Activity of Catalase in serum of patients with acute respiratory syndrome.C. Activity of superoxide dismutase (SOD) in serum of patients with acute respiratory syndrome. Concentration of GCSF (D), Interleukin 1 beta (IL-1β) ( E), Interleukin 6 ( IL-6) (F), Interleukin 10 ( IL-10) ( G), Monocyte-chemotactic protein 3 (MCP3) (H), and Tumor necrose factor ( TNF) (I) in serum level of patients with acute respiratory syndrome. (* P < 0.05; ** P < 0.01; **** P< 0.001)
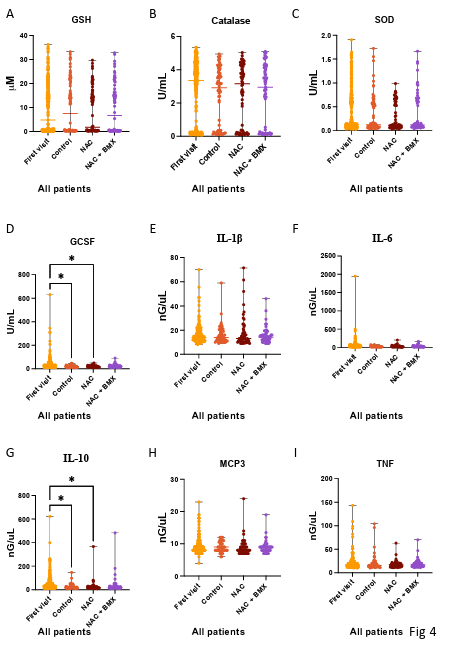
Figure 4. Serum Antioxidant activity and cytokine levels of patients with acute respiratory syndrome treated with placebo (Control), N-Acetyl cysteine (NAC), or N-acetyl cysteine combined with bromhexine (NAC+BMX) for 3-5 days was compared to serum collected during the first visit (First visit). A. Concentration of Glutatione (GSH) in serum of patients with acute respiratory syndrome. B. Activity of Catalase in serum of patients with acute respiratory syndrome. C. Activity of superoxide dismutase (SOD) in serum of patients with acute respiratory syndrome. Concentration of GCSF (D), Interleukin 1 beta (IL-1β)(E), Interleukin 6 ( IL-6) (F), Interleukin 10 ( IL-10) ( G), Monocyte-chemotactic protein 3 (MCP3) (H), and Tumor necrose factor ( TNF) (I) in serum level of patients with acute respiratory syndrome. (* P < 0.05)
Supplementary table 1 shows demographic and clinical characteristics of patients with mild and moderate respiratory syndrome collected during the first visit of the study protocol. During the first visit was observed a median of six signs and symptoms and an IQR of 5-7. Most patients had one or more comorbidities (241/383; 63%), the highest to lowest frequency were listed as obesity, former smoker, smoking, hypertension, asthma, diabetes, and other diseases with frequencies ≤1%. Twenty percent of the patients enrolled in the study were positive for SARS-CoV-2 (83/419), 13% (55/420) were positive for influenza A, 0.2% (1/420) were positive for influenza B and 1% were positive for respiratory syncytial virus. In Supplementary table 2 we show the general clinical characteristics among patients with mild and moderate respiratory syndrome at their first visit. In general, patients with mild and moderate respiratory syndrome had headache (366/417; 88%), cough (368/419; 88%), rhinorrhea (356/416; (86%), weakness (367/419; 88%). 88%), sore throat (365/419; 87%), fever (≥37.8°C) (310/420; 74%), muscle pain/arthralgia (309/419; 74%), and with a frequency <30% for shortness of breath, ageusia or dysgeusia, anosmia or dysosmia and skin irritation.
The results of clinical score in the treated population positive for COVID-19 (SARS-CoV-2) are shown in Table 1. Note that the overall clinical score in the ordinal score of a symptoms or signs was reduced in the group treated with NAC + BMX compared to control group with Vit. C (2/26; 8% vs 7/18; 39%; p < 0.05). Analyzing the details of which specific signs and symptoms were relevant in reducing the score, we observed that fever (≥37.8°C) was reduced by NAC + BMX treatment when compared to control treatments (Figure 5F). Treatment with NAC+BMX also increased cough in patients with COVID-19 (Figure 5B).
Treatment with NAC alone decreased muscle pain / arthalgia in COVID-19 patients (Figure 6A), furthermore NAC treatment alone also increased ageusia / dysgeusia in COVID-19 patients (Figure 6C). Adverse events are shown in Supplementary table 3. Overall adverse events occurred with a frequency of less than 3% (12/420) and comparison between experimental treatment groups did not show significant differences. There was a serious adverse event related to the hospitalization of a patient due to worsening of the patient's health, this patient was allocated in the control group of Vit. C.
|
Clinical scores at the second visit in the study protocol |
Total* N (%) |
N-acetylcysteine + bromhexine & N (%) |
N-acetylcysteine + vitamin C N (%) |
Vitamin C N (%) |
|
Score on the ordinal scale – no. (%) RT- |
|
|
|
|
|
PCR positive for SARS-CoV-2 §: |
|
|
|
|
|
1. No symptoms or signs |
7 / 69 (10) |
3 / 26 (12) a |
3 / 25 (12) a |
1 / 18 (6) a |
|
2. A symptom or sign |
13 / 69 (19) |
2 / 26 (8) a |
4 / 25 (16) a, b |
7 / 18 (39) b |
|
3. Two symptoms or signs |
10 / 69 (15) |
5 / 26 (19) a |
3 / 25 (12) a |
2 / 18 (11) a |
|
4. Three or more symptoms or signs |
39 / 69 (57) |
16 / 26 (62) a |
15 / 25 (60) a |
8 / 18 (44) a |
|
Clinical signs and symptoms |
|
|
|
|
|
Headache |
33 / 74 (45) |
10 / 26 (39) a |
13 / 28 (46) a |
10 / 20 (50) a |
|
Cough |
53 / 74 (72) |
22 / 26 (85) a |
17 / 28 (61) a |
14 / 20 (70) a |
|
Rhinorrhea |
36 / 74 (49) |
15 / 26 (58) a |
13 / 28 (46) a |
8 / 20 (56) a |
|
Weakness |
29 / 72 (40) |
11 / 26 (42) a |
11 / 27 (41) a |
7 / 19 (37) a |
|
Sore throat |
20 / 74 (27) |
7 / 26 (27) a |
8 / 28 (29) a |
5 / 20 (25) a |
|
Fever (≥37.8°C) |
8 / 74 (11) |
0 / 26 (0) a |
4 / 28 (14) b |
4 / 20 (20) b |
|
Muscle pain/arthralgia |
22 / 74 (30) |
9 / 26 (35) a |
6 / 28 (21) a |
7 / 20 (35) a |
|
Shortness of breathe |
12 / 72 (17) |
4 / 26 (15) a |
4 / 27 (15) a |
4 / 19 (21) a |
|
Ageusia or dysgeusia |
18 / 73 (25) |
7 / 26 (27) a |
8 / 27 (30) a |
3 / 20 (15) a |
|
Anosmia or dysosmia |
16 / 74 (22) |
5 / 26 (19) a |
7 / 28 (25) a |
4 / 20 (20) a |
|
Skin irritation |
3 / 74 (4) |
1 / 26 (4) a |
2 / 28 (6) a |
0 / 20 (0) a |
* The percentage may not add up to 100 due to rounding. § RT-PCR positive by CT < 37. & Treatment dosage by experimental group: N-acetylcysteine 1,800 mg/day for 10 days + bromhexine 32 mg/day PO; N-acetylcysteine 1800 mg/day PO for 10 days; Control placebo (Vitamin C 500 mg/Day PO for 10 days) + juice vehicle. + Statistical tests used: Chi-Square Likelihood Ratio Test. Each subscript letter denotes a subset of treatment categories whose column proportions do not significantly differ from each other at the 0.05 level.
Table 1. Results according to clinical scores in the treated population and positive RT-PCR test for SARS-CoV-2.
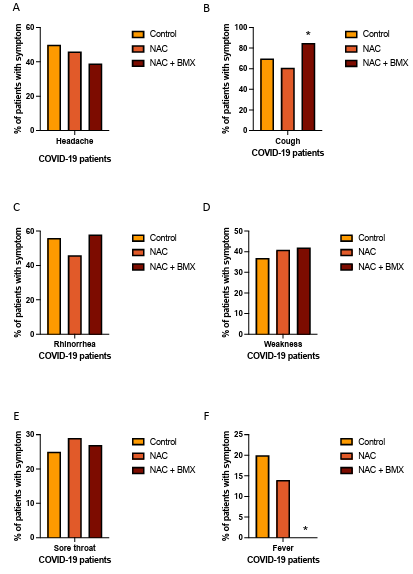
Figure 5. Prevalence of reported symptoms of patients with acute respiratory syndrome diagnoses with COVID-19 treated with placebo (Control), N-Acetyl cysteine (NAC), or N-acetyl cysteine combined with bromhexine (NAC+BMX) for 3-5 days. A. Prevalence of Headache in each treatment group. B. Prevalence of cough in each treatment group. C. Prevalence of Rhinorrhea in each treatment group. D. Prevalence of weakness in each treatment group. E. Prevalence of sore throat in each treatment group. F. Prevalence of fever in each treatment group. (* P < 0.05)
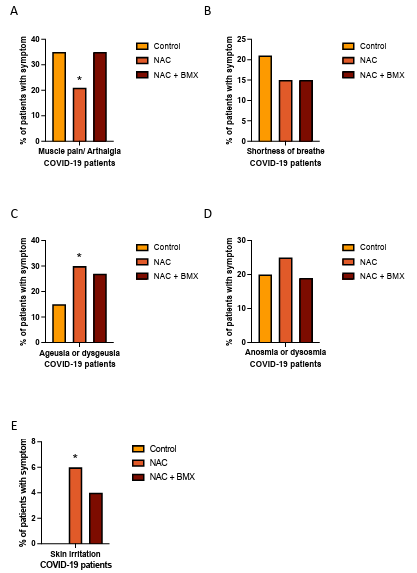
Figure 6: Prevalence of reported symptoms of patients with acute respiratory syndrome diagnoses with COVID-19 treated with placebo (Control), N-Acetyl cysteine (NAC), or Nacetyl cysteine combined with bromhexine (NAC+BMX) for 3-5 days. A. Prevalence of muscle pain / arthalgia in each treatment group. B. Prevalence of shortness of breath in each treatment group. C. Prevalence of Ageusia or dysgeusia in each treatment group. D. Prevalence of Anosmia or dysosmia in each treatment group. E. Prevalence of skin irritation in each treatment group. (* P < 0.05)
Discussion
Antiviral drugs for the treatment of mild to moderate COVID-19 is extremely limited. The CDC, USA indicates only two drugs for the treatment of COVID-19, nirmatrelvir combined with ritonavir (Paxlovid) and Remdesivir, both approved by Food and Drug Administration (FDA). There is strong evidence that patients at risk for severe COVID-19 have a reduced risk for hospitalization and death when taking these drugs [15, 16]. However, these medications are not easily available in the pharmaceutical market, especially in developing countries. This unavailability is mostly due to the high economic cost of making these drugs, turning this treatment unavailable for the population. This randomized clinical trial evaluated the activity of drugs available at lower costs, such as Nacetylcysteine and bromhexine. Pre-clinical experiments revealed that BMX significantly inhibits SARS-CoV-2 infection and replication in vitro. Molecular docking showed that bromhexine binds to the extracellular domain of TMPRSS2 (-5.7 kcal/mole binding energy). Bromhexine significantly reduces the production of SARS-CoV-2 progeny in Caco-2 cells in a dose-dependent manner. The outcome observed in Caco-2, which are known to express TMPRSS2 (mRNA and protein) [17], were not reproduced with HUH7 and Vero CCL-81 cells. This difference could be explained by a relatively low level of expression of ACE2 in hepatocytes (e.g., HUH7), and by differences in the nonhuman primate TMPRSS2 on Vero CCL-81 cells [18]. Furthermore, bromhexine showed a higher inhibitory effect than camostat, a widely known TMPRSS2 inhibitor. These results strongly indicate that the anti-SARS-CoV-2 action of bromhexine is mediated by TMPRSS2 inhibition.
Corroborating with our pre-clinal results, the randomized clinical trial showed that the combination N-acetylcysteine plus bromhexine significantly reduced the clinical score of COVID-19 patients, associated specifically with the reduction of fever (≥37.8 oC) in patients. These results suggest a beneficial action upon bromhexine treatment in the SARS-CoV-2 infection. Clinical improvement in treatment groups was mainly due to abolition of fever. The results also emphasize the absence of adverse events and serious adverse and the high tolerability of patients taking these medications. As we did not observe an isolated benefit of N-acetylcysteine on the clinical score, we believe that the beneficial effect on the observed clinical improvement was more associated with the use of bromhexine. These medications are already widely used in children and older people, showing the broad potential benefit in the treatment of COVID-19 in these populations considering their risk for developing severe COVID-19. Bromhexine is a mucolytic compound approved by FDA and derived from the alkaloid vasicine, which has been identified as a potential inhibitor of TMPRSS2 [11,19]. We observed an increase in cough in patients treated with bromhexine, this increase may be due to its mucolytic activity stimulating the expectorant response aiding in the removal of virus and debris from the airways.
Two others related bromhexine randomized clinical trial reports were found [20,21]. Ansarin et al. (2020) was using bromhexine (24mg p.o./day, 14 days) to reduced patient transfer to the intensive care unit, intubation, and mortality rate in patients with COVID-19. In the study by Méndez et al. (2022), they were also using open-label randomized design, where bromhexine was used at a dose of 48 mg p.o./day for 7 days. This 2022 study showed no reduction in viral load for SARS-CoV-2 measured at 4, 7 and 14 days compared to the control standard of care. In our study, we observed a drastic reduction in viral load as early as the third day of evolution in all treatment groups, thus making it difficult to assess this secondary endpoint. Our study seems to be the only one so far with a randomized, double-blind clinical trial design with placebo control and having a clinical score of signs and symptoms for COVID-19 as the primary outcome parameter.
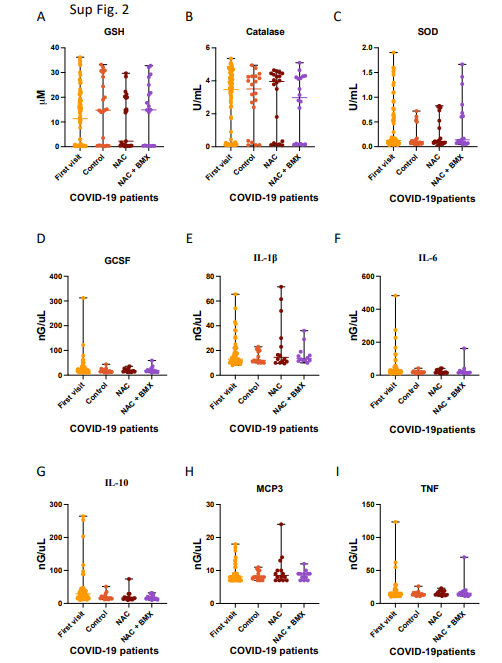
SARS-CoV-2 blocks the activity of the ACE2 receptor leading to an increased activity of angiotensin II (ANGII). ANGII binds to AT1R activating NADPH oxidases which generates several reactive oxygen species [22]. ROS mediated oxidative stress leads to activation of NF-kB in a signaling cascade to create a cytokine storm associated with tissue damage and poor prognosis [23]. GSH is an antioxidant enzyme known as a master regulator of oxidative stress in all tissues. In COVID-19 GSH seems to inhibit NF-kB activation by reducing ROS production, consequently GSH antioxidant activity helps to control cytokine storms [24]. Serum level of patients diagnosed with COVID-19 or Influenza A infection showed higher levels of GSH in comparison to a group of patients without either of those viruses (Control). Furthermore, Influenza A patients also showed higher levels of catalase and SOD activity in their plasm when compared to the control groups and COVID-19 patients. The damage that is occasioned by oxidative stress is not peculiar to SARS infection, it is shared by all inflammatory lung diseases. This study shows that influenza A patients can count on the activity of 3 different antioxidant enzymes while SARS-CoV-2 infected patients rely only on GSH. However, is important to mention that GSH deficiency has been observed in severe COVID-19 disease [25,26], which lead us to hypothesize that those patients presented a mild case of this disease due to their high levels of GSH activity.
Out of the cytokines analyzed, only IL-10 was increased in plasma of Influenza A patients when compared to control or COVID-19 patients. IL-10 works as an anti-inflammatory cytokine mediating the activity of Th1 cells, NK cells, and macrophages. This anti-inflammatory response is important to prevent tissue damage. Thus, proper activation of this cytokine is important for optimal removal of pathogens and prevention of tissue damage. Influenza A patients are actively producing and releasing IL-10 in their plasma, suggesting the inflammatory response to the virus, while COVID-19 patients do not show the same response. However, higher level of IL-10 has been found in severe COVID-19 patients, these studies hypothesize that IL- 10 has an inflammatory role in these cases [27,28].
In conclusion, our results demonstrate that BMX effectively inhibits SARS-CoV-2 infection in vitro and the randomized trial data suggest that NAC + BMX treatment is beneficial in reducing ordinal clinical score, specifically by the reduction of fever in patients with mild and moderate respiratory syndrome and with COVID-19.
Data Availability Statement: Study protocol is available on request to the corresponding author and researchers support team.
Funding Statement: This study received support from the Cearense Foundation for Supporting Scientific and Technological Development - Funcap grant number 03041300/2021, Brazilian National Council for Scientific and Technological Development (CNPq) (grant number 403201/20209), and the São Paulo Research Foundation (FAPESP) (grant number 2019/26119-0).
Conflict of Interest Disclosure: There are no conflicts of interest to disclose.
Author Contributions:
R. B. Martins, I. Ferreira, J. P. Souza, M. Pontelli, I. A. Castro, T. M. Lima, R. M. M. Viana, D. M. Jorge, P. D. Marcato and E. Arruda participated in the conceptualization of the pre-clinical experiments.
A.A. Santos, P.J.C. Magalhães, A. Havt, H. P. Monteiro, E. Arruda-Neto, A.A.M. Lima participated in the conceptualization of the randomized trial.
R. B. Martins, I. Ferreira, J. P. Souza, M. Pontelli, I. A. Castro, T. M. Lima, R. M. M. Viana, D. M.
Jorge, P. D. Marcato and E. Arruda participated in the methodology of the pre-clinical experiments;
M. Clementino, E.A.G. Arruda, R.J. Pires-Neto, M.S. Medeiros, J. Quirino-Filho, R.N.D.G. Gondim, José K. Souza, Xhaulla M.Q.C. Fonseca, L.M.V.C. Magalhães, K.F. Cavalcante, V.A.F. Viana, Liana Perdigão Mello, A. Havt, and A.A.M. Lima participated in the methodology of the randomized trial.
M. Clementino, E. Arruda, J. Quirino-Filho, R.N.D.G. Gondim, A. Havt, and A.A.M. Lima. Data curation: R. B. Martins, E. Arruda, J. Quirino-Filho, M. Clementino, R.N.D.G. Gondim, L.M.V.C. Magalhães, K.F. Cavalcante, V.A.F. Viana, L. P. Mello, A. Havt, A.A.M. Lima participated in data analysis. M.Clementino, R. B. Martins, I. Ferreira, J. P. Souza, M. Pontelli, I. A. Castro, T. M. Lima, R. M. M.
Viana, D. M. Jorge, P. D. Marcato and E. Arruda, A.A. Santos, P.J.C. Magalhães, A. Havt, H. P. Monteiro, E. Arruda-Neto, A.A.M. Lima participated in the writing and submission of the manuscript. All authors have read and agreed to the published version of the manuscript.
Ethics Approval Statement:
This trial was approved by the Brazilian National Research Ethics Commission (no. 47715321.9.0000.5054)
Patient Consent Statement: Patients with mild to moderate respiratory infection and clinical suspicion of COVID-19 were invited to participate in the study after signing a consent form.
Permission to Reproduce Material from Other Sources: Not applicable.
Clinical trial Registration: ClinicalTrials.gov Identifier: NCT04928495
Acknowledgements
We thank the patients for their availability to participate in this study. We also thank all staff participating in the randomized trial including administrators, coordinators at various levels of research, health workers and laboratory technical assistants. The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Cao, B., Wang, Y., Wen, D., Liu, W., Wang, J., Fan, G., ...& Wang, C. (2020). A trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. New England journal of medicine, 382(19), 1787-1799.
- Borba, M. G. S., Val, F. F. A., Sampaio, V. S., Alexandre, M.A. A., Melo, G. C., Brito, M., ... & Lacerda, M. V. G. (2020).Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial. JAMA network open, 3(4), e208857-e208857.
- Beigel, J. H., Tomashek, K. M., Dodd, L. E., Mehta, A.K., Zingman, B. S., Kalil, A. C., ... & Lane, H. C. (2020).Remdesivir for the treatment of Covid-19. New EnglandJournal of Medicine, 383(19), 1813-1826.
- Shi, Z., & Puyo, C. A. (2020). N-acetylcysteine to combat COVID-19: an evidence review. Therapeutics and Clinical Risk Management, 1047-1055.
- Poppe, M., Wittig, S., Jurida, L., Bartkuhn, M., Wilhelm, J., Müller, H., ... & Kracht, M. (2017). The NF-κB-dependent and-independent transcriptome and chromatin landscapes of human coronavirus 229E-infected cells. PLoS pathogens, 13(3), e1006286.
- Geiler, J., Michaelis, M., Naczk, P., Leutz, A., Langer, K., Doerr, H. W., & Cinatl Jr, J. (2010). N-acetyl-L- cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochemical pharmacology, 79(3), 413-420.
- HO, W. Z., & DOUGLAS, S. D. (1992). Glutathione and N-acetylcysteine suppression of human immunodeficiency virus replication in human monocyte/macrophages in vitro. AIDS research and human retroviruses, 8(7), 1249-1253.
- Mata, M., Sarrion, I., Armengot, M., Carda, C., Martinez, I., Melero, J. A., & Cortijo, J. (2012). Respiratory syncytial virus inhibits ciliagenesis in differentiated normal human bronchial epithelial cells: effectiveness of N-acetylcysteine. PloS one, 7(10), e48037.
- Hoffmann, M., Kleine-Weber, H., Schroeder, S., Krüger, N., Herrler, T., Erichsen, S., ... & Pöhlmann, S. (2020). SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. cell, 181(2), 271-280.
- Maggio, R., & Corsini, G. U. (2020). Repurposing themucolytic cough suppressant and TMPRSS2 proteaseinhibitor bromhexine for the prevention and management of SARS-CoV-2 infection. Pharmacological research, 157, 104837.
- Fu, Q., Zheng, X., Zhou, Y., Tang, L., Chen, Z., & Ni,S. (2021). Re-recognizing bromhexine hydrochloride: pharmaceutical properties and its possible role in treating pediatric COVID-19. European Journal of Clinical Pharmacology, 77, 261-263.
- Habtemariam, S., Nabavi, S. F., Ghavami, S., Cismaru, C. A., Berindan-Neagoe, I., & Nabavi, S. M. (2020). Possible use of the mucolytic drug, bromhexine hydrochloride, as a prophylactic agent against SARS-CoV-2 infection based on its action on the Transmembrane Serine Protease 2. Pharmacological Research, 157, 104853.
- Lucas, J. M., Heinlein, C., Kim, T., Hernandez, S. A., Malik,M. S., True, L. D., ... & Nelson, P. S. (2014). The androgen- regulated protease TMPRSS2 activates a proteolytic cascade involving components of the tumor microenvironment and promotes prostate cancer metastasis. Cancer discovery, 4(11), 1310-1325.
- Arruda, E. A., Pires-Neto, R. J., Medeiros, M. S., Quirino- Filho, J., Clementino, M., Gondim, R. N. D. G., ... & Magalhães, P. J. C. (2021). Clinical features, pathobiology, efficacy, and toxicity of tenofovir disoproxil fumarate and emtricitabine for mild to moderate SARS-CoV-2 infections. Eur J Respir Med., 3(3), 238-248.
- Hammond, J., Leister-Tebbe, H., Gardner, A., Abreu, P., Bao, W., Wisemandle, W., ... & Rusnak, J. M. (2022). Oral nirmatrelvir for high-risk, nonhospitalized adults with Covid-19. New England Journal of Medicine, 386(15), 1397-1408.
- Hashemian, S. M. R., Sheida, A., Taghizadieh, M., Memar,M. Y., Hamblin, M. R., Baghi, H. B., ... & Mirzaei, H. (2023). Paxlovid (Nirmatrelvir/Ritonavir): A new approach to Covid-19 therapy?. Biomedicine & Pharmacotherapy, 162, 114367.
- Bertram, S., Glowacka, I., Blazejewska, P., Soilleux, E., Allen, P., Danisch, S., ... & Pöhlmann, S. (2010). TMPRSS2 and TMPRSS4 facilitate trypsin-independent spread of influenza virus in Caco-2 cells. Journal of virology, 84(19), 10016-10025.
- Dong, M., Zhang, J., Ma, X., Tan, J., Chen, L., Liu, S., ...& Zhuang, L. (2020). ACE2, TMPRSS2 distribution and extrapulmonary organ injury in patients with COVID-19. Biomedicine & Pharmacotherapy, 131, 110678.
- Shen, L. W., Mao, H. J., Wu, Y. L., Tanaka, Y., & Zhang,W. (2017). TMPRSS2: A potential target for treatment ofinfluenza virus and coronavirus infections. Biochimie, 142,1-10.
- Ansarin, K., Tolouian, R., Ardalan, M., Taghizadieh, A., Varshochi, M., Teimouri, S., ... & Chapman, K. R. (2020). Effect of bromhexine on clinical outcomes and mortality in COVID-19 patients: A randomized clinical trial. BioImpacts: BI, 10(4), 209.
- Vila Méndez, M. L., Antón Sanz, C., Cárdenas García, A. D. R., Bravo Malo, A., Torres Martínez, F. J., Martín Moros, J. M., ... & Soler López, B. (2022). Efficacy of Bromhexine versus Standard of Care in Reducing Viral Load in Patients with Mild-to-Moderate COVID-19 Disease Attended in Primary Care: A Randomized Open-Label Trial. Journal of Clinical Medicine, 12(1), 142.
- Vajapey, R., Rini, D., Walston, J., & Abadir, P. (2014). The impact of age-related dysregulation of the angiotensin system on mitochondrial redox balance. Frontiers in physiology, 5, 439.
- Zhang, X., Wu, K., Wang, D., Yue, X., Song, D., Zhu, Y., & Wu, J. (2007). Nucleocapsid protein of SARS-CoV activates interleukin-6 expression through cellular transcription factor NF-κB. Virology, 365(2), 324-335.
- Silvagno, F., Vernone, A., & Pescarmona, G. P. (2020). The role of glutathione in protecting against the severe inflammatory response triggered by COVID-19. Antioxidants, 9(7), 624.
- Kumar, P., Osahon, O., Vides, D. B., Hanania, N., Minard,C. G., & Sekhar, R. V. (2021). Severe glutathione deficiency, oxidative stress and oxidant damage in adults hospitalized with COVID-19: Implications for GlyNAC (Glycine and N-Acetylcysteine) supplementation. Antioxidants, 11(1), 50.
- Labarrere, C. A., & Kassab, G. S. (2022). Glutathione deficiency in the pathogenesis of SARS-CoV-2 infection and its effects upon the host immune response in severe COVID-19 disease. Frontiers in Microbiology, 13, 979719.
- Dhar, S. K., Vishnupriyan, K., Damodar, S., Gujar, S., & Das, M. (2021). IL-6 and IL-10 as predictors of disease severity in COVID-19 patients: results from meta-analysis and regression. Heliyon, 7(2).
- SALEH, M. A., TAMIM, H. H., MARAWAN, M., SAMEH,A., & SELIM, M. (2022). TNF-a and IL-10 Serum Levels in COVID-19 Patients and their Relation to Disease Severity. The Medical Journal of Cairo University, 90(9), 1459-1467.
- Neumann, J., Prezzemolo, T., Vanderbeke, L., Roca, C. P., Gerbaux, M., Janssens, S., ... & Yserbyt, J. (2020). Increased IL-producing regulatory T cells are characteristic of severe cases of COVID-19. Clinical & translational immunology, 9(11), e1204.
Supplementary table 1. Demographic and clinical characteristics of patients at basel *+.
|
Characteristics |
Total N (%) |
N-acetylcysteine + bromhexine & N (%) |
N-acetylcysteine + vitamin C N (%) |
Vitamin C + N (%) |
|
Age – year |
34 (25-42) |
34 (25-42) |
33 (25-42) |
33 (25-40) |
|
Male – no. (%) |
179 (43) |
65 (47) |
60 (43) |
54 (40) |
|
Schooling score – no. /total (%) |
|
|
|
|
|
1. First series completed |
79 / 410 (19) |
25 / 137 (18) |
31 / 137 (23) |
23 / 136 (17) |
|
2. Second series completed |
271 / 410 (66) |
89 / 137 (65) |
88 / 137 (64) |
94 / 136 (69) |
|
3. Complete university degree |
56 / 410 (14) |
21 / 137 (53) |
17 / 137 (12) |
18 / 136 (13) |
|
4. Graduate degree completed |
4 / 410 (1) |
2 / 137 (2) |
1 /137 (1) |
1 / 136 (1) |
|
Median time (IQR)** from onset of symptoms/signs to randomization - days |
3 (2-4) |
3 (2-4) |
3 (2-4) |
3 (2-4) |
|
Total number of symptoms and signs – median (IQR) |
6 (5-7) |
6 (5-7) |
7 (5-7) |
6 (5-7) |
|
Number of coexisting |
|
|
|
|
|
conditions – no. /total (%) |
|
|
|
|
|
None |
142 / 383 (37) |
47 / 130 (36) |
42 / 128 (33) |
53 / 125 (42) |
|
One |
140 / 383 (37) |
50 / 130 (39) |
47 / 128 (37) |
43 / 125 (34) |
|
two or more |
101 / 383 (26) |
33 / 130 (25) |
39 / 128 (31) |
29 / 125 (23) |
|
Coexisting conditions – no. |
|
|
|
|
|
/total (%) |
|
|
|
|
|
Obesity |
73 / 420 (18) |
22 / 137 (16) |
25 / 139 (18) |
26 /136 (19) |
|
Ex smoker |
60 / 416 (14) |
25 / 138 (18) |
21 / 140 (15) |
14 / 138 (10) |
|
Smoke |
48 / 420 (11) |
16 / 140 (11) |
18 / 140 (13) |
14 / 140 (10) |
|
Hypertension |
45 / 420 (11) |
11 / 140 (8) |
16 / 140 (11) |
18 / 140 (13) |
|
Asthma |
34 / 420 (8) |
12 / 140 (9) |
12 / 140 (9) |
10 / 140 (7) |
|
Type 1 or 2 diabetes |
19 / 420 (5) |
4 / 140 (3) |
8 / 140 (6) |
7 / 140 (5) |
|
Heart disease |
3 / 420 (1) |
1 / 140 (1) |
1 / 140 (1) |
1 / 139 (1) |
|
Liver disease |
6 / 419 (1) |
2 / 140 (1) |
2 / 139 (1) |
2 / 140 (1) |
|
HIV / AIDS |
5 / 411 (1) |
2 / 137 (2) |
2 / 138 (1) |
1 / 136 (1) |
|
Other respiratory diseases++ |
19 / 417 (5) |
8 /138 (6) |
9 / 138 (7) |
2 / 138 (1) |
|
Other chronic diseases+++ |
98 / 399 (25) |
32 / 131 (24) |
33 / 135 (24) |
33 / 133 (25) |
|
# of positive RT-PCR for SARS- CoV-2§ - no. /total (%) |
83 / 419 (20) |
29 / 140 (21) |
32 / 139 (23) |
22 /140 (16) |
|
Viral load: SARS-CoV-2 (RNA copies/mL) – median (IQR) |
23 (0,60-472) |
56 (0,46-398) |
6 (0,52-299) |
59 (0,67- 580) |
|
Influenza A no. /total (%) |
55 / 420 (13) |
17 / 140 (12) |
16 / 140 (11) |
22 /140 (16) |
|
Influenza B no. /total (%) |
1 / 420 (0,2) |
1 / 140 (1) |
- |
- |
|
Respiratory syncytial virus no. /total (%) |
4 / 420 (1) |
2 / 140 (1) |
2 / 140 (1) |
- |
* Values are in median and IQR. ** IQR denotes interquartile range. The percentage may not add up to 100 due to rounding. § RT-PCR positive by CT < 37.
+ No statistical difference between all treatment groups. Statistical tests used: Kruskal-Wallis test or Chi-square Likelihood Ratio test. ++ Other respiratory diseases included rhinitis 2% (9/420) and sinusitis 0.5% (2/420). +++
Other chronic illnesses included: allergic rhinitis 8% (34/420), anxiety 2% (9/420), hypothyroidism 1%, and various others <1%; & Treatment dosage by experimental group: N-acetylcysteine 1,800 mg/day for 10 days
+ bromhexine 32 mg/day PO; N-acetylcysteine 1800 mg/day PO for 10 days; Control placebo (Vitamin C 500 mg/Day PO for 10 days) + juice vehicle.
Supplementary table 1
Supplementary table 2. Clinical characteristics of mild to moderate acute respiratory infections with and without SARS-CoV-2 infection.
|
Clinical signs and symptoms |
Total N (%) |
|
Patients with SARS-CoV-2 confirmed by laboratory RT-PCR N (%) |
Patients who tested negative for SARS-CoV-2 N (%) |
Values of p + |
|
Headache |
366 / 417 (88) |
|
74 / 82 (90) |
291 / 334 (87) |
0.287 |
|
Cough |
368 / 419 (88) |
|
74 / 83 (89) |
293 / 335 (88) |
0.418 |
|
Rhinorrhea |
356 / 416 (86) |
|
72 / 82 (88) |
283 / 333 (85) |
0.325 |
|
Weakness |
367 / 419 (88) |
|
74 / 82 (90) |
292 / 336 (87) |
0.269 |
|
Sore throat |
365 / 419 (87) |
|
75 / 83 (90) |
289 / 335 (86) |
0.211 |
|
Fever (≥37.8°C) |
310 / 420 (74) |
|
64 / 83 (77) |
245 / 336 (73) |
0.264 |
|
Muscle pain/arthralgia |
309 / 419 (74) |
|
67 / 83 (81) |
241 / 335 (72) |
0.066 |
|
Shortness of breathe |
85 / 405 (21) |
|
18 / 80 (23) |
67 / 324 (21) |
0.412 |
|
Ageusia or dysgeusia |
67 / 415 (16) |
|
15 / 83 (18) |
52 / 331 (16) |
0.354 |
|
Anosmia or dysosmia |
61 / 416 (15) |
|
13 / 82 (16) |
48 / 333 (14) |
0.429 |
|
Skin irritation |
3 / 404 (1) |
|
0 / 81 (0) |
3 / 322 (1) |
0.509 |
|
Treatment |
|
|
|
|
|
|
Analgesic and antipyretic |
264 / 420 (63) |
|
52 / 83 (63) |
211 / 336 (63) |
0.538 |
|
Vitamins |
12 / 420 (3) |
|
1 / 83 (1) |
11 / 336 (3) |
0.276 |
|
Antiemetic |
10 / 420 (2) |
|
2 / 83 (2) |
8 / 336 (2) |
0.621 |
|
Antihistamine |
5 / 420 (1) |
|
3 / 83 (4) |
2 / 336 (1) |
0.055 |
|
bronchodilator |
5 / 420 (1) |
|
2 / 83 (2) |
3 / 336 (1) |
0.258 |
|
Anxiolytic |
3 / 420 (0.7) |
|
1 / 83 (1) |
2 / 336 (1) |
0.485 |
|
antispasmodic |
3 / 420 (0.7) |
|
3 / 83 (4) |
0 / 336 (0) |
0.008 |
|
Antibiotic |
1 / 420 (0.2) |
|
0 / 83 (0) |
1 / 336 (0.3) |
0.802 |
|
Anti-inflammatory |
1 / 420 (0.2) |
|
0 / 83 (0) |
1 / 336 (0.3) |
0.802 |
|
Syrup |
1 / 420 (0.2) |
|
0 / 83 (0) |
1 / 336 (0.3) |
0.802 |
+ Statistical tests used: Chi-Square Test of Likelihood Ratio.
Supplementary table 2
Supplementary table 3. Number of adverse events and serious adverse events total and by study groups in patients with mild to moderate respiratory syndrome up to the 10th day visit.
|
Parameters |
Total* N (%) |
N-acetylcysteine + bromhexine & N (%) |
N-acetylcysteine + vitamin C N (%) |
Vitamin C N (%) |
|
Adverse events |
|
|
|
|
|
Nausea |
12 / 48 (25) |
3 / 12 (25) a |
4 / 18 (22) a |
5 / 18 (28) a |
|
Diarrhea |
10 / 48 (2) |
4 / 12 (33) a |
3 / 18 (17) a |
3 / 18 (17) a |
|
Abdominal pain |
5 / 48 (10) |
0 / 12 (0) a |
3 / 18 (17) a |
2 / 18 (11) a |
|
Pharmacodermia |
5 / 48 (10) |
0 / 12 (0) a |
1 / 18 (6) a |
0 / 18 (0) a |
|
Dyspepsia |
3 / 48 (6) |
0 / 12 (0) a |
0 / 18 (0) a |
3 / 18 (17) a |
|
Somnolence |
2 / 48 (4) |
0 / 12 (0) a |
1 / 18 (6) a |
1 / 18 (6) a |
|
Cough |
2 / 48 (4) |
1 / 12 (8) a |
1 / 18 (6) a |
0 / 18 (0) a |
|
Spots on the skin |
1 / 48 (2) |
3 / 12 (25) a |
0 / 18 (0) a |
2 / 18 (11) a |
|
Palpitation |
1 / 48 (2) |
0 / 12 (0) a |
0 / 18 (0) a |
1 / 18 (6) a |
|
Sinusitis |
1 / 48 (2) |
1 / 12 (8) a |
0 / 18 (0) a |
0 / 18 (0) a |
|
Sweating |
1 / 48 (2) |
0 / 12 (0) a |
1 / 18 (6) a |
0 / 18 (0) a |
|
Gum ulcer |
1 / 48 (2) |
0 / 18 (0) a |
0 / 18 (0) a |
1 / 18 (6) a |
|
Serious adverse events |
|
|
|
|
|
Hospitalization |
1 / 420 (0.24) |
- |
- |
1 / 140 (0.71) |
* The percentage may not add up to 100 due to rounding. & Treatment dosage by experimental group: N- acetylcysteine 1,800 mg/day for 10 days + bromhexine 32 mg/day PO; N-acetylcysteine 1800 mg/day PO for 10 days; Control placebo (Vitamin C 500 mg/Day PO for 10 days) + juice vehicle. + Statistical tests used: Chi-Square Likelihood Ratio Test. Each subscript letter denotes a subset of treatment categories whose column proportions do not significantly differ from each other at the 0.05 level.
Supplementary table 3
Study Group Members - AVANTI.
|
NAME |
|
Function |
IES |
Department and centers |
|
Aldo Ângelo Moreira Lima |
Coordinator |
UFC |
Fisiologia e Farmacologia, NUBIMED, UFC |
|
|
Alexandre Havt Bindá |
Collaborator |
UFC |
Fisiologia e Farmacologia, NUBIMED, UFC |
|
|
Armênio Aguiar dos Santos |
Collaborator |
UFC |
Fisiologia e Farmacologia, NUBIMED, UFC |
|
|
Bruno Lins de Souza |
2st Typing |
UFC |
Nubimed, FAMED-UFC |
|
|
Charles Roberto Sousa de Melo |
Accounting Assistant |
UFC |
Nubimed, FAMED-UFC |
|
|
Charliene Sousa de Melo |
Laboratory technique |
UFC |
Nubimed, FAMED-UFC |
|
|
Dayane Feitosa Guedes de Melo |
Technique in nursing |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Djayne Rodrigues de Sousa |
Health Agent Coordinator |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Érico Antônio Gomes de Arruda |
Collaborator |
HSJ |
Hospital São José, SESA, Fortaleza, CE, Brasil |
|
|
Eurico de Arruda Neto |
Collaborator |
USP |
Biologia Celular, FMRP, USP, RB, São Paulo |
|
|
Fabiana Maria da Silva Nascimento |
Study Coordinator and Ethics Committee |
UFC |
Nubimed, FAMED-UFC |
|
|
Francisca Mônica da Silva Nascimento |
Health Agent Coordinator |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Francisco de Sousa Junior |
Programming Technician |
UFC |
Nubimed, FAMED-UFC |
|
|
Hugo Pequeno Monteiro |
Collaborator |
EPM, UNIFESP |
Escola Paulista de Medicina, UNIFESP, São Paulo, SP |
|
|
José Amadeus Sousa |
Laboratory Technical Maintenance |
UFC |
Nubimed, FAMED-UFC |
|
|
José Quirino da Silva Filho |
Programming Technician |
UFC |
Nubimed, FAMED-UFC |
|
|
Kátia Maria Lima Nogueira |
Project Administrator |
UFC |
Nubimed, FAMED-UFC |
|
|
Lucia de Fátima Alves |
luciadefatimaalves60@gmail. com |
Health Agent |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
Luciana França da Silva |
Laboratory technique |
UFC |
Nubimed, FAMED-UFC |
|
|
Lyvia M.V.C. Magalhães |
Pharmacist |
UFC |
Nubimed, FAMED-UFC |
|
|
Marco Clementino |
Marco.Clementino22@gmail. com |
Collaborator |
UFC |
Nubimed, FAMED-UFC |
|
Magina Maria Justa da Silva |
Nurse |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Marenilda Justa da Silva |
Technique in nursing |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Maria Jacinilda Rodrigues Pereira |
Nurse |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Maria Luzia Sousa de Melo |
Health Agent |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Mariana Silva Lima |
Clinical researcher |
UFC |
Nubimed, FAMED-UFC |
|
|
Melissa Soares Medeiros |
Collaborator |
HSJ |
Hospital São José, SESA, Fortaleza, CE, Brasil |
|
Pedro Jorge Caldas Magalhães |
Collaborator |
UFC |
Fisiologia e Farmacologia, NUBIMED, UFC |
|
|
Rafael Ferreira Mesquita |
Clinical researcher |
UFC |
Nubimed, FAMED-UFC |
|
|
Rafhaella N.D.G. Gondim |
Pharmacist |
UFC |
Nubimed, FAMED-UFC |
|
|
Renan Lobo Cavalcanti |
1nd Typing |
UFC |
Nubimed, FAMED-UFC |
|
|
Roberto da Justa Pires Neto |
Collaborator |
UFC |
Saúde Comunitária, UFC |
|
|
Rosania Maria de Paula Silva |
Health Agent |
HSJ |
Hospital São José, Nubimed, FAMED-UFC |
|
|
Terezinha Freire França |
terezinhafrana45@yahoo. com.br |
Laboratory technique |
UFC |
Nubimed, FAMED-UFC |
IES: Instituições de Ensino Superior; UFC: Universidade Federal do Ceará; HSJ: Hospital São José; USP: Universidade de São Paulo; FAMED: Faculdade de Medicina; NUBIMED: Núcleo de Biomedicina; EPM, UNIFESP: Escola Paulista de Medicina, UNIFESP, São Paulo, SP.



