Research Article - (2020) Volume 2, Issue 1
Potential Impact of Petroselinum Sativum on the Treatment of Urolithiasis in Rats
2Chemistry Department, Faculty of Science, Zagazig University, Egypt
3Medical Biochemistry Department, Medical Research Division, National Research Centre, Giza, Egypt
Received Date: Jan 01, 2020 / Accepted Date: Jan 25, 2020 / Published Date: Jan 28, 2020
Copyright: ©Eman R Youness, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Petroselinum sativum (PS) belongs to Apiaceae family and is widely used as herb, spice and vegetable. We aimed to investigate the anti-urolithiatic effect of PS extract against ethylene glycol inducing urolithiasis and kidney stones in rats. Serum levels of creatinine, urea, uric acid, sodium, potassium, calcium and magnesium before and after treatment were measured. Protein gel electrophoresis was also assessed. Serum uric acid and creatinine were significantly decreased, magnesium level was significantly increased and calcium level was significantly decreased in treated groups compared with the group induced by ethylene glycol only without receiving treatment. Histopathology showed significant decrease in calcium oxalate deposits in kidneys of the treated groups compared with the ethylene glycol group. Protein gel electrophoresis showed significant decrease in both low and high molecular weight proteins in treated groups and significant increase in high molecular weight proteins in the group induced by ethylene glycol.
Keywords
Petroselinum sativum (PS), Cystone, Ethylene glycol (EG), SDS Protein gel electrophoresis
Introduction
Urolithiasis is the third most common disorder of the urinary tract, the others being frequently occurring urinary tract infections and benign prostatic hyperplasia [1].The worldwide incidence of urolithiasis is quite high and in spite of tremendous advances in the field of medicine, there is no truly satisfactory drug for the treatment of renal calculi [2]. Some patients still have to undergo surgery to get rid of this stones. Hyperoxaluria is the main initiating factor for urolithiasis [3]. It is estimated that about 12% of men and 55 % of women have at least one episode of kidney stone during their life time. Once kidney stone develops the recurrence rate is estimated to be 14% at 1 year, 35% at 5 years, and 52% at 10 years. The incidence in general population is about 1 in 1000 adults per year. The main cause of urolithiasis is still unknown but most probably due to positive family history, overweight, obesity, or increased body mass index (BMI). Low urine volume < 1500 ml/ day, high dietary animal protein intake, increased urine excretion of calcium oxalate, uric acid, cystine, urinary tract structural abnormalities leading to stasis of urine flow [4]. More than 80% of urinary calculi are calcium oxalate stones alone or calcium oxalate mixed with calcium phosphate [5]. Kidney stone formation is a complex process, including supersaturation, nucleation, growth aggregation and retention within the renal tubules [6]. The recurrence of urolithiasis represents a serious problem and thus stone treatment is highly recommended. Additionally, uses of ESWL (Extracorporeal shock-wave lithotripsy) method may cause acute renal injury and an increase in stone recurrence [7]. Furthermore, some drugs used to prevent and treat the disease are not effective in all patients and often have adverse effects that compromise their use in long-term medical treatment. Thus, treatment with herbal drugs has been suggested. The toxic effects of EG have been linked with an increase in free radical production [8]. Petroselinum sativum (PS) or parsley, a member of the family of Umbelliferae, have been reported to be, antioxidant, anti-inflammatory, anti-edema, anti-hypertensive, anti-diabetic, anti-microbial and reconstruct kidney tissue after nephrotoxicity [9]. Calcium oxalate crystals in urinary tubules can produce damages in the epithelial cells [10, 11] and consequently, these cells may produce free radicals, inducing heterogenous crystal nucleation and cause aggregation of crystals [12]. Ethanolic extract of PS have glycosidea flavonoids such as apiine, apiol, apigenin, myristisine ,tanin, palmitic acid, etc. [13] and the flavonoides have antioxidant effects [14]. It can be speculated that the role of the herb extract in treatment of calcium oxalate calculi is due to the antioxidant effects of the different compounds of the PS [15]. Calcium salts are insoluble at physiological pH inducing calcium oxalate (CaOx) nephrolithiasis [16]. We believed that the herb extract provide the optimum pH which it can maintain CaOx particles dispersed in the solution and thus allow them to be eliminated easily from the kidney. Therefore, we decided to investigate the effect of the ethanolic extract of Petroselinum sativum or parsley on the treatment of calcium oxalate calculi in rat model.
Materials and Methods
Phytochemical Screening
The phytochemical components of the plant were analyzed for alkaloids, carbohydrate, phenolic compound and tannins, flavonoids, proteins and amino acids, steroids, saponins and triterpenoids using standard methods described previously[17].
Experimental Animals
Forty adult male rats of Sprague Dawley strain (average 200-230 g in weight) were obtained from the Laboratory of Animal Farm of Veterinary Medicine at Zagazig University, Egypt. The animals were housed under hygienic conditions at a room temperature of about 25±2°C with relative humidity of 50-60% and 12h light/ 12h dark cycles in the Animal House of Scientific and Medical Research Center (ZSMRC), Zagazig, Egypt. Basal diet and water were allowed.
Petroselinum sativum
The plant was purchased from the local supermarket; about 3kg of the plant were dried in air and under shadow to be crispy and finely grinded into fine powders to give about 301.8 g powder. It was packed in to the soxhlet column and extracted with pure ethanol at 30-40°C for 24h. The extract obtained was evaporated at 40°C in the rotary evaporator then dried to yield about 25.4 g extract and stored in airtight container then freezed to be used [18].
Cystone
Cystone, a polyherbal formulation, mainly comprising plant drugs, which are widely used for antilithic activity in traditional medicine, was evaluated for its effects on experimentally induced urolithiasis in rats. It is produced by Himalaya Drug Company Makali, Bangalore 562, 123 (India).
Ethylene Glycol
Ethylene glycol is a relatively non-volatile, slightly viscous, sweet-tasting liquid. It was used for inducing the formation of kidney stones. It was purchased from El-Gomhouria Company for chemical industries, Zagazig, Egypt [19].
Gum Acacia
It is the dried exudate from the trunks and branches of Acacia senegal or Vachellia (Acacia) seyal in the family leguminosae (fabaceae). Gum arabic’s mixture of polysaccharides and glycoproteins gives it the properties of a glue and binder that is edible by humans. Pharmaceutical drugs and cosmetics use the gum as a binder, emulsifying agent, and a suspending or viscosity increasing agent [20].
Experimental Design
Forty adult male Sprague Dawley rats were randomly divided into five equal groups each of 8 rats. Group (1) received saline and served as control group negative control group. Group (2) received 1/10 LD50 (LD50): 4700 mg/kg [Rat] of the ethylene glycol and served as positive control group [21] for 30 days. Group (3), Group (4) and Group (5) also received ethylene glycol for 30 days but from the 14th day of the experiment started to receive treatments. Group (3) treated with Petroselinum sativum 250 mg/kg [22]. Group (4) treated with Cystone 750 mg/kg [23]. Group (5) treated with both Petroselinum sativum and Cystone with the previous doses. As the ethanolic extract of PS is not water soluble, so it was dissolved in Gum acacia to be like a suspension. Gum acacia was also added to Cystone and was given to both the negative and positive control groups to be sure that gum acacia has no effect on a group than the other. All the doses were fed orally by gastric gavage.
Biochemical Assays
The blood was collected from the retro-orbital sinus under anaesthetic condition using thiopental and the serum was separated by centrifugation and analyzed for creatinine, urea, uric acid, sodium, potassium, calcium and magnesium. These parameters were estimated using standard reagent kits (Biomed Diagnostics, Hannover, Germany) for creatinine, urea and uric acid, (Spectrum Diagnostics, Hannover, Germany) for magnesium and (Labcare Diagnostics(ɪ)PVT.LTD, India) for sodium, potassium and calcium.
Histopathological Assessment Studies
Both kidneys were isolated, and then rinsed in ice-cold physiological saline. The right kidney was fixed in 10% neutral buffered formalin, processed in a series of graded alcohol and xylene, embedded in paraffin wax, sectioned at 5µm and stained with haematoxylin (H) and eosin (E) for histopathological examination. The slides were examined under binocular light microscope (magnification of x 200, x 400) to study light microscopic architecture of the kidney and calcium oxalate deposits [24].
Sds- Page for Kidney Proteins
The left kidney was finely minced and 20% homogenate was prepared in phosphate buffer (pH 7.4). Total kidney homogenate then centrifuged to give very clear supernatant that contain kidney proteins. The purpose of SDS-PAGE is to separate proteins according to their size, and no other physical feature. Proteins first were denatured from their secondary, tertiary or quaternary structure in to their primary amino acid structure by using SDS (sodium dodecyl sulfate) which is a detergent (soap) that can dissolve hydrophobic molecules but also has a negative charge (sulfate) attached to it. The negatively charged proteins were put into an electric field through a polyacrylamide gel, which is a polymer of acrylamide monomers. When this polymer is formed, it turns into a gel and we will use electricity to pull the proteins through the gel so the entire process is called polyacrylamide gel electrophoresis (PAGE). A polyacrylamide gel is not solid but is made of a laberynth of tunnels through a meshwork of fibers. So, different sized proteins move at different rates [25].
Statistical Analysis
Data were checked, entered and analysed by using (SPSS version 22). Data were expressed as mean ±SD. (ANOVA test) was used for analysis of several means and post hoc test was used to fine significant between groups. PË?0.05 was statistically considered significant [26].
Results
The phytochemical analysis of the current study revealed the presence of large number of bioactive components in the PS including flavonoids, Table (1) showed significant increase in serum creatinine, urea, uric acid and calcium while there was a significant decrease in magnesium level. On the other hand, the treated groups showed a significant decrease in serum creatinine, uric acid and calcium while there was significant increase of magnesium level to normal values. Serum sodium and potassium levels mainly remained within normal values.
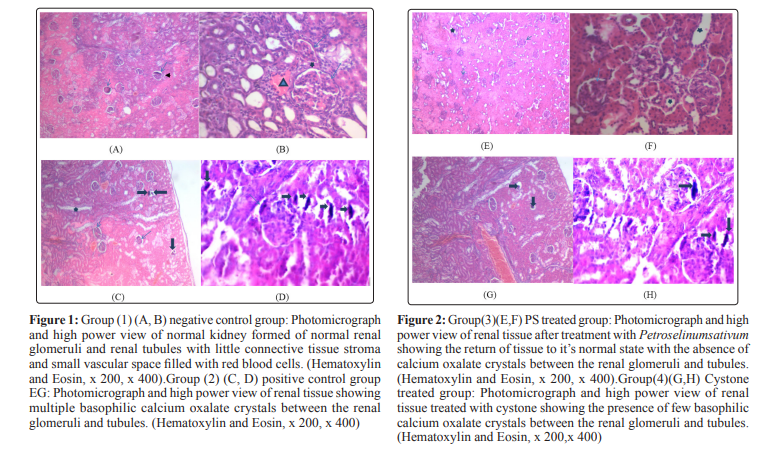
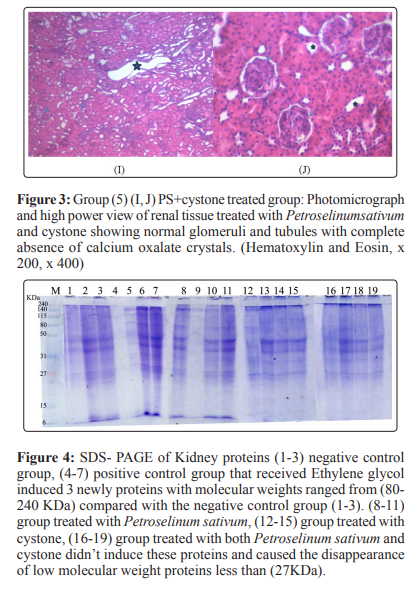
Histopathological analysis revealed no calcium oxalate deposits or other abnormalities in the nephron segments of treated groups with Petroselinum sativum extract group(3) and PS extract+cystone group(5) while the presence of only very few calcium oxalate deposits in the cystone group (4). On the other hand, multiple basophilic calcium oxalate deposits inside the renal tubules and dilation of the proximal tubules along with interstitial inflammation were observed in the renal tissue of urolithiatic rats group(2).
SDS-PAGE for protein: Protein gel electrophoresis showed that the untreated positive control (4-7) group induced 3 newly proteins with molecular weights ranged from (80-240 KDa) compared with the negative control (1-3). On the other hand, treated groups (8-11) PS extract group, (12-15) cystone group, (16-19) PS extract + cystone group caused the disappearance of low molecular weight proteins less than (27 KDa) compared with the negative control group(1-3).
Table 1: Concentration of Sodium, Potassium, Calcium, Magnesium, Creatinine, Urea and Uric acid of different groups
|
Rats Group |
Sodium **(mEq/l) |
Potassium NS (mmol/l) |
Calcium * (mg/dl) |
Magnesium ** (mg/dl) |
Creatinine ** (mg/dl) |
Urea NS (mg/dl) |
Uric acid * (mg/dl) |
|
(1)-ve |
150.3±9.4 |
6.0±0.9 |
47.2±33.4 |
0.89±0.05 |
2.47±0.05 |
16.7±1.0 |
8.5±1.6 |
|
(2)+ve |
154.3±7.7 |
10.6±4.5 |
73.3±16.3 |
2.0±0.43 |
1.7±0.7 |
22.4±5.0 |
8.0±1.4 |
|
(3)PS |
149.3±12.4 |
6.4±1.7 |
72.25±19.4 |
1.06±0.17 |
2.66±0.1 |
15.6±3.6 |
9.3±1.6 |
|
(4)Cystone |
156.3±4.4 |
5.5±3.3 |
79.6±2.7 |
1.01±0.27 |
2.64±0.1 |
17.0±3.7 |
8.8±1.1 |
|
(5)PS+Cystone |
153.2±4.1 |
4.2±1.15 |
70.1±2.6 |
0.97±0.34 |
2.63±0.03 |
16.6±3.1 |
9.3±1.3 |
Discussion
In the present study, hyperoxaluria was induced in rats by employing ethylene glycol (EG). On administration, EG gets absorbed rapidly and metabolized into glycolic acid, glyoxylic acid, and finally converted into glycolate and oxalate by coupling of oxidation-reduction reactions. Glycolic acid oxidase and lactate dehydrogenase are enzymes involved in its metabolic pathways [27].The process of calcium oxalate renal stone formation is started from calcium phosphate plaque. It is located in interstitium which is known as Randall’s plaque [28]. Oxalate is attracted to cations to form insoluble CaOx and soluble magnesium oxalate salts. CaOx salt deposits on Randall’s plaque. Initially calcium oxalate monohydrate (COM) crystal attaches to renal papilla which leads to epithelial damage causing aggregation of crystals [29]. Subsequently, hyperoxaluria leads to nucleation, crystal growth and further aggregation, and ultimately retention in renal tubules [30].The damages in the epithelial cells of renal tubules lead to the production of free radicals that induce heterogenous crystal nucleation and cause aggregation of crystals [31].
It can be speculated that the role of the herb extract of Petroselinum sativum in the prevention of calcium oxalate calculi is due to the antioxidant effects of different compounds of the PS extract 16. The calcium salts are insoluble in physiological pH [32].The herb extract provided the optimum pH which could maintain calcium oxalate particles dispersed in the solution and thus allowed them to be eliminated easily from the kidney. Probably the mechanism of herb extract on nephrolithiasis was related to the increase of diuresis via inhibition of the Na+–K+ pump in renal epithelial cell leading thus to an osmotic water flow into the lumen and diuresis. It has been reported that calcium oxalate calculi may have a bacterial origin [33, 34].
Antimicrobial activity of PS materials against natural microflora may be effective in this mechanism. Ethylene glycol induction produces oxalic acid which leads to the acidosis with ultimate reduction of pH. Due to the acidosis condition, 0.5% of removable calcium on the extra cellular fluid causes the calcium oxalate stones in the kidney and this may elevate the level of Ca2+[35]. Ethylene glycol increases oxalate production by way of increasing substrate availability which induces the activity of oxalate synthesizing liver enzyme, glycolate oxidase [36]. Cystone significantly reversed the ethylene glycol induced urolithiasis, presumably by preventing the urinary supersaturation of lithogenic substances, especially of oxalate and calcium. The reduction of urinary and kidney oxalate levels by cystone may be due to its inhibitory action on oxalate synthesizing liver enzyme glycolate oxidase [37]. It normalizes urinary pH and alleviates burning during urination. Its antimicrobial property combats common urinary pathogens. Its demulcent and anti-inflammatory properties are beneficial in soothing an irritated bladder [38].
In urolithiasis, the glomerular filtration rate decreases due to the obstruction of the outflow of urine by stones in urinary system. Due to this, the waste products particularly nitrogenous substances such as proteins, urea and uric acid accumulate in blood.Serum magnesium level significantly decreases in positive control group compared to treated groups. So that magnesium deficiency accelerates renal stone formation in rats and that the administration of magnesium results in the prevention of calcium oxalate crystallization in their kidneys [39]. Magnesium complexes with oxalate, thus reduces calcium oxalate super saturation in urine [40]. Oral administration of PS extract, cystone and PS+cystone normalized the decreased levels of sodium and potassium electrolytes in the serum when compared with EG treated group [34, 41].
Histopathological examination showed that the ethylene glycol treated rat kidney showed the presence of multiple basophilic calcium oxalate deposits between the renal glomeruli and tubules which caused further widening of the tubules. The dilatation of kidney tubules and the deposition of crystals caused a compression on the glomerulus. In the group (5) of PS+cystone, there was complete absence of calcium oxalate deposits with normal renal glomeruli and tubules. This approved the diuretic effect of PS and cystone. The protein (PAGE) showed that, Proteins formed a discontinuous coat around the crystals ranging in thickness from 10 to 20 nm. Proteins have a strong affinity for CaOx crystals [42]. It has been suggested that newly formed crystals with a macromolecular coat are less likely to dissolve during the routine urinary ionic and pH changes and therein may lay the importance of matrix in stone formation [43].
Some investigators have obtained evidence that oxalate and CaOx crystals may be injurious to renal tubular cells. Cultured renal tubular cells exhibited evidence of damage after exposure to CaOx crystals. Addition of CaOx crystals to monolayers of Madin-Darby canine kidney (MDCK) cells led to a marked increase in the release of lysosomal enzymes, prostaglandin E2, and, to a lesser extent, cytosolic enzymes [44,45]. This membranous debris appears together with focal loss of the brush border from proximal tubular cells and the appearance of proximal tubular enzymes in the urine, suggesting that proximal tubular cells are the source of the membranous debris [46]. It was reported that individual cell injury and generalized cell monolayer injury result in the presentation of different cell surfaces, and that both types of injury result in increased affinity for crystal adhesion.
Conclusion
In conclusion, our results indicated that administration of the ethanolic extract of Petroselinum sativum, at dose of 250 mg/ kg, to rats with ethylene glycol-induced lithiasis, reduced and prevented the growth of urinary stones in different parts of the renal tubules. This may be due to the presence of strong bioactive antioxidant, anti-inflammatory and anti-microbial compounds like saponins, alkaloids, flavonoids, triterpenoids, glycosides and tanines. Ethanolic extract of Petroselinumsativum has antiurolithiatic effect.
References
- Priyanka Kantivan G, Rashmi S, Srivastava, Mayuri S (2013) Urolithiasis: An Overview. International Journal of Pharmaceutical & Biological Archives 4: 1119-1123.
- Narendra V, Ameeta A (2013) Antiurolithiatic effect of Oleanolic acid isolated from Lantana Camara on glycolic acid-induced urolithiasis in rats. International Journal of Experimental Pharmacology 3: 43-46.
- Robertson WG, Peacock M (1980) The course of idiopathic calcium disease: Hypercalciuria or hyperoxaluria. Nephron 26, 105-110.
- Melissa AC, Cadnapaphornchai P (2008) Common surgical disease, 4th edition, Springer, New York 142-183.
- Hardik G, Maunik C, Pinakin J (2015) Diuretic and antiurolithiatic activities of an ethanolic extract of Acoruscalamus L. rhizome in experimental animal models. Journal of Traditional and Complementary Medicine 6: 431-436.
- Andrew PE, Elaine MW, Fredric LC, James W, James EL, et al. (2015) Mechanisms of human kidney stone formation 43: 19-32.
- Jayesh M, Pranjal M, Bipinchandra P, Jyoti B, Suresh K, et al. (2015) Role of Vitamin C and E supplementation in reduction of serum level of renal injury marker following shock wave lithotripsy: Prospective single centre experience. Urol Ann 7: 350-354.
- IA A (2012) Chemoprotection of ethylene glycol monoethyl ether-induced reproductive toxicity in male rats by kolaviron, isolated biflavonoid from Garcinia kola seed. Hum Exp Toxicol 31: 506-517.
- Saeidi J, Bozorgi H, Zendehdel A, Mehrzad J (2012) Therapeutic effects of aqueous extracts of Petroselinumsativum on ethylene glycol-induced kidney calculi in rats. Urol J 9: 3616.
- Robertson WG (2015) Potential role of fluctuations in the composition of renal tubular fluid through the nephron in the initiation of Randall’s plugs and calcium oxalate crystalluria in a computer model of renal function. Urolithiasis 43: 93-107.
- Elaine MW, Andrew PE, Fredric LC, James EL, Amy K, et al. (2013) A test of the hypothesis that oxalate secretion produces proximal tubule crystallization in primary hyperoxaluria typeI. Am J Ren Physiol 305: 00382.
- Saeed RK (2012) Reactive Oxygen Species as the MolecularModulators of Calcium Oxalate Kidney Stone Formation: Evidence from Clinical and Experimental Investigations. J Urol 5: 78.
- Mejdi S, Ameni D, Emira N, Guido F, Adele P, et al. (2015) Chemical composition and antibiofilm activity of Petroselinum crispum and Ocimum basilicum essential oils against Vibrio spp. strains. J MicroPatho11: 4.
- Jesus V, Kim R, Michael G, Nora MO (2015) Antioxidant Activity, Total Phenolic and Total Flavonoid Content in Sixty Varieties of Potato (Solanum tuberosum L.) Grown in Ireland. Potato Research 58: 221-244.
- Mudhir SS, Trifa FI, Falah MA (2015) Anti-Urolithiatic and Anti-Oxidant Effects of Fenugreek on Ethylene Glycol-Induced Kidney Calculi in Rats. Jordan Journal of Biological Sciences 8: 1995-6673.
- Rathod N, Chitme HR, Chandra R (2014) In Vivo And In Vitro Models for evaluating antiurolithiasisactivity of Herbal Drugs. Int J Pharmaceut Res Bio-Sci 3: 309-329.
- Pandey S, Pandey R, Singh R (2014) Phytochemical screening of selected medicinal plant Cinnamon zeylanicum bark extract. Area of research; Uttarakhand, India. Int J Sci Res Publi 4: 4.
- Pawar A T, Vyawahare N S (2015) Protective effect of methanolic extract of Abelmoschus moschatus seeds against calcium oxalate urolithiasis in rats. J Chem Pharmaceut Res 7: 269-278.
- Gehan SE M (2016) Evaluation of anti-urolithiatic effect of aqueous extract of parsley (Petroselinum sativum) using ethylene glycol-induced renal calculi. Wor J Pharmaceut Res 5: 1721-1735.
- Smolinske, Susan C (1992) Handbook of Food, Drug, and Cosmetic Excipients.
- John W E, J Thomas L (2012) Reduced toxicity ethylene glycol-based antifreeze/heat transfer fluid concentrates and antifreeze/ heat transfer fluid concentrates and antifreeze/heat transfer fluids. US8206607 B2.
- Nermien Z A Kamal M El-D, Mahgoub M A (2010) Studies on Curcuma longa, Chicoriumintybus and Petroselinumsativum Water Extracts against Cisplatin-Induced Toxicity in Rats. Journal of American Science 6: 545-558.
- Atul Makasana, Vishavas Ranpariya, Dishant Desai, Jaymin Mendpara, Vivek Parekha, et al. (2014) Evaluation for the antiurolithiatic activity of Launaea procumbens against ethylene glycol-induced renal calculi in rats. Toxicology Reports 1: 46-52.
- Hodgkinson A (1970) Determination of oxalic acid in biological material. J Clini Chem 16: 547-557.
- Shapiro AL, Viñuela E, Maizel JV (1967) Molecular weight estimation of polypeptide chains by electrophoresis in SDS-polyacrylamide gels. Biochem Biophys Res Commun 28: 815-20.
- Jaccard J, Becker M A, Wood G (1984) Pairwise multiple comparison procedures: A review. Psychol Bull 96: 589.
- Apexa BS, Divyesh RM, Manish JB, Seema NB, Chandrabhanu RT, et al. (2014) Evaluation of anti-urolithiatic effect of aqueous extract of Bryophyllum pinnatum (Lam.) leaves using ethylene glycol-induced renal calculi. Avicen J Phytomed 4: 151-159.
- Kim SC, Coe FL, Tinmouth WW (2005) Stone formation is proportional to papillary surface coverage by Randall’s plaque. J Urol 173: 117-119.
- Andrew PE, Elaine MW, Fredric LC, James W, James EL, et al (2015) Mechanisms of human kidney stone formation.Urolithiasis 43: 19-32.
- Saeed RK (2014) Reactive oxygen species, inflammation andcalcium oxalate nephrolithiasis .Trans Androl Urol 3: 256-276.
- Kanu PA, Shifa N, Monica K, Chanderdeep T (2013) Nephrolithiasis: Molecular mechanism of renal stone formation and the critical role played by modulators. BioMed Res Int, Article ID 292953, 21 pages.
- Aml F El, AlaaOA (2013) Nephroprotective and Diuretic effects of three medicinal herbs against gentamicin-induced nephrotoxicity in male rats. Pak J Nutr 12: 715-722.
- Xiangling W, Amy EK, James CW, Xiaojing T, Andrew DR, et al. (2014) Distinguishing characteristics of idiopathic calcium oxalate kidney stone formers with low amounts of Randall’s Plaque. Clin J Am Soc Nephrol 9: 1757-1763.
- Bhavani R, Nandhini S, Rojalakshmi B, Shobana R, Rajeshkumar S, et al. (2014) Effect of noni (morindacitrifolia) extract on treatment of ethylene glycol and ammonium chloride induced kidney disease. Int J Pharma Sci Res l5: 0975-9492.
- Suresh BS, Chitr AC K, Venkat RN, Vasant RC (2014) Antiurolithic activity of aqueous extract of roots of cissampelospareira in albino rats. Asi J Pharmaceut Clini Res l7: 49-53.
- Amol LS, Venkatesh R, QadriS Y H, Arjun LK (2013) Exploring Antiurolithic Effects of Gokshuradi Polyherbal Ayurvedic Formulation in Ethylene-Glycol-Induced Urolithic Rats. Evidence-Based Complement Alternat Med, Article ID 763720, 9 pages.
- Dilip K, Pralhad SP (2010) Evaluation of efficacy and safety of a herbal formulation Cystone in the management of urolithiasis: Meta-analysis of 50 clinical studies. Int J Alternat Med 8: 1-10.
- Jignesh GS, Bharat GP, Sandip BP, Ravindra KP (2012)Antiurolithiatic and antioxidant activity of Hordeumvulgare seeds on ethylene glycol-induced urolithiasis in rats. Ind J Pharmacol 44: 672-677.
- Lakshmi B V S, Divya V (2014) Antiurolithiaticand antioxidant activity of Zingiberofficinalerhizomes on ethylene glycol-induced urolithiasisin rats. Int J Adv Pharma med Bioall sci 2: 148-153.
- Sridhar N, Surya K B V V S, Rudrapal M (2014) Diuretic potential of Aervalanata and Ecboliumligustrinum root extracts. Asi J Pharmaceut Res Heal Care 6: 12-14.
- Aggarwal K P, Tandon S, Singh S K, Tandon (2013) C2D map of proteins from human renal stone matrix and evaluation of their effect on oxalate induced renal tubular epithelial cell injury. Int Brazil J Urol 39: 128-136.
- Khan S R, Hackett R L (1993) Role of organic matrix in urinary stone formation: an ultrastructural study of crystal matrix interface of calcium oxalate monohydrate stones. J Urol 15: 239-245.
- Khan S R (1995) Calcium oxalate crystal interaction with renal tubular epithelium, mechanism of crystal adhesion and its impact on stone development. Urol Res 23: 71-79.
- Wiessner J H, Hasegawa A T, Hung L Y, Mandel N S (1999) Oxalate-induced exposure of phosphatidylserine on the surface of renal epithelial cells in culture. J Am Soc Nephrol 10: 441-445.
- Khan S R, Hackett R L (1993) Hyperoxaluria, enzymuria and nephrolithiasis. Contributions to Nephrology 101: 190-193.
- Khan S R, Finlayson B, Hackett R L (1982) Experimental calcium oxalate nephrolithiasis in the rat. Am J Pathol 107: 59-69.



