Research Article - (2022) Volume 4, Issue 2
Polyphenols from Prunus armeniaca.L as Promising Anticancer (Cervical Cancer): In silico studies and in vivo safety assessment
2Team of Ethnopharmacology and Pharmacognosy, Faculty of Sciences and Techniques Errachidia, , Moulay Ismail University of Meknes, BP 509, Boutalamine, Errachidia, 52000, Morocco
3Molecular Chemistry and Natural Substances Laboratory, Faculty of Science, , Moulay Ismail University of Meknes, Morocco and EST Khenifra, Sultan Moulay Sliman University, Khenifra 23000, Morocco
4LABASE Laboratory, Faculty of Science of Meknes, Moulay Ismail University, Meknes 52202, Morocco
Received Date: Oct 10, 2022 / Accepted Date: Oct 17, 2022 / Published Date: Oct 25, 2022
Copyright: ©Moualij Benaissa. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Moujane Soumia, Bouadid Ismail, Bouachrine Mohammed, Benlyas Mohamed, Filali Zegzouti Younes, Eddouks Mohamed and Moualij Benaissa. (2022) Polyphenols from Prunus armeniaca.L as Promising Anticancer (Cervical Cancer): In silico studies and in vivo safety assessment. Adv J Uro Nephro 4(2): 50-59.
Abstract
Some anogenital tract malignancies have high-risk human papillomavirus (HPV) infections as their etiological cause. Although many HPV preventative vaccines have been licensed, there is still a need for medication that targets the infection and its carcinogenic effects. One of the important elements in cell immortalization and tumor development in HPV-positive cells has been identified as the viral oncoprotein E6. The cellular ubiquitin ligase E6AP interacts with E6, which can facilitate the degradation of the tumor suppressor protein p53. One of the best ways to prevent the maintenance and growth of infected cells is to block the creation of the E6-E6AP complex. The present study aims to determine the ability of polyphenols identified in Prunus armeniaca.L, to target the HPV16 virus by virtual high-throughput screening and molecular docking, and to evaluate the safety of this plant in vivo. In silico, the PDB: 4GIZ structure of E6HPV16 was prepared as a target by Discovery Studio 2021. Virtual screening of 47 polyphenols was performed by the iGEMDOCK program, followed by an evaluation of potential inhibitors based on docking affinities obtained from the The SYBYL-X Surflex-Dock module v2.0, 21. In vivo toxicity studies of Prunus armeniaca. L aqueous extract was also conducted in Wistar rats. Of all the polyphenols investigated in this study, the compounds 3-pCoumaroylquinic, 5-pCoumaroyloquinic, Epicatechin, and Dimethoxyflavone were predicted to have the highest binding affinity for E6HPV16, also revealed several interactions with the E6 binding site area. A study on acute in vivo toxicity of Prunus armeniaca .L aqueous extract was conducted and didn't produce any harmful effects. Moreover, Epicatechin, a dimethoxyflavone from Prunus armeniaca.L, 3-pCoumaroylquinic, 5-pCoumaroylquinic, and 5-pCoumaroyloquinic were chosen as possible E6HPV16 inhibitors for novel medication development.
Keywords
Cervical cancer; E6-E6AP; Docking; Prunus armeniaca.L; ADMET properties; toxicity
Introduction
Human papillomaviruses (HPV) are small, double-stranded, cir-cular DNA viruses of the HPV family. They are part of the Papil-lomaviridae family. There are more than 200 different HPV gen-otypes that infect mucosal and epithelial cells of human skin, a subset of which are tumorigenic[1] .The viral genome consists of seven genes classified as early genes (E1, E2, E3, E4, E5, E6, and E7), which control viral transcription and genome replication, and two Late genes (L1 and L2) encode the structural proteins in¬volved in capsid formation [2]. HR-HPV16 is the major etiologi¬cal factor in most cases of cervical cancer [3]. HR-HPV-induced carcinogenesis depends primarily on the expression of HPV-en-coded E6 and E7 oncogenes, which are synergistically involved in the malignant conversion of infected cells due to their ability to degrade p53 and Rb, respectively [4]. The HPV E6 oncoprotein cooperates with several cellular proteins activating various onco-genic pathways that lead to blockage of apoptosis, changes in the transcriptional machinery, interference with cell-cell interactions, and cell immortalization [5]. HR E6 oncoproteins are involved in the regulation of p53 gene transactivation and can abolish the tran¬scriptional transactivation activity of p53 [6]. HR E6 oncoproteins are also able to interact with p300/CBP co-activators to control p53-dependent gene regulation [7].
HPV E6 proteins are rather small (about 150 amino acids), are cysteine-rich, and share a common architecture consisting of two zinc-binding domains (E6N and E6C) with a conserved fold that is connected by a helical linker [8]. The E6 amino-terminal zinc-binding domain and the carboxy-terminal zinc-binding do-main have a globally conserved fold in the crystal [9]. The two zinc domains, along with an alpha helix tube that connects them, form a deep pocket in which the LXXLL peptide makes close con-tact [10]. LxxLL is a 20 amino acid peptide in E6AP, which results in recruitment and polyubiquitination of p53. The LxxLL peptide isolated from E6AP is sufficient to make E6 susceptible to inter-action with p53 [11]. The central pocket of E6 binds to the LxxLL motif of the ubiquitin ligase E6AP, resulting in a conformational change of the E6 HR proteins allowing the creation of a complex with p53. In this ternary complex, called E6/E6AP/p53 [12, 13].
To date, three prophylactic vaccines have been validated and ef-fectively used to control persistent viral infections and HPV-as-sociated cervical lesions. As a result, the management of cervical cancer and precancerous lesions is still limited to the use of che-motherapeutic agents and/or surgical and ablative techniques to remove developed tumors. Both of these treatments are invasive, non-specific, and tend to be expensive, making their availability limited for millions of patients, usually in developing countries [14]. Therefore, the development of affordable drug therapies targeting the onco-virus is essential for the specific treatment of HPV-related diseases and improved management of cervical can¬cer and precancerous lesions.
The population of Deraa Tafilalt region has great experience in using medicinal plants to fight against various diseases. Indeed, UNESCO has declared this region a biosphere reserve Medicinal and aromatic plants are a real source of phytocompounds that have been used in the treatment of various diseases caused by micro¬organisms. Through numerous pharmacological tests, plants and their derived components have proven their antibiotic, antimitotic, and antiviral activity [15, 16] .
With this in mind, we wanted to determine if medicinal plants are used by the population. We used molecular docking techniques to evaluate the efficacy of Prunus armeniaca. L compounds against E6HPV16. In addition, a safety assessment through acute and sub chronic in vivo studies was performed.
Materials and Methods
In silico studies
Data set
A series of 47 phenolic compounds that were extracted in the plant Prunus armeniaca. L, their molecular structures were obtained from the database pubchem these molecules were considered for the study of in-silico [17].
Preparation of the target
The 3D X-ray crystal structure of Crystal structure of full-length human papillomavirus oncoprotein E6 in complex with LXXLL peptide of ubiquitin ligase E6AP (PDB ID: 4GIZ), was obtained from the Protein Data Bank[18]. They were prepared by Discov¬ery Studio 2020 by removing water molecules and adding polar hydrogen [19]. The structure of 4GIZ was extracted in PDB format and saved for inclusion as a target in docking.
Virtual screening
High-speed virtual screening was performed using the iGEM-DOCK (Generic Evolution Method for Docking) program [20]. The in silico screening of 47 phytocompounds was performed us¬ing the PDB code of the targets (PDB ID: 4GIZ) chain “C”, the screening score, which is based on total energy calculations (total energy = VdW + HBond + electrostatic), was calculated using iG-EMDOCK v2.1.11.
The standard parameters used for screening; population size, gen¬erations, and number of solutions were set at 300, 70 and 2, respec¬tively. Energy-based results were analyzed and 12 potential inhib¬itors were selected based on stability for further detailed analyses.
Prediction of ADMET
To develop a drug, it is necessary to go through several steps, start¬ing with target identification and ending with ADMET prediction. Early detection of these properties is therefore very necessary to decrease the cost and duration of the drug development process. To define the passage of this drug in the organism, an evaluation of pharmacokinetic and ADMET parameters (adsorption, distri¬bution, metabolism, excretion and toxicity) was performed, in this perspective, 16 phenolic compounds that were selected in the screnning step were evaluated to determine these pharmacokinetic parameters in silico using ADMETSAR[21] . and pkCSM, in or¬der to prevent the failure of these compounds in clinical trials and increase their chances of reaching the stage of drug candidates in the future [22].
Molecular docking
Molecular docking is a technique for predicting receptor-ligand interaction in drug discovery. Using this method, several studies have proposed certain molecules as good candidates for treating several pathologies. The ligands 3-pCoumaroylquinic, 5-pCoumaroyloquinic, and Epi-catechin, Dimethoxyflavone, were docked to the 'C' chain of the unit cell of the crystal structure (PDB: 4GIZ). The SYBYL-X Sur-flex-Dock module v2.0, 21 were used to generate bioactive bind-ing positions of ligands in the E6HPV16 active site [23]. After completing docking, the ligand pose gave the minimum binding energy. Discovery studio and PyMOL were used to visualize the results. The results of the molecules that showed an interesting docking score were analyzed and their positioning inside the ac-tive site was compared [19, 24]. The type of interactions estab-lished by each molecule inside the active site was also compared.
In vivo toxicity studies
Plant material
The Prunus armeniaca L. leaves were collected from the Tafilalet region (semi-arid area) of Morocco in May 2022, and air-dried at 40 °C. The plant was taxonomically identified and authenticated, and a voucher specimen was deposited at the herbarium of the Fac¬ulty of Sciences and Techniques of Errachidia under the number PA22.
Preparation of the aqueous extract
The aqueous extract of the plant material was prepared according to the most traditional method used in Morocco (decoction): 1 g of powdered leaves mixed with 100 ml distilled water, was boiled for 10 min and then cooled for 20 min. Thereafter, the aqueous extract was filtered using a Millipore filter (Millipore 0.2 mm, St Quentin en Yvelines, France) to remove particulate matter. Final¬ly, the filtration of the extract was lyophilized in a lyophilizator (LABCONCO, G.BOYER, materiel de laboratoire, Casablanca). Doses administered for acute toxicity were 1, 2 and g freeze-dried aqueous extract per kg body weight [25] .
Experimental animals
Healthy albino adult male rats (Wistar strain) with a weight ranged between 150 and 210 g were housed under standard environmen¬tal conditions (23 ± 1 oC with 55 ± 5% humidity and a 12 h/12 h light/dark cycle) and maintained with free access to water and ad libitum standard laboratory diet.
Acute toxicity study
In the current study, safety assessment was carried as described previously, with slight modifications and in accordance with the or¬ganization for Economic Cooperation and Development (OECD) [26, 27]. Briefly, healthy female rats were treated orally by a limit dose of 2000 mg/kg of Prunus armeniaca aqueous extract (PAAE). Firstly, one overnight fasted rat was treated with PAAE (2000 mg/ kg body weight) then mortality and signs of toxicity were moni¬tored hourly follow-up for three hours after the dosing and then periodically throughout 48 h. If the first rat dosed survived sequen¬tially other rats were treated, and finally observation was made for 14 days. If three or more rats survived, the LD50 was predicted to be above 2000 mg/kg. All applicable guidelines for the care and use of animals were followed (FSTE/2015) [28].
Sub-chronic toxicity study
Twelve female Wistar rats were divided into two groups and their weights were recorded. The first group received 2000 mg/kg body weight of PAAE orally once daily for 28 days. The control group received distilled water. During the experimental period, toxic manifestations and mortality were observed. After 28 days of treat¬ment all animals were fasted overnight, body weight was recorded and all rats were anaesthetized, and blood samples were collected for biochemical analyses and blood pressure parameters measure¬ment. Additionally, heart, lungs, liver, spleen and kidneys were dissected and weighed at the end of the experiment.
Relative organ weight
After sacrificed by cervical dislocation, organs were weighed and their index in relation to body weight was calculated as described in: ROW (g) = weight of organ/bodyweight of rats on the day of sacrifice × 100% [29].
Effect on liver enzymes and chlorides
Serum alanine aminotransferase (ALT) activity was estimated us¬ing the modified kinetic method of Srivastava et al., using a kit sup¬plied by SGM, Italia, according to the instructions of the supplier. Serum aspartate aminotransferase (AST) activity was assessed us¬ing the modified kinetic method of Schumann and Klauke, using a kit supplied by SGM, Italia, according to the instructions of the supplier. Serum chlorides were estimated using SGM, Italia, ac¬cording to the instructions of the supplier [30].
Blood pressure measurement
For estimation of blood pressure parameters, systolic blood pres¬sure (SBP), mean blood pressure (MBP), and heart rate (HR) were measured using a tail-cuff and a computer-assisted monitoring de¬vice (Harvard, Boyer, Casablanca, Morocco) as described previ¬ously [31]. In brief, Systolic blood pressure (SBP), mean blood pressure (MBP) and heart rate (HR) were measured directly using pulse tracing while diastolic blood pressure (DBP) was calculated from SBP and MBP using the formula: DBP= (3MBP-SBP)/2.
Glycemia determination
Blood glucose levels were determined by the glucose oxidase method using a reflective glucometer (Contour™ TS) from Bayer Diabetes Care (ref).
Statistical analysis
Data were expressed as mean ± SEM. Statistical differences among the means studied were assessed by two-way ANOVA with GraphPad Prism 6 software. Differences were considered to be significant when p<0.05. Whereas, data obtained from the relative organs weights were analyzed using unpaired sample T-test at a 95% confidence interval, with Welch’s correction.
Results
In silico studies
Virtual screening
In the present study 47 phytocompounds of Prunus armeniaca. L was screened against the E6 HPV16 protein. In this step, 16 mol-ecules were selected based on their binding affinity to E6 HPV16 protein for further evaluation (Table 3).
Prediction of ADMET
Lipinski's Rule of Five (Ro5) prediction results of these com-pounds are shown in Table 1. The results of logP values and molec¬ular weights of all designed compounds except 3-O-Caffeoylqui-nic compound, indicating that they have reasonable absorption and are moderately soluble in water, were in perfect agreement with the most important rules of drug similarity. In addition, the HBA of Quercetin-3- O - glucoside, Quercetin-3-O_6-acetyl-glu-coside, Kaempferol-3- O - rutinoside,Proanthocyanidin A2,Pro-cyanidin B1,Procyanidin B2, Quercetin-3-O_6-acetyl-gluco-side, Kaempferol-3- O - rutinoside. HBD of Chlorogenic acid, Cis-5-Caffeoylquinic acid, Neochlorogenic acid the number of ro¬tational bonds of Kaempferol-3- O - rutinoside indicated that these eleven compounds do not conform to Lipnski's rules (Table 1).
Table 1: Molecular properties for predicting the drug sensitivity of the potential inhibitors.
|
Compound name |
MW |
Log P |
HB Acceptor |
HB donor |
Rotating bonds |
|
Procyanidine B1 |
578.526 |
2.995 |
12 |
10 |
3 |
|
Épicatéchine |
290.271 |
1.5461 |
6 |
5 |
1 |
|
Chlorogenic acid |
364,39 |
-0.4 |
8 |
6 |
4 |
|
Cis-5-Caffeoylquinic acid |
354.311 |
-0.4 |
9 |
6 |
5 |
|
Neochlorogenic acid |
354.311 |
-0.6459 |
8 |
6 |
4 |
|
5-p-Coumaroylquinic acid |
338.312 |
-0.3515 |
8 |
5 |
5 |
|
5-oCaffeoylquinic acid |
354.311 |
-0.6459 |
9 |
6 |
5 |
|
3-p-Coumaroylquinic acid |
338.312 |
-0.3515 |
8 |
5 |
5 |
|
3-OCaffeoylquinic |
678.599 |
0.4 |
8 |
4 |
4 |
|
Kaempférol-3- O - rutinoside |
594.5 |
-0.9 |
15 |
9 |
6 |
|
Acide 4-O-caféoylquinique |
354.31 |
-0.6459 |
8 |
6 |
4 |
|
Caféoyl-glucoside |
342.3 |
-1.5459 |
9 |
6 |
4 |
|
caryophyllene |
204.357 |
4.7252 |
0 |
0 |
0 |
|
Quercetin |
302.23 |
1.5 |
7 |
5 |
1 |
|
Rosmarinic |
360.3 |
2.4 |
8 |
5 |
7 |
|
Dimethoxyflavone |
282.29 |
2.2 |
4 |
0 |
3 |
The results of the predictions of absorption, distribution, metab-olism, excretion and toxicity are presented in Table 2. For the pkCSM predictive model, the compounds 3-pCoumaroylquinic, 5-pCoumaroyloquinic, and Epicatechin, caryophyllene, Querce-tin, Dimethoxyflavone show good absorption and excellent dis-tribution properties, this could be envisioned as permeable mol-ecules with low distribution in the brain. They also have good clearance and no inhibition of the hERG system or AMES toxicity.
The cytochrome P450 subtypes CYP2D6 and CYP3A4 indicate that 3-pCoumaroylquinic, 5-pCoumaroyloquinic, and Epicate¬chin, caryophyllene, Quercetin, Dimethoxyflavone, could not be substrates or inhibitors for the two major subtypes, and therefore probably could not be metabolized, resulting in a low risk of drug interactions. We suggest that they are promising inhibitors and were selected for the docking.
Table 2: ADMET in silico prediction of identified E6HPV16 inhibitors
|
|
Épicatéchine |
5-p-Coumaroylquinic acid |
3-p-Coumaroylquinic acid |
Dimethoxyflavone |
|
Absorption and Distribution |
||||
|
Blood-brain barrier (logBB) |
-1.054 |
-1.16 |
-1.16 |
0.429 |
|
Intestinal absorption (human) |
68.829 |
43.925 |
43.925 |
97.629 |
|
Caco-2 permeability |
-0.283 |
-0.656 |
-0.656 |
1.33 |
|
P-glycoprotein substrate |
Yes |
yes |
yes |
yes |
|
P-glycoprotein inhibitor |
no |
no |
no |
no |
|
Metabolism |
||||
|
Substrat CYP2D6 |
no |
no |
no |
no |
|
Substrat CYP3A4 |
no |
no |
no |
yes |
|
Inhibiteur CYP2D6 |
no |
no |
no |
no |
|
Inhibiteur CYP3A4 |
no |
no |
no |
yes |
|
Excretion and Toxicity |
||||
|
Clearence |
0.183 |
0.453 |
0.453 |
0.561 |
|
hERG I inhibitor |
no |
no |
no |
no |
|
Carcinogens |
no |
no |
no |
no |
|
AMES toxicity |
no |
no |
no |
no |
|
Hepotoxicity |
no |
no |
no |
no |
Molecular docking
The 4 compounds with the highest affinity selected after the virtu¬al screening approach were docked to the HPV16 E6 active site. Based on their score values, these molecules were scored and ranked. Drugs with docking scores between -6.2 and -6.7 were considered interesting compounds that can be proposed as poten¬tial candidates to inhibit the HPV16 E6 receptor The 4 candidates with the highest docking score are presented in Table 3. The posi¬tioning of these 4 molecules with HPV16 E6 was visualized.
Table 3: Docking results showing binding affinities of phytocompounds and established hydrogen interactions with amino acids.
|
Compound name |
PubChem CID |
Hydrogen bonds |
Total Energy |
Binding affinity |
|
3-p-Coumaroylquinic acid |
9945785 |
Ser(74), Ser(71), Cys(51), Tyr(70), Tyr(32) |
-70.7856 |
-6.7 |
|
5-pCoumaroyloquinic |
90478782 |
Gln(107), Tyr(32), Ser(74) |
-73.7024 |
-6.9 |
|
Épicatéchine |
72276 |
Cys(51), Ser(74), |
-66.5173 |
-6.2 |
|
Dimethoxyflavone |
88881 |
Tyr(32) |
-66.6708 |
-6.6 |
Visualization and analysis of results
we observed that all four candidate compounds share common interactions and are involved in H-bond type interactions with Ser(74), Cys(51), Tyr(32) residues, which is consistent with recent studies [32]. Figure 1.
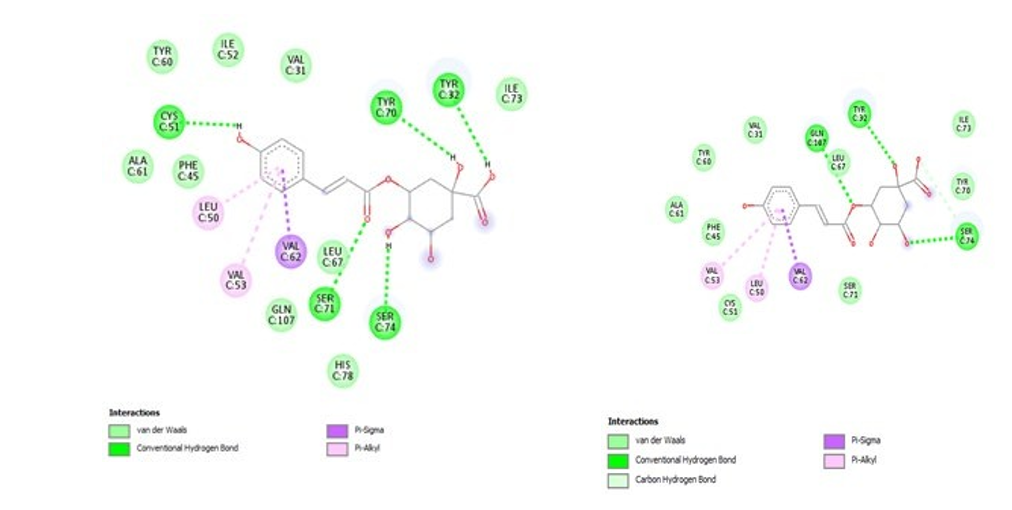
3-p-Coumaroylquinic acid 5-pCoumaroyloquinic
Épicatéchine Dimethoxyflavone
Figure 1: Types of interactions between the E6HPV16 active site and selected potent Prunus armeniaca L inhibitors.
In vivo toxicity studies
Acute toxicity
Prunus armeniaca aqueous extract (PAAE) was given orally once at a dose of 2000 mg/kg b.w without producing any toxicity symp-toms or fatalities. Additionally, there was no significant difference in body weight between treated and control rats. However, it was shown that this herb's LD50 value was greater than 2000 mg/kg.
Sub-chronic toxicity
Sub-chronic toxicity Daily oral administration of PAAE at a dose of 2000 mg/kg b.w for 28 days did not cause any toxicity signs or death. When compared to the control group, also the body weight of the treated rats did not increase significantly, in addition to relative organs weight (ROW) of heart, Kidneys, Spleen. In contrast, the ROW of Lungs (p<0.05) and Liver (p<0.0001) were significantly increased in PAAE treat¬ed group (Table 4). Table 5 represents the biochemical and blood pressure parameters of the treated and control groups. The results showed that PAAE at a high dose of 2000 mg/kg b.w did not sig¬nificantly alter systolic, mean and diastolic blood pressure, heart rate, chlorides, plasma AST and ALT levels by compared to the untreated group. However, blood glucose level (p<0.05) slightly decreased significantly after 28 days of dosing.
Table 4: Body weight and relative organ weights of rats treated orally with PAAE.
|
groups |
Day |
Body weight (g) |
Heart (%) |
Lungs (%) |
Kidneys (%) |
Liver (%) |
Spleen (%) |
|
Control |
D0 |
186.2 ± 27.71 |
0.34 ± 0.02 |
0.62 ± 0.06 |
0.59 ± 0.05 |
2.77 ± 0.19 |
0.22 ± 0.04 |
|
D28 |
201.4 ± 18.82 |
||||||
|
PA 2 g/kg |
D0 |
166.66 ± 3.5 |
0.41 ± 0.03 |
0.92 ± 0.08* |
0.7 ± 0.03 |
3.85 ± 0.08**** |
0.23 ± 0.02 |
|
|
181.66 ± 6.72 |
||||||
Values expressed as mean ± SEM, n=6. ****p<0.0001.
Table 5: Biochemical values and blood pressure parameters of rats treated with PAAE for 28 days.
|
Biological parameter |
Day |
Control |
PAAE 2 g/Kg |
|
Glycemia (mg/dL) |
D0 |
96.6±10.06 |
102.00±5.69 |
|
D28 |
97.80±8.04 |
76.33±1.60* |
|
SBP |
D0 |
130.25±8.99 |
127.83±1.88 |
|
D28 |
132.5±14.93 |
119.22±1.81 |
|
|
MBP |
D0 |
110.75±12.28 |
96.83±2.00 |
|
D28 |
115.75±15.52 |
92.5±4.41 |
|
|
DBP |
D0 |
101±14.74 |
81.33±2.68 |
|
D28 |
107.38±16.11 |
77.33±6.17 |
|
|
HR |
D0 |
293.25±19.65 |
298.8±15.79 |
|
D28 |
302.25±3.77 |
297.6±12.77 |
|
|
AST |
D0 |
20.97±5.75 |
28.15±2.07 |
|
D28 |
26.19±8.61 |
37.68±3.27 |
|
|
ALT |
D0 |
30.96±5.26 |
23.54±1..93 |
|
D28 |
26.31±6.58 |
18.26±1.43 |
|
|
Chlorides |
D0 |
76.70±13.70 |
102.15±13.13 |
|
D28 |
84.39±14.81 |
109.50±12.18 |
Values expressed as mean ± SEM, n=6. *p<0.05.
Discussion
HPV is the agent responsible for the development of cervical cancer in women. It is among the most severe and deadly malignancies in women that we find, the formation of complexes between the HPV oncoprotein, E6 with the cellular ubiquitin ligase E6AP allows to trigger the implementation of modulation, such as attenuation of telomere shortening, immortalization, host cell differentiation, control of cellular pathways, regulation of growth factors, degra¬dation and inactivation of tumor suppressors, disruption of DNA repair efficiency and apoptosis and facilitate cell transformation and hTERT gene increment, Therefore, suppression of the creation of the E6-E6AP complex is one of the essential strategies to inhibit the survival and proliferation of infected cells [33]. Although sur¬gery, radiation therapy, hormone therapy, combination chemother¬apy and immunotherapy as well as the validation of several pro¬phylactic vaccines, there is a lack of effective prognosis, therefore there is no effective treatment for persistent HPV infection. On the other hand Medicinal plants have received much attention to treat many diseases, studies have explored the role of plant metabolites in cancer treatment, and these metabolites may be one of the solu¬tions to target drugs against HPV induced cancers. In our present study, we screened 47 polyphenols from P. Armeniaca L for their efficacy in targeting E6HPV16 using an in silico approach. In a previous study, the authors revealed the beneficial action of Prunus Armeniaca L seed extract used in liver cancer therapy[34] . Based on their total binding energy, this step allowed us to select 16 mol¬ecules. These molecules were selected as the compounds with the highest stability and affinity (binding energy higher than -50.8508 and lower than -93.6087kcal/mol). According to Lipinski, a drug with promising potential is one that meets the five adopted rules, namely, Log P partition coefficient less than 5, weight (MW) less than 500 Da, HBA number<10, HBD number<5, and rotational bonds less than 10. Among the 15 molecules, 3-pCoumaroylqui-nic, 5-pCoumaroyloquinic, and Epicatechin, Dimethoxyflavone, have chemical and physical properties that allow them to be used hand, the other molecules indicated that these 12 compounds do not comply with the Lipinski rules revealing violations towards OR5. The 4 compounds selected by the virtual screening approach and showed the best pharmacokinetic profiles were docked to the active site of E6HPV16, using Autodock vina and MGL Tools programs, the results of the current study showed a higher dock¬ing score (-6.2and -6.9) of the selected polyphenols and revealed several interactions with the active site of E6HPV16. Compound toxicology testing is a crucial step in the development of pharma¬ceutical products. Preclinical toxicity tests reveal adverse or tox¬ic effects specific to the species and the dose of an experimental product [35]. Acute toxicity tests provide preliminary information on the toxic nature after administration of a single dose of a test substance to determine the dose that will cause serious toxicolog¬ical effects that occurs either immediately or at a short-term. They also serve to provide information on doses that should be used in subsequent studies, such that the estimation of LD50 is critical in carrying out toxicological investigations on chemicals, including plant extracts [36, 37]. In the present study, female Wistar rats re¬ceived a single oral dose of 2000 mg/kg b.w of PAAE, and signs of toxicity and mortality were observed for 14 days. During this pe¬riod, the results revealed no deaths, changes in animal behavior or signs of toxicity developed in both the control and treated group. Therefore, the LD50 of PAAE was above 2000 mg/kg b.w. As per UN Classification, any substance which has oral LD50 of more than 2000 mg/kg b.w is considered as low hazard potential and cat¬egorized as UN 6.1 PG III [38]. Sub-chronic studies aim to assess adverse effects of continuous or repeated exposure of plant ex¬tracts or compounds over for a specified period up to the expected lifespan of the test species. They provide information on general characteristics of the toxicity, the toxicity to specific target organs, responses to toxic metabolites formed in the organism, delayed re¬spon [39]. To achieve this goal a sub-chronic study was established at the same dose of the extract during 28 days of daily treatment.
Similarly, the sub-chronic oral administration of PAAE caused no mortality and no behavioral changes in the animals throughout the 28-day study period. Thus, no change in body weight during the acute (14 days) and sub-chronic (28 days) toxicity study was ob-served. Likewise, changes in body weight have been used as an indicator for detecting adverse effects of drugs and chemicals [40]. Significant changes in relative organ weights are considered a rela-tive indicator of potential toxicity of the substance[41]. The results of this study revealed that the relative organs weights (ROW) of the heart, kidneys, spleen in the treated groups were not signifi-cantly different. In contrast, the ROW of Lung and Liver were sig-nificantly increased in PAAE treated (2000mg/kg b.w) group when compared to the control group, that may reflect hypertrophy of this plant at this high dose used. Assessment of biochemical parame¬ters is critical in assessing organ function, most especially kidney and liver. They have significant roles as a marker because of their response to clinical signs and symptoms produced by toxicants. Evaluation of hepatic and renal function is of prime importance to assess the toxic properties of extracts and drugs [42]. Non-signifi-cant differences were seen in biochemical parameters (AST, ALT, and chloride values) except mean values of serum glucose showed significant decrease compared with control. The result is consis¬tent with previous studies which showed that polyphenol-rich Prunus armeniaca leaf extract has an ant-diabetic effect by inhib¬iting α-glucosidase and α-amylase [43]. However, further studies need to be conducted on the effect of PAAE on the liver and lungs. Blood pressure parameters had no impact after 28 days from treat¬ment, indicating its safety on the cardiovascular system despite the high dose used. De plus, la dose utilisée dans l'activité antihyper-tensive précédemment démontrée était de 100 mg/kg b.w, soit 20 fois inférieure à la dose utilisée dans les études de toxicité [43].
Conclusions
By applying the in silico approach to detect protein-ligand inter-actions, we identified four potential candidates with the highest binding energy scores that would be potential inhibitors of HR E6 oncoproteins, presumably without significant side effects. Further-more, this study highlights the value of using the virtual screening approach as a time- and cost-efficient strategy to identify chemi-cals with potential biological effects as well as Administration of an aqueous extract of Prunus armeniaca (PAAE) did not result in mortality or clinically significant changes in the biological param-eters tested, with the exception of hypoglycemia, and the relative organ weights (ROW) of the lungs and liver were significantly in-creased during 28 days of PAAE administration.
Author Contributions: All authors have confirmed that they con-tributed to the intellectual content of this article and have met the following criteria: significant contributions to the conception and design, data acquisition, or data analysis and interpretation; drafting or revising the article for intellectual content; and final approval of the published article. Author involvement by task: M.S. and B.S. designed, developed, and implemented the study; M.S., B.S., F.Z.Y., and M.S., B.S., F.Z.Y., and B.M. developed the methodology; M.S., B.M., and M.B. (Moualij Benaissa) wrote the original version; M.S. and B.S. validated the obtained data; B.S., M.S., B.M., M.B. (Moualij Benaissa) and M.B. (Mohammed Bouachrine) analyzed and validated the data; all authors reviewed, edited, and approved the submitted article; B.M., M.B. (Moual¬ij Benaissa), and M.B. (Mohammed Bouachrine) supervised the study. All authors read and approved the published version of the manuscript.
Data Availability Statement: No new data were created or ana¬lyzed in this study. Data sharing is not applicable to this article.
Acknowledgments: The authors would like to thank the anony¬mous reviewers for their valuable comments and suggestions to improve the paper’s quality.
Conflicts of Interest: We wish to confirm that there are no known conflicts of interest associated with this publication, and there has been no significant financial support for this work that could have influenced its outcome.
References
- Buck, C. B., Day, P. M., & Trus, B. L. (2013). The papilloma-virus major capsid protein L1. Virology, 445(1-2), 169-174.
- Burk, R. D., Chen, Z., & Van Doorslaer, K. (2009). Human papillomaviruses: genetic basis of carcinogenicity. Public health genomics, 12(5-6), 281-290.
- Peng Guan,Rebecca Howell-Jones,Ni Li,Laia Bruni, Silvia de Sanjosé, et al., (2012) Human papillomavirus types in 115,789 HPV-positive women: A meta-analysis from cervical infection to cancer, Int. J. Cancer, 131(2349-2359).
- Yim, E. K., & Park, J. S. (2005). The role of HPV E6 and E7 oncoproteins in HPV-associated cervical carcinogenesis. Cancer research and treatment: official journal of Korean Cancer Association, 37(6), 319-324.
- Chakrabarti, O., & Krishna, S. (2003). Molecular interactions of ‘high risk’human papillomaviruses E6 and E7 oncoproteins: implications for tumour progression. Journal of biosci-ences, 28(3), 337-348.
- Pim, D., Storey, A., Thomas, M., Massimi, P., & Banks, L. (1994). Mutational analysis of HPV-18 E6 identifies domains required for p53 degradation in vitro, abolition of p53 transactivation in vivo and immortalisation of primary BMK cells. Oncogene, 9(7), 1869-1876.
- Patel, D., Huang, S. M., Baglia, L. A., & McCance, D. J. (1999). The E6 protein of human papillomavirus type 16 binds to and inhibits co-activation by CBP and p300. The EMBO journal, 18(18), 5061-5072.
- Kumar, A., Rathi, E., Hariharapura, R. C., & Kini, S. G. (2020). Is viral E6 oncoprotein a viable target? A critical analysis in the context of cervical cancer. Medicinal research reviews, 40(5), 2019-2048.
- Nominé, Y., Masson, M., Charbonnier, S., Zanier, K., Ristri-ani, T., Deryckère, F., ... & Travé, G. (2006). Structural and functional analysis of E6 oncoprotein: insights in the molecular pathways of human papillomavirus-mediated pathogene-sis. Molecular cell, 21(5), 665-678.
- Pol, S. B. V., & Klingelhutz, A. J. (2013). Papillomavirus E6 oncoproteins. Virology, 445(1-2), 115-137.
- Martinez-Zapien, D., Ruiz, F. X., Poirson, J., Mitschler, A., Ramirez, J., Forster, A., ... & Zanier, K. (2016). Structure of the E6/E6AP/p53 complex required for HPV-mediated degradation of p53. Nature, 529(7587), 541-545.
- Zanier, K., Charbonnier, S., Sidi, A. O. M. H. O., McEwen, A. G., Ferrario, M. G., Poussin-Courmontagne, P., ... & Travé, G. (2013). Structural basis for hijacking of cellular LxxLL motifs by papillomavirus E6 oncoproteins. Science, 339(6120), 694-698.
- Sailer, C., Offensperger, F., Julier, A., Kammer, K. M., Walker-Gray, R., Gold, M. G., ... & Stengel, F. (2018). Structural dynamics of the E6AP/UBE3A-E6-p53 enzyme-substrate complex. Nature communications, 9(1), 1-12.
- Hampson, L., Martin-Hirsch, P., & Hampson, I. N. (2015). An overview of early investigational drugs for the treatment of human papilloma virus infection and associated dysplasia. Expert opinion on investigational drugs, 24(12), 1529-1537.
- Eddouks, M., Ajebli, M., & Hebi, M. (2017). Ethnopharma-cological survey of medicinal plants used in Daraa-Tafilalet region (Province of Errachidia), Morocco. Journal of ethnopharmacology, 198, 516-530.
- Barreca, D., Gattuso, G., Bellocco, E., Calderaro, A., Trom-betta, D., Smeriglio, A., ... & Nabavi, S. M. (2017). Flava-nones: Citrus phytochemical with healthâ?ÂÂpromoting properties. BioFactors, 43(4), 495-506.
- PubChem, « PubChem ». https://pubchem.ncbi.nlm.nih.gov/ (consulté le 9 juillet 2020).
- R. P. D. Bank, RCSB PDB: Homepage
- « BIOVIA Discovery Studio | System Requirements for Discovery Studio 2020 ».
- « iGEMDOCK ». http://gemdock.life.nctu.edu.tw/dock/igem-dock.php (consulté le 11 mai 2020).
- Yang, H., Lou, C., Sun, L., Li, J., Cai, Y., Wang, Z., ... & Tang,Y. (2019). admetSAR 2.0: web-service for prediction and optimization of chemical ADMET properties. Bioinformatics, 35(6), 1067-1069.
- Pires, D. E., Blundell, T. L., & Ascher, D. B. (2015). pkCSM: predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. Journal of medicinal chemistry, 58(9), 4066-4072.
- Jain, A. N. (2007). Surflex-Dock 2.1: robust performance from ligand energetic modeling, ring flexibility, and knowledge-based search. Journal of computer-aided molecular design, 21(5), 281-306.
- « PyMOL | pymol.org ». https://pymol.org/2/ (consulté le 12 mai 2020).
- Ajebli, M., & Eddouks, M. (2017). Buxus sempervirens L improves streptozotocin-induced diabetes mellitus in rats. Cardiovascular & Haematological Disorders-Drug Targets (Formerly Current Drug Targets-Cardiovascular & Hematological Disorders), 17(2), 142-152.
- Ahmad, M., Lim, C. P., Akowuah, G. A., Ismail, N. N.,Hashim, M. A., Hor, S. Y., ... & Yam, M. F. (2013). Safety assessment of standardised methanol extract of Cinnamomum burmannii. Phytomedicine, 20(12), 1124-1130.
- OECD, Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure. OECD, 2022. doi: 10.1787/9789264071049-en.
- Mohamed, E. A. H., Lim, C. P., Ebrika, O. S., Asmawi, M. Z., Sadikun, A., & Yam, M. F. (2011). Toxicity evaluation of a standardised 50% ethanol extract of Orthosiphon stamineus. Journal of ethnopharmacology, 133(2), 358-363.
- Wu, J. Y., Chan, Y. C., Guo, H., Chen, Y. J., Liu, Y. X., Yi, H.,& Yu, Z. L. (2020). Twenty-four-week oral dosing toxicities of Herba Siegesbeckiae in rats. BMC complementary medicine and therapies, 20(1), 1-9.
- Schumann, G., & Klauke, R. (2003). New IFCC reference procedures for the determination of catalytic activity concentrations of five enzymes in serum: preliminary upper reference limits obtained in hospitalized subjects. Clinica chimica acta, 327(1-2), 69-79.
- Bouadid, I., Akdad, M., & Eddouks, M. (2022). Antihyperten-sive Activity of Prunus armeniaca in Hypertensive Rats. Cardiovascular & Hematological Agents in Medicinal Chemistry.
- Nabati, F., Moradi, M., & Mohabatkar, H. (2020). In silico analyzing the molecular interactions of plant-derived inhibitors against E6AP, p53, and c-Myc binding sites of HPV type 16 E6 oncoprotein. Molecular Biology Research Communications, 9(2), 71.
- Luo, G. G., & Ou, J. H. J. (2015). Oncogenic viruses and cancer. Virologica Sinica, 30(2), 83-84.
- Hosny, S., Sahyon, H., Youssef, M., & Negm, A. (2021). Prunus Armeniaca L. Seed Extract and Its Amygdalin Containing Fraction Induced Mitochondrial-Mediated Apoptosis and Autophagy in Liver Carcinogenesis. Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Cancer Agents), 21(5), 621-629.
- Parasuraman, S. (2011). Toxicological screening. Journal of pharmacology & pharmacotherapeutics, 2(2), 74.
- Asare, G. A., Addo, P., Bugyei, K., Gyan, B., Adjei, S., Otu-Nyarko, L. S., ... & Nyarko, A. (2011). Acute toxicity studies of aqueous leaf extract of Phyllanthus niruri. Interdisciplinary toxicology, 4(4), 206.
- Nalimu, F., Oloro, J., Peter, E. L., & Ogwang, P. E. (2022). Acute and sub-acute oral toxicity of aqueous whole leaf and green rind extracts of Aloe vera in Wistar rats. BMC complementary medicine and therapies, 22(1), 1-14.
- Patel, A. G., Nariya, M. B., & De, S. (2018). Acute toxicity and repeated dose 28-day oral toxicity study of metriviv syrup in female rats. Ayu, 39(2), 107.
- K. Yuet Ping, I. Darah, Y. Chen, S. Sreeramanan, et S. Sasidha-ran, « Acute and Subchronic Toxicity Study of Euphorbia hir-ta L. Methanol Extract in Rats », BioMed Res. Int., vol. 2013,p. 1-14, 2013, doi: 10.1155/2013/182064.
- El Hilaly, J., Israili, Z. H., & Lyoussi, B. (2004). Acute andchronic toxicological studies of Ajuga iva in experimental an-imals. Journal of ethnopharmacology, 91(1), 43-50.
- Kluwe, W. M. (1981). Renal function tests as indicators of kidney injury in subacute toxicity studies. Toxicology and Applied Pharmacology, 57(3), 414-424.
- Loha, M., Mulu, A., Abay, S. M., Ergete, W., & Geleta, B. (2019). Acute and subacute toxicity of methanol extract of Syzygium guineense leaves on the histology of the liver and kidney and biochemical compositions of blood in rats. Evidence-Based Complementary and Alternative Medicine, 2019.
- Wojdylo, A., & Nowicka, P. (2021). Profile of phenolic compounds of Prunus armeniaca L. leaf extract determined by LC-ESI-QTOF-MS/MS and their antioxidant, anti-diabetic, anti-cholinesterase, and anti-inflammatory potency. Antioxi-dants, 10(12), 1869.



