Research Article - (2024) Volume 2, Issue 9
Photons do not Exist, Light does not Move, it is Transmitted by Electromagnetic Resonance
Received Date: Sep 01, 2024 / Accepted Date: Oct 01, 2024 / Published Date: Oct 10, 2024
Copyright: ©Â©2024 Luis Rodolfo Cabrera Juarez. This is an open- access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Juarez, L. R. C. (2024). Photons do not Exist, Light does not Move, it is Transmitted by Electromagnetic Resonance. Int J Med Net, 2(9), 01-03.
Abstract
A new theory about light is presented, according to which it does not move in space, like particles or self-sustaining waves. It is transmitted as a resonance phenomenon between two tuned electromagnetic oscillators. This is demonstrated by using the helical model of the electron to calculate the frequency of the first quantum transition of the Lymann series of the emission spectrum of the hydrogen atom.
Introduction
Historically, light has been considered as a material flow of particles or waves that propagate through space carrying energy.The contradictions between both models led to the adoption of a hybrid approach called “wave-particle” that currently prevails,and which postulates the existence of “photons”, luminous particles with wave characteristics, and that travel in space at a constant speed, independent of the chosen reference system. However, this model cannot explain certain phenomena such as the Doppler-Fizeau effect , the displacement of the frequency of light according to the speed of distance from its source with respect to the receiver, nor the “double slit” phenomenon.
In this article, a model of light transmission is proposed as a phenomenon of electromagnetic resonance, between two coupled or tuned oscillators, in the same way that occurs in the lower part of the electromagnetic spectrum, used for the transmission of information. For this, the helical model of the electron is used, postulated since the third decade of the 20th century by various authors and the frequency of the first quantum transition of the Lymann series of the emission spectrum of the atom is calculated. of hydrogen, with an accuracy greater than 90% [1].
Deduction of the Helical Model of the Electron
The simplest electromagnetic oscillator consists of two elementary electric charges, of opposite sign, between which there must be equality of electric and magnetic forces. The electric force F e between two particles is expressed
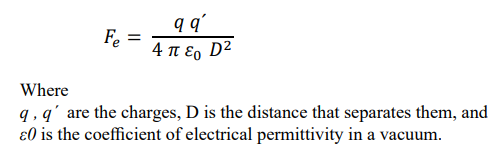
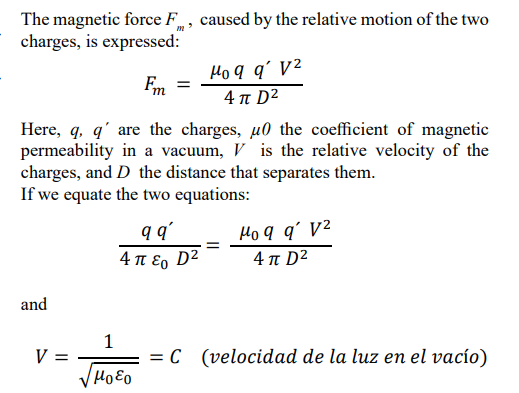
This result indicates that in the electromagnetic oscillator composed of the two elementary charges, they must move with a relative speed equal to that of light, to remain in equilibrium. The most basic electromagnetic oscillator, composed of two elementary charges, is the hydrogen-1 atom, in which an electron, with an electric charge of – 1.6 x 10 -19 C, moves around a proton, whose charge is + 1.6 x l0 -19 C.
If we accept that the electron moves around the proton in a circular orbit with a constant tangential speed C, of approximately 3 x 10 8 m/s, what will be its radius of gyration Re and its frequency of rotation f e ?
According to Bohr's theory, the centripetal force that keeps the electron rotating in a stable circular orbit is equal to the electrical force of attraction between said electron and the proton that constitutes the nucleus.
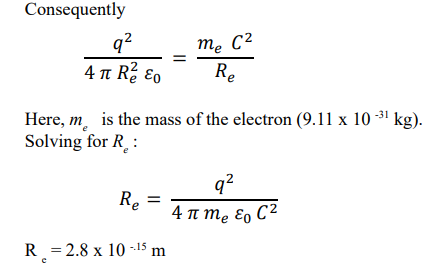
This is the value of the “classical electron radius”, which has been calculated and interpreted in various ways.
Now, Bohr's theory establishes that the radius of gyration (α0) of the electron around the nucleus, in the hydrogen atom , in its basal state, is equal to 5.31 x 10 -11 m .
This inconsistency can be solved by postulating the following:
The electron moves around the nucleus following a helical path, defined by its motion in a principal circular orbit, with a radius of gyration α0 , at a tangential velocity Vt , and by a circular motion perpendicular to the principal orbit, with a radius of gyration Re and a tangential speed Ve. The total speed of the electron is equal to C (speed of light in vacuum).
Since the two trajectories are perpendicular to each other, we can define this equation:

This is the value of the “ fine structure constant ”, which has been interpreted in various ways, but originally had the meaning that we have expressed: the numerical relationship between the tangential velocity in its first circular orbit, and the speed of light.
With the data obtained, we can define the parameters of the electromagnetic resonator made up of the proton and the electron of the hydrogen atom , which is the simplest that we can conceive.
For every electromagnetic resonator, its resonance frequency ( fr) is defined by the equation
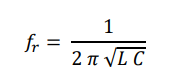
where L is the inductance and C is the capacitance, measures of the capacity to store energy in the form of a magnetic and electric field, respectively, in a physical system. The inductance L , at a macroscopic level, is a function of the magnetic permeability constant ( µ ) of the system, and a geometric factor. The same is valid for the capacitance C, dependent on the electrical permittivity constant of the system ( ε ) and their respective geometry.
In macroscopic terms, the electrostatic energy ( E c ) stored in a capacitance C , is expressed by this equation:

This is the “ self-capacitance ” of the hydrogen atom , if it is considered as a hollow sphere of radius α 0- The calculated value of this self-capacitance is 5.9 x l0 -21 F.
To calculate the inductance L of the hydrogen atom, we assimilate the helical path of the electron around the nucleus, constituted by the proton, to a circular solenoid (or toroidal coil) of average radius α0, with N turns, and a length equal to 2 π α0.
To calculate the inductance L of this “circular coil”, we use the formula

Experimentally, it is known that the first frequency of light emitted by the excited hydrogen atom, in the so-called “ Lymann series ”, is 2.4677 Hz , which coincides exactly with the value calculated by means of the Rydberg formula , in the transition from the first to the second quantum state:
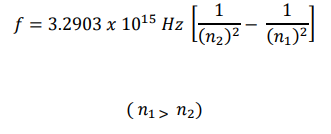
The result obtained by calculation, using the helical model of the electron, differs from the experimental one by approximately 6%.
This difference can be attributed to two factors:
1) The “toroidal solenoid” model is not totally valid to describe the helical path of the electron around the atomic nucleus, since it cannot be assimilated to an electric current distributed uniformly along that solenoid.
2) The system formed by the electron and the proton in the hydrogen atom behaves like a parametric oscillator, since when it oscillates, the radius of gyration (the most important parameter) increases and decreases cyclically, taking the circular orbit to a trajectory elliptical.
Conclusions
The helical model of the electron allows us to predict the frequency of the first quantum transition of the electron in the hydrogen atom, demonstrating that it is like any electromagnetic resonator.
The quantum transitions of an electron have a duration determined by the distance they are from the source of electromagnetic energy, since this varies linearly as a
function of the former. The quantum transition occurs when the electron energy reaches a value determined by the resonance frequency, multiplied by Planck's constant ( E = hf).
The so-called “speed of light”, whose value is 3 x 10 8 m/s, is actually the constant ratio of the distance between the emitter and the receiver of electromagnetic energy in resonance, and the time necessary for the energy to in the receiver allows the electronic quantum transition.