Review Article - (2022) Volume 7, Issue 1
Oral Vasoactive Medications: A Summary of Midodrine and Droxidopa as Applied to Orthostatic Dysfunction
2Pennsylvania Hospital of the University of Pennsylvania Health System, Philadelphia, PA, USA
3Murray Waitzer Endowed Chair for Diabetes Research, Professor of Medicine/Pathology/Neurobiology, Director of Research & Neuroendocrine Unit, Eastern Virginia Medical School, The Strelitz Diabetes Center, Norfolk, VA, USA
4Penn State Milton S. Hershey Medical Center, Hershey PA, USA
5Neuro-Cardiology Research Corporation, LLC, Wilmington, DE, USA
6CTO and Sr. Medical Director, Physio PS, Inc., Atlanta, GA, USA
Received Date: Mar 05, 2022 / Accepted Date: Mar 17, 2022 / Published Date: Mar 25, 2022
Copyright: ©Joe Colombo, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Nicholas L DePace, Aaron I Vinik, Cesar R Acosta, Howard J Eisen, Joe Colombo (2022) Oral Vasoactive Medications: A Summary of Midodrine and Droxidopa as Applied to Orthostatic Dysfunction. Cardio Open, 7(1): 216-232.
Abstract
This is a summary of the uses and applications of two vasoactive drugs, Midodrine (ProAmatine) and Droxidopa (Northera), in the treatment of Orthostatic Dysfunction (OD) disorders, including Neurogenic Orthostatic Hypotension, Postural Orthostatic Tachycardia Syndrome (POTS). This summary based on 15 years of experience monitoring and serially treating over 4000 patients with OD from all around the world and supportive literature. OD caused by an abnormal (decrease) in α-adrenergic, or α-Sympathetic, activity when assuming a head-up posture (e.g., sitting or standing). OD symptoms typically include lightheadedness, brain-fog, neurocognitive loss, fatigue, sleep difficulties, anxiety, and Syncope. Decreasing α-adrenergic activity upon head-up postural change often underlies OD and may be relieved with vasoactive medications. Midodrine and Droxidopa are the only medications that are FDA approved and approved for NOH. Except for their specific uses with NOH, these medications are used off-label with other forms of OD, including POTS, Orthostatic Hypotension, Orthostatic Intolerance and Vasovagal Syncope when co-morbid with OD. This summary will expand the knowledge of Midodrine and Northera, describe their advantages and disadvantages, and describe their comparative data. Hope this summary will help to make the clinician more comfortable diagnosing these disorders as well as using these treatments for this large population of patients with poor Quality of Life and poor functioning.
Keywords
Orthostatic Dysfunction, Parasympathetic, Sympathetic, Autonomic Dysfunction, Midodrine (ProAmatine), Droxido- pa (Northera)
Introduction
The medical term “orthostatic” is defined as relating to, or caused by, an upright (sitting or standing) postural change. Or- thostatic Dysfunction (OD) is an umbrella term for several con- ditions in which symptoms made worse by upright posture and improve with recumbence. OD includes Postural Orthostatic Tachycardia Syndrome (POTS), Orthostatic Hypotension (OH), Neurogenic OH (NOH), and Orthostatic Intolerance. In addi- tion, Vasovagal Syncope is often co-morbid with OD, as it pres- ents with similar symptoms because both lead to poor cerebral perfusion and often-poor coronary perfusion. For OD, there are only two USFDA approved drugs: Midodrine (ProAmatine) and Northera (Droxidopa). They approved specifically for OH and NOH, respectively. The management of these two drugs requires specialization and is why most common therapies for OD and Syncope have been non-pharmacologic, including: fluids with electrolytes, compression garments at multiple levels, r-Alpha Lipoid Acid (r-ALA), isometric maneuvers, elevating head at night, and avoidance of known triggers, such as large meals, heat exposure and emotional stress. In many cases, the non-pharma- ceutical measures are not sufficient to meet the clinical goals of improving symptoms and function of patients.
Here the focus will be on the pharmacological therapies. Mi- dodrine and Northera have been used successfully off-label to treat POTS and other OD disorders [1-4]. OD caused by an ab- normal (decrease) in α-adrenergic, or α-Sympathetic (αSNS), activity when assuming a head-up posture (e.g., standing from sitting). OD symptoms typically include lightheadedness, brain- fog, neurocognitive loss, fatigue, sleep difficulties, anxiety, and Syncope. This summary will expand the knowledge of Midodrine, North- era, and other vasoactive, and describe their advantages and disadvantages. Hope this summary will help to make clinicians more comfortable diagnosing these disorders and using these treatments as first-line pharmacological therapy for this large population of patients with poor Quality of Life (QOL) and poor functioning. With treatment, these patients have improved QOL and improved productivity. With serial autonomic (P&S) testing, OD disorders can detected, documented, diagnosed by all clini- cians [5]. However, treatment of OD tends to be in the realm of the Cardiologist, Neurologist, and Endocrinologist.
Currently, diagnoses of OD are also in the domain of Cardiolo- gists, Neurologists, and Endocrinologists, although all clinicians trained to diagnose. The Cardiologist, however, is typically the most experienced and trained to titrate these vasoactive medica- tions. Some Cardiologists are also trained in the additional an- cillary medication(s) that are often needed, such as volume-ex- panding agents (e.g., Florinef (Fludrocortisone)), Desmopressin (DDAVP), and in some POTS cases beta-blockers or Corlanor (Ivabradine) to limit excessive increases in HR. They specifical- ly trained in serially treating and following such patients in mon- itoring their HR, BP, and weight responses. These patients are complicated, and diagnosed by Family Medicine practitioners, they need to be referred to Cardiology should prescribed and need to be followed appropriately, especially as hypertension, and chest tightness, palpitations, and shortness of breath are present in high percentages of these patients. The use of these medications may reduce or, in some cases, increase these symp- toms. The Cardiologist is best suited to exclude significant in- trinsic cardiac disease as the cause of these symptoms, making it easier for them to titrate medications to reach an acceptable clinical endpoint.
Orthostatic Dysfunction
Orthostasis is a combination of two Greek root words: Ortho meaning “change,” and Stasis meaning “the same.” There is a complex interplay between the heart, blood vessels, and the nervous system (primarily the Parasympathetic and Sympathet- ic (P&S) branches of the autonomic nervous system) that must all coordinate properly in the struggle against gravity to simply stand or sit up (collectively, herein referred to as “stand”). There is the consequence of having a brain that is often located higher than the heart. Upon standing (or assuming any head-up pos- ture), venous pooling increases in the peripheral veins below the heart. This is in part due to the inertia of blood being higher than the electrical activity involved with stand.
Note: This is a reason for Respiratory Sinus Arrhythmia. With every inhalation, Heart Rate (HR) normally increases a little and then, with every exhalation, HR decreases a little. People nor- mally inhale just before standing, which begins to increase HR to begin to move blood to overcome the inertia to defeat gravity.
Upon assuming a head-up posture, 500-1000 ml of blood needs to be shifted up to the abdomen to support the heart in pump- ing blood to the brain (maintaining normal cerebral and cardiac perfusion, see Figure 1) [6]. Under abnormal conditions, this amount of blood may pool in the lower, peripheral vasculature causing OD. With OD, decreased venous return decreases arteri- al pressure, ventricular filling, and cardiac output, and decreases Baroreceptor and Cardiac Mechano-receptor activity. Prevent- ing these events are the “ortho” (the change) that then drives the temporary increase in HR and blood pressure (BP) that helps to maintain normal cardiac and brain perfusion (the “stasis”) to maintain a same state of heart and brain perfusion and function while standing. The neurophysiology of Norm tension and the pathophysiology of Neurogenic Orthostatic Hypotension (NOH) depicted in Figure 2 [7].
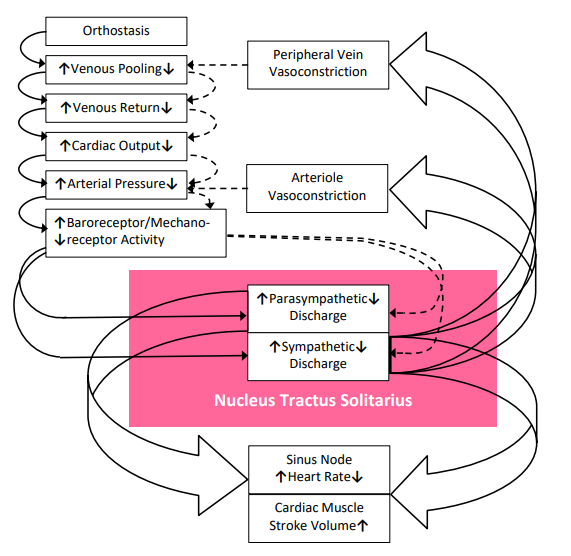
Figure 1: Circulatory Response to Orthostasis (pink represents Nucleus Tractus Solitarius). See text for details [6, Slide 2].
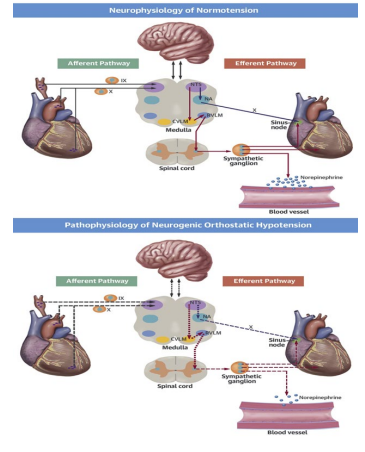
Figure 2: The anatomy and physiology of BP control: the upper panel displays the neurophysiology of normal orthostatic func- tion, and the lower panel displays the pathophysiology of Ortho- static Dysfunction. Abbreviations: IX, 9th Cranial Nerve (Glos- sopharyngeal Nerve); X, 10th Cranial Nerve (Vagus N.); CVLM, Caudal Ventrolateral Medulla; NA, Nucleus Accumbens; NTS, Nucleus Tractus Solitarius; RVLM, Rostral Ventrolateral Me- dulla [7].
Diagnosis
The common neurological cause of OD is a lack of a proper α1-adrenergic, or α1-Sympathetic (SNS), stimulation (decreased αSNS activity). This neurologic cause, decreased αSNS activi- ty, measured as a decrease in SNS activity upon standing (see Figure 3, top) [8]. Normal αSNS activity (see Figure 3, bottom) causes vasoconstriction of the peripheral vasculature. This vaso- constriction supports norm tension with stand. Decreased αSNS activity is the autonomic dysfunction underlying POTS, NOH, OH, and OI disorders [8,9]. Decreased αSNS activity, contribut- ing to the lack of vasoconstriction upon standing, may also be a component of the mechanism that causes the sudden drop in BP with Vasovagal Syncope (VVS).
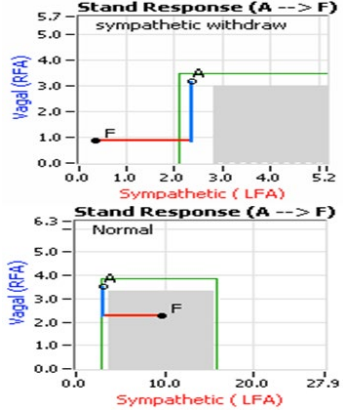
Figure 3: Stand Response plots from P&S Monitoring: Normal (bottom) and SW (top) [8]. The abscissa represents Sympathetic activity and the red portion of the curve documents the patient’s Sympathetic response to stand in (beats per minute)2 (bpm2). The ordinate represents Parasympathetic activity and the blue portion of the curve documents the patient’s Parasympathetic response to stand in bpm2. Point ‘A’ represents the patient’s rest- ing (sitting) P&S responses and point ‘F’ represents the patient’s stand P&S responses. The grey area of the graph represents the range of normal Stand responses given the patient’s own resting response (point ‘A’, which is always in the upper left corner of the normal area).
OD includes orthostatic conditions that do not meet the specif- ic clinical criteria of POTS* or NOH**, but are either trending in that direction or carry the symptoms of those disorders (i.e., pre-clinical conditions). In these pre-clinical conditions, the ad- ditional information of decreased αSNS activity with the trend in HR towards POTS or BP towards NOH, OH, or OI, respectively, enables early diagnosis and treatment. Many times, BP or HR does not change significantly, and they just have evidence of de- creased αSNS activity. Decreased αSNS activity can be detected with simple outpatient testing known as Cardio-Respiratory, or P&S, testing [8]. Decreased αSNS activity is additional informa- tion that could potentially reduce morbidity and mortality risk [10,11].
Normally upon standing, the Parasympathetic (PSNS) first de- crease (as indicated by the blue portion of the response curve in Figure 3, bottom, labeled “Normal”), followed by the (normal) αSNS- increase (as indicated by the red portion of the response curve in Figure 3, bottom, labeled “Normal”). This PSNS-de- crease begins the process of vasoconstriction to move blood up to the abdomen to help the heart pump blood to the brain. Then, the αSNS-increase follows. This αSNS-increase sustains the va- soconstriction and continues to shift the majority of the blood volume from the feet, against gravity, to the abdomen it properly perfuse the heart, so that the heart may more easily pump the blood to the brain to maintain proper cerebral perfusion.
This normal stand αSNS-increase is shown at the beginning of section ‘F’ in the P&S Trends plot (the red response, Figure 4). This SNS-increase should be significantly less than that for Val- salva (section ‘D’ in the P&S Trends plot, Figure 4); at least a 3:1 ratio of Valsalva to stand [8]. In other words, it should not take as much SNS-activity to stand as it does to manage other significant stressors.
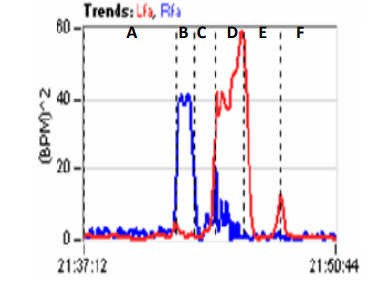
Figure 4: P&S Trends plots from a healthy, young, adult subject [8]. Time is represented on the abscissa and P&S (RFa, blue & LFa, red, respectively) is represented on the ordinate. The six sections of the graph (‘A’ through ‘F’) are the patient’s responses to the six phases of the clinical study: A) Resting baseline, B) Deep Breathing, C) Baseline, D) Valsalva Challenge, E) Base- line, and F) Stand [8].
While a normal PSNS-response (a decrease with respect to rest) is typically assumed to occur in response to a SNS-chal- lenge (i.e., Valsalva or stand), in dysfunctional cases, a normal PSNS-response is not always the case [12]. However, this is not the focus of this study. Abnormal αSNS-activity may produce symptoms of lightheadedness, syncope (fainting), pre-synco- pe, fatigue, brain-fog, difficulties finding words, cognitive and memory difficulties, and sleep difficulties [8]. In the USA, 20 million people are estimated to have OD symptoms which sig- nificantly affect individuals’ ability to function, and, thereby, their QOL and work and disability status [6].
Patients with OD who are younger are predisposed to develop the extreme condition of POTS (see OD Algorithm, Figure 5.). There is a higher prevalence of POTS in women. Figure 6. Dis- plays data from a typical POTS patient. The OD demonstrated by the decreased αSNS activity as displayed in Figure 6, right panel, and the Tachycardia demonstrated by the cardiogram (Figure 6, left panel, red, upper, trace). Figure 6, right panel, is the Cardio-Respiratory Coupling graph for this patient with the lower (gray) trace displaying the instantaneous Respiratory Ac- tivity and the upper (red) trace displaying the HR during the first five minutes of standing from a relaxed seated posture [13]. Note how the HR does not return to baseline, as would be normal, but increases and continues to increase throughout the stand period and, for the most part, exceeds 120 bpm.
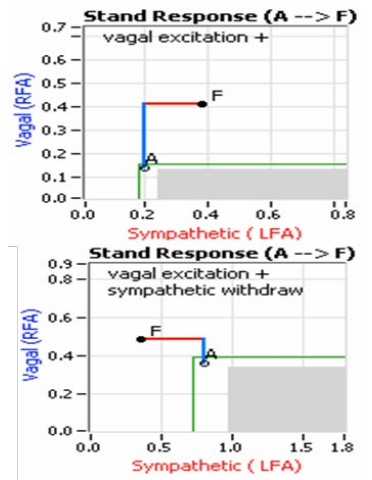
Figure 5: OI associated with the Autonomic Dysfunction: SW. Orthostatic Dysfunction starts as SW (typically asymptomatic), due to disease or trauma (mental or physical) and progresses to OI (SW with symptoms), then progresses to POTS (SW with Tachycardia upon standing) in younger patients, or NOH (SW with a drop in BP upon standing) in older patients [14]. Corlanor is an option for POTS only.
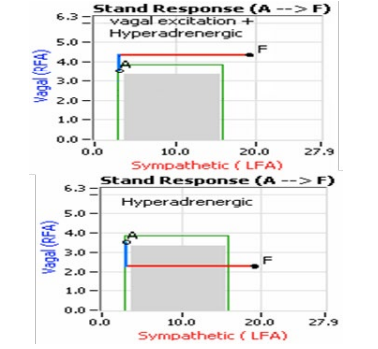
Figure 6: POTS patient’s Cardio-Respiratory Coupling graph (CCG, left) and Stand Response plot (right, see Figure 3 for details). The red curve in the CCG is the cardiogram (bpm2) and the gray curve is Respiratory Activity (mV). The Cardiogram demonstrates Tachycardia and the Response plot demonstrates Orthostatic Dysfunction (Sympathetic Withdrawal). Together the two graphs in- dicate POTS [8].
NOH or OH typically occurs in older people (see Figure 5) and often accompanies other disorders or diseases, such as Parkin- son’s disease, Diabetes, and Amyloid disease, as well as many other chronic diseases or disorders (including chronic pain dis- orders). Chronic diseases or disorders often involve autonomic dysfunction, and a first symptom of autonomic dysfunction is OD often in the form of OH or NOH. OH is arguably the most debilitating symptom of autonomic dysfunction [10,11]. Based on waveform assessment with beat-to-beat BP and tilt-table test- ing, NOH differentiated into three sub-types:
1) Classical NOH,
2) Delayed NOH, and
3) Initial NOH (see Figure 7) [7].
Figure 7: Beat-to-beat BP waveforms (blue curves) with accompanying cardiograms (brown curves) from tilt-table testing demon- strating responses from patients with: 1) Classical NOH, upper left; 2) Delayed NOH, upper right; and 3) Initial NOH; re-printed from [7].
Often, VVS is co-morbid with OD. A beat-to-beat BP with tilt test of a patient demonstrating VVS presented in Figure 8. [15]. VVS documented in approximately one-third of POTS patients [16]. While OD characterized by decreased αSNS activity, Syn- cope characterized by an excessive βSNS response to the synco- pe during stand. Syncope causes poor cerebral perfusion and the excess βSNS activity is the result of the body’s attempt to main- tain normal cerebral perfusion. Therefore, it is possible for both to co-exist (e.g., see Figure 9). With VVS or Reflex Syncope, there is often an abrupt decrease in αSNS activity or vasodepres- sor response as BP falls. HR may drop as well, possibly due to a secondary decrease in βSNS-activity due to the orthostatic vol- ume shift. Tilt testing may demonstrate both abnormalities. Car- dio-Respiratory testing is a more practical, less invasive method to demonstrate the co-existence of both entities in a patient [8].
Figure 8: VVS beat-to-beat BP (mmHg) response to tilt-table test. Head-up tilt at “Start,” for duration of “Upright.” Syncope occurred just before the end of “Upright” [15].
Figure 9: Stand Response plot (see Figure 3 for details) and P&S Trends plot (see Figure 4 for details) demonstrating multiple Au- tonomic Dysfunctions leading to multiple co-morbidities, in this case POTS with VVS. Compare Trends plot with a normal Trends plot in Figure 4. See text for details.
VVS also known as "simple fainting." A neurological reflex that leads to a temporary loss of consciousness mediates it. This re- flex causes a sudden dilatation of the blood vessels of the legs causing blood pooling in the lower extremities. It may also slow HR, sometimes down to 20 bpm, which may also reduce cardiac output. At times, both mechanisms may occur simultaneously. Oftentimes, VVS known as Neuro-Cardiogenic Syncope or Re- flex Syncope. While tilt-table testing will consider the test of choice for differentiating VVS, the simple placement of the pa- tient on the tilt-table may cause anxiety and a SNS surge, which may counter the Vagal excess and give a false negative result for VVS.
Therapy
Currently, three oral vasoactive medications (Midodrine, Drox- idopa, and Pseudoephedrine) that often used to treat Dysau- tonomia. Mestinon (Pyridostigmine) occasionally used and considered by some as an indirect-acting vasoactive drug. These medications symptoms of OD, including:
1. POTS typically early in the progression of Autonomic Dys- function and
2. OH typically later in the progression of Autonomic Dys- function (see Figure 5).
They use in Vagal or Reflex Syncope to prevent the abrupt drop in BP that may occur.
Compression stockings, fluids, and electrolytes (sodium-based electrolytes for those with normal to low BP and potassi- um-based electrolytes for those with high BP) are therapeutic and can be useful adjuncts in these instances, but rarely are cura- tive. OD is usually due to a decreased ability of post-ganglionic SNS-neurons to release Norepinephrine. This is often seen in OD disorders where patients complain of “brain fog,” especially when standing or sitting upright for long periods. Brain fog may include cognitive or concentration abnormalities, word-finding abnormalities, and short-term memory loss, as well as fatigue, symptoms of depression or pre-Syncope, and even occasional frank Syncope. Many times, patients have to lie down to get re- lief from these symptoms, and they feel giddy and dizzy. OD is the main contributor to persistent fatigue*** and Chronic Fa- tigue Syndrome (CFS). It is also a main contributor to exercise intolerance.
Midodrine (Proamatine)
Midodrine is an oral, peripherally acting, α1-Adrenergic ago- nist. The FDA approved it in 1996 for OH [7]. Also used in the hospital in intra-dialysis for hypotension or BP drops that occur with end-stage renal disease patients on dialysis, in post-surgery cases, in septic shock patients, in a syndrome called Hepatorenal Syndrome, where it improves renal function and others with se- verely low BP, to increase BP [17-27]. In the Intensive Care Unit (ICU), Midodrine substituted for low dose intravenous Phenyl- ephrine, Vasopressin, or Norepinephrine [28,29]. Midodrine used in Cirrhosis-related hemodynamic complications [17,27]. Another agent, Octreotide also used in these instances with suc- cess. Further studies are included in this area. In these acute sit- uations, Midodrine administered in high doses.
There has been conflicting data on the adjunctive use of Mi- dodrine. It has been a useful adjunctive to IV-vasopressor agents in adults with shock; however, in this study, Midodrine did not have an effect on ICU or hospital length of stay [25]. This was a negative study for Midodrine. Other studies have shown a bene- fit. In critically ill patients, Midodrine shown to be effective and safe as an adjunctive therapy to increase mean arterial pressure in patients on IV-vasopressors [28]. Another study demonstrated the utility of Midodrine in the recovery phase of septic shock [24]. This study also demonstrated that Midodrine may reduce the dose of IV-vasopressin from septic shock and may be associ- ated with reduction in length of stay.
There are ongoing clinical trials of Midodrine for the treatment of refractory Hypotension in patients otherwise ready for dis- charge from the ICU [29]. Potential for bias increased by single center and observational design studies in the past and, therefore, multicenter studies needed. One study investigated the prescrip- tion of high dose Midodrine started at 2.5mg every eight hours titrated to 40mg every eight hours in the ICU. For autonomic dysfunction, including OD (such as OI & OH, and POTS) and VVS or Reflex Syncope, Midodrine is the most commonly used first-line drug [6,30-41]. It is administered in very low doses to treat the decreased αSNS activity and other causes of OD, which are mostly vascular in nature. Midodrine started at 1.25mg three times a day, titrated very slowly in six steps over 12 weeks, from half a pill in the morning to the full dose of 2.5mg three times a day. Note, at this dose, the patient should not lie flat for at least two hours (preferably four hours) after dosing. If there is the need to lie down, make sure there are two pillows under the head, or preferably, a wedge pillow under the whole body. In more severe cases, Midodrine advanced to 5mg, three times a day. For the most severe cases, dosing up to 10-15mg three times a day needed, but this is rare. (At these higher doses, patients should not lie flat for four hours after dosing without at least two pillows under the head, or preferably a wedge pillow under the patient.) Of course, Midodrine is contra-indicated for supine hypertension and high BP, especially at the higher doses [9]. One hundred pills of Midodrine at the low dose generally cost a little over $100.00.
The most frequent side effects of Midodrine are piloerection (“Goose Bumps” on the skin), chills, scalp pruritus (“itchy” or “crawly” scalp), numbness and tingling (paresthesia’s) on the face or throughout the body, and urinary retention or, at times, urethra discomfort, especially in males with benign prostatic hy- pertrophy. While very little Midodrine crosses the Blood-Brain Barrier, occasionally, headaches have reported, especially in the beginning, due to some of its vasoconstrictor effects. This may include additional head pressure due to more blood flow to the head and brain. Please note that these symptoms are indications that the medicine is working. These symptoms may be over- whelming. That is why, as above, the prescribed dosage should be titrated very slowly, perhaps over as much as three months. This seems to be especially necessary in cases where patients prescribed high doses of Midodrine (10mg or more tid). Giv- en these side effects, patients’ expectations need to be set and stressed to help ensure compliance.
Still, many patients are frightened with the initial side effects of Midodrine. Droxidopa, which we will discuss later, has been associated with better treatment adherence, compared to Mi- dodrine, because it does not have these early side effects. Again, patients may need multiple reassurances that these are not side effects, but may be an indication that the vasoconstriction is actually working. This also reassures that the patient has been compliant, the Midodrine is being absorbed, and it is working to restore vasoconstriction. For those without decreased αSNS ac- tivity (i.e., people with normal αSNS activity), vasoconstriction is common, frequent, and largely ignored. It is important that the patient become accustomed to the therapy for treatment success.
In 2010, the FDA proposed the withdrawal of Midodrine (ProAmatine) because of lack of studies that verified the clinical benefits of the drug. It was proposed because neither the original manufacturer nor any generic manufacturer had demonstrated the drug’s clinical benefit. However, studies had shown its effi- cacy, and the withdrawal was repealed. Further studies contin- ue to demonstrate efficacy. ICU Studies showed that Midodrine was effective to wean IV-pressers and shorten ICU and hospital length of stays [28,29]. At the time, over 100,000 patients were taking Midodrine for conditions that would otherwise be dis- abling and complaints were numerous. Subsequent studies have confirmed the efficacy of Midodrine in OI disorders [7]. A sys- tematic review of Midodrine addressed health-related QOL in patients with recurrent reflex Syncope (VVS), and in patients with OH [35]. The evidence was low or moderate, but the con- fidence was present that suggested Midodrine showed a clini- cal and outcome improvement. Midodrine continues to be used off-label for OD such as POTS [33].
Midodrine should only be considered when non-pharmacologic strategies (e.g., high-dose r-ALA) with or without volume ex- panding drugs have failed to alleviate the patient's symptoms [42]. However, pharmacologic agents, life-style changes, and even volume expansion drugs, such as Fludrocortisone and DDAVP, are oftentimes not effective as a stand-alone therapy. For example, there may be problems with Fludrocortisone, in- cluding possible long-term use causing fibrosis [43]. Therefore, take care in prescribing Fludrocortisone, and only prescribe it if further therapy is needed, adjunctive to Midodrine. Thus, Mi- dodrine is a first-line agent. Still, Fludrocortisone potentiates the effects of Midodrine, and vice versa, enabling lower dosages of both when used together, thereby limiting the side effects of both agents. Midodrine may lower HR, especially with α-Adrenergic blocking agents or β-Blockers. In these cases, HR should monitor carefully. Oftentimes, this is beneficial and welcomed, as many patients with OD have a form of POTS with high HRs (about 1/3 of the OD patients present with POTS). With Midodrine, another warning for patients is to avoid over-the-counter drugs, such as Pseudoephedrine or other α-Adrenergic agonists. The optimal morning dose of Midodrine is 5 to 10 minutes before rising, right at the bedside to counter early morning (severe) symptoms. The final dose should be taken two (2) to four (4) hours before lying down, including in bed, depending on dose. In general, it is pre- scribed for 8am, 12pm, and 4pm. This may vary depending on time of waking.
A common condition requiring Midodrine with adjunctive ther- apy is OD with VVS. Both decreased αSNS activity and PSNS excess, must treat simultaneously. Decreased αSNS activity may be reversed with Midodrine or Northera together with r-Al- pha-Lipoic Acid (r-ALA) and PSNS excess may be reversed with very low-dose anticholinergics or low and slow exercise [9,42,44]. The anti-cholinergic therapy is very, very low-dose anti-depressant therapy (e.g., no more than**** 10.0mg Nor- triptyline, qd, dinner). This is prophylactic in treating co-morbid VVS and adjunctive to Midodrine treats the decreased αSNS ac- tivity. Midodrine is the first line therapy, helping to relieve both dysautonomias (see Figure 10). The primary effect of Midodrine for patients whose BP drops upon standing is to prevent the drop upon standing. Relief of brain-fog, memory or cognitive difficul- ties, trouble finding words, lightheadedness, and fatigue typical- ly reported, and in some sleep difficulties and Anxiety as well.
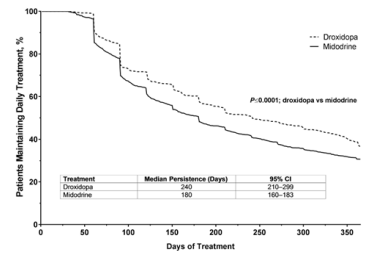
Figure 10: Mechanisms of neutrally mediated Orthostatic Dys- function: Orthostatic Stress caused by chronic disease, exposure, injury, surgery, etc., may induce SW leading to OVVS, NMH, NOH, or POTS, as indicated, or OIS, which is a diagnosis by omission, indicating condition due to SW that does not fit the above three classifications. Orthostatic Intolerance (OI) de- scribes the symptoms of SW and includes one of the above four diagnoses. Abbreviations: NE, Norepinephrine; Epi, Epineph- rine.
Midodrine Mechanism of Action and Adjunctive Inter- actions
The mechanism of action of Midodrine is actually quite com- plex. Upon ingestion, Midodrine undergoes an enzymatic re- action producing a metabolite, Desglymidodrine. Following an oral administration, 90% of Midodrine is rapidly absorbed. It may even be absorbed through the buccal mucosa, and patients have even chewed it to get a quick response, rather than swal- lowing it. So, it is absorbed through the mouth directly. The me- tabolite Desglymidodrine peaks within 1-2 hours with the subse- quent half-life of 3-4 hours and excreted in the urine.
It has a similar mechanism of action compared to Norepineph- rine. There may be an increase in BP due to the increased return of blood from the lower extremities to the heart while the heart is continuing to pump harder to draw blood from the lower ex- tremities. This increase in BP is relatively short-lived, perhaps up to nine to twelve months. Again, the initial dosing of Mi- dodrine should be avoided in patients with supine hypertension and restricted to patients with resting BPs less than 160/90, ac- cording to one study [9]. Otherwise, the increase in BP due to Midodrine may potentially lead to stroke. Patients with NOH often present with compensatory systemic Hypertension. This is the reason anti-hypertensive may be used concurrently with Mi- dodrine, such as Clonidine or Hydralazine. A very high BP does not preclude the use of Midodrine if the systemic Hypertension adequately treated with these agents. A notable exclusion is pa- tients with Pure Autonomic Failure, where Clonidine may raise BP [45,46]. Therefore, in conditions with OH and hypertension selective therapies must cautiously tailored in specific instances. Typically, patients with OH treated during the day with pressor agents and patients should remain upright to prevent hyperten- sion.
Midodrine is a selective α1-Adrenergic Agonist and stimulates α-Adrenergic receptor activation on both arteries and veins. This causes vasoconstriction of blood vessels and a decrease in ve- nous pooling, especially in the extremities; therefore, increas- ing BP with upright postures. During changes in posture, α1 receptors assist in raising BP temporarily to counter the effects of gravity and the inertia of blood, helping the heart to maintain proper blood flow and profusion pressures within the brain (and heart) to prevent lightheadedness and reductions in brain and nervous system function and the function of other structures at the level of the heart or higher. The primary effect of Midodrine is to prevent the BP drop upon standing. Relief of brain-fog, memory or cognitive difficulties, trouble finding words, light- headedness, and fatigue typically reported.
Many patients are reluctant to take Midodrine because they be- lieve it will raise their BP and exacerbate their hypertension. Given the low dosages of Midodrine used, while it usually does raise BP slightly, it typically eventually reduces BP once the lower vasculature begins to constrict, properly supporting the heart and, thereby, reducing cardiac workload [9]. Midodrine is contraindicated for supine hypertension. The safeguard at these low doses is to have them lie down on a wedged pillow or an incline or with at least two pillows under their heads.
Midodrine does not stimulate beta-Adrenergic receptors. With more blood returning to the heart, the pre-Midodrine, requisite, compensatory rise in BP to counter the lack of a rise in peripher- al resistance upon standing that is OD is also relieved, and BP is ultimately reduced once proper peripheral vasculature function is re-integrated with proper cardiac blood flow [9]. Midodrine lacks central nervous side effects since it does not cross the Blood-Brain Barrier in contrast to other nonselective Sympath- omimetic agents. Midodrine does not cause cardiac stimulation, palpitations, or arrhythmias. It has no known effects on the pul- monary, renal, or blood coagulation function or changes in blood glucose or lipids.
The efficacy of Midodrine shown in phase-4, double blind and placebo-controlled, randomized tilt-table study [36]. The mini- mally effective dose is 1.25-2.5mg a day. The maximum phar- maceutically recommended dose is 30mg a day (10mg, tid), but rarely may be exceeded for severe OH. For patients with Supine Hypertension and severe OH, careful consideration and fol- low-up, titration from very low doses, and an additional anti-hy- pertensive agent considered. Clonidine or Hydralazine recom- mended, depending on history, for BP control in these patients [47]. Midodrine with beta-blockers or Digoxin requires care to not significantly lower HR. Midodrine with Monoamine Oxi- dase (MAO) Inhibitors are not recommended, as this can result in Hypertensive crises. Midodrine’s active metabolite, Desgly- midodrine eliminated. Therefore, for Chronic Kidney Disease and patients with other types of renal impairment, Midodrine should initiated cautiously and at lower dosages, such as 1.25mg per day in the morning titrating to 2.5mg tid in six increments over 12 weeks. It is very successful in treating patients with he- modialysis who drop their BP.
While Midodrine is taking effect, and to augment the effect of midodrine as needed, additional therapies may be considered, including: avoiding triggers (e.g., large meals, heat exposure, alcohol), increasing water and salt in-take, and “low and slow” exercise, compression garments and sleeping with the head of the bed elevated (Figure 11) [7]. Volume builders, such as Flud- rocortisone (Florinef) or a Vasopressin analogue such as Desmo- pressin (DDAVP) considered as adjunctive therapy to increase central blood volume. Additional sympathomimetic therapy, such as Pyridostigmine (Mestinon) or Atomoxetine (Strattera), must consider as adjunctive therapy.
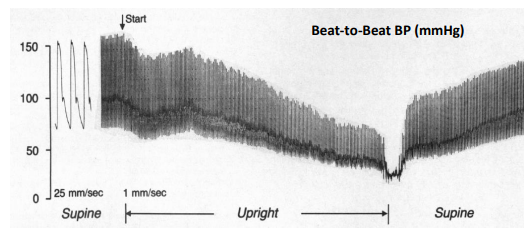
Figure 11: NOH Therapy Algorithm [modified from 7, open access].
In a dose-dependent manner, Midodrine increases standing Sys- tolic BP, reaching peak efficacy in one hour (see Figure 12) [48]. Up-titration is based on the patient's clinical symptoms and BP responses (see Figure 13) [48]. Midodrine may increase stand- ing BP by 21-22mmHg, especially at higher doses, and leads to improved scores in lightheadedness and other symptoms as pre- viously described. By restoring proper brain perfusion, depres- sion and anxiety-like symptoms relieved as well. BP increases of 21-22 mmHg are higher than what are often see in clinical practice, and for patients that present with the higher dosages, the dosage is often reduced to affect a better outcome; especially if they are still symptomatic [personal communication, DePace]. Due to the tendency for BP to rise during the day, many have found it very useful to recommend decreasing the dose of Mi- dodrine as the day goes on. For example, prescribing 5mg in the morning and 2.5mg for the next 2 doses. This is especially true with patients who are symptomatic when arising in the morning, such as with POTS patients.
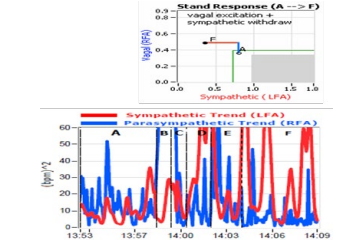
Figure 12: Midodrine dose response curves [reprinted from 48].
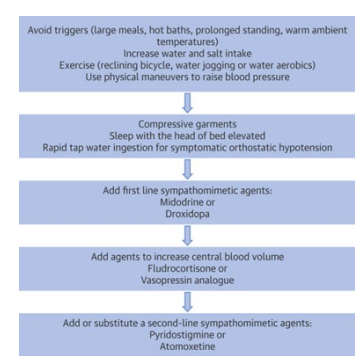
Figure 13: Midodrine-BP time course of action [reprinted from 48].
Midodrine has been especially useful in children with POTS with peripheral neuropathy [39]. Some patients with POTS ben- efit from Theophylline, which is a vasoconstrictor and used to treat Dysautonomia. Others have used Methylphenidate in POTS patients with hypotension. Methylphenidate stimulates α-recep- tors, causing increased peripheral vascular resistance. Howev- er, there is a risk of addiction and increased HR with Methyl- phenidate should avoid, if possible. Clonidine, a direct-acting, central, α2-adrenergic agonist, as well as Ivabradine (Corlanor) and β-blockers used adjunctively in POTS patients [49,50]. Of- tentimes, they used to control orthostatic tachycardia when Mi- dodrine which is prescribed to correct decreased αSNS activity. In POTS patients, Midodrine has shown to have better effects than intravenous saline and shown to be effective in neuropathic POTS due to its effects on vascular resistance [33,37].
Midodrine reported in the prevention of VVS [41]. It was very effective in the prevention of head-up tilt-induced Syncope, but its results in this study were somewhat poor in terms of dou- ble-blinded trials. However, Midodrine seems to be very effec- tive in treating people with VVS, especially when used with low-dose anticholinergic agents, such as Tricyclics (e.g., 10mg, qd, dinner, Nortriptyline), or SNRIs (e.g., 20mg, qd, Duloxetine) [51]. Rarely these recommend dosages titrated higher; thereby, preventing additional symptoms. Midodrine has a shelf half-life of only eight weeks from opening. It prescribed for OD or Syn- cope. It is relatively contra-indicated in severe Hypertension, severe cardiac disorders such as Hypertrophic Obstructive Car- diomyopathy, certain cardiac valve disorders, Thyrotoxicosis, Pheochromocytoma, acute Nephritis, Urinary Retention, Hy- perthyroidism, Narrow Angle Glaucoma, pregnancy or breast feeding, proliferative Diabetic Retinopathy, and serious Prostate disorders. In addition to SSRIs and drugs that can cause Bra- dycardia, such as Digitalis and beta-blockers (as mentioned be- fore), patients receiving Antiarrhythmic drugs, Decongestants, some appetite suppressants, or occasionally Clonidine, may have an exaggerated response to Midodrine and further lower HR.
Midodrine and Pyridostigmine both compared in patients with Heart Failure (HF) [52]. While both may be beneficial, only Mi- dodrine is beneficial in Hypotension. For Hypotensive patients with HF, Midodrine was associated with a two-fold decrease in medication-load and hospitalization days, with improvement to optimal medication for HF, Left Ejection Ventricular Frac- tion (LVEF, indicating improved mortality risk), and associated symptoms (improved morbidity risk). Pyridostigmine use in HF was associated with beneficial effects in cardiac remodeling, im- provement in LVEF, and improved exercise tolerance. Midodrine in Congestive HF improves the LVEF and clinical outcomes, with significant reduction in total hospitalizations, as it allowed for up-titration of neurohormonal antagonist (i.e., Angiotensin Converting Enzyme-Inhibitor, or ACE-I) therapy leading to im- proved outcomes) [20]. Ten consecutive patients with HF due to Systolic Dysfunction and symptomatic Hypotension interfering with optimal medical therapy has started on Midodrine. After six months, a higher percentage of these patients were on optimal HF therapy. Therefore, Midodrine is routinely use in patients with very low BP when ACE-I, β-blockers, Aldosterone antagonists cannot be instituted in patients who will need these to improve their prognosis and LVEF. Reversible Cerebral Vasoconstriction Syndrome due to Midodrine in a patient with Autonomic Dysre- flexia has been described [21]. This rare neurological condition typically presents with sudden onset Thunderclap Headaches as- sociated with or without focal neurologic defects.
As indicated above, patients with long standing decreased αSNS activity often present with elevated to moderately high, resting BP. Often, in these patients the higher-than-normal resting BP is compensatory for the associated drop in BP upon standing. Un- fortunately, without the additional information from P&S Mon- itoring, the cardiologist perceives the higher BP as the primary diagnosis and treats as such. However, oftentimes, the patient feels worse (more lightheaded or exercise intolerant), or the pa- tient’s BP becomes difficult to control, the patient is perceived to be non-compliant, and the relationship degrades.
Midodrine is a first-line drug for the treatment of OH. It may also be used, off-label, as a first-line drug for treatment of POTS, OD syndromes in general (conditions that include decreased αSNS activity), and VVS. Midodrine must be prescribed and titrated under the auspices, experienced specialist who has used this in the past for Autonomic Dysfunction syndromes. R-ALA, a su- per-antioxidant selective for nerves, and supports and may po- tentiate the actions of Midodrine. Dysautonomia is often a result of oxidative stress, including on autonomic nerves. Oxidative stress is often a result of serious illness or injury, surgery, mental or physical trauma, virus or other severe infection, etc. Often the first, and most debilitating, symptom of Dysautonomia is OD [10,11]. R-ALA helps to heal the effects of oxidative stress, including the Mitochondria in the nerves, leaving Midodrine to “re-train” the nerves to properly vasoconstrictor. Midodrine (ProAmatine), remains the only FDA approved, oral, α1-Adren- ergic receptor agonist.
Northera (Droxidopa)
The FDA approved the vasoactive drug, Northera (Droxidopa) for NOH in 2014. Northera approved to treat NOH in Japan in 1982. Northera also carries the warning about the risk of supine Hypertension. This is common in patients that have Dysautono- mia and can cause stroke. As with Midodrine, individuals must sleep with their heads up and their upper body elevated. North- era (Droxidopa) is a synthetic amino acid analogue. Upon oral intake, it is metabolized by Dopa-Decarboxylase to Norepineph- rine, both peripherally and centrally (Figure 14) [53].
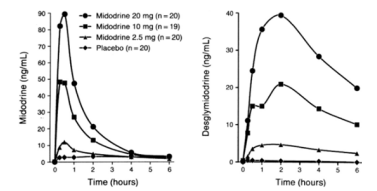
Figure 14: Metabolism and pathways of Northera (Droxidopa)[modified from 53].
It is therefore a synthetic Norepinephrine pro-drug. The time course of action of Droxidopa shown in Figure 15. Norepineph- rine is vasoactive in that it increases BP through peripheral ar- tery and venous constriction. Droxidopa used for the treatment of orthostatic dizziness (lightheadedness) and pre-Syncopal feelings of "blackouts" in adults with symptomatic NOH caused by Primary Autonomic Failure, including due to Parkinson's dis- ease, Multiple System Atrophy (MSA), Pure Autonomic Fail- ure, Dopamine Beta-Hydroxylase deficiency, and non-Diabetic Autonomic Neuropathy. These latter disorders and diseases in-clude other systemic diseases such as Amyloidosis. Droxidopa is available in 100, 200, and 300mg hard gelatin capsules. It starts at 100mg three times a day (every four hours) and taken with or without food. One should take the last dose at least 3-4 hours before going to bed (lying down). The maximum dose is 600mg three times a day [6].
Figure 15: Time course of action of Northera (Droxidopa) and Orthostatic BP (Mean Arterial Pressure, MAP, mmHg). Dosing starts at time 0 hours [modified from 6, Slide 8].
Droxidopa is an analogue of Levodopa (Figure 16) and is very similar. Dopa-Decarboxylase, which converts Droxidopa to Norepinephrine, is the same enzyme that converts Levodo- pa to Dopamine in the treatment of Parkinson's disease. As a Norepinephrine pro-drug, it activates α1-Adrenergic receptors. Droxidopa, unlike Midodrine, will cross the Blood-Brain Barrier and enhance Norepinephrine production in the Central Nervous System. Droxidopa can be metabolized into Norepinephrine and Epinephrine (Figure 17) to stimulate both α- and β-adrenergic receptors and has various physiological responses depending on the tissue it affects. Droxidopa compared to Midodrine in the treatment of NOH [54].
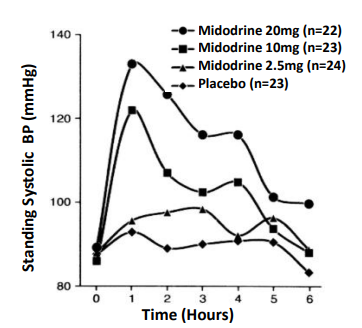
Figure 16: Droxidopa is an analog of Levodopa. [Modified from 6, Slide 6].
Figure 17: Catecholamine Metabolism [modified from 6, Slide 5].
Patients using Droxidopa were more likely to remain on treat- ment longer than patients on Midodrine, presumably due to few- er side-effects and possibly due to enhanced efficacy (see Figure 18) [53]. However, the cost per dose of Droxidopa is consider- ably higher than Midodrine, and pre-certification with the in- surance company is required. Patients on Midodrine were 27% more likely to become non-compliant than those on Droxidopa, which is indicative of better compliance with Droxidopa (Figure 19) [53].
Figure 18: Potential sites of action of Northera (Droxidopa). Catalysts are indicated in rectangle boxes: Aromatic L-amino acid decarboxylase & Phenyl ethanolamine N-methyltranferase) [modified from 6, Slide 7].
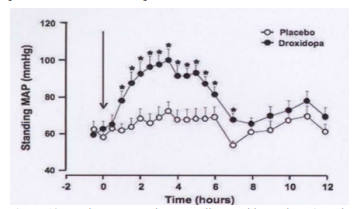
Figure 19: Persistence or patient compliance with Northera (Droxi-dopa as compared with Midodrine (ProAmatine) [modified from 53].
Droxidopa well tolerated and improves symptoms of NOH [55- 57]. Some patients fail to respond and benefited by the addition of Midodrine, which works as a pro-drug and results in vasoac- tivation. Together, the two may be synergistic, and both agents used together successfully. However, care taken not to produce significant supine Hypertension. Long-term safety of Droxidopa in patients with symptomatic NOH was established [58]. Rates of adverse cardiovascular side effects, including supine Hyper- tension, were low. Most of the side effects related to the patient is underlying neurological disease (e.g., Parkinson’s or Pure Au- tonomic Failure) [59]. Droxidopa has a favorable benefit-to-risk ratio in patients with NOH. The most frequently recorded ad- verse effects were falls, urinary tract infections, headaches, Syn- cope, and dizziness or lightheadedness.
The long-term effects of Droxidopa do not appear to be use-pro- hibitive. There were low incidences of cardiovascular events in patients with NOH on Droxidopa [44,60]. Comparative analysis with Droxidopa and Midodrine showed that supine Hyperten- sion was significantly greater for Midodrine but not for Droxido- pa when compared to placebo [61,62]. Both Droxidopa and Mi- dodrine, however, did improve standing systolic BP. Droxidopa shown to be effective in treatment of POTS, apparently improv- ing some symptoms of OD in patients with POTS [63]. VVS was relieved by Droxidopa [4]. It improves both SNS Splanchnic ar- terial and venous constriction in POTS and VVS patients.
Droxidopa shown effective for the short-term treatment of symp- tomatic NOH and Parkinson's disease [64]. Droxidopa shown to improve symptomatic OH, MSA, and Pure Autonomic Failure patients and provide a hemodynamic and symptomatic benefit. One case report of adding Droxidopa to Fludrocortisone and Mi- dodrine in a patient with NOH and Parkinson's disease demon- strated how three agents could be effective in patients with re- fractory OH disorders [65].
Amyloid may infiltrate the nervous and cardiac systems. A very uncommon, but rarely recognized disorder is Transthyretin Am- yloidosis. These patients may present with diarrhea, OH, cardiac disease, CHF, and bi-lateral carpal tunnel syndrome [66]. OH in patients with this disorder can be a consequence of heart fail- ure due to Amyloid Cardiomyopathy or volume depletion due to diarrhea, drug effects, or Amyloid Autonomic Neuropathy. Diagnosis is usually made with positive biopsy in affected or- gans or genetic testing for the mutations associated with the spe- cific type of Amyloid, for example, assessing for mutations in the TTR gene for Transthyretin Amyloidosis. Immuno-fixation, Serum Protein Electrophoresis, Serum and Quantitative Free κ and λ Light-Chain (FLC), and Serum FLC Ratio test for amyloid Autonomic Neuropathies due to immunoglobulin abnormalities. Treatment with Droxidopa shown to be effective in these disor- ders, improving NOH [23].
Cardiovascular autonomic testing, with beat-to-beat BP, often shows absent BP overshoot after release of the Valsalva strain. The lack of overshoot indicates impaired Baroreflex-mediated SNS-activation. These patients also have both sensory and Auto- nomic Neuropathies and can have abnormalities in EMG testing and small fiber assessment (e.g., Sudomotor testing or skin biop- sy). When gastrointestinal (GI) symptoms are present, biopsy of the GI tract may often yield positive results. When cardiac dis- ease is present, biopsy of the heart can also show abnormalities. Many of these patients get post-prandial Hypotension, which re- sults in blood pooling within the splanchnic system. Droxidopa shown to be helpful here also. Off-label uses of Droxidopa for Hypotension, Diabetic Autonomic Neuropathy, and Hypoten- sion related to Autonomic Dysfunction and Rheumatoid Arthri- tis have also been described [60,67,68].
The main obstacle in using Northera (Droxidopa) is the cost, and insurance companies always require pre-certification for pay- ment. It is a very expensive drug. Whereas Midodrine is not very expensive, Droxidopa can cost thousands of dollars per month if not covered by insurance. Given Droxidopa’s mechanism of action, Physicians must be cautious if patients are on medica- tions that increase Norepinephrine, such as Sympathomimetic, α1, or α2 adrenergic agonists, as negative synergistic effects may occur.
Pseudoephedrine
Pseudoephedrine used at 30-60mg every six hours to treat OD disorders. It is also used in Dysautonomia with decreased release of Norepinephrine from postganglionic SNS-neurons. It acts as a direct agonist in both adrenergic branches, specifically through α1, β1, and β2 adrenergic receptors. It is rapidly absorbed in the gastrointestinal tract. Onset is within 15-30 minutes, its duration is 4-6 hours, and is very inexpensive. Pseudoephedrine is a sym- pathomimetic drug of the Phenethylamine and Amphetamine chemical classes. It can be used as a nasal or sinus decongestant, as a stimulant, or as a wakefulness-promoting agent in the high- er doses. It is used both orally, and as a topical decongestant. Pseudoephedrine is used as a first-line prophylactic for recurrent Priapism [69]. Erection is largely a PSNS-response, which must reversed by Pseudoephedrine. SNS-action of Pseudoephedrine may serve to relieve this condition. It is often also used off-label for urinary incontinence. It is used in the ICU for refractory or severe hypotension [70,71]. It is contraindicated in patients with Diabetes, cardiovascular disease, significant Hypertension, sig- nificant Coronary Artery Disease, Prostate Hypertrophy, Hyper- thyroidism, Closed-Angle Glaucoma, and in pregnant women. It also relaxes the smooth muscle in the bronchi because of its β2 adrenergic receptor affect. It is a chemical precursor to Meth- amphetamine, and there is increasing Regulatory restriction on this drug.
A comparison of Ephedrine and Pseudoephedrine should be not- ed. Both drugs have an adrenergic effect. They are both Ephedra Alkaloids, classified as stimulants that mimic what Adrenalin does to the body. Ephedrine and Pseudoephedrine are both pre- cursors of Methamphetamine. With Ephedrine, however, a dif- ferent chemical structure found in a hydroxyl branch. Ephedrine is used to counteract OD, while Pseudoephedrine is used to de- congest nasal passageways. One is a cis-variant chemical and one is a trans-variant chemical (very similar structures). Both have the capacity to increase BP in patients, and Ephedrine is more powerful; however, Pseudoephedrine is more available.
Use of over-the-counter Ephedra Alkaloids has been associat- ed with potentially serious cerebral vascular events, especially Hemorrhagic Stroke [72]. The compound Phenylpropanolamine is substituted by Pseudoephedrine. Ephedra Alkaloids were shown to increase BP significantly in individuals with impaired Baroreflex function [72]. These individuals are largely Dysau- tonomia patients. The concomitant ingestion of Ephedra Alka- loids and water produces a greater increase in BP [72]. It is well known that drinking water rapidly can induce a SNS-response (see Figure 20). This interaction of drinking water with Ephedra Alkaloids can be beneficial in the treatment of OD. However, it could contribute to cardiovascular complications associated with diffuse use of Ephedra Alkaloids, given that Baroreflex function varies widely in normal individuals, and it was impaired in sev- eral medical conditions.
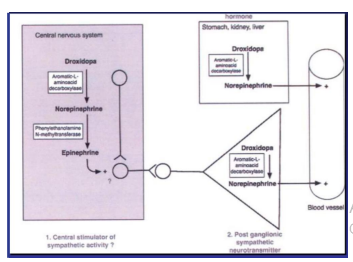
Figure 20: Change in systolic BP in patients with autonomic failure after administration of phenylpropanolamine (PPA). Pressor effect of phenylpropanolamine taken with 50 mL of wa- ter is compared with pressor effect of same dose of phenylpro- panolamine taken with 480 mL of water and with pressor effect of 480 mL of water alone. P<0.0001 between interventions by ANOVA. [Reprinted from 72, open access]
The use of Ephedra Alkaloids and Pseudoephedrine is avoided, when possible, in patients because of stroke risk and Hemor- rhagic risk. In addition, the regular chronic dosing of Pseudo- ephedrine (30-60mg tid or qid) and Ephedrine (15-40mg tid) to treat Hypotension or OD increases the probability of significant, adverse effects such as Tachycardia and Anxiety. Pseudoephed- rine and related Sympathomimetic cause modest increases in BP. Use with Droxidopa not advised. Use with caution or not at all recommended in open angle glaucoma, CAD, CHF, Be- nign Prostatic Hyperplasia, Hypertension, and Urinary retention. Based on quality of evidence and safety issues, Midodrine and Droxidopa (Northera) are preferred. Midodrine and Droxidopa reach a strong recommendation level for NOH compared with other agents where the strength is weak and quality of evidence is low [73].
Alternate Agents
The other agents with vasoconstriction properties that used to treat OD include Atomoxetine, Yohimbine, Pyridostigmine, and Octreotide, as well as volume expansion agents, such as Flud- rocortisone and DDAVP, as previously mentioned. Fludrocorti- sone has some vasoconstrictive properties but viewed as a vol- ume-expansion agent. The selective Norepinephrine transport inhibitor Atomoxetine increases seated and standing BP and im- proves OD even when given in pediatric doses of 18mg [74-76]. It shown to be effective in Multiple System Atrophy patients due to the presence of residual SNS-tone. It should be avoided with structural cardiac diseases, liver diseases and psychiatric diseas- es. Suicidal Ideation, Sudden Cardiac Death, Stroke, Myocardial Infarction, Hepatotoxicity and Depression have been reported.
The α2 adrenergic antagonist Yohimbine shown to increase seated and standing BP and reduces pre-Syncopal episodes in patients with Autonomic Failure [77]. This drug acts centrally on the SNS-outflow and Norepinephrine release from post-gan-glionic SNS-neurons. It works well in MSA patients. It is avail- able only from compounding pharmacies. Pyridostigmine (Mes- tinon), in 60mg doses, preferentially increases upright BP and improves orthostatic symptoms without worsening supine Hy- pertension. Mestinon may be the drug of choice for patients who have very severe, high BP and severe supine Hypertension when Midodrine and Northera cannot be used. However, Mestinon is not as efficacious as Midodrine and Northera and, oftentimes, if one has severe Autonomic Failure, it is not sufficient. It is an excellent add-on medicine to Midodrine or Northera in patients with NOH. It is also an excellent add-on agent in the treatment of POTS when Midodrine and Beta-Blockers are ineffective in controlling symptoms and HR.
The Somatostatin analogue, Octreotide, which is subcutaneously used, is also very effective in the treatment of OD by constrict- ing Splanchnic circulation to prevent venous pooling [78,79]. However, it has side effects of Hyperglycemia and abdominal pain and can cause diarrhea. Oftentimes, combination therapy with Midodrine used with this agent in people with post-prandial Hypotension. One study showed sympathomimetic agents, such as Midodrine and Ephedra Alkaloids, have a synergistic effect on BP when combined with 480mL bolus of water [31]. DDAVP used to improve symptoms in patients with POTS [80]. It is used chronically in these patients at a dosage of 0.1 to 0.2 mg once a week and not daily to avoid too much free water accumulation with hyponatremia, increased headache, and edema [personal communication, DePace]. DDAVP is an agonist to the Vasopres- sin type-2 receptor, not type-1 receptor, and therefore does not raise BP [7]. DDAVP is often valuable in patients who cannot consume sufficient quantities of water due to Gastroparesis or find no relief with 48-64oz of water a day, including with elec- trolytes [personal communication, DePace].
As mentioned, Fludrocortisone is a powerful mineralocorticoid that acts as a volume expander, which is not recommended in pa- tients with CHF, Hypertension, edema, or hypokalemia. DDAVP may be safer in these patients, but not in patients with a history of blood clots as it can theoretically increase Factor 8 and Von Willebrand factor. It is prescribed prn as a “pill in the pocket” for occasional decompensation episodes and during the men- strual cycle when female patients may be symptomatic due to fluid loss. Patients with recurrent Syncope and positive head-up tilt were prescribed Methylphenidate (Ritalin, 10mg, tid) which resulted in six of the seven patients (85.7%) responding favor- ably [81]. Rarely has Methylphenidate been used off-label in OD patients. It is used in the treatment of Narcolepsy, Bipolar and Depressive disorders, Attention Deficit Disorders, and for enhancing performance, especially in cognition and memory. Methylphenidate carries addiction and dependence potential and is avoided when possible.
One very important point is that many patients are taking cen- tral stimulants, such as Ritalin and Adderall, for treatment of so- called ADHD. However, in reality, these patients have impaired memory and cognitive function, often from a lack of blood sup- ply to their head due to OD, including decreased αSNS activity. Oftentimes, these patients are treated with vasoactive agents first to see if their cognition improves, and brain fog lessens before even considering them for treatment with central stimulants by Psychiatry. In the United States, Methylphenidate is classified as Schedule II controlled substance. It is recognized to have medi- cal value but presents a high potential for abuse.
Conclusions
Midodrine is approved for the treatment of OH. However, signif- icant data now supports use off-label in certain situations, such as intra-Dialysis, Hypotension, Cirrhosis and Ascites Hepatorenal Syndrome, VVS, and POTS. It is used in Heart Failure patients with low BPs in order to titrate their medications up more fully. It is used in ICUs to have patients both come off vasopressors early and decrease length of stay when they have Hypotension postoperatively or Septic Shock. In those instances, much higher doses are used than for Autonomic Dysfunction treatment. Mi- dodrine appears to be the mainstay of treatment in OD symptoms with Dysautonomia and is a first-line pharmacological agent and cost-effect. Decreased αSNS activity is a common mechanism that underlies the Dysautonomias discussed in this paper (OI, OH, NOH, POTS, & VVS) and is the reason why Midodrine (and perhaps secondarily Northera), are effective therapy and priority treatment for all. Many patients discontinue the use of Midodrine initially because of side-effects. However, if the pa- tient is properly warned that these side-effects may occur, and they should be interpreted as though the medicine is working properly, they will continue use until they eventually adapt to the medication; starting at very low doses, as low as 1.25mg once a day, may be necessary. Midodrine is a pure α-adrenergic agonist similar to Phenylephrine, which is given intravenously. Howev- er, unlike Phenylephrine, in which a tachyphylaxis can be seen when patients get used to that medication, this is not the case with Midodrine, and it is useful long-term without losing its ef- fectiveness. Oftentimes, add-on medications are needed, such as volume expanders like DDAVP and Fludrocortisone, Mestinon (Pyridostigmine), and other agents. Fludrocortisone is avoided when possible.
For patients who cannot tolerate Midodrine or are contraindicat- ed for Midodrine, Mestinon is the second-line pharmaceutical therapy. If Mestinon is not tolerated, then Northera is prescribed. While it is possible to anticipate those patients who will even- tually tolerate Northera [personal communication, DePace], its cost and whether insurances will reimburse for it requires that Midodrine and Mestinon be attempted and found ineffective first. While r-ALA is a useful adjunct to pharmaceutical therapy, it may also be able to replace pharmaceutical therapy in cases where no pharmaceutical therapy is tolerated or preferred. In most cases, pharmaceutical therapy helps to accelerate the relief of Dysautonomias that r-ALA is known to treat.
Northera (Droxidopa) is a very efficacious drug in treating OD disorders, especially NOH (for which it has FDA approval) and in some select cases of POTS. Northera has a low side-ef- fects profile, is better tolerated, and produces less supine Hy- pertension than Midodrine. If need be, the two agents can be used together, carefully, by experienced physicians. Mestinon (Pyridostigmine) can also be added onto these agents and has little effect on supine Hypertension. Pseudoephedrine is also a possibility for Orthostatic Dysfunction therapy, however, rarely in the clinical setting; unless in those rare cases where there is decreased ability of postganglionic SNS-neurons to release Nor- epinephrine. The limited utility is further necessary due to its contraindications in diabetes, heart disease, and the others listed above. Pseudoephedrine is most often used (when it is used) in the intensive or critical care setting.
All of these therapies, once titrated appropriately for the indi- vidual patient, have been found to be very effective in ultimately relieving the Dysautonomia(s) and related symptoms. Titration is based on symptoms and P&S Monitoring. Dysautonomia therapy may take up to 24 months because it takes small, gen- tle changes to correct the P&S nervous systems. Unfortunately, faster, larger doses cause more problems by pushing P&S bal- ance too far in the other direction. This is in part why multiple modalities are used when possible (e.g., low-and-slow exercise with very low-dose anti-cholinergics, or high dose r-ALA with low-dose Midodrine or Northera). Together, with the coopera- tion of the patient, these therapy plans restore these complex patients to health and function, improving QOL and returning them to productive lifestyles.
Abbreviations
NOH: Neurogenic Orthostatic Hypotension
OD: Orthostatic Dysfunction
OH: Orthostatic Hypotension
OI: Orthostatic Intolerance
P&S: Parasympathetic and Sympathetic
PSNS: Parasympathetic Nervous System or Parasympathetic or Vagal or Cholinergic
QOL: Quality of Life
r-ALA: r-Alpha-Lipoic Acid
SNS: Sympathetic Nervous System or Sympathetic or Adren- ergic
VVS: Vasovagal Syncope
Conflicts of Interest
Only Dr. Colombo has any conflicts of interest, specifically as inventor of P&S Monitoring technology and officer of Physio PS, Inc. *The clinical criteria for POTS is a rise in HR upon standing (or tilt-table testing – postural change) of 30 bpm for adults (40 bpm for pediatrics), or a HR of over 120 bpm within the first 10 minutes of postural change, after ruling out OH.
**The clinical criteria for OH (including NOH) is a drop of 20 mmHg in systolic BP or a drop of 10 mmHg in diastolic BP within two to five minutes of standing (or tilt-table testing – pos- tural change), or if postural change causes signs or symptoms of lightheadedness, nausea, etc.
***Persistent Fatigue is fatigue that has some, but not all, of the symptoms of Chronic Fatigue Syndrome.
****Higher, typically clinical doses of anti-cholinergics (e.g., > 25mg Nortriptyline, qd, dinner) aggravate symptoms or cause additional symptoms, often being the very symptoms that are being treated or prevented.
References
- Benarroch, E. E. (2012). Postural tachycardia syndrome: a heterogeneous and multifactorial disorder. In Mayo Clinic Proceedings, 87(12), 1214-1225.
- Zhao, J., Tang, C., Jin, H., & Du, J. (2014). Plasma copeptin and therapeutic effectiveness of midodrine hydrochloride on postural tachycardia syndrome in children. The Journal of Pediatrics, 165(2), 290-294.
- Ruzieh, M., Dasa, O., Pacenta, A., Karabin, B., & Grubb,B. (2017). Droxidopa in the treatment of postural orthostat- ic tachycardia syndrome. American journal of therapeutics, 24(2), e157-e161.
- Northera Improves Postural Tachycardia Syndrome (POTS) and Postural Vasovagal Syncope (VVS). https://www.cen- terwatch.com/clinical-trials/listings/78506/postural-tachy- cardia-syndrome-pots-northera-improves-postural-tachy- cardia/.
- Vinik, A. I., Camacho, P. M., Davidson, J. A., Handelsman,Y., Lando, H. M., & et al. (2017). Task force to develop an AACE position statement on autonomic testing. Ameri- can Association of Clinical Endocrinologists and American College of Endocrinology Position Statement on testing for autonomic and somatic nerve dysfunction. Endocr Pract, 23(12), 1472-1478.
- Grant AO. Pharmacological management of POTS. www. slideshare.net/DukeHeartCenter/4b-pots-summit.
- Freeman, R., Abuzinadah, A. R., Gibbons, C., Jones, P., Mi- glis, M. G., & et al. (2018). Orthostatic hypotension: JACC state-of-the-art review. Journal of the American College of Cardiology, 72(11), 1294-1309.
- Colombo, J., Arora, R., DePace, N. L., & Vinik, A. I. (2015). Clinical Autonomic Dysfunction. Switzerland: Springer In- ternational Publishing.
- Arora, R. R., Bulgarelli, R. J., Ghosh-Dastidar, S., & Co- lombo, J. (2008). Autonomie mechanisms and therapeutic implications of postural diabetic cardiovascular abnormali- ties. Journal of diabetes science and technology, 2(4), 645- 657.
- Vinik, A. I., Maser, R. E., & Nakave, A. A. (2007). Diabet- ic cardiovascular autonomic nerve dysfunction. US Endocr Dis, 2, 2-9.
- Vinik, A. I., & Ziegler, D. (2007). Diabetic cardiovascular autonomic neuropathy. Circulation, 115(3), 387-397.
- Tobias, H., Vinitsky, A., Bulgarelli, R. J., Ghosh-Dastidar, S., & Colombo, J. (2010). Autonomic nervous system mon- itoring of patients with excess parasympathetic responses to sympathetic challenges–clinical observations. US Neurol, 5(2), 62-66.
- Bloomfield, D. M., Kaufman, E. S., Bigger Jr, J. T., Fleiss, J., Rolnitzky, L., & et al. (1997). Passive head-up tilt and actively standing up produce similar overall changes in au- tonomic balance. American heart journal, 134(2), 316-320.
- DePace, N. L., & Colombo, J. (2019). Autonomic and mito- chondrial dysfunction in clinical diseases: diagnostic, pre- vention, and therapy.
- Osswald, S., & Trouton, T. G. (1993). Neurocardiogenic (Vasodepressor) Syncope. New England Journal of Medi- cine, 329(1), 30-30.
- Nwazue, V. C., & Raj, S. R. (2013). Confounders of vaso- vagal syncope: postural tachycardia syndrome. Cardiology clinics, 31(1), 101-109.
- Prakash, S., Garg, A. X., Heidenheim, A. P., & House, A. A. (2004). Midodrine appears to be safe and effective for dial- ysis-induced hypotension: a systematic review. Nephrology Dialysis Transplantation, 19(10), 2553-2558.
- Kaufmann H. Treatment of orthostatic and postprandial hypotension. UpToDate. 2018. https://www.uptodate.com/ contents/treatment-of-orthostatic-and-postprandial-hypo- tension.
- Gutman, L. B., & Wilson, B. J. (2017). The role of mi- dodrine for hypotension outside of the intensive care unit. Journal of Population Therapeutics and Clinical Pharmacol- ogy, 24(3), e45-e50.
- Zakir, R. M., Folefack, A., Saric, M., & Berkowitz, R. L. (2009). The use of midodrine in patients with advanced heart failure. Congestive Heart Failure, 15(3), 108-111.
- Kikkeri, N. S., Nagarajan, E., Premkumar, K., & Nattana- mai, P. (2019). Reversible Cerebral Vasoconstriction Syn- drome Due to Midodrine in a Patient with Autonomic Dys- reflexia. Cureus, 11(3), e4285.
- Wecht, J. M., Rosado-Rivera, D., Handrakis, J. P., Radu- lovic, M., & Bauman, W. A. (2010). Effects of midodrine hydrochloride on blood pressure and cerebral blood flowduring orthostasis in persons with chronic tetraplegia. Ar- chives of physical medicine and rehabilitation, 91(9), 1429- 1435.
- Palma, J. A., Gonzalez-Duarte, A., & Kaufmann, H. (2019). Orthostatic hypotension in hereditary transthyretin amyloi- dosis: epidemiology, diagnosis and management. Clinical Autonomic Research, 29(1), 33-44.
- Whitson, M. R., Mo, E., Nabi, T., Healy, L., Koenig, S., & et al. (2016). Feasibility, utility, and safety of midodrine during recovery phase from septic shock. Chest, 149(6), 1380-1383.
- Hammond, D. A., Smith, M. N., Peksa, G. D., Trivedi, A. P., Balk, R. A., & et al. (2020). Midodrine as an adjuvant to intravenous vasopressor agents in adults with resolving shock: systematic review and meta-analysis. Journal of in- tensive care medicine, 35(11), 1209-1215.
- Splete H. (2016) Midodrine cuts ICU days in septic shock patients. CHEST Physician.
- Mattos, Â. Z., Schacher, F. C., & Mattos, A. A. (2019). Va- soconstrictors in hepatorenal syndrome–A critical review. Annals of Hepatology, 18(2), 287-290.
- Rizvi, M. S., Trivedi, V., Nasim, F., Lin, E., Kashyap, R., & et al. (2018). Trends in use of midodrine in the ICU: a sin- gle-center retrospective case series. Critical care medicine, 46(7), e628-e633.
- Anstey, M. H., Wibrow, B., Thevathasan, T., Roberts, B., Chhangani, K., & et al. (2017). Midodrine as adjunctive support for treatment of refractory hypotension in the in- tensive care unit: a multicenter, randomized, placebo con- trolled trial (the MIDAS trial). BMC anesthesiology, 17(1), 1-7.
- Arnold, A. C., & Raj, S. R. (2017). Orthostatic hypotension: a practical approach to investigation and management. Ca- nadian Journal of Cardiology, 33(12), 1725-1728.
- Arnold, A. C., & Shibao, C. (2013). Current concepts in orthostatic hypotension management. Current hypertension reports, 15(4), 304-312.
- Figueroa, J. J., Basford, J. R., & Low, P. A. (2010). Prevent- ing and treating orthostatic hypotension: as easy as A, B, C. Cleveland Clinic journal of medicine, 77(5), 298-306.
- Raj, S. R. (2013). Postural tachycardia syndrome (POTS). Circulation, 127(23), 2336-2342.
- Cambridgeshire and Peterborough. Midodrine for ortho- static hypotension and neurocardiogenic syncope. www. cambridgeshireandpeterboroughccg.nhs.uk/easysiteweb/ getresource.axd?assetid=10918&type=0&servicetype=1.
- Izcovich, A., Malla, C. G., Manzotti, M., Catalano, H. N., & Guyatt, G. (2014). Midodrine for orthostatic hypotension and recurrent reflex syncope: a systematic review. Neurolo- gy, 83(13), 1170-1177.
- Smith, W., Wan, H., Much, D., Robinson, A. G., & Mar- tin, P. (2016). Clinical benefit of midodrine hydrochloride in symptomatic orthostatic hypotension: a phase 4, double blind, placebo-controlled, randomized tilt-table study. Clin- ical Autonomic Research, 26(4), 269-277.
- Jacob, G., Shannon, J. R., Black, B., Biaggioni, I., Mosque- da-Garcia, R., & et al. (1997). Effects of volume loading and pressor agents in idiopathic orthostatic tachycardia. Cir- culation, 96(2), 575-580.
- Ross, A. J., Ocon, A. J., Medow, M. S., & Stewart, J. M. (2014). A double-blind placebo-controlled cross-over study of the vascular effects of midodrine in neuropathic com- pared with hyperadrenergic postural tachycardia syndrome. Clinical Science, 126(4), 289-296.
- Miller, A. J., & Raj, S. R. (2018). Pharmacotherapy for pos-tural tachycardia syndrome. Autonomic Neuroscience, 215, 28-36.
- Hale, G. M., Valdes, J., & Brenner, M. (2017). The treat- ment of primary orthostatic hypotension. Annals of Phar- macotherapy, 51(5), 417-428.
- Lei, L. Y., Raj, S. R., & Sheldon, R. S. (2022). Midodrine for the prevention of vasovagal syncope: a systematic re- view and meta-analysis. EP Europace. Canada J Cardiol, 35(10), S46.
- Murray, G. L., & Colombo, J. (2020). Maintenance (r) alpha lipoic acid reduces sudden cardiac death in geriatric diabe- tes mellitus II patients. Age (mean yrs), 67(64), 0-100.
- Grijalva, C. G., Biaggioni, I., Griffin, M. R., & Shibao, C.A. (2017). Fludrocortisone is associated with a higher risk of allâ?cause hospitalizations compared with midodrine in patients with orthostatic hypotension. Journal of the Ameri- can Heart Association, 6(10), e006848.
- Raj, S. R., & Hewitt, L. A. (2019). Use of droxidopa for the long-term treatment of neurogenic orthostatic hypotension. European Heart Journal, 40(1), 69-70.
- Jordan, J., Shibao, C., & Biaggioni, I. (2015). Multiple sys- tem atrophy: using clinical pharmacology to reveal patho- physiology. Clinical Autonomic Research, 25(1), 53-59.
- Robertson, D., Goldberg, M. R., Hollister, A. S., Wade, D., & Robertson, R. M. (1983). Clonidine raises blood pressure in severe idiopathic orthostatic hypotension. The American Journal of Medicine, 74(2), 193-200.
- Gibbons, C. H., Schmidt, P., Biaggioni, I., Frazier-Mills, C., Freeman, R., & et al. (2017). The recommendations of a consensus panel for the screening, diagnosis, and treatment of neurogenic orthostatic hypotension and associated supine hypertension. Journal of neurology, 264(8), 1567-1582.
- Wright, R. A., Kaufmann, H. C., Perera, R., Opfer-Gehrk- ing, T. L., McElligott, M. A., & et al. (1998). A double blind, dose-response study of midodrine in neurogenic orthostatic hypotension. Neurology, 51(1), 120-124.
- Gee, M. E., Watkins, A. K., Brown, J. N., & Young, E. J. (2018). Ivabradine for the treatment of postural orthostatic tachycardia syndrome: a systematic review. American Jour- nal of Cardiovascular Drugs, 18(3), 195-204.
- Messerli, F. H., Seiler, C., & Rimoldi, S. F. (2021). Ivabra- dine in POTS: Increase in Central Pressure Rather Than Down-Regulation of Sympathetic Nervous System. Journal of the American College of Cardiology, 77(24), 3141-3142.
- Benditt, D. G., Fahy, G. J., Lurie, K. G., Sakaguchi, S., Fa- bian, W., & et al. (1999). Pharmacotherapy of neurally me- diated syncope. Circulation, 100(11), 1242-1248.
- Al Turk, A. A., Odanovic, N. R., Waller, A. H., & Mazza,V. J. (2017). Midodrine and Pyridostigmine; Novelties in Heart Failure Therapy. Journal of Cardiovascular Disease Research, 8(4).
- Hewitt, L. A., Kymes, S., Jackson, K., Widolff, M., Sulli- van, C., & et al. (2019). Droxidopa and Midodrine Treat- ment Persistence in Patients with Orthostatic Hypotension (P3. 6-047).
- Patrick, K., & Martin, T. (2017). Effectiveness of droxido- pa compared to midodrine in standing blood pressure and orthostatic tolerance in adults with neurogenic orthostatic hypotension: a systematic review protocol. JBI Evidence Synthesis, 15(9), 2287-2294.
- Kaufmann, H. (2017). Droxidopa for symptomatic neuro- genic orthostatic hypotension: what can we learn?. Clinical Autonomic Research, 27(1), 1-3.
- Biaggioni, I., Freeman, R., Mathias, C. J., Low, P., Hewitt,L. A., & et al. (2015). Randomized withdrawal study of pa-tients with symptomatic neurogenic orthostatic hypotension responsive to droxidopa. Hypertension, 65(1), 101-107.
- Ross, A. J., & Stewart, J. M. (2015). Prospects for droxi- dopa in neurogenic orthostatic hypotension. Hypertension, 65(1), 34-35.
- Isaacson, S., Vernino, S., Ziemann, A., Rowse, G. J., Kalu, U., & et al. (2016). Long-term safety of droxidopa in pa- tients with symptomatic neurogenic orthostatic hypoten- sion. Journal of the American Society of Hypertension, 10(10), 755-762.
- What's Hot in PD? Could Northera (Droxidopa) Be an Alternative Treatment for Low Blood Pressure and Pass- ing Out Symptoms in Parkinson's Disease? https://www. parkinson.org/blogs/whats-hot/northera-droxidopa-alterna- tive-treatment.
- White, W. B., Hauser, R. A., Rowse, G. J., Ziemann, A., & Hewitt, L. A. (2017). Cardiovascular safety of droxidopa in patients with symptomatic neurogenic orthostatic hypo- tension. The American Journal of Cardiology, 119(7), 1111- 1115.
- Chen, J. J., Han, Y., Tang, J., Portillo, I., Hauser, R. A., & et al. (2018). Standing and supine blood pressure outcomes associated with droxidopa and midodrine in patients with neurogenic orthostatic hypotension: a Bayesian meta-anal- ysis and mixed treatment comparison of randomized trials. Annals of Pharmacotherapy, 52(12), 1182-1194.
- Olshansky, B., & Muldowney, J. (2020). Cardiovascular safety considerations in the treatment of neurogenic ortho- static hypotension. The American Journal of Cardiology, 125(10), 1582-1593.
- Ruzieh, M., Dasa, O., Pacenta, A., Karabin, B., & Grubb,B. (2017). Droxidopa in the treatment of postural orthostat- ic tachycardia syndrome. American journal of therapeutics, 24(2), e157-e161.
- Hauser, R. A., Isaacson, S., Lisk, J. P., Hewitt, L. A., & Rowse, G. (2015). Droxidopa for the S hortâ?T erm T reat- ment of S ymptomatic N eurogenic O rthostatic H ypoten- sion in P arkinson's D isease (n OH 306 B). Movement Dis- orders, 30(5), 646-654.
- Kremens, D., Lew, M., Claassen, D., & Goodman, B. P. (2017). Adding droxidopa to fludrocortisone or midodrine in a patient with neurogenic orthostatic hypotension and Parkinson disease. Clinical Autonomic Research, 27(1), 29- 31.
- Donnelly, J. P., Hanna, M., Sperry, B. W., & Seitz Jr, W. H. (2019). Carpal tunnel syndrome: a potential early, red-flag sign of amyloidosis. The Journal of Hand Surgery, 44(10), 868-876.
- Oommen, J., Chen, J., Wang, S., Caraccio, T., & Hanna, A. (2019). Droxidopa for hypotension of different etiologies: two case reports. Pharmacy and Therapeutics, 44(3), 125- 144.
- Weinstock, L. B., Brook, J. B., Myers, T. L., & Goodman B. (2018). Case Report: Successful treatment of postural orthostatic tachycardia and mast cell activation syndromes using naltrexone, immunoglobulin and antibiotic treatment. BMJ Case Reports, 2018.
- Mocniak, M., Durkin, C. M., & Early, K. (2012). The use of sudafed for priapism in pediatric patients with sickle cell disease. Journal of Pediatric Nursing, 27(1), 82-84.
- Van Berkel, M. A., Fuller, L. A., Alexandrov, A. W., & Jones, G. M. (2015). Methylene Blue, Midodrine, and Pseu- doephedrine. Critical Care Nursing Quarterly, 38(4), 345- 358.
- Patterson, A., Bourgault, A. M., & Crawford, B. R. (2008). Correction of severe hypotension by oral pseudoephedrine in a patient with idiopathic autonomic dysfunction. Ameri- can Journal of Critical Care, 17(5), 484-481.
- Jordan, J., Shannon, J. R., Diedrich, A., Black, B., Robert- son, D., & et al. (2004). Water potentiates the pressor effect of ephedra alkaloids. Circulation, 109(15), 1823-1825.
- Eschlböck, S., Wenning, G., & Fanciulli, A. (2017). Evi- dence-based treatment of neurogenic orthostatic hypoten- sion and related symptoms. Journal of Neural Transmission, 124(12), 1567-1605.
- Green, E. A., Raj, V., Shibao, C. A., Biaggioni, I., Black,B. K., & et al. (2013). Effects of norepinephrine reuptake inhibition on postural tachycardia syndrome. Journal of the American Heart Association, 2(5), e000395.
- Okamoto, L. E., Shibao, C. A., Gamboa, A., Diedrich, A., Raj, S. R., & et al. (2019). Synergistic pressor effect of ato- moxetine and pyridostigmine in patients with neurogenic orthostatic hypotension. Hypertension, 73(1), 235-241.
- Byun, J. I., Kim, D. Y., Moon, J., Shin, H. R., Sunwoo, J. S., & et al. (2020). Efficacy of atomoxetine versus midodrine for neurogenic orthostatic hypotension. Annals of clinical and translational neurology, 7(1), 112-120.
- Shibao, C., Okamoto, L. E., Gamboa, A., Yu, C., Diedrich, A., & et al. (2010). Comparative efficacy of yohimbine against pyridostigmine for the treatment of orthostatic hy- potension in autonomic failure. Hypertension, 56(5), 847- 851.
- Ong, A. C. L., Myint, P. K., Shepstone, L., & Potter, J. F. (2013). A systematic review of the pharmacological man- agement of orthostatic hypotension. International journal of clinical practice, 67(7), 633-646.
- Hoeldtke, R. D., Bryner, K. D., Hoeldtke, M. E., & Hobbs,G. (2006). Treatment of postural tachycardia syndrome: a comparison of octreotide and midodrine. Clinical Autonom- ic Research, 16(6), 390-395.
- Coffin, S. T., Black, B. K., Biaggioni, I., Paranjape, S. Y., Orozco, C., & et al. (2012). Desmopressin acutely decreases tachycardia and improves symptoms in the postural tachy- cardia syndrome. Heart rhythm, 9(9), 1484-1490.
- Grubb, B. P., Kosinski, D., Mouhaffel, A., & Pothoulakis,A. (1996). The use of methylphenidate in the treatment of refractory neurocardiogenic syncope. Pacing and clinical electrophysiology, 19(5), 836-840.




