Research Article - (2025) Volume 3, Issue 5
Nephroprotective Activity of Ethanolic Leaves Extract of Vachellia origena (Hunde) Kyal. & Boatwr. (Fabaceae) on Paracetamol-Induced Nephrotoxicity in Male Guinea Pigs
2Biology Department, Science Collage, Ibb University, Ibb, Yemen
3Department of Pharmacy, Faculty of Medical Sciences, Al-Jazeera University, Ibb City, Yemen
Received Date: Aug 22, 2025 / Accepted Date: Oct 25, 2025 / Published Date: Oct 31, 2025
Copyright: ©©2025 Warda Mohamed Abdu Kaidama, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Saif, M. S. M., Kaidama, W. M. A., Alahdal, A. A., Al-Nawah, A. A. A., Al-Hajj, Y. A. A., et al. (2025). Nephroprotective Activity of Ethanolic Leaves Extract of Vachellia origena (Hunde) Kyal. & Boatwr. (Fabaceae) on Paracetamol-Induced Nephrotoxicity in Male Guinea Pigs. Int Internal Med J, 3(5), 01-08.
Abstract
Vachellia origena is a medicinal plant found in J. Saber, Alturba, Ibb, Dhamar, Sanaa, Haraz, Shibam and Yafea. They are utilized therapeutically to address many conditions, including antibacterial, antifungal, and antioxidant applications. This study aimed to investigate the preventive potential of V. origena against paracetamol-induced kidney injury in male guinea pigs. Thirty-five male guinea pigs (350–650 g) were randomly assigned to seven groups, each including five guinea pigs. Group I served as the control group. Group II was administered PCM (500 mg/kg) exclusively, Group III received of PCM and proximol (0.8 mg/kg), Group IV was given V. origena extract at a dosage of 100 mg/kg alone, and Group V received V. origena extract at a dosage of 200 mg/kg alone. In group VI, we received PCM (500 mg/kg) and 100 mg/kg of V. origena extract. Simultaneously, group VII received PCM (500 mg/kg) and 200 mg/kg of V. origena extract. The treatment duration was ten days, following which sera were collected and analyzed for serum renal indices using conventional procedures. The results indicated that the PCM-only group exhibited elevated serum urea and creatinine levels, alongside reduced total protein and albumin levels. Additionally, it induced several histological alterations. Treatment with ethanolic leaf extract of V. origena (100 mg/kg and 200 mg/kg) mitigated renal intoxication generated by PCM and reduced the extent of histological lesions in guinea pigs. Our investigation indicates that the ethanolic extract of V. origena leaves has protective properties against PCM-induced renal damage.
Keywords
Paracetamol, Vachellia origena, Serum Kidney Indices, Nephroprotective Activity
Introduction
Paracetamol (PCM), also known as acetaminophen (C8H9NO2) or N-acetyl-p-aminophenol, is a pharmaceutical agent utilized for its analgesic and antipyretic properties. Nevertheless, it possesses restricted anti-inflammatory capabilities [1]. Paracetamol is usually recognized as safe for human usage when taken at authorized levels. However, potentially fatal liver damage can develop in cases of acute overdose or, in rare occasions, even when standard doses are consumed by specific individuals [2]. Hepatotoxicity is a known consequence of an overdose of PCM, which typically results in nephrotoxicity. Renal insufficiency is reported to occur in 1-2% of patients who are exposed to PCM toxicity [3]. After oral administration, approximately 63% of paracetamol is metabolized through glucuronidation, while 34% is metabolized via sulfation, primarily in the liver. The resulting water-soluble compounds from these metabolic processes are eliminated by the kidneys. N-acetyl-p-benzoquinone imine (NAPQI) is a reactive intermediate generated when the microsomal P-450 enzyme system oxidizes 55% of paracetamol. Intracellular glutathione (GSH) detoxifies NAPQI at therapeutic levels [3]. NAPQI has been identified as the causative metabolite of PCM poisoning [4]. In cases of PCM overdosing, glutathione stores are depleted, and necrosis is caused by a rapid increase in the concentration of NAPQI [5]. Furthermore, this condition leads to the production of substantial metabolites, which generate substantial quantities of unbound reactive species. PCM toxicity induces acute tubular necrosis, which is one of the primary causes of acute renal failure. Serum urea and creatinine levels may serve as indicators of acute tubular necrosis resultant from PCM [3]. Free radicals are generated as a result of drug toxicity in an organism, and oxidative damage is a significant factor in hepatorenal injuries caused by PCM [6]. Consequently, natural substances with antioxidant properties may be employed as substitute therapies for PCM toxicity [3].
The genus Vachellia Wight & Arn. (Acacia Mill.) is among the largest genera within the Fabaceae family. It has approximately 1350 species, categorized into three subgenera: Acacia, Aculeiferum and Phyllodineae. The Acacia is mostly a tropical tree, yet it also inhabits subtropical regions. Approximately 1000 species inhabit Australia, while 170 species are indigenous to Africa, of which 18 are widespread and 152 are endemic to the African continent, specifically Ethiopia, Eritrea and Saudi Arabia [7-9]. The genus Acacia Mill. is represented in Yemen by 32 species: 5 introduced and cultivated as ornamental trees (A. auriculiformis, A. calcicola, A. cyanophylla, A. Cyclops and A. farnesiana); 25 natives to the mainland, including A. origena and 2 endemics to Socotra Island, A. sarcophylla and A. pennivenia [10].
Vachellia origena Hunde (synonym: Acacia origena) is a flat- topped tree up to 12 meters tall with a yellow-brown, papery peeling of layers; paired, straight spines; yellow-brown branchlets; bipinnate leaves; 10–25 pairs of leaflets; flowers in globular cream heads; and glabrous green-brown pods [11,10]. It is prevalent on the escarpment over 1900 m and also located on mountains around the high plateau between 2300 m and 2900 m; hence, it thrives at altitudes greater than any other Acacia in Yemen [10]. Previous studies indicate that Vachellia origena trees have medicinal properties due to the presence of various phytochemicals, such as phenols, flavonoids, tannins, saponins, alkaloids, steroids, and carbonyls [12]. As a result, they are used in medical applications to treat a variety of ailments, functioning as antioxidants and antibacterial agents [13,14]. This study utilized silymarin as a standard phytotherapeutic agent. Similar to silymarin, it is essential to identify additional plant- based sources for treating renal illnesses and other conditions with minimal or no adverse effects. The use of cost-effective and beneficial medications to prevent renal toxicity reduces hospitalization costs and helps lower morbidity and mortality rates.
The study was based on the nephroprotective properties of related species and their polyphenolic chemicals in animal models, as well as the in vivo protective effects of Vachellia origena. The current study aimed to investigate the preventive/ curative potential of V. origena against paracetamol-induced kidney injury in guinea pigs. We expected that flavonoids would prevent PCM-induced kidney injury due to their antioxidant properties. In this work, we sought to determine what effect V. origena might have on the nephrotoxic consequences of PCM treatment.
Materials and Methods
Collection of Plant
Green leaves of V. origena were obtained in Waqash Village, Jiblah, Ibb City, Yemen. Plant specimens were identified and authenticated by Dr. Esam Aqlan, Assistant Professor of Plant Taxonomy and Flora, Department of Biology, Faculty of Sciences, Ibb University. A voucher specimen was placed at the Biology Herbarium, Faculty of Sciences, Ibb University, with the code CM202115.
Plant Extraction
The obtained fresh plant leaf material was thoroughly cleaned with tap water to remove dirt before being dried in an oven at 40°C until brittle. After drying, the leaves were crushed roughly and pulverized with a blender. 30 grams of leaf powder and 3000 milliliters of 70% ethanol were used. The extraction was placed in the electric shaker for 24 hours, then the extract was filtered, and the ethanol was removed by evaporating the extract in an oven at 40°C to obtain the extract powder.
Preliminary Phytochemical Screening
Phytochemical screening was conducted following established methodologies outlined in the literature [15].
Experimental Protocol
All methods utilizing laboratory animals were evaluated and sanctioned by the Institutional Animal Ethics Committee of Aljazeera University-Yemen. Thirty-five male guinea pigs (350– 650 g) were maintained in a controlled setting with a regulated room temperature and a 12-hour light-dark cycle, allowing unrestricted access to food and water ad libitum. Guinea pigs were randomly allocated into seven groups of five animals each, with all treatments administered daily for a duration of 10 days. Paracetamol (PCM) and plant extracts at 100 and 200 mg/kg of body weight were administered orally. Guinea pigs in group I acted as the control group, receiving just distilled water. Group II got PCM (500 mg/kg) alone; Group III received PCM (500 mg/kg) and proximol (0.8 mg/kg); Group IV received 100 mg/kg V. origena ethanolic leaves extract alone; and Group V received 200 mg/kg V. origena ethanolic leaves extract alone. In Group VI, we received 500 mg/kg PCM and 100 mg/kg V. origena ethanolic leaf extract. Meanwhile, Group VII was given 500 mg/ kg PCM and 200 mg/kg V. origena ethanolic leaf extract. On day 11, all animals were sedated with chloroform and blood was drawn.
Sample Preparation
The blood sample placed in tubes has no anticoagulant, allowing the blood to clot at room temperature for thirty minutes. To obtain the serum, the blood was centrifuged at 3000 rpm for 15 minutes. The serum samples were collected into clean tubes and stored in the deep freezer for biochemical analysis. The kidney was extracted when the guinea pig was slaughtered, cleaned for histological examination, and kept in 10% formalin.
Biochemical Analysis
Total proteins, albumin, blood urea nitrogen and creatinine were all estimated from serum samples. All estimates were performed using a diagnostic kit (Span Diagnostics Ltd., India) in accordance with the manufacturer's instructions [16].
Histological Analysis
Kidney histology was examined using conventional histology procedures. Briefly, when the animal was slaughtered, the kidney portion was taken, cleaned in normal saline, and cut into minute pieces. The sectioned tissue was then fixed in 10% formalin, gradually dehydrated with increasing percentages of ethanol solution (50% to 100%), and embedded in paraffin. Tissue slices of 4-μm thickness were generated using a microtome, fixed overnight on slides, stained with hematoxylin and eosin (H&E), and subsequently examined under a light microscope (Olympus BX41, Japan).
Statistical Analysis
Data expressed as mean ± standard error of the mean (n=5). The statistical variances across treatment groups were analyzed using one-way analysis of variance (ANOVA) and Tukey’s test using SPSS (version 20). The significance levels were established at p<0.05.
Ethics Approval and Consent to Participate
The research proposal was initially submitted to the Department of Pharmacy at Aljazeera University, which then forwarded it to the Ethical Review Committee of the Faculty of Medical Sciences at the same institution. This study utilizes guinea pig models, and the experimental protocol for animal use received approval (Reference Number: 0114 on September 29, 2024).
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Results
Phytochemical's Screening of Vachellia origena Leaves
Qualitative analyses for several phytochemical elements were conducted on the leaf extracts, and the findings are delineated in Table 1.
|
Phytocjemical |
Result |
|
Flavonoids |
+ |
|
Tannins |
+ |
|
Alkaloids |
+ |
|
Steroids |
+ |
|
Glycosides |
_ |
|
Triterpenoids |
+ |
|
Saponins |
_ |
|
(+) Present; (-) Absent |
|
Table 1: Phytochemical Screening of Alcoholic Extracts of V. origena Leaves
The initial phytochemical analysis of V. origena ethanolic leaf extracts revealed the presence of flavonoids, tannins, alkaloids, steroids and triterpenoids; however, glycosides and saponins were not detected.
Effect of Treatment with Ethanolic Extract of V. origena Leaves on Serum Biochemical Parameters
The serum profile demonstrates adequate renal function. Urea, serum creatinine, and uric acid concentrations were regarded as indicators of functional nephrotoxicity. PCM-induced nephrotoxicity was evidenced by the increased (p<0.05) levels of blood urea nitrogen and creatinine, but serum albumin and total protein (p<0.05) diminished compared to the control group (Table 2). Treatment groups mitigated the harmful effects of PCM by significantly (p<0.05) increasing serum urea nitrogen, creatinine, albumin, and total protein levels.
Treatment with V. origena ethanolic extract resulted in significant (p<0.05) restoration of the aforementioned parameters in guinea pigs. V. origena ethanolic extracts alone in the treatment group exhibited no significant difference in serum parameters compared to the control group, supporting the non-toxic impact of the chosen dosage.
|
|
Urea (mg/dl) |
Creatinine (mg/dl) |
Total protein (g/dl) |
Albumin (g/dl) |
|
Mean ± SD |
Mean ± SD |
Mean ± SD |
Mean ± SD |
|
|
Control |
19.00±1.41 |
0.38±0.05 |
5.18±0.09 |
3.60±0.29 |
|
Paracetamol (PCM) |
36.67±1.69# |
0.73±0.09# |
3.60±0.29# |
2.70±0.16# |
|
PCM +Proximol(0.8 mg/kg) |
21.33±0.94* |
0.60±0.01* |
4.93±0.12* |
3.20±0.16* |
|
V. origena (100 mg/kg) |
19.67±1.05* |
0.38±0.05* |
5.08±0.38* |
3.60±0.21* |
|
V. origena (200 mg/kg) |
18.33±1.35* |
0.38±0.05* |
5.05±0.26* |
3.53±0.23* |
|
PCM + V. origena (100 mg/kg) |
24.00±1.94* |
0.50±0.01* |
4.83±0.33* |
3.40±0.28* |
|
PCM+ V. origena (200 mg/kg) |
20.67±1.05* |
0.45±0.05* |
4.80±0.08* |
3.40±0.21* |
|
All value represents mean± SD of five animals. # P < 0.05 compared with normal control value. * P < 0.05 compared with non-treated control values. |
||||
Table 2: Effect of V. origena Extracts Treatment on the Biochemical Parameters Involved in Nephrotoxicity induced by Paracetamol
Figure 1: Effect of PCM and V. origena Extract on Urea Levels (mg/dl) in PCM- Induced Nephrotoxicity In Guinea Pigs
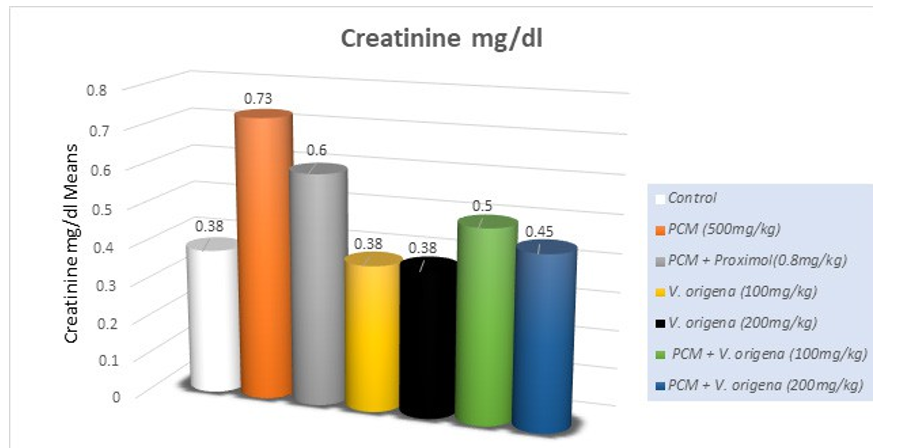
Figure 2: Effect of PCM and V. origena Extract on Creatinine Levels (mg/dl) in PCM- Induced Nephrotoxicity in Guinea Pigs
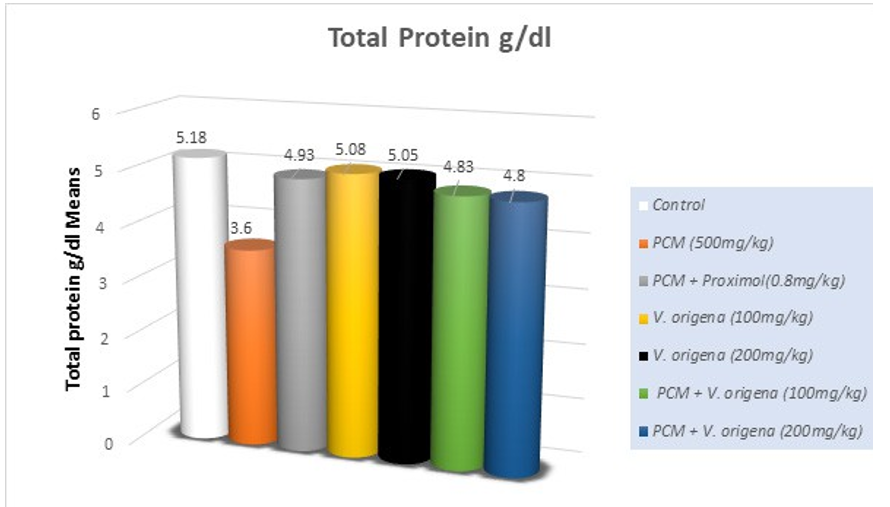
Figure 3: Effect of PCM and V. origena Extract on Total Protein (g/dl) in PCM- Induced Nephrotoxicity in Guinea Pigs
Figure 4: Effect of PCM and V. origena Extract on Albumin (g/dl) in PCM- Induced Nephrotoxicity in Guinea Pigs
Effect of V. origena Extracts on Kidney Histopathology in PCM- Induced Nephrotoxicity in Guinea Pigs
Histopathological analysis of kidney sections of the control group revealed normal morphology of glomeruli, tubules, blood vessels and interstitium (Figures 5a, b, and c). After 10 days of PCM administration, acute renal injury occurred, resulting in significant congestion of the glomerular tuft, renal blood vessels and interlobular blood capillaries. Perivascular and periglomerular leukocytic infiltrate was also seen (Figures 5.d and e). The analysis of kidney sections from guinea pigs fed PCM and the proximal treatment group for 10 days revealed a normal appearance, exhibiting only little congestion in the renal blood vessels and interlobular capillaries. The glomeruli exhibited congestion in the glomerular tuft, accompanied by vacuolation of the glomerular endothelium. Atrophy of some renal glomeruli was also seen (Figure 5.f).
The kidney tissue section of guinea pigs administered 100 mg/ kg of ethanol leaf extract from V. origena alongside PCM for 10 days exhibited minimal histological alterations compared to the PCM-only group, including mild congestion of the glomerular tufts and interlobular blood capillaries. The glomeruli exhibit a small enlargement of the glomerular tuft, accompanied by eosinophilic material in the glomerular space (Figure 5.g). The analysis of kidney tissue sections from guinea pigs treated with PCM and 200 mg/kg of ethanol extract from V. origena for 10 days revealed minimal histological alterations compared to the PCM-only group, including mild congestion of the glomerular tufts and interlobular blood capillaries. The glomeruli exhibit a small enlargement of the glomerular tuft, accompanied by eosinophilic material in the glomerular space (Figure 5.h.).
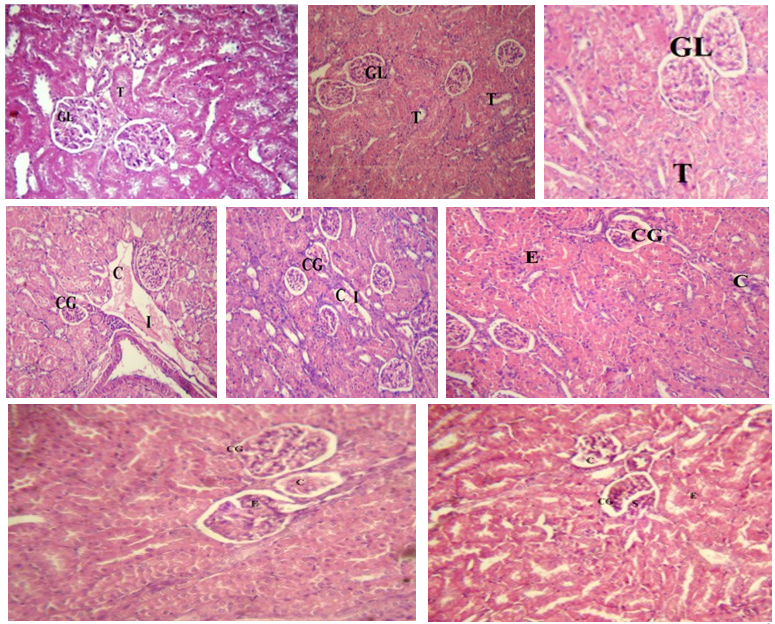
Figure 5: Histopathological Study of Kidney Tissue of Guinea Pigs
(a) Control group given distilled water for ten days showed normal kidney architecture(H&E×100), (b and c) group given V. origena (100 and 200 mg/kg) showed the normal appearance of glomerulus (GL) and renal tubules (T) (H&E×100), (d and e) PCM intoxicated kidney tissue showed shrinking and congestion of the glomerular tuft (CG), renal tubules and renal blood vessels (C) as well as interlobular blood capillaries. Also, mononuclear leukocytic infiltration(I), (f) effect of Proximol treatment on PCM intoxicated kidney tissue showing little congestion and shrinkage of some of the renal glomeruli (CG) and renal tubules (C). The glomeruli show presence of eosinophilic (E)debris in the glomerular space, (g) effect of 100 mg/kg V. origena treatment on PCM intoxicated kidney tissue showing little congestion and shrinkage of some of the renal glomeruli (CG) and renal tubules (C). The glomeruli show presence of eosinophilic (E)debris in the glomerular space, (h) effect of 200 mg/kg V. origena treatment on PCM intoxicated kidney tissue showing little congestion and shrinkage of some of the renal glomeruli (CG) and renal tubules (C). The glomeruli show presence of eosinophilic (E)debris in the glomerular space
Discussion
Several investigations have previously established PCM- induced nephrotoxicity [17]. A notable increase in urea and creatinine levels in the PCM-alone group in this study indicates that administering 500 mg/kg of PCM caused kidney injury. Creatinine is generated through protein metabolism in muscles, with the majority being filtered from the blood by the kidneys and eliminated in urine. In renal disease, serum urea accumulates, resulting in uremia, when the generation rate exceeds the clearance rate [18]. The markedly elevated blood urea in the PCM-treated group indicates renal impairment. N-acetyl-p- benzoquinone imine's (NAPQI) harmful action is what causes PCM-induced nephrotoxicity. PCM undergoes oxidation by cytochrome P-450, resulting in the formation of the reactive intermediate metabolite NAPQI [19]. Another factor contributing to PCM toxicity is the production of reactive oxygen species (ROS), particularly superoxide anions. Glutathione significantly reduces the nephrotoxicity caused by ROS and NAPQI during the early phases of toxicity [20]. However, after glutathione is used up, NAPQI chemically binds to the sulfhydryl groups of proteins during the later stages [21]. The significant reduction (P<0.05) in serum total protein and albumin in the PCM-treated group (Table 2) may be attributable to the arylation of protein by NAPQI [21]. Protein level in the blood in V. origena-treated groups (100 and 200 mg/kg) was considerably higher than in the PCM-treated group, indicating that V. origena may be able to reduce the harmful effects of PCM. Also, the creatinine clearance in urine improved with the Acacia senegal treatment [22].
The current investigation found that proximol had comparable effects in regulating serum biochemical parameters in guinea pigs with PCM-induced toxicity. Also, administering V. origena at 100 mg/kg and 200 mg/kg concurrently with PCM significantly inhibited the rise in kidney injury markers, i.e., urea and creatinine, compared to the PCM-treated group, revealing V. origena ability to eliminate creatinine from blood into urine, eventually normalizing creatinine content in the blood, which may be attributed to its cell-protective effect by preventing free radical production. Lipid peroxidation is defined as the oxidative debasement of polyunsaturated fatty acids, which leads to impaired membrane function and structural integrity. V. origena may reduce the number of free radicals responsible for lipid peroxidation, lowering the quantity of malondialdehyde. This demonstrates that V. origena has the ability to scavenge free radicals and reduce PCM-induced free-radical damage, which is supported by histological data.
Administration of V. origena extract reduces oxidative stress through various mechanisms, including a reduction in free radicals such as superoxide and the preservation of total antioxidant capacity by maintaining near-normal activity levels of endogenous enzymatic/non-enzymatic antioxidants. The latter effects may be attributed to a higher amount of total phenolic compounds, flavonoids, tannins, saponins, alkaloids, steroids, and carbonyl in the V. origena extract, as demonstrated by phytochemical investigation [12].
The biochemical results were also confirmed by histological findings, which showed that oral administration of PCM caused severe damage to the kidney, with tubular degeneration, wide lumen, damaged glomeruli, interstitial vascular congestion, and epithelial degeneration, whereas V. origena pretreatment resulted in significant dose-dependent nephroprotection against PCM-induced nephrotoxicity. Most medicines cause kidney damage to the proximal tubules, glomerulus, or more distal regions of the nephron [23]. The results of this study were in agreement with the results of another study carried out on Acacia sp. by Osman et al., (2022) [24]. They showed that there was a significant (p < 0.05) increase in serum creatinine and blood urea levels in the PCM-treated group compared to the control groups due to nephrotoxicity. Treatment with Acacia senegal resulted in a substantial reduction in serum urea levels as compared to the PCM group. Histopathological analysis of the rat kidneys indicated severe degeneration in the PCM-treated group, but the plant extract-treated groups showed considerable protection from PCM-induced alterations. The biochemical results of blood and urine, along with histopathological investigation of the kidney, demonstrated the nephroprotective efficacy of Acacia senegal extract against PCM-induced nephrotoxicity. Furthermore, the current study's findings were consistent with Ahmed et al., (2022) [25], who discovered that administering A. senegal aqueous extract followed by gentamicin improved protein and albumin activity to normal levels in plant extract- treated groups, whereas it was significantly reduced in the group treated only with gentamicin [25]. The gentamicin-treated group had significantly higher urea and creatinine levels, but the Acacia senegal-treated groups had significantly lower values at 250 and 500 mg/kg, respectively.
The current study provided the first experimental evidence that the ethanol extract of V. origena prevents kidney parameters such as urea, creatinine, total proteins, and albumin concentrations from gradually increasing after PCM induction, and they were kept at mean normal values when compared to the positive control, which was induced by PCM and caused kidney cell damage.
Conclusion
To date, this is the first scientific proof that V. origena protects against PCM nephrotoxicity in guinea pigs. The findings suggest that V. origena may protect against PCM-induced kidney damage by inhibiting PCM-mediated degradation in guinea pig renal tubular cells. V. origena had a stronger protective effect against PCM-induced nephrotoxicity. V. origena nephroprotective mechanism might be attributed to its antioxidant and free radical scavenging properties. The current findings showed that V. origena might be a promising option for reducing PCM nephrotoxic effects. However, no appropriate herbal medicine capable of preventing PCM nephrotoxicity has been clinically endorsed.
Acknowledgement
The authors would like to thank the expert doctors and colleagues from Al Jazeera University's Pharmacy Department in Ibb, Yemen, for their cooperation. The authors would also like to thank Dr. Esam Aqlan, Department of Biology, Faculty of Sciences, Ibb University, Yemen, for assisting with plant identification
Author Contributions
Mohammed S. M. Saif and Warda M. A. Kaidama contributed to writing the manuscript and interpreting the data. Abdulrahman A. Alahdal, Ahmed A.A. Al-Nawah, Ali M.A. Alshaweri, Ameen N.A. Al-Hasani, Ayman A.M. Al-Muntasir, Basem A.A. Al- Badwi, Hesham A.S. Shams Aldeen, Mohammed A.M. Mahdi, Osama E.A. Al-Duais, Taher E.S. Al faqeeh and Yasser A.A. Al-Hajj conceived the study. Mohammed S. M. Saif and Warda M. A. Kaidama supervised the study. All authors have read and approved the final version of the manuscript.
Funding
There is no participating funding.
Declarations
Competing interests
The authors declare no competing interests.
Consent for Publication
All authors have reviewed and approved the final manuscript and consented to its publication.
Additional Information
Correspondence and requests for materials should be addressed to Warda Mohamed Abdu Kaidama,
References
- Ibrahim, T., Agnihotri, S., & Agnihotri, A. K. (2013). Paracetamol toxicity-an overview. Emergency Medicine, 3(158), 1-3.
- Hegazy, A., Abd Al Hameed, E. A., El-Wafaey, D., & Khorshed, O. (2021). Effect of Paracetamol administration on the Rat kidney structure: A Morphological Study. Zagazig University Medical Journal, 27(4), 567-576.
- Canayakin, D., Bayir, Y., Kilic Baygutalp, N., Sezen Karaoglan, E., Atmaca, H. T., Kocak Ozgeris, F. B., ... & Halici, Z. (2016). Paracetamol-induced nephrotoxicity and oxidative stress in rats: the protective role of Nigella sativa. Pharmaceutical biology, 54(10), 2082-2091.
- Waring, W. S. (2012). Novel acetylcysteine regimens for treatment of paracetamol overdose. Therapeutic advances in drug safety, 3(6), 305-315.
- Bessems, J. G., & Vermeulen, N. P. (2001). Paracetamol(acetaminophen)-induced toxicity: molecular and biochemical mechanisms, analogues and protective approaches. Critical reviews in toxicology, 31(1), 55-138.
- Das, J., Ghosh, J., Manna, P., & Sil, P. C. (2010). Taurine protects acetaminophen-induced oxidative damage in mice kidney through APAP urinary excretion and CYP2E1 inactivation. Toxicology, 269(1), 24-34.
- Negash, L. (2010). Ethiopias indigenous trees. Addis Ababa University, Press, Addis Ababa.
- Farzana, M. U. Z. N., & Tharique, I. A. L. (2014). A review of ethnomedicine, phytochemical and pharmacological activities of Acacia nilotica (Linn) willd. Journal of Pharmacognosy and Phytochemistry, 3(1).
- Al-Khulaidi, A. W., Rabei, S. H., & Al-Gifri, A. N. (2024). The Genus Acacia sl.(Fabaceae) in Yemen. Sci. J. Damietta Fac. Sci, 14(2), 90-97.
- Al-Khulaidi, A. W. (2013). Flora of Yemen. The Sustainable Natural Resource Management Project (SNRMP II) EPA and UNDP. Republic of Yemen; 2013.
- Nasser, R. A., & Aref, I. M. (2014). Fuelwood characteristics of six acacia species growing wild in the southwest of Saudi Arabia as affected by geographical location. BioResources, 9(1), 1212-1224.
- Shaikhe, I. A., Muddapur, U. M., Bagewadi, Z. K., Chiniwal, S., Ghoneim, M. M., Mahnashi, M. H., Alsaikhan, F., Yaraguppi, D., Niyonzima, F. N. More, S. S., Mannasaheb,B. A., Al Ali, A., Asiri, A.; Khan, A. A. and Iqubal, S. M.S. Characterization of bioactive compounds from Acacia Concinna and Citrus Limon, silver nanoparticles production by A. concinna extract, and their biological properties. Molecules, 27(9) (2022).
- Ibrahim, H., Humaid, A. A., Thabit, A. A. M., Rizq, E. A., & Al-awadhi, B. H. (2023). Phytochemical screening, antioxidant and antimicrobial activities of acacia origena hunde. J. Chem. Biol. Phys. Sci, 13(3), 308-321.
- Mahmoud, M. F., & Alrumman, S. A. (2016). Biological activities of some Acacia spp.(Fabaceae) against new clinical isolates identified by ribosomal RNA gene-based phylogenetic analysis. Pakistan journal of pharmaceutical sciences, 29(1), 221-230.
- Apoorva, M., Pooja, S., & GM, V. (2021). Phytochemical screening for secondar y metabolites and nutraceutical value of Sesbania grandiflora (L) pers leaf extract. Indo Glob. J. Pharm. Sci, 11(01), 28-32.
- Parameshappa, B., Ali Basha, M. S., Sen, S., Chakraborty, R., Kumar, G. V., Sagar, G. V., ... & Lakshmi, A. V. S.M. (2012). Acetaminophen-induced nephrotoxicity in rats: protective role of Cardiospermum halicacabum. Pharmaceutical Biology, 50(2), 247-253.
- Abdul Hamid, Z., Budin, S. B., Wen Jie, N., Hamid, A., Husain, K., & Mohamed, J. (2012). Nephroprotective effects of Zingiber zerumbet Smith ethyl acetate extract against paracetamol-induced nephrotoxicity and oxidative stress in rats. Journal of Zhejiang University Science B, 13(3), 176-185.
- Adeneye, A. A., Olagunju, J. A., Benebo, A. S., Elias, S. O., Adisa, A. O., Idowu, B. O., ... & Alana, E. O. (2008).Nephroprotective effects of the aqueous root extract of Harungana madagascariensis (L.) in acute and repeated dose acetaminophen renal injured rats. Int J Appl Res NatProd, 1(1), 6-14.
- Parmar, S. R., Vashrambhai, P. H., & Kalia, K. (2010). Hepatoprotective activity of some plants extract against paracetamol induced hepatotoxicity in rats. J Herbal Med Toxicol, 4(2), 101-106.
- Miettinen, T. P., & BjoÌ?rklund, M. (2014). NQO2 is a reactive oxygen species generating off-target for acetaminophen. Molecular pharmaceutics, 11(12), 4395-4404.
- Lahon, K., & Das, S. (2011). Hepatoprotective activity of Ocimum sanctum alcoholic leaf extract against paracetamol- induced liver damage in Albino rats. Pharmacognosy research, 3(1), 13.
- Za’abi, A., Al Salam, S., Al Suleimani, Y., Manoj, P., Nemmar, A., & Ali, B. H. (2018). Gum acacia improves renal function and ameliorates systemic inflammation, oxidative and nitrosative stress in streptozotocin-induced diabetes inrats with adenine-induced chronic kidney disease. CellularPhysiology and Biochemistry, 45(6), 2293-2304.
- Nashar, K., & Egan, B. M. (2014). Relationship between chronic kidney disease and metabolic syndrome: current perspectives. Diabetes, metabolic syndrome and obesity: targets and therapy, 421-435.
- Walla’a, A. O., Mostafa-Hedeab, G., Osman, A. H., Rashed,L. A., & Labib, D. A. A. (2022). Effect of arabic gum (Acacia senegal) on paracetamol-induced chronic nephrotoxicity in albino rats. International Journal of Pharmacology, 18(8), 1593-1604.
- Ahmed, H. E., Abdlrahman, S. H., Ahmed Mohamed, S., Bilal, T. A. H., Elghazali, F. E., & Elbadwi, S. M. (2022). Nephroprotective effect of acacia senegal (Gum Arabic) against gentamicin induced nephrotoxicity in rats. Int. J. Res.-Granthaalayah, 10, 120-128.




