Research Article - (2025) Volume 4, Issue 4
Multicomponent Mixtures - Stability Study to Support Carbon Metrology
Received Date: Jul 02, 2025 / Accepted Date: Aug 04, 2025 / Published Date: Aug 11, 2025
Copyright: ©2025 Florbela A. Dias, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Dias, F. A., Palma, C., Costa, C. J. (2025). Multicomponent Mixtures - Stability Study to Support Carbon Metrology. J Electrical Electron Eng, 4(4), 01-09.
Abstract
The European project EPM SRT-v01 – MetCCUS (Metrology for Carbon Capture, Utilization and Storage) represents the first metrology-focused initiative dedicated specifically to CCUS (Carbon Capture, Utilization, and Storage). Developed within the scope of the European Green Deal, this project addresses the urgent need for accurate and traceable measurements to support the deployment of CCUS technologies. CCUS is identified as a strategic area for achieving the EU’s climate objectives, namely the reduction of greenhouse gas emissions by 55 % by 2030 and the attainment of carbon neutrality by 2050. By strengthening the metrological infrastructure related to CO2 measurement and analysis, MetCCUS plays a key role in enabling reliable monitoring, verification, and regulation of carbon capture processes. Funded by EURAMET, the MetCCUS project addresses the metrological challenges identified by industry. It aims to provide primary standards, measurement methods, and good practice guides to support the essential measurements required in this field. Additionally, it seeks to develop a robust metrological infrastructure for monitoring and detecting carbon dioxide leaks in energy and industrial processes, as well as in transport networks. This infrastructure will also contribute to a better understanding of the carbon dioxide life cycle. As part of the project, four binary gas mixtures were successfully characterized: two of SO2 in a CO2 matrix and two of H2S in a CO2 matrix. Moreover, four multicomponent mixtures were also characterized: two containing SO2 + CO + O2in a CO2 matrix, and two with H2S + CO + CH4 + O2 in a CO2 matrix. All mixtures were prepared in cylinders at approximately 40 bar and subjected to a one-year stability study. This stability assessment was conducted using statistical analysis based on the Normalized Error (En) function, in accordance with ISO 13528:2022, which specifies statistical methods for interlaboratory comparisons and performance evaluation.
Keywords
Carbon Metrology, Multicomponent Mixtures, Gravimetric Method, Certification, Stability Study
Introduction
Climate change has driven a significant expansion of environmental monitoring efforts worldwide. To ensure the reliability of collected data and to minimize measurement uncertainties, the demand for traceable measurements has increased considerably. In this context, reliable gas mixtures play a crucial role, as they are essential for the calibration of analytical instruments used to measure atmospheric pollutants and greenhouse gases (GHGs). These calibrated instruments enable accurate determination of the amount fraction of GHGs in the atmosphere, which is fundamental for climate research and policy development. The excessive accumulation of GHGs, particularly carbon dioxide (CO2), contributes to global warming, ocean acidification, extreme weather events, and loss of biodiversity. In response to these environmental challenges, decarbonization has emerged as a key strategy. It involves the progressive reduction of CO2 emissions resulting from human activities, with the objective of achieving a low-carbon or carbonneutral economy. An important technological pathway to support decarbonization is Carbon Capture, Utilization, and Storage (CCUS). This set of technologies is designed to capture CO2 emissions from industrial and energy-related sources, enabling either its utilization in industrial processes or its permanent storage in suitable geological formations, thereby preventing its release into the atmosphere. Decarbonization and CCUS represent fundamental components of global efforts to mitigate climate change. Despite existing technological, economic, and regulatory challenges, continuous innovation, supportive policy frameworks, and international collaboration provide a viable path forward. Together, these initiatives aim to significantly reduce carbon emissions and promote a sustainable, low-carbon future [1].
The European initiative EPM (European Partnership on Metrology) SRT-v01 - MetCCUS (Metrology for Carbon Capture Utilization and Storage) [1] represents the first metrology project dedicated to CCUS (Carbon Capture Utilization and Storage) in Europe. This initiative is a key element of the European Commission’s Green Deal, an ambitious plan outlined in the European ecological pact. CCUS is identified as one of the priority areas for achieving the overarching goals of a 55 % reduction in greenhouse gas emissions by 2030 and achieving carbon neutrality by 2050. The project, funded by EURAMET, addresses significant metrological challenges faced by industry stakeholders. It aims to develop primary standards, methodologies and best practice guidelines to ensure precise and reliable measurements essential to support CCUS projects. A critical aspect of this initiative lies in the preparation and use of primary gas mixtures, which plays a key role in environmental monitoring and decarbonization research. Reliable gas mixtures are essential for calibrating instruments that measure atmospheric pollutants and greenhouse gases, ensuring accuracy and consistency in data collection. Decarbonization itself refers to the process of significantly reducing carbon dioxide (CO2 ) emissions resulting from human activities, with the goal of transitioning to a low-carbon or carbon-neutral economy. This transformation is a key strategy for mitigating climate change by directly reducing greenhouse gas concentrations in the atmosphere.
The MetCCUS project is a collaborative European initiative aimed at developing advanced metrological techniques to support the accurate measurement and monitoring of carbon dioxide (CO2) emissions, as well as the processes of its capture, utilization, and storage. Launched on October 1, 2022, the project involves 21 partners and will run for a duration of 36 months. It responds to the growing need for reliable and precise data to ensure the effectiveness and safety of carbon management technologies, which are crucial in addressing climate change. The main objectives of the project include the development of accurate CO2 measurement techniques, the improvement of calibration procedures and metrological standards, the support of carbon capture and storage (CCS) technologies, and the promotion of CO2 utilization, particularly in chemical manufacturing and enhanced oil recovery. The project encompasses several key activities, such as research and development, field testing and validation, standardization and harmonization of methodologies, as well as stakeholder engagement and training initiatives. MetCCUS represents a significant advancement in the metrology of CO2 measurement, with important implications for the performance and credibility of carbon capture, utilization, and storage technologies. By enhancing the accuracy and reliability of CO2 measurements, the project contributes to the broader goal of reducing greenhouse gas emissions and combating climate change [1].
As part of the MetCCUS project, the Reference Gas Laboratory (LGR) at the Portuguese Institute for Quality (IPQ) is collaborating in the development of gas metrological capabilities. Specifically, the laboratory is preparing Certified Reference Materials (CRMs) to enable precise measurement of impurities in CO2 with full metrological traceability [1-5]. These materials are vital for validating analytical methods and calibrating the instruments used in carbon capture and storage processes.
The production of the mixtures is done according to a primary method that is a highly accurate technique for preparing standard gas mixtures. It involves accurately measuring the mass of each gas component and combining them with well-defined ratios to achieve the target amount fraction. Ensuring accuracy and precision throughout the preparation process is essential, which requires meticulous calibration of the equipment and careful handling of the gases to minimize errors and ensure the reliability of the resulting standards. This method enables metrological traceability, which guarantees that measurements are consistent and comparable over time and across different locations. Such traceability is vital for maintaining accuracy in a wide range of scientific and industrial applications [3-5].
To date, the LGR has prepared eight gas mixtures. These include four bicomponent mixtures (two consisting of SO2 in a CO2 matrix and two of H2 S in a CO2 matrix) and four multicomponent mixtures (two containing SO2 , CO, and O2 in a CO2 matrix, and two with H2 S, CO, CH4 , and O2 in a CO2 matrix). These mixtures were successfully characterized to measure four pollutant gases and oxygen in a CO2 matrix under high pressure, approximately 40 bar. The analysis of these gas mixtures employs analytical techniques such as gas chromatography (GC), paramagnetic sensor, non-dispersive infrared (NDIR), and non-dispersive ultraviolet (NDUV) spectroscopy.
The certification of these mixtures required an extensive stability study. The stability of these mixtures was evaluated over one year using statistical methods. The Normalized Error (En) function, as specified by the ISO 13528 standard for statistical analysis, was employed to ensure accuracy and reliability in the results [6]. This rigorous approach underscores the project's commitment to providing industry and research sectors with robust and reliable metrological solutions.
Production of Reference Gas Mixtures
The standard ISO 17034 ensures that the gas mixtures produced are of high quality, reliable, and suitable for use in calibration and analytical applications [7]. The production of reference gas mixtures follows the requirements set out in this standard, which defines the criteria for the development of Certified Reference Materials (CRMs). The process includes comprehensive documentation, strict quality control procedures, and adherence to traceability principles aligned with the International System of Units (SI) [7-9].
Producers are required to validate their preparation methods, evaluate measurement uncertainties, and perform stability studies to confirm the long-term reliability of the mixtures. By complying with ISO 17034, the CRMs produced are recognized as meeting internationally accepted standards, supporting consistency and comparability in calibration, testing, and measurement processes across different sectors and laboratories.
Preparation of Gas Mixtures
The process of preparation of gas mixtures involves the use of gravimetric methods, which rely on the accurate weighing of each component in accordance with ISO 6142-1 [10]. The procedure begins with the careful selection of an appropriate gas cylinder, which must be meticulously cleaned and evacuated to eliminate any potential contaminants. This step is critical to ensure the purity and integrity of the final mixture. The gravimetric approach allows for high accuracy in determining the composition of the mixture, contributing to its traceability and reliability as a reference material. The cylinder is then filled with the desired gas components, introduced sequentially, starting with the minor components (trace gases) and finishing with the balance gas, often nitrogen (N2) or carbon dioxide (CO2).
The mixture is prepared by gravimetric addition of each component. The mole fractions of the components in the final mixtures are calculated using the following equation [10]:

Each gas is added using a high-precision mass comparator balance capable of measuring mass with a very low uncertainty. The mass of each component is calculated, considering the buoyancy effect of the surrounding air on the balance. Once all components are added, the total composition of the mixture is determined from the masses of the individual components and their respective molar masses.
The method described in the relevant section of ISO 6142 standard applies specifically to mixtures composed of gaseous or fully vaporized components, which can be introduced into the cylinder in either gaseous or liquid form. These mixtures may consist of either two components (binary mixtures) or multiple components (multicomponent mixtures).
The calculation of the associated uncertainty with the amount fraction of each component involves evaluating contributions from several key factors, namely the weighing of the source gases, the purity of the source gases, and the molar masses. Once the uncertainties related to these three categories have been assessed, they are combined to determine the overall uncertainty associated with the mole fraction, using the following expression:
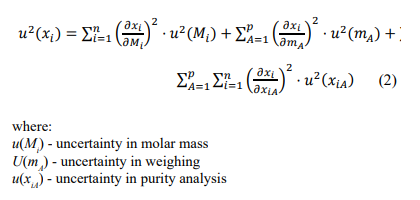
To obtain the expanded uncertainty, the combined standard uncertainty is multiplied by a coverage factor k, which, for a t-distribution with νef (effective degrees of freedom), corresponds to a coverage probability of approximately 95 %. During the preparation of gas mixtures, aluminium cylinders with a special internal coating are used to minimize the adsorption of mixture components onto the inner walls, thereby preserving the integrity and stability of the gas composition. Following the selection of the cylinder, a rigorous cleaning procedure is carried out to eliminate any potential residues that could affect the composition and, consequently, the uncertainty of the final gas mixture. This step is especially critical when preparing mixtures with very low concentration levels. Another essential stage in the filling process involves the transfer of each component gas from its respective parent cylinder to the target cylinder where the mixture is being prepared. The addition of each gas is performed at a dedicated filling station, which is equipped with electropolished tubing, high-purity valves, vacuum and pressure gauges, and oil-free turbomolecular vacuum pumps (Figure 1).

Figure 1: Filling Station
The precise mass of each gas component introduced into the cylinder is determined using a mass comparator, with reference to calibrated weights that are traceable to the national standard. Traceability of gas measurements to the International System of Units (SI) is ensured through the use of calibrated and verified instrumentation. By combining the results from the purity certificates of the source gases with the data obtained from the weighing process, the exact composition of the gas mixture can be calculated, along with the associated uncertainties related to the amount fractions of each component (Figure 2).

Figure 2: Mass Comparator Balance
After preparation, the mixture undergoes a homogenization process (Figure 3), which typically involves rotating the cylinder to ensure uniform distribution of gases within the cylinder

Figure 3: Rolling Cylinder System
Certification of Gas Mixtures
The prepared gas mixture is calibrated with others gas standards, by a suitable analytical method, to confirm its composition and validate the gravimetric calculations. The multi-point calibration method [11, 12] involves measuring the response of an analytical instrument against a series of calibration gas mixtures with precisely known compositions, known as reference standards, according to the international standard ISO 6143 [11]. The purpose is to create a dependable correlation between the instrument’s signal and the concentrations of the target gases in the sample. This ensures accurate, consistent, and traceable measurements during gas analysis.
The process begins by selecting reference gas mixtures that cover the range of concentrations expected in the sample. These reference standards must be traceable to SI and prepared with high accuracy, often using gravimetric methods according to ISO 6142-1 [10]. The instrument response to each reference mixture is recorded, typically using signals such as peak areas or voltages, depending on the type of analyzer is used. The specific analytical methods used were GC, paramagnetic sensor, NDIR, and NDUV.
A mathematical model, such as a linear or polynomial regression is then applied to describe the relationship between the instrument response and the concentration of the analyte. ISO 6143 [11, 12] emphasizes the evaluation of uncertainties for both the reference mixtures and the instrument response, ensuring the calibration curve is accurate and reliable.
Finally, the established calibration curve is used to determine the composition of unknown gas mixtures by measuring their instrument response and interpolating within the range of the calibration standards.
The certification process is carried out using an automated multichannel sampling system, to which all the cylinders scheduled for analysis are connected (Figure 4).

Figure 4: Certification of Gas Mixtures Facility
During the analysis, the system automatically selects each cylinder, allowing the individual flow of gas through the analyzer. The entire sampling and measurement process is managed by the custom-developed IPQAnaliseQui software, which controls the system operation and records all measurement data obtained by the analyzer. All collected data, including standard and sample analysis records, are subsequently compiled into a spreadsheet. The final results are corrected using zero readings and pressure data.
The determination of the calibration function is performed using XGENLINE, a software tool developed by the National Physical Laboratory (NPL). This software identifies the most appropriate low-degree polynomial calibration function (first-, second-, third-, or fourth-order) for a given set of measurement data (X, Y), taking into account the associated uncertainties. In this case, XGENLINE identified a first-order polynomial as the most suitable calibration function for each calibration performed.
The resulting calibration function is then used to calculate the estimated amount fraction values of the analyzed samples, along with their associated uncertainties. XGENLINE, which complies with the ISO 6143 standard, handles the propagation of uncertainty and provides a standard uncertainty value for each analytical result.
The final results are presented together with their respective uncertainties in accordance with the Guide to the Expression of During the analysis, the system automatically selects each cylinder, allowing the individual flow of gas through the analyzer. The entire sampling and measurement process is managed by the custom-developed IPQAnaliseQui software, which controls the system operation and records all measurement data obtained by the analyzer. All collected data, including standard and sample analysis records, are subsequently compiled into a spreadsheet.
The final results are corrected using zero readings and pressure data. The determination of the calibration function is performed using XGENLINE, a software tool developed by the National Physical Laboratory (NPL). This software identifies the most appropriate low-degree polynomial calibration function (first-, second-, third-, or fourth-order) for a given set of measurement data (X, Y), taking into account the associated uncertainties. In this case, XGENLINE identified a first-order polynomial as the most suitable calibration function for each calibration performed.
The resulting calibration function is then used to calculate the estimated amount fraction values of the analyzed samples, along with their associated uncertainties. XGENLINE, which complies with the ISO 6143 standard, handles the propagation of uncertainty and provides a standard uncertainty value for each analytical result. The final results are presented together with their respective uncertainties in accordance with the Guide to the Expression of Uncertainty in Measurement [13].
Traceable gas mixtures are widely used for applications requiring high precision such as equipment calibration, environmental monitoring, and industrial quality control.
The quality control of these measurements is enhanced through active participation in projects and international comparisons [14-19]. Furthermore, recognition and inclusion in the Bureau International des Poids et Measures (BIPM) database of Calibration and Measurement Capabilities (CMC) strengthens the commitment to quality [9].
Stability Study
Gas mixtures are often used in calibration and analytical processes where accuracy is critical. The stability study of the eight bi- component and multi-component gas mixtures conducted over approximately one year were essential to ensure the accuracy and reliability of these reference materials over time. A one-year study allows for the assessment of potential chemical interactions, adsorption effects, or decomposition within the cylinders. This helps determine shelf-life, proper storage conditions, and suitability for long-term use. Stability data supports the certification of gas standards, ensuring traceability to international references. It also minimizes measurement uncertainty in critical applications such as environmental monitoring, industrial safety, and laboratory analysis. Detecting any instability early avoids risks in operational settings. Overall, the study confirms the integrity and usability of the mixtures throughout their intended lifespan.
The analyzed parameters included H2S, CO, O2, CH4, and SO2 in a CO2 matrix. This research aimed to assess the chemical interactions and long-term stability of these gas combinations. Understanding their behavior is crucial for industrial applications, storage, and transportation safety.
This study was conducted using Primary Reference Material (PRM) of several multicomponent mixtures (Table 1, Table 2, Table 3, Table 4, Table 5). All these reference gas mixtures were prepared in carbon dioxide matrix. The calibration curves were done using primary standards in nitrogen matrix. With these primary standards we can have traceability to the standards of these impurities in CO2. In this case, the matrix will not influence the analysis because the analyzers in question do not detect CO2 just as they do not detect nitrogen. Each of them detects only the respective gas.
The purpose is to determine the shelf life of this type of mixtures, that is, the actual period during which a mixture can be considered stable concerning its original metrological specifications.
The study was conducted according to ISO 13528 standard [6], which provides various statistical evaluation methods that can be used in specific tests or measurements and for monitoring the ongoing performance of laboratories. In this context, it will be used to compare two concentrations: the concentration resulting from the initial certification and the concentration obtained in subsequent certifications according to the international standard ISO 6143 [11], over the lifetime of the standard.
In this study, we used the normalized error (En) statistical tool that is typically applied in comparison of measurement systems, calculated according to the following equation:

Results
The objective of this preparation was to enable a comprehensive characterization of these gas mixtures, supporting their use as accurate reference materials in analytical applications. Four binary gas mixtures, classified as Primary Reference Materials (PRM), were meticulously prepared for analysis. Among them, two were binary combinations of hydrogen sulfide (H2S) in a carbon dioxide (CO2) matrix, designated as PRM108595 and PRM108596. Additionally, two mixtures of sulfur dioxide (SO2) in CO2 were prepared, identified as PRM408326 and PRM108593. Each of these four mixtures was stored in individual cylinders, maintained at an approximate pressure of 40 bar, allowing for proper containment and stability during the study [20].
Furthermore, four multicomponent mixtures were prepared in a CO2 matrix, PRM608395 and PRM308978 with the impurities SO2, CO and O2; and two, PRM202557 and PRM302530, with the impurities H2S, CO, O2, and CH4. These mixtures were contained within four cylinders at an approximate pressure of 40 bar each. No stability study was conducted on PRM308978 because this cylinder was sent for analysis to one of the participants of the MetCCUS project.
Each of these multicomponent mixtures was stored in individual cylinders, also at approximately 40 bar. Nevertheless, it is important to note that no stability study was carried out for PRM308978, as this specific cylinder was sent to a participant laboratory, Research Institutes of Sweden (RISE) from Sweden, involved in the MetCCUS project for immediate analysis in March 2024. The remaining seven gas mixtures were retained for long- term monitoring and characterization. However, two of them, PRM408326 and PRM202557, were requested for analysis by two participants of the project, Technical University of Denmark (DTU) and Danish National Metrology Institute (DFM) from Denmark, in March 2025.
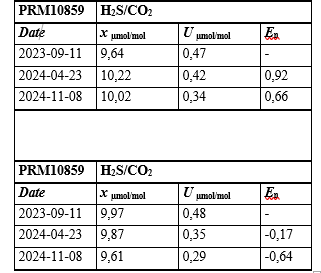
Table 1: Results of the Stability Study for the Prepared Binary Mixtures of H2S/CO2
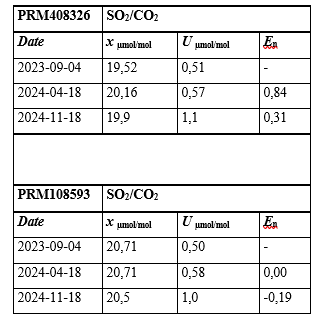
Table 2: Results of the Stability Study for the Prepared Binary Mixtures of SO2/CO2
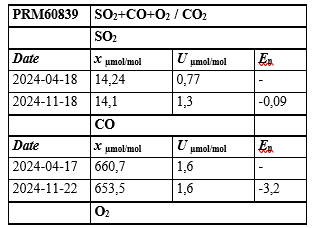
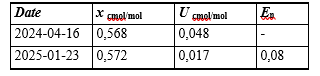
Table 3: Results of the Stability Study for the Prepared Multicomponent Mixture of SO2 + CO + O2 / CO2
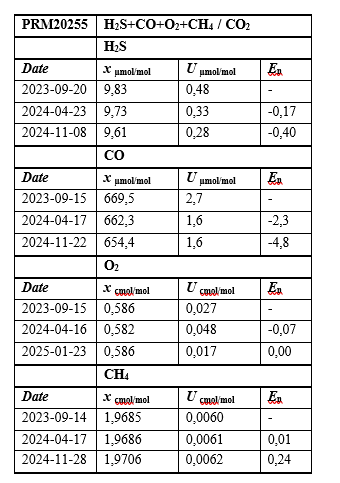
Table 4: Results of the Stability Study for the Prepared Multicomponent Mixture of H2S +CO+O2+CH4 / CO2
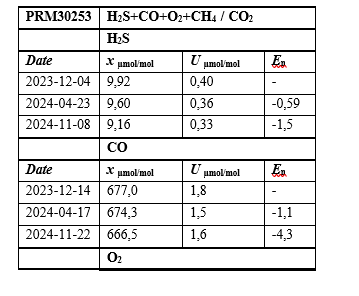
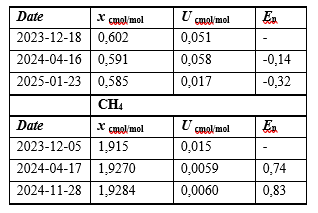
Table 5: Results of the Stability Study for the Prepared Multicomponent Mixture of H2S +CO+O2+CH4 / CO2
Summary
As part of the MetCCUS project, LGR plays a key role in the preparation of Certified Reference Materials (CRM) designed to support stability studies of CO2-based gas mixtures with full metrological traceability. This work supports the calibration and validation of analytical instruments used in carbon capture processes, ensuring accurate and reliable measurements. By developing traceable and stable reference mixtures, LGR helps enhance the quality and comparability of data across laboratories and monitoring systems involved in CO2 capture, transport, and storage.
In conclusion, IPQ has demonstrated its ability to prepare and certify reference materials (CRM) for measuring impurities of H2 S, SO2 , CO, O2 and CH4 in CO2 , within the requested concentration with metrological traceability. The uncertainties obtained were as expected.
The stability study has confirmed that, except for CO, all components remain stable for about one year within the associated uncertainties. To further refine our understanding, a longer study will be conducted to determine the stability period of each type of mixture.
This work is being carried out in collaboration with project partners, involving a joint study in which some of the prepared multicomponent mixtures were sent to participating laboratories (RISE, DTU, DFM) for analysis. The results obtained by the different laboratories will be compared to assess consistency and reliability on measurement. Based on this comparative analysis, conclusions will be drawn regarding the performance and stability of the mixtures, and the findings will be published as part of the project's outcomes.
Looking ahead, this research may be extended to mixtures containing additional components. Additionally, to enhance the scope of the project, these mixtures will be analyzed using alternative analytical methods to gather more data for further characterization of the mixtures.
These standard multicomponent gas mixtures are essential tools for calibrating the analyzers used to measure the purity of carbon dioxide throughout the entire carbon capture and storage (CCS) process. This includes the stages of CO2 capture, compression, transport, and final storage, as well as its application in industrial processes. Accurate monitoring of CO2 purity is critical to ensure the safety, efficiency, and compliance of CCS operations with environmental and technical standards.
The use of these reference mixtures allows for more reliable and comparable measurements across different laboratories and facilities. By providing metrological traceability, they help reduce uncertainties and improve the overall quality of the analytical results. Furthermore, the implementation of such standards supports harmonization at the international level, promoting confidence in data reporting and regulatory verification. Ultimately, these mixtures contribute to the advancement of carbon management technologies and to the effectiveness of climate mitigation strategies.
Acknowledgments
This project (21GRD06 MetCCUS) has received funding from the EPM program co-financed by the Participating States and from the European Union's Horizon 2020 research and innovation program.
This article is a revised and expanded version of a conference oral presentation “Stability Study of Multicomponent Mixtures to Support Carbon Metrology”, which was presented at CIM2025, Lyon, 11-14 March 2025.
Conflicts of Interest
Declare conflicts of interest or state “The authors declare no conflicts of interest.”
References
- MetCCUS Project. (2025).
- Dias, F. A., Palma, C., & Costa, C. J. (2025). Preparation of multicomponent mixtures to support carbon metrology. Measurement, 239, 115453.
- Amico di Meane, E., Plassa, M., Rolle, F., & Sega, M. (2009). Metrological traceability in gas analysis at IN Ri. M: gravimetric primary gas mixtures. Accreditation and quality assurance, 14(11), 607-611.
- Milton, M. J. T., Guenther, F., Miller, W. R., & Brown, A. S. (2006). Validation of the gravimetric values and uncertainties of independently prepared primary standard gas mixtures. Metrologia, 43(3), L7.
- Milton, M. J. T., Vargha, G. M., & Brown, A. S. (2011). Gravimetric methods for the preparation of standard gas mixtures. Metrologia, 48(5), R1.
- ISO, I. (2005). ISO 13528: 2005. Statistical methods for use in proficiency testing by interlaboratory comparisons.
- ISO. (2016). General requirements for the competence of reference material producers.
- ISO. (2017). General requirements for the competence of testing and calibration laboratories.
- KCDB. (2025).
- ISO. (2015). Gas analysis Preparation of calibration gas mixtures Part 1: Gravimetric method for Class I mixtures. 6142-1:
- ISO. (2001). Gas analysis. Comparison methods for determining and checking the composition of calibration gas mixtures. 6143.
- Guenther, F. R., & Possolo, A. (2011). Calibration and uncertainty assessment for certified reference gas mixtures. Analytical and bioanalytical chemistry, 399(1), 489-500.
- Evaluation of measurement data. (2008). Guide to the expression of uncertainty in measurement JCGM 100:(GUM 1995 with minor corrections).
- Kim, D., Kim, Y., Lee, S., Kang, J., Smeulders, D., Wu, H., & Murugan, A. (2021). International comparison CCQM-K41. 2017, hydrogen sulfide in nitrogen. Metrologia, 58(1A), 08010.
- Van der Hout, J. W., van der Veen, A. M., Ziel, P. R., Kipphardt, H., Tuma, D., Maiwald, M., & Engin, E. (2017). International comparison Euramet. QM-K111—propane in nitrogen. Metrologia, 54(1A), 08020.
- Dias, F. A. (2013). Final report on international comparison EURO.QM-S5/1166: Carbon dioxide mixtures in nitrogen, Metrologia. 50 1A 08017.
- Guenther, F. R. (2011). International comparison CCQM-K76: Sulphur dioxide in nitrogen, Metrologia, 48, Tech. Suppl. 08015.
- Botha, A. (2010). International comparison CCQM-K51: Carbon monoxide (CO) in nitrogen (5 µmol mol–1), Metrologia, 47, Tech. Suppl. 08008.
- Nieuwenkamp, G. (2010). Final report on international comparison CCQM-K71: Measurement of stack gas, Metrologia, 47 Tech. Suppl. 08021.
- Dias, F. A., Palma, C., Costa, C. J. (2025). “Stability Study of Multicomponent Mixtures to Support Carbon Metrology”. CIM 2025, Lyon, France, 11-14 March.