Research Article - (2024) Volume 8, Issue 1
Morpho-Anatomical Changes in the Leaves of Alstonia Scholaris (L.) R. Br. In Response to Particulate Matter Pollution
Received Date: Feb 16, 2024 / Accepted Date: Mar 18, 2024 / Published Date: Mar 23, 2024
Copyright: ©©2024 Asad Ullah, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ullah, A., Siddiqi, M. R., Bhatti, A. G. (2024). Morpho-Anatomical Changes in the Leaves of Alstonia Scholaris (L.) R. Br. In Response to Particulate Matter Pollution. Stem Cell Res Int, 8(1), 01-06.
Abstract
Leaves of Alstonias cholaris (L.) R. Br. are sensitive to particulate matter pollution. Thus these leaves show morphological and anatomical changes to counter the effect of particulate matter pollution. To evaluate this, samples of the said leaves were collected from three site of Orange Line Train Route (polluted). Peeling and cross sections were used to study morphological and anatomical features of the leaves of the said tree. The samples were preserved using FAA (Formaline, Acetic acid and Alcohol solution). Sections of the leaves were cut using free hand sectioning techniques and stained using toluidine blue. Then their anatomy was compared to the leaves obtained from Botanical garden of Forman Christian College (unpolluted). Studied carried out on these leaves showed that no significant changes are there in the epidermal cells of both types. But size and number of stomata and size of mesophyll cells showed marked differences. It is necessary to understand the molecular mechanism behind defense mechanism employed by the plant to mitigate the negative effects of particulate matter pollution.
Introduction
Environmental pollution is one of the biggest threats that the industrialized world is facing today. This pollution not only affects human health but also other aspects of human life are negatively affected. There is a vast literature available on the effects of environmental pollution on morphology and anatomy of various plants [1,2]. The most susceptible organ- to environmental pollution- of plants is leaf. It is because of two reasons. Firstly, leaf has direct contact with air as gaseous exchange occurs through stomata of the leaves. Secondly, the internal structure of leaf is more delicate than other plant organs which make it more susceptible to injury. Parameters which are affected by environmental pollution include structure, size, frequency and distribution of stomata [3]. Other scientists have identified some other important parameters which alter with environmental pollution. These include stomatal index, stomatal frequency, number and frequency of trichomes [4].
Distribution and development of stomata in environmentally polluted areas have been investigated by various authors and they have found out that the type of stomata remains the same. But number and size of stomata change with increasing environmental pollution. Stomatal index is somewhat constant for a given species or vary within a given range for a particular species, but in environmentally polluted areas, the stomatal index have been found out to be varying considerably. This variation has been attributed to the clogging of stomata [5,6]. Stomatal Index has been found out to be decreased in wheat, Linseed and Isabgol. Researchers have also found out that size of stomata decrease while number of stomata increases [7]. They have concluded that morphology of leaves of Berberis microphylla change with increase in pollution. Calotropis gigantitia and Ipomoea fistulosa are two important plants that grow in Indian subcontinent. The leaf morphology changes with increasing air pollution. Size of stomata and stomatal index of both plants have been observed to be reduced while number of stomata increases. The author has come across an interesting observation that the stomata on both sides change with pollution though pollutants do not get deposited on the lower side of the leaf [8].
Among the internal structures of the leaves, mesophyll is the most delicate tissue and it gets affected the most. There is a significant decrease in the size of palisade cells in the polluted areas. Width of outer walls of epidermal cells has been found to be changing with environmental pollution [9]. Other scholars have worked on the adaptability of leaves of Acer saccharinum in industrial pollution. The author has come out with the results that there is reduction in the size of palisade cells but the ration of spongy to palisade cells has been constant in control and polluted sites. The anatomical features of leaves of Acer platanoids has been investigated intensively by different researchers (Dineva). There have been some anatomical features which get affected by environmental pollution. There is a significant decrease in size of the cells of mesophyll tissue [10].
have shown that there is a significant reduction in the length of palisade tissue of the leaves of Salix alba in the highly polluted area [11].
The study was conduct on leaves of A. scholaris to see the The study was conduct on leaves of A. scholaris to see the
• To study effects of particulate matter pollution on morphological parameters of A. scholaris leaves.
• To determine the effects of particulate matter pollutants on the anatomy of leaves of A. scholaris.
• To observe the adaptability of the leaves of A. scholaris to high concentration of particulate matter pollution.
Materials and Methods
Site Description
The project was carried in the city of Lahore, Pakistan. The area targeted for the project was the orange line train route which was under construction during the time when opted for the project. The reason for choosing Orange Line Train route was the extraordinary amount of particulate matter pollution. There are three main sites from where the samples were collected i.e Lakshmi Chowk, Haji Camp Chowk and Marri Chowk in the city of Lahore. Control samples were collected from the campus of Forman Christain College, Lahore.
Sample collection
Four plants from each site were chosen on random basis and five leaves from each plant were collected on random. The leaves were the gently shaken to remove dust particles and then put in ethylene bags and transported to the laboratory. In the laboratory, leaves were washed in running water so that all the dust particles are washed away and the fresh leaves were then put into distilled water and used as such for study of morphological traits.
Study of Morphological Traits
The morphological traits were studied using fresh leaves. It is quite easy to have peels of the leaves of Alstonia scholaris. Slides of peels were prepared using toluidine blue and observed under 100X and measurements were taken with the help of stage micrometer. Number of stomata and epidermal cells counted in per unit area
(mm2). Stomatal frequency is the number of stomata per unit area (mm2).
Stomatal index was calculated using the following equation:

Where, S represents the number of stomata per unit area (mm2) and E is the number of epidermal cells per unit area (mm2).
Anatomical Studies
A sizeable portion of the leaf material was cut from the midrib region each leaf and they were preserved in FAA- formaline, glacial acetic acid and ethanol (5%:5%:90%). The preserved samples were then used for free hand sectioning. Sections were cut and put in a watch glass containing toluidine blue. The sections were placed on a clean glass slide. The stained sections were then observed under binocular microscope.
Measuring Technique
Measurements of both morphological and anatomical parameters were recorded using stage micrometry technique. An ocular micrometer was inserted into the eye piece of the microscope. Measurements were recorded under 100X of the microscope. Calibration was made in accordance to the calculations given in appendix 2. The measurements taken were then multiplied with ocular constant with respect to the objective under which the measurements were carried out. 100 readings were taken for each parameter.
Data Analysis
For each parameter, 100 measurements were taken and the data was analyzed using one-way analysis variance (ANOVA) through online software called as Social Science Statistics. P level was selected as p>0.05. It was then followed by post-hoc Tukey HSD Test.
Results
Measurement of pollution
Particulate matter pollution was measured using petri dish method. Details of the formula used is given in appendix 1. Concentration of pollution is given in Fig.1

Figure 1: Concentration of pollution in the investigated areas
The leaves of Alstonia scholaris are dorsiventral with a moderately thick cuticle on the upper as well as lower epidermis. Cells of the upper epidermis are somewhat smaller than those of the lower epidermis. There is clear differentiation between spongy mesophyll and palisade mesophyll. Cells of the spongy mesophyll are round while those of palisade are long and narrow. Leaves are hypostomatic with stomata on the adxial surface of the leaves. Stomata are paracytic in structure. Trichomes are abundantly present on the upper epidermis of the leaves (Fig.2) shows that structure of stomata of Alstonia scholaris and (Fig. 3 and 4) shows the anatomical structures of the leaves of A. Scholaris.

Figure 2: Structure of stomata of leaves of A. scholaris (100X)
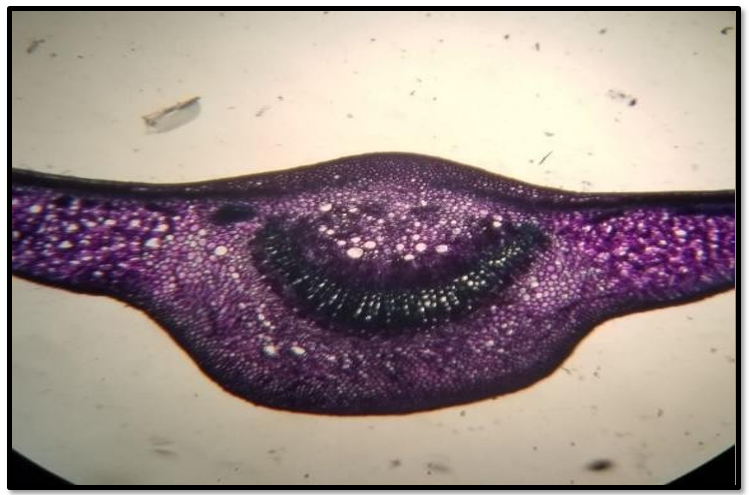
Figure 3: Cross section through the leaf of A. scholaris (10X)

Figure 4: Cross section through the leaf of A. scholaris (40X)
Table 2 shows Morphological parameters from leaves of Alstonia scholaris, size of guard cells, number of stomata, stoamatal frequency and stoamtal index both in control as well as in all the polluted sites. Length and width of the guard cells does not change significantly (at P>0.05). Length to width ratio does not change significantly. Area of the guard cells decrease (at P>0.05) and it is very significant. It changes from 125.656±2.993 (control) to 118.237±5.851 (high pollution site). Stomatal frequency changes significantly (at P.0.05) as shown in the table 1.2. Similarly, stomatal index increases very significantly from 7.845±0.432 (control) to 10.034±0.705 (high pollution site). Other values of stoamtal index are given in the table 2.
Transverse sections of the leaves of A. scholaris showed that there is significant reduction in the size of palisade mesophyll cells which reduces from 526.43±41.32 (control) to 41.826±2.217 (high pollution). Significant reduction in the length of the cells occur while width of palisade mesophyll cells showed some mixed results which meant that this feature is not affected by the environmental pollution. Other parameters like number of spongy and palisade cells does not alter with increasing pollution and so does the ratio of spongy to palisade cells. The results of anatomical features are given in the table 3.
|
Parameters |
S 0 |
S I |
S II |
S III |
|
Length of guard cells (µm) |
22.009±0.788 |
21.105±0.849 |
21.930±0.192 |
21.098±0.341 |
|
Width of guard cells (µm) |
5.665±0.342 |
6.054±0.089 |
5.486±0.054 |
6.097±0.019 |
|
Area of guard cells (µm2) |
125.656±2.993 |
118.035±5.851* |
118.307±1.787* |
126.302±1.878 |
|
Length/Width of guard cells |
3.950±0.342 |
3.520±0.250 |
3.886±0.094 |
3.563±0.059 |
|
No. of stomata /(mm2) |
18.200±0.832 |
23.350±2.265** |
21.000±0.663* |
19.950±0.718* |
|
No of epidermal cells/(mm2) |
207.600±3.871 |
207.950±5.584 |
200.450±4.174 |
201.700±3.189 |
|
Stomatal frequency |
18.200±0.832 |
23.350±2.265** |
21.300±0.663* |
21.150±0.718* |
|
Stomatal Index |
7.695±0.438 |
10.034±0.705** |
9.865±0.290** |
9.692±0.111** |
Table 2: Morphological parameters from leaves of Alstonia scholaris.
Note: Data is given as mean± standard deviation.
*= 0.05
**= 0.001
Stomata frequency is the number of stomata per unit area (mm2). Stomatal Index was calculated using equation (1), given in the meth- odology section.
|
Parameters |
S 0 |
S I |
S II |
S III |
|
Length of palisade cells (µm) |
51.7474±6.420 |
41.826±2.217** |
43.852±0.478* |
45.437±0.588 |
|
Width of palisade cells (µm) |
10.183±0.758 |
10.135±0.517 |
8.550±0.232** |
10.245±0.284 |
|
Area of palisade cells (µm2) |
526.43±41.32 |
427.086±35.018* |
375.015±13.616** |
464.568±12.140* |
|
No. of spongy cells (/mm2) |
17.413±1.596 |
17.950±0.192 |
18.350±0.378 |
18.200±0.326 |
|
No. of palisade cells (/mm2) |
15.297±0.572 |
13.711±0.698* |
14.600±0.673 |
14.800±283 |
|
Spongy/Palisade ratio |
1.276±0.232 |
1.322±0.053 |
1.265±0.049 |
1.235±0.038 |
Table 3: Anatomical parameters for the leaves of Alstonia scholaris.
Note: Data is given as mean± standard deviation
*= 0.05
**= 0.001
All measurements are given in micrometers (µm).
Discussion
The relatively high concentration of particulate matter pollution on the Orange Line Train route is due to the construction which was going on during the course of the study. Heavy traffic also contributes its part. Various authors have shown that both anatomical and morphological parameters change with increasing pollution [12]. This study also suggests that environmental pollution alter anatomical and morphological traits of the leaves of A. scholaris. On one hand, size of the guard cells showed statistical decrease in the polluted sites as compared to control site. On the other hand, number of stomata and stomatal index has shown to increase. Reduced size of guard cells means a reduction in size of the pore of stomata and hence there are less chances of particulate matter pollutants to get inside the leaf via stomata. But the reduction in size will also affect the rate of respiration. To compensate for that, leaves increase the number of stomata per unit area. This is one of the urgent and most successful adaptations to environmental pollution by plants.
However, this is not common to all plants as researchers have found out a decrease in number of stomata in leaves of Ipomea pes-tigridis [13]. Other researchers have reported no change in stomatal index and stomatal frequency and the possible reason for that has been suggested that the concentration of pollution was not that high to induce changes to morphological parameters [14]. Difference in the area of the guard cells is not significant in case of S I and S II as compared to the control group which suggests that there is a minimum threshold of concentration of particulate matter pollutants to induce changes in the size of guard cells.
Other morphological traits like number of epidermal cells, type of stomata, length and width of guard cells and length to width ratio of guard cells showed no significant variation due to particulate matter pollution. This is in consistency with results reported by other researchers. Although some researchers have reported that number of epidermal cells increase with environmental pollution [15]. This shows that micro-morphological changes in response to environmental pollution are species specific. Some plant species are resistant while others are susceptible to environmental pollution have shown that there is significant reduction in the chlorophyll content [16]. Since, chlorophyll is mainly present in the mesophyll cells of the leaves. So, there are two possibilities for this; on the one hand, there is a possibility of reduction in the number of mesophyll cells, while on the other hand, the size of mesophyll cells may reduce. This study found out that there is a significant reduction in the length and size of the palisade mesophyll cells. This is in agreement to the results reported by other researchers who found out that reduction in size of mesophyll cells do happen in response to environmental pollution [17]. The palisade mesophyll are thin walled cells which make these cells more vulnerable to environmental pollution. Some researchers have also noted that environmental pollution does not alter size or number of mesophyll cells which may be due to the reason that those species are much more resistant to environmental pollution or that the threshold concentration of pollutants does not exist at those sites. Leaves growing in shade are usually monomorphic (there is no distinction between palisade and spongy mesophylls) while those in the sunlight are dimorphic (having distinct palisade and spongy mesophylls). With decreasing light intensity, the length of palisade cells decreases to allow sunlight to reach the lower layer of spongy cells [18]. In the current case, the intensity of light was depleted by dust particles both in the air as well as on the leaf surface which caused reduction in the length of the palisade cells to allow sunlight to reach the spongy cells.
Conclusion
Changes in size of various tissues in the leaves of A. scholaris are due to the effect of particulate matter pollution. Number of stomata has increased while size of stomata has decreased due to particulate matter pollution. Size of palisade mesophyll has also shown reduction in size. To conclude with, particulate matter pollution induces morpho-anatomical changes in leaves of A. scholaris. Biochemical and physiological changes due to environmental pollution of the said plant have also been recorded. A study of the molecular mechanism behind adaptation to high level of particulate matter pollution is suggested for A. scholaris.
Acknowledgement
We are thankful for the financial support for the lab work and analysis provided by Department of Biological sciences, Forman Christian College (A Chartered University), Lahore, Pakistan
References
- Raina, A. K., & Bala, C. (2011). Effect of vehicular pollution on Duranta repens L. in Jammu City. Journal of Applied and Natural Science, 3(2), 211-218.
- Tiwari, P., & Pandey, S. (2017). Impact of cement dust pollution on leaf anatomical features of Lantana camara and Calotropis procera. Curr Sci Int, 6, 34-40.
- Dickison, W. C. (2000). Integrative plant anatomy. Academic press.
- Lee, J. K., Kim, D. Y., Park, S. H., Woo, S. Y., Nie, H., &Kim, S. H. (2021). Particulate matter (PM) adsorption and leaf characteristics of ornamental sweet potato (Ipomoea batatas L.) cultivars and two common indoor plants (Hedera helix L. and Epipremnum aureum Lindl. & Andre). Horticulturae, 8(1), 26.
- Jain, P., & Jain, P. Effect Of Slate Pencil Dust Pollution On Leaf Morphology Of Triticum Aestivum (L), Linum Usitatissimum (Forsk.) AND Plantago Ovata (L).
- Saadabi, A. M. A., & El-Amin, A. (2011). Effects of environmental pollution (auto-exhaust) on the micro- morphology of some ornamental plants from Sudan.
- Rahul, J., & Jain, M. K. (2014). An investigation in to the impact of particulate matter on vegetation along the national highway: a review. Res. J. Environ. Sci, 8(7), 356-372.
- Shweta, T. (2012). Air Pollution Induced changes in Foliar Morphology of two shrub species at Indore city, India. Research Journal of Recent Sciences ISSN, 2277, 2502.
- Gostin, I. N. (2009). Air pollution effects on the leaf structure of some Fabaceae species. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 37(2), 57-63.
- Mitrovic, M., Pavlovic, P., Djurdjevic, L., Gajic, G., Kostic, O., & Bojovic, S. (2006). Differences in Norway maple leaf morphology and anatomy among polluted (Belgrade city parks) and unpolluted (Maljen Mt.) landscapes. Ekológia (Bratislava), 25(2), 126-137.
- Gostin, I. (2016). Air pollution stress and plant response. Plant responses to air pollution, 99-117.
- Ekpemerechi, S. E., Ajao, A. A., Jimoh, M. A., & Saheed, S. A. (2017). Variation in leaf anatomical characters in response to air pollution in some Euphorbiaceae species. West African Journal of Applied Ecology, 25(1), 21-31.
- VERMA, R. B., Siddiqi, T. O., & IQBAL, M. (2006). Foliar response of Ipomea pes-tigridis L. to coal-smoke pollution.Turkish Journal of Botany, 30(5), 413-417.
- Ogunkunle, C. O., Abdulrahaman, A. A., & Fatoba, P. O. (2013). Influence of cement dust pollution on leaf epidermal features of Pennisetum purpureum and Sida acuta. Environmental and Experimental Biology, 11(1), 73-79.
- Pawar, A. (2016). Impact of Urban Air Pollution on Epidermal Traits of Amaranthus viridis Growing along the Road Side. Journal of Pure & Applied Science & Technology, 6(1).
- Muhammad, S., Khan, Z., Zaheer, A., Siddiqui, M. F., Masood,M. F., & Sarangzai, A. M. (2014). Alstonia scholaris (L.) R. Br.- planted bioindicator along different road-sides of Lahore city. Pak. J. Bot, 46(3), 869-873.
- Dineva, S. B. Assessing the Adaptability of Acer saccharinum L. to industrially contaminated environment according to its leaf blade structure.
- Dörken, V. M., & Lepetit, B. (2018). Morpho-anatomical and physiological differences between sun and shade leaves in Abies alba Mill.(Pinaceae, Coniferales): a combined approach. Plant, cell & environment, 41(7), 1683-1697.


