Research Article - (2023) Volume 5, Issue 2
MMF as Immunosuppressive Therapy in IgA Nephropathy
2Brighton and Sussex Medical School, United Kingdom
Received Date: Nov 21, 2023 / Accepted Date: Dec 20, 2023 / Published Date: Dec 26, 2023
Copyright: ©Â©2023 Farid Ghalli, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Murphy, C., Barrington-White, H., Pomeranc, A., Ghalli, F. (2023). MMF as Immunosuppressive Therapy in IgA Nephrop-athy. Adv J Uro Nephro, 5(2), 36-43.
Abstract
Introduction: IgA Nephropathy (IgAN) is the most common cause of primary glomerulonephritis in developed countries. Treatment with ACEI/ARB has strong evidence in managing IgAN. If there is evidence of progression, immunosuppression is recommended. KDIGO guidelines do not advise Mycophenolate Mofetil’s (MMF) use in non-Chinese population currently.
Methods: In this study, we reviewed immunosuppression with MMF retrospectively in IgAN patients managed at the Sussex Kidney Unit (SKU) – Brighton – United Kingdom. This was assessed using the primary measures of renal survival without requiring renal replacement therapy (RRT) and proteinuria reduction to >50% of the diagnosis baseline. Twenty-five patients diagnosed with IgAN between 2011 and 2020 and had been treated with MMF were retrospectively reviewed. Data was collected until January 2023, including laboratory results, histopathology, clinic letters, and medication. For those on RRT, data was collected up until the start of RRT.
Results: Twenty- Five patients were reviewed; 24 were white Caucasians, and 1 was ethnically Asian. MMF was used in all 25 patients. Three patients were treated with MMF alone and 17 in combination with steroids. Five patients had prednisolone and cyclophosphamide for three months, followed by MMF maintenance. The average treatment duration was 2 years, and the average dose was 1g BD. Five patients progressed to end-stage renal disease (ESRD), and 3 had renal transplants. Twenty patients maintained renal survival; the mean eGFR at diagnosis was 45.6+/-34.1 and, at the time of review, was 56.1+/-26.6. Of the 17 patients who presented with AKI, 5 recovered to normal renal function, 5 had end-stage renal disease (ESRD), 4 had improvements, and 3 showed a decrease in eGFR. Overall, 70.6% of AKI patients recovered to normal or CKD levels. Eighty % of the patients had renal survival without RRT during the review. Twenty patients achieved more than 50% reduction of proteinuria, with five patients having proteinuria less than 0.3 g/24 hours and nine patients less than 0.5 g/24 hours. Comparison between the proteinuria at diagnosis and at the assessment time was significant (P<0.001).
Conclusions: Mycophenolate mofetil effectively maintained renal survival and improved proteinuria in IgA nephropathy patients indicated for immunosuppression. The treatment was well tolerated by all patients.
Introduction
IgA nephropathy (IgAN) is the most common glomerulonephritis globally, with an incidence of ≥2.5 cases per 100,000 individuals each year [1]. Its presenting features are highly variable, ranging from asymptomatic microscopic haematuria to significant protein¬uria, acute kidney injury and even rapidly progressive kidney fail¬ure. The condition is generally characterised by the deposition of IgA in the mesangium with mesangial proliferation [2,3].
Many patients have a benign disease course. However, IgAN re¬mains a common cause of chronic kidney disease (CKD), and up to 40% of patients progress to end-stage renal failure within 20 years [4]. The UK National Registry of Rare Kidney Diseases (RaDar) study recently investigated 2299 adults and 140 children with IgA nephropathy. It concluded that ‘outcomes are generally poor with few patients expected to avoid kidney failure in their lifetime' [5]. The international IgAN Prediction Tool uses histo¬logical and clinical data at the time of biopsy to determine the risk of disease progression. Prognostic variables include proteinuria, blood pressure, eGFR, age, sex and ethnicity. Also, the use of ACE inhibitor (ACEi) or angiotensin receptor blocker (ARB), immu-nosuppression at or before biopsy, and the Oxford MEST-C score are included [6,7]. While this tool can aid clinical trial design and aid patient discussions, it is not currently used to make treatment decisions [8].
Management of IgAN primarily involves control of blood pres-sure and proteinuria (supportive care) as set out by the KDIGO 2021 guidelines. There is extensive evidence for ACEI and ARBs, which should be given at their maximum tolerated dose to those with proteinuria of >0.5g/day, regardless of whether hypertension is present or not [8]. More recent evidence from the DAPA-CKD trial demonstrates that adding dapagliflozin to ACEI or ARB ther¬apy significantly reduces the risk of CKD progression [9].
Immunosuppression should be considered for those with persistent proteinuria >0.75-1g/day despite 3-6 months of optimised sup-portive care [8]. The best immunosuppressive option is yet to be established, with numerous recent and ongoing trials leading to new perspectives in this rapidly evolving field [10]. Due to the un-certainty of the efficacy and safety profiles of immunosuppressive treatment options, the KDIGO 2021 guidelines suggest offering clinical trial enrolment and for those with eGFR>/=30 ml/min to consider a 6-month course of glucocorticoid therapy depending on careful risk stratification for adverse effects [6-8]. Since these guidelines were written, the TESTING 2.0 trial demonstrated that lower-dose glucocorticoid therapy combined with Pneumocystis jiroveci prophylaxis also has a clinical benefit, with fewer adverse events than the higher-dose group [10,11]. There is also evidence for targeted-release formulation of budesonide as a disease-modi-fying drug, which has a significant prolonged benefit on eGFR and proteinuria and was shown to be well tolerated [4].
Mycophenolate Mofetil (MMF) is a reversible inhibitor of inosine monophosphate dehydrogenase (IMPDH). This prevents T-and-B-cell proliferation and production of cytotoxic T-cells and an-tibodies [12]. Several randomised controlled trials conducted on Chinese patients have demonstrated robust evidence for mycophe¬nolate mofetil (MMF) in managing IgAN [13-15]. This evidence has not been replicated in non-Chinese populations, with clinical trials from North America and Belgium failing to show the ben¬eficial impact of MMF on renal function or proteinuria [16-18]. As such, the KDIGO guidelines only recommend using MMF as a steroid-sparing agent in Chinese patients [8]. However, recent evidence from a retrospective study of 30 patients (29 Caucasian 1 Asian) compared MMF plus glucocorticoids with a conventional glucocorticoid regimen and found equal reductions in proteinuria in both groups, with a cumulative steroid-sparing effect of 6g in the MMF-based regimen [19].
This retrospective single-centre study evaluated the efficacy of MMF as a therapeutic agent in 25 patients (24 were white Cau¬casians) with IgAN, in whom immunosuppression was indicated.
Methods
Twenty-five patients diagnosed with IgAN between 2011 and 2020 who were treated with MMF at Sussex Kidney Unit (SKU) – Uni¬versity Hospitals Sussex in Brighton – United Kingdom, were ret¬rospectively reviewed. Data was collected until January 2023. The review included laboratory results, histopathology reports, clinic letters, and medications. Data collected for those patients eventu¬ally requiring RRT was collected up to the point of commencing RRT. The collected data was analysed statistically using means, percentages, standard deviations and p value calculated using t-test to determine statistical significance. P value was considered signif-icant <0.05.
Inclusion Criteria
1. Age >18 years
2. Patient with primary IgA nephropathy – biopsy-proven
3. Patients managed with MMF for at least six months
4. Patients had a clear indication for immunosuppression and high risk of IgA progression:
i. Worsening renal function
ii. AKI in presentation
iii. Progressive worsening of proteinuria
iv. Significant biopsy findings: immune complex deposits or cres¬cents
Exclusion Criteria
1. Age < 18 years
2. Secondary IgA
3. Patients who had no treatment with MMF
4. Patients treated with MMF less than six months
Results
Twenty- five patients were reviewed, 13 male and 12 females. Twenty-four were white Caucasian, and one was Asian. The mean age was 42.4+/-16 years. Oxford classification showed M1 (21 pa¬tients), E1 (5), S1 (19), T1 (10), T2 (4), C1 (13) and C2 (6) (Table 1). MMF was used in all 25 patients; the average treatment dura¬tion was 2 years and the average dose of MMF was 1g BD.
|
Parameters |
Value |
|
|
Number of patients |
|
25 |
|
Mean Age |
|
42.4+ 16 years |
|
Gender |
Male |
13 |
|
Female |
12 |
|
|
Mean eGFR at the diagnosis |
45.6+34.1 ml/min |
|
|
Mean eGFR at the time of the study |
56.1+26.6 ml/min |
|
|
Mean Proteinuria at the diagnosis |
4.8+5 gm |
|
|
Mean Proteinuria at the time of the study |
1.2+1.4 gm |
|
|
Oxford Classification at the diagnosis |
M0 |
4 |
|
M1 |
21 |
|
|
E0 |
20 |
|
|
E1 |
5 |
|
|
S0 |
16 |
|
|
S1 |
9 |
|
|
T1 |
10 |
|
|
T2 |
4 |
|
|
C0 |
6 |
|
|
C1 |
13 |
|
|
C2 |
6 |
|
|
Patients on ACEI/ARBs |
|
22 (13 on full dose) |
|
Patients managed with cyclophosphamide |
|
5 |
|
Patients managed with Steroids |
|
21 |
Table 1: Baseline Characteristics of Patients Involved in this Study
Five had prednisolone and cyclophosphamide for three months, followed by MMF maintenance. Three patients treated with cyclo-phosphamide had crescentic IgA with crescent percentages (24%, 29% and 73%). The other two patients had crescents (14 % and 19%) with AKI and nephrotic range proteinuria. Three patients were treated with MMF alone and 17 in combination with ste-roids. Twenty-one patients had steroid treatment on weaning dose with titration of MMF dose. The starting dose of steroid treatment ranged between 40 -60 mg. Twenty-two patients were also on con¬tinuous ACEI/ARB with 13 patients on full-dose ACE inhibitors. Three patients were not on ACEI/ARB. Looking into the reason for this, one had facial swelling and dizziness using losartan 50 mg. No clear cause for not being on ACEI/ARB was documented in the other 2 patients.
Twenty patients maintained renal survival; the mean eGFR at diag¬nosis was 45.6+/-34.1 and, at the time of review, was 56.1+/-26.6. Eighty % of the patients had renal survival without RRT during the review (Figure 1). Five patients progressed to ESRD, and 3 of them had renal transplant. Seventeen patients presented with AKI, 5 recovered to normal renal function, 8 recovered to CKD and 5 had ESRD. Four CKD patients had improvements in eGFR, and three showed a decrease in eGFR (Figure 2). Overall, 70.6% of AKI patients recovered to normal or CKD levels.
Twenty patients achieved more than 50% reduction in proteinuria, with five patients having proteinuria less than 0.3 g/24 hours and 9 patients less than 0.5 g/24 hours. Comparison between the pro¬teinuria at diagnosis and at the assessment time was significant (P<0.001) (Figure 3).
Figure 1: Renal Survival Percentage of Patients Managed with MMF
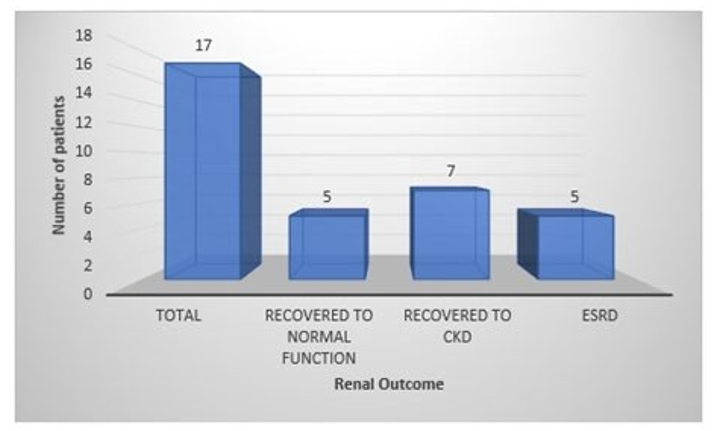
Figure 2: Renal Recovery of Patients Presenting in AKI and Managed with MMF
Figure 3: Improvement in Proteinuria Values in Patients Managed with MMF
Discussion
This single-centre retrospective study involved 25 patients with renal biopsy-proven IgA nephropathy treated with MMF therapy (average dose 1g BD and average duration of two years of treat-ment). It demonstrated positive results concerning renal survival and proteinuria in patients who were indicated for immunosup-pression based on histopathology and risk of progression. Eighty % achieved a 50% reduction in proteinuria, and 80% had renal survival (not requiring transplant or dialysis) at the 6.1-year aver-age assessment point.
KDIGO guidelines 2021 advocate using glucocorticoid therapy in those considered high risk of progression- defined as proteinuria ≥ 0.75g/day despite adequate ACEI or ARB therapy and cardio-vascular risk factor modification for 3-6 months [8]. However, the use of MMF as an alternative agent is currently only supported in Chinese patients.
Two randomised controlled trials in Chinese populations have demonstrated statistically significant reductions in proteinuria and reduction in the progression of CKD using MMF versus best sup-portive care [14,15]. A further multicentre RCT in China demon-strated non-inferiority of MMF plus prednisone versus full-dose prednisone in a population with active proliferative lesions [13]. However, RCTs in Caucasian populations with IgA nephropathy have failed to demonstrate statistically significant improvements with MMF use [16-18]. This study, in an almost exclusive Cau-casian population, adds to the evidence base supporting the use of MMF in IgA nephropathy in non-Chinese populations.
Baseline Characteristics
IgA nephropathy is a highly heterogeneous disease with prediction tools risk stratifying patients based on clinical and histopathologi¬cal features at diagnosis [7]. The KDIGO guidelines advocate us- ing the IgA prediction tool; this uses features such as age, eGFR at the time of biopsy, proteinuria per day and MEST score to quantify the risk of progression [8].
Reviewing the demographics of our study, our population was ma¬jority white Caucasian (96%). This is similar to our population de¬mographics for Sussex, which, according to the 2021 census infor¬mation, is 88.3% white ethnic in East Sussex and 91% white ethnic in West Sussex [20,21]. The mean age for our patients was 42.4± 16; this is very similar to the census data again (East 42.9 years and West Sussex 44.8). Our cohort had a comparable mean age of participants (42.4 +/- 16 years) to previous studies [13-18]. The baseline eGFR of 47.5 was significantly lower than three previous studies that had documented this, but would have fallen within the inclusion criteria of all previous studies on this topic [13,16,17]. Forty-four % of participants in our study had baseline proteinuria in the nephrotic range. The mean proteinuria documented in pre-vious studies ranged between 1.59 and 2.6 g/day, suggesting that our participants had more advanced disease and were at higher risk of progression.
Five out of six previous studies assessing MMF in IgA nephrop¬athy displayed histopathological data, with 3 using the Oxford classification [13,14,16] Eighty-four % of patients in our cohort demonstrated mesangial hypercellularity, 76% segmental sclerosis and 76% had crescents (24% with crescents in more than 25% of glomeruli- a score of C2). The MEST-C scores from our cohort appear to be higher than previous studies, particularly regarding the presence of crescents (76% vs 44% and 58% of participants). Crescents are a poor prognostic feature, suggesting our cohort was at greater risk of disease progression than in previous studies. Crescents less than 25% (C1) carry poor prognosis when untreated with immunosuppression [22].
Intervention
In this study, the average MMF dose was 1g BD for an average duration of 2 years of treatment. Twenty-one patients in our co-hort also received steroid treatment. There is significant diversity in the dose and duration of MMF treatment in previous RCTs. The 2 Chinese studies with results supportive of MMF treatment used between 1-2g per day for 6 months to 3 years; therefore, our study falls within this range [14,15]. Mycophenolate is not currently rec¬ommended as a treatment for IgA nephropathy except as a ste¬roid-sparing agent in Chinese patients [8].
There is increasing evidence in recent studies that it can be used in the treatment for IgA nephropathy in non-Chinese populations. An Italian single-centre study using MMF regimen in IgA nephropa¬thy with active lesions on biopsy demonstrated similar responses compared to conventional steroid treatment whilst reducing cu¬mulative glucocorticoid dose [19]. A multi-centre RCT in South Korea of patients with IgAN at high risk of disease progression concluded that a combination of MMF and corticosteroid thera¬py was superior to supportive therapy in reducing proteinuria and preserving renal function [23]. A meta-analysis conducted in 2018 combining studies in Caucasian and Chinese populations conclud¬ed that 'MMF was superior to placebo for renal remission, but not for ESRD but that further studies were required [24].
Duration of Treatment
In our study, the average MMF treatment was 2 years. This is consistent with the duration of Beckwith et al. study, which demonstrated histopathological improvement in IgAN with MMF treatment alone in a majority Caucasian population. This study demonstrated statistically significant improvement in the average proportion of glomeruli showing hypercellularity, cellular/fibro cellular crescents and mesangial IgA deposition. It also showed that histopathological improvement persisted after cessation of MMF therapy, suggesting that 2-year duration of treatment may be sufficient to achieve beneficial outcomes [25].
Renal Survival and Proteinuria
Eighty % of patients in our study maintained renal survival (not requiring dialysis or renal transplant). Of those achieving renal survival, the mean eGFR was 45.6 +/- 34.1 at baseline and 56.1 +/- 26.6 at assessment time. Five patients progressed to ESRD, all of them presented with AKI at baseline.
All six previous studies that used MMF in IgA nephropathy de-scribed changes in eGFR or renal survival, with only two studies reporting statistically significant results [13,14]. Hou et al. 2023 found only 7.1% of the MMF group had a doubling of creatinine from baseline or onset of ESRD at three years. Hou et al. 2017 compared MMF plus low-dose prednisone versus full-dose predni-sone. In this study, eGFR decline of >30% was seen in 12% of pa¬tients in the MMF group at one year. The rates of significant eGFR decline or progression to ESRD in our cohort are higher than the findings of these two studies (7.1% and 12% versus 20%); how¬ever, less favourable baseline characteristics could explain these differences and the longer follow-up time in our study.
From the statistical analysis of our results, we showed that 80% of patients had more than 50% reduction in proteinuria, five pa¬tients (20%) had proteinuria less than 0.3 g/24 hours and 9 (36%) patients less than 0.5 g/24 hours. Comparison between the pro¬teinuria at diagnosis and at the assessment time was significant (P<0.001). Proteinuria is a well-known risk factor for renal func¬tion worsening. Recent studies, including the RaDaR study, con¬cluded that almost all patients are at risk of kidney failure within their lifetime unless a rate of eGFR loss ≤1 ml/min per 1.73m2 per year was maintained [5]. Therefore, we can conclude the impor¬tance of this result as reducing proteinuria in the long term has a great impact on the achievement of renal survival. Comparison be¬tween proteinuria at the time of diagnosis and time of assessment was statistically significant (p<0.001).
All six previous studies described effects on proteinuria or UPCR. Two studies demonstrated statistically significant reductions in proteinuria in MMF groups compared to control [14,15]. Hou et al. 2023 demonstrated an average 57% reduction in urinary protein excretion rate (UPER) in the MMF group at three years [14]. Sim¬ilarly, Tang et al. 2005 demonstrated 80% remission of protein¬uria in the MMF group defined by proteinuria <0.3g/d at 72 weeks [15]. Our results on reduction in proteinuria appear comparable to Hou et al. 2023 study but not as impressive as Tang et al. findings.
The patient group tolerated MMF well; the average course was 2 years. The most common reason for stopping MMF was the cli¬nician's decision for cessation of the treatment course. Review¬ing the patients' notes showed no major side effects in the patient group, resulting in patients stopping taking the medication
Conclusion
Mycophenolate mofetil effectively maintained renal survival and improved proteinuria in IgA nephropathy patients, with histo¬pathological and clinical features predicting risk of disease pro¬gression. A large randomised controlled trial is needed to compare the MMF effect with currently available therapies.
Limitations and Future Studies
This study provides evidence of possible efficacy of MMF treat¬ment in non-Chinese populations with IgA nephropathy. Howev¬er, there are some limitations to our study and its generalisability which must be considered. It was a retrospective study. Patients in our cohort appeared to have baseline characteristics of more se¬vere disease (lower baseline eGFR, higher level of proteinuria and higher MEST-C scores) than previous studies. There remains in¬sufficient evidence to guide timing and treatment decisions based on the MEST score or the presence of crescent on biopsy. Our study adds to this evidence base and suggests that those with more severe disease may benefit from MMF intervention. Most of our cohort in this retrospective study also had concomitant corticoste¬roid treatment, making isolating the effect of MMF alone more difficult. However, it does suggest after weaning of steroid, MMF maintained and achieved the desired treatment effect. Our data suggest a further prospective RCT study is needed to assess and isolate the effect of MMF in non-Chinese patients with features of more severe IgA nephropathy, as these patients appear to be more likely to benefit from this intervention.
Author Contributions
Conor Murphy: First author. He participated in data collection, data analysis at the start of the project, writing the abstract, the dis-cussion and conclusion, references and paper review and editing.
Henry Barrington White: Second author. He participated in data collection, data analysis, writing the abstract, writing the discus-sion, writing reference, paper reviews and editing.
Abigail Pomeranc: Third author. She participated in data collec-tion, data analysis, writing the abstract, writing the introduction, paper reviews, editing and references.
Farid Ghalli: The senior author. He had the research idea and monitored all the work and paper-writing steps. He participated in data analysis, writing the abstract, writing the patient and methods and results sections with the design of the table and figures. Also, he edited and reviewed the whole paper.
References
- Storrar, J., Chinnadurai, R., Sinha, S., & Kalra, P. A. (2022). The epidemiology and evolution of IgA nephropathy over two decades: A single centre experience. Plos one, 17(9), e0268421.
- Tarafdar, S. (Ed.). (2020). Nephrology: a comprehensive guide to renal medicine. John Wiley & Sons.
- Hou, J. H., Le, W. B., Chen, N., Wang, W. M., Liu, Z. S., Liu,D., ... & Liu, Z. H. (2017). Mycophenolate mofetil combined with prednisone versus full-dose prednisone in IgA nephropathy with active proliferative lesions: a randomized controlled trial. American Journal of Kidney Diseases, 69(6), 788-795.
- Rajasekaran, A., Julian, B. A., & Rizk, D. V. (2021). IgA nephropathy: an interesting autoimmune kidney disease. The American journal of the medical sciences, 361(2), 176-194.
- Pitcher, D., Braddon, F., Hendry, B., Mercer, A., Osmaston, K., Saleem, M. A., ... & Barratt, J. (2023). Long-term outcomes in IgA nephropathy. The Clinical Journal of the American Society of Nephrology, 18(6), 727-738.
- Storrar, J., Chinnadurai, R., Sinha, S., & Kalra, P. A. (2022). The epidemiology and evolution of IgA nephropathy over two decades: A single centre experience. Plos one, 17(9), e0268421.
- Barbour, S. J., Coppo, R., Zhang, H., Liu, Z. H., Suzuki, Y., Matsuzaki, K., ... & Cattran, D. C. (2019). Evaluating a new international risk-prediction tool in IgA nephropathy. JAMA internal medicine, 179(7), 942-952.
- Rossing, P., Caramori, M. L., Chan, J. C., Heerspink, H. J., Hurst, C., Khunti, K., ... & de Boer, I. H. (2022). KDIGO 2022 clinical practice guideline for diabetes management in chronic kidney disease. Kidney International, 102(5), S1-S127.
- Wheeler, D. C., Toto, R. D., Stefansson, B. V., Jongs, N.,Chertow, G. M., Greene, T., ... & Committees, D. C. T. (2021). A pre-specified analysis of the DAPA-CKD trial demonstrates the effects of dapagliflozin on major adverse kidney events in patients with IgA nephropathy. Kidney international, 100(1), 215-224.
- Noor, S. M., Abuazzam, F., Mathew, R., Zhang, Z., Abdipour, A., & Norouzi, S. (2023). IgA nephropathy: a review of existing and emerging therapies. Frontiers in Nephrology, 3, 1175088.
- Lv, J., Wong, M. G., Hladunewich, M. A., Jha, V., Hooi, L. S., Monaghan, H., ... & Zhou, Y. (2022). Effect of oral meth-ylprednisolone on decline in kidney function or kidney failure in patients with IgA nephropathy: the TESTING randomized clinical trial. Jama, 327(19), 1888-1898.
- Allison, A. C. (2005). Mechanisms of action of mycophenolate mofetil. Lupus, 14(3_suppl), 2-8.
- Hou, J. H., Le, W. B., Chen, N., Wang, W. M., Liu, Z. S., Liu,D., ... & Liu, Z. H. (2017). Mycophenolate mofetil combined with prednisone versus full-dose prednisone in IgA nephropathy with active proliferative lesions: a randomized controlled trial. American Journal of Kidney Diseases, 69(6), 788-795.
- Hou, F. F., Xie, D., Wang, J., Xu, X., Yang, X., Ai, J., ... &MAIN Trial Investigators. (2023). Effectiveness of mycophenolate mofetil among patients with progressive IgA nephropathy: a randomized clinical trial. JAMA Network Open, 6(2), e2254054-e2254054.
- Tang, S. C., Tang, A. W., Wong, S. S., Leung, J. C., Ho, Y.W., & Lai, K. N. (2010). Long-term study of mycophenolate mofetil treatment in IgA nephropathy. Kidney international, 77(6), 543-549.
- Hogg, R. J., Bay, R. C., Jennette, J. C., Sibley, R., Kumar,S., Fervenza, F. C., ... & Wyatt, R. J. (2015). Randomized controlled trial of mycophenolate mofetil in children, adolescents, and adults with IgA nephropathy. American Journal of Kidney Diseases, 66(5), 783-791.
- Frisch, G., Lin, J., Rosenstock, J., Markowitz, G., D'Agati, V., Radhakrishnan, J., ... & Appel, G. (2005). Mycophenolate mofetil (MMF) vs placebo in patients with moderately advanced IgA nephropathy: a double-blind randomized controlled trial. Nephrology Dialysis Transplantation, 20(10), 2139-2145.
- Maes, B. D., Oyen, R., Claes, K., Evenepoel, P., Kuypers, D., Vanwalleghem, J., ... & Vanrenterghem, Y. F. C. (2004). Mycophenolate mofetil in IgA nephropathy: results of a 3-year prospective placebo-controlled randomized study. Kidney international, 65(5), 1842-1849.
- Roccatello, D., Careddu, A., Ferro, M., Naretto, C., Quattroc-chio, G., Fenoglio, R., & Sciascia, S. (2023). The steroid-spar-ing effects of a mycophenolate mofetil-based regimen in the management of immunoglobulin A nephropathy in patients with histologically active lesions: A comparison with a control cohort receiving conventional therapy. Journal of Nephrology, 1-9.
- East Sussex Council (2021) Census Briefing, Research andIntelligence Team, Governance Services.
- West Sussex JSNA, Census 2021: Ethnicity, language and religion, Public Health and Social Research Unit; 2023.
- Jia, Q., Ma, F., Yang, X., Li, L., Liu, C., Sun, R., ... & Sun,S. (2022). Long-term outcomes of IgA nephropathy patients with less than 25% crescents and mild proteinuria. Clinical and Experimental Nephrology, 1-9.
- Han, S. Y., Jung, C. Y., Lee, S. H., Lee, D. W., Lee, S., Kim,C. D., ... & Kim, B. S. (2022). A multicenter, randomized, open-label, comparative, phase IV study to evaluate the efficacy and safety of combined treatment with mycophenolate mofetil and corticosteroids in advanced immunoglobulin A nephropathy. Kidney Research and Clinical Practice, 41(4), 452.
- Zheng, J. N., Bi, T. D., Zhu, L. B., & Liu, L. L. (2018). Effica-cy and safety of mycophenolate mofetil for IgA nephropathy: An updated meta-analysis of randomized controlled trials. Experimental and therapeutic medicine, 16(3), 1882-1890.
- Beckwith, H., Medjeral-Thomas, N., Galliford, J., Griffith, M., Levy, J., Lightstone, L., ... & Cairns, T. (2017). Mycophenolate mofetil therapy in immunoglobulin A nephropathy: histological changes after treatment. Nephrology Dialysis Transplantation, 32(suppl_1), i123-i128.



