Case Report - (2025) Volume 4, Issue 4
Mesenchymal Stem Cell Implants for Chondropathies: A Promising Approach in Joint Regeneration? Retrospective Study
2University of Campania "Luigi Vanvitelli”, Italy
3University of Salerno, Italy
4Saint Camillus International University of Health and Medical Sciences, Rome, Italy
Received Date: Aug 22, 2025 / Accepted Date: Oct 20, 2025 / Published Date: Nov 10, 2025
Copyright: ©2025 Francesco Saturno, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Saturno, F., Monaco, G., Salerno, A., Secondulfo, C., Cianciola, A., et al. (2025). Mesenchymal Stem Cell Implants for Chondropathies: A Promising Approach in Joint Regeneration? Retrospective Study. J Surg Care, 4(4), 01-02.
Abstract
Osteoarthritis (OA) is the most prevalent joint disorder and is primarily responsible for a greater number of disability cases among the elderly than any other clinical condition. OA affects approximately 4 million people and represents the leading cause of temporary disability and the third leading cause of hospitalization. The estimated incidence is about 100,000 new cases per year. The probability of developing OA increases proportionally every 10 years starting from the age of 45. It is estimated that over 50% of the population over 60 years of age is affected by OA. Currently, this pathology is no longer considered a purely degenerative process related solely to biomechanical alterations and/or aging, but rather as a pathological expression of wear, inflammation, and immunological imbalance of the joint.
Introduction
Osteoarthritis (OA) is the most prevalent joint disorder and is primarily responsible for a greater number of disability cases among the elderly than any other clinical condition. OA affects approximately 4 million people and represents the leading cause of temporary disability and the third leading cause of hospitalization. The estimated incidence is about 100,000 new cases per year. The probability of developing OA increases proportionally every 10 years starting from the age of 45. It is estimated that over 50% of the population over 60 years of age is affected by OA. Currently, this pathology is no longer considered a purely degenerative process related solely to biomechanical alterations and/or aging, but rather as a pathological expression of wear, inflammation, and immunological imbalance of the joint.
Case Series
We conducted a retrospective analysis of our case series, which included 46 patients who attended our pain therapy clinic from January 10, 2023, to January 3, 2024, with a mean age of 50 years (range 42-75 years), comprising 30 males and 16 females. During the pre-operative assessment, all patients with advanced OA, who were eligible for prosthetic surgery, were excluded, while those with mild to moderate intra-articular lesions were selected.
Harvest and Implantation
Patients were pre-treated with three intra-articular injections (local anesthetic and triamcinolone 40 mg) on a weekly basis to reduce joint inflammation. In summary, mononuclear cells were harvested from the iliac crest in a sterile operative field and under local anesthesia. Using a trocar inserted into the spongy bone, a bone marrow aspiration of up to 60 ml was performed. Utilizing the Sepax Cell Processing System, mononuclear cells were directly selected in the operating room, eliminating red blood cells and plasma, thus reducing the volume of aspirated material (figure 1
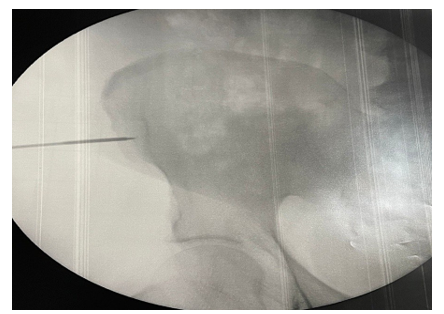
Figure 1: This Image Illustrates the Procedure of Bone Marrow Aspiration from the Iliac Crest. A Trocar is Inserted Into the Spongy Bone to Collect Mononuclear Cells, Which are Essential for the Subsequent Stem Cell Therapy in Joint Regeneration
The collected material was then injected into the collection bag, and the entire kit was mounted on the separator, activating a specific program. At the end of the process, after approximately 15 minutes, the majority of red blood cells and plasma were separated into the bag, while the final vial contained the stem cells in an adequate volume of plasma necessary for cell resuspension. Subsequently, a dry infiltration of the separated and concentrated mesenchymal stem cells was performed. All patients received prophylactic pre- operative antibiotic therapy (cefazolin 2g); post-intervention, they were advised to refrain from sports activities for one week.
Results
No patient showed known postoperative side effects reported in the literature, such as severe pain, edema, surgical site infections, or bruising at the site of harvest. However, patients reported a sensation of "heaviness" in the joint for approximately 15 days. Patients were evaluated 12 months post-procedure. The International Knee Documentation Committee questionnaire and Visual Analog Scale were used for pain assessment, and an MRI was performed for instrumental evaluation of OA. Additionally, the Short Form Health Survey and the Western Ontario and McMaster Universities Osteoarthritis Index were administered to assess the impact of the treatment on overall quality of life and function. After 12 months post-intervention, 35 patients (76%) experienced a 50% reduction in pain, and significant improvements were observed in Short Form Health Survey scores, particularly in the Physical Functioning and Role-Physical domains.
Conclusions
The introduction of intra-articular stem cells represents a valid alternative therapy for lesions compared to high molecular weight hyaluronic acid injections and Platelet-Rich Plasma. The intra- articular administration of mesenchymal preparations indeed results in a reduction of inflammation in the joint and regeneration of the cartilage itself. In line with the scientific literature, the preliminary data from this case series, albeit with a limited follow- up, demonstrate the notable efficacy of the procedure, which despite being technologically advanced and seemingly complex, has proven safe and free from significant side effects in our experience [1-7].
References
- Saturno, F., Monaco, G., Tripari, G., Salerno, A., Lombardi,M. C., Guadagno, P., Cianciola, E. (2024). Implantation of Mesenchymal Stem Cells for the treatment of knee chondropathies: retrospective results of a multi-centric study. Journal of Anesthesia, Analgesia and Critical Care, 4(1), A137.
- Raghavan, S., Karthik, K. (2023). Role of Mesenchymal Stem Cells in Osteoarthritis: A Review. Stem Cells International.
- Smith, R. L., Jones, T. A. (2022). Advances in Cartilage Regeneration: The Promise of Stem Cells. Journal of Orthopedic Research, 40(3), 555-566.
- Chen, X., Wang, Y. (2023). Intra-Articular Injection of Stem Cells for Osteoarthritis: Current Evidence and Future Directions. Clinical Rheumatology, 42(5), 1401-1410.
- Albrecht, S., Möller, A. (2022). Stem Cell Therapy in Osteoarthritis: A Review of Current Evidence. Current Opinion in Rheumatology, 34(2), 145-152.
- D’Ambrosio,A., Catania, V. (2023). The Effect of Mesenchymal Stem Cells on Cartilage Repair and Regeneration. Tissue Engineering and Regenerative Medicine, 17(1), 19-29.
- Lee, J. H., Kim, H. S. (2023). Recent Advances in Regenerative Medicine for Osteoarthritis: Focus on Stem Cells. International Journal of Molecular Sciences, 24(5), 2750