Research Article - (2023) Volume 1, Issue 1
Long Covid and Neurodegenerative Disease
Copyright: ©©2023 Patrick Chambers. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Chambers, P. (2023). Long Covid and Neurodegenerative Disease. J Path Lab Med, 1(1), 11-24.
Abstract
Brain fog with compromised ability to concentrate has been the most frequent Long Covid (LC) complaint. This is due to an increased TGF beta/IFN gamma with consequently increased bradykinin (BKN), especially in Caucasian females. Brain and lung blood vessels “leak.” This same ratio is increased in Alzheimer’s disease (AD), but decreased in Parkin- son’s disease (PD), because CD4+ and CD8+ T cells are differentially affected by the invading associated viruses, e.g., SARS CoV2, HIV, Varicella zoster (VZV).... In Covid-19 CD147 receptors on immune cells are critical in generating the increased TGF beta/IFN gamma and those on endothelial cells, platelets, and erythrocytes are critical to the abnormal microvascular blood flow. ACE2 receptors on pneumocytes and enterocytes enable pulmonary and GI entry, initiating gut dysbiosis. Epigenetics, methylation, magnesium, vitamin D, the B vitamins, and antioxidants suggest that these is- sues can be surmounted. Biochemical, physiologic, and epidemiologic data are analyzed to answer these questions. An LC model is presented and discussed in the context of the most recent research. Suggestions to avoid these and other worrisome concerns are included. Other topics discussed include estrogen, the gut microbiome, type 2 diabetes (T2D), and homocysteine.
Keywords
Homocysteine, Estrogen, Bradykinin, Magnesium, Vitamin D, CD147Introduction
Long Covid (LC) has replaced Covid-19 as the topic du jour. Long term LC risks are unknown but have stoked growing con-cern. The neurodegenerative and tumorigenic implications are at the top of this list. Unfortunately, the wide spectrum of LC symptoms has defied mechanistic attempts to link their patho-genesis. There are clearly multiple factors involved, compli-cating these attempts. The male dominated Covid-19 stands in stark contrast to the female dominated LC. “Evidence based” efforts investigating such issues have traditionally relied on Ran-dom Clinical Trials (RCTs) and meta-analyses - the top down approach. Two inherent problems are loss of timeliness (RCTs) and diluted results (meta-analyses). A bottom up approach based on biochemistry, physiology, and epidemiology may be more advantageous, given the urgency of and universal interest in LC. This is an opinion piece aided by the deluge of recent research on this burgeoning problem.
LC Model
• CD147 receptors on T cells bind CD147 epitopes (the falci-parum antigen) on the spike protein S (no ACE2 receptors on circulating immune cells or on erythrocytes) [1-5].
• Subsequently SARS CoV2 overwhelms and exhausts CD4+ and CD8+ T cells and Natural Killer cells (NKs) • Persistent chronic lymphopenia after Covid-19 lowers secre-tion of IFN gamma (type II IFN), produced only by T cells and NK cells, especially CD4+ T cells [6-8].
• Decreased secretion of IFN gamma implies less hepatic syn- thesis of C1-INH [9] Uninhibited C1 triggers the Classic Complement Pathway (CCP) and crosstalk with the Kallikrein Kinin System (KKS) [10].
• The consequently increased bradykinin (BKN) is normally catabolized by angiotensin converting enzyme (ACE)
• Estrogen downregulates ACE and prolongs BKN half life. This makes estrogen an ACE inhibitor of sorts and increases the risk of some cancers [11,14].
• BKN enhances vascular permeability creating “leaks” primar¬ily in lungs15 and brain16 linking brain fog and dyspnea/post exertional malaise [15,16].
• IL-1beta, prominent in LC, potentiates the BKN induced mi¬crovascular leakage18 and brain fog primarily in Caucasian fe¬males [17,18].
• The ACE DD genotype in African Americans, an evolutionary adaptation to falciparum malaria, downregulates this leakage (tighter endothelial junctions) and elevates the relative frequen¬cy of LC in Caucasian females
• IFN gamma and TGF beta counterbalance each other and the loss of IFN gamma secreted by CD4+ and CD8+ T cells leaves an environment of unopposed TGF beta [19,20].
• Chronic low-grade IL-1beta and TNF alpha redirect pleiotro¬pic TGF beta from wound healing fibrosis to endothelial mesen¬chymal transition (End MT)/epithelial mesenchymal transition (EMT) and from tumor suppressor to tumor promoter [21-23].
• The switch of pleiotropic TGF beta from anti-inflammatory to proinflammatory appears to be more organ specific, e.g., neuro-vascular pericytes [24].
• Implications
• Long term LC may drive an increase in sporadic/late onset AD due to an elevated TGF beta/IFN beta [25-27].
• Late onset AD may appear earlier
• AD frequency in Caucasian females, especially in those also on HRT, may approach that in African American females [28, 29].
• Cancer risk/progression and fibrosis may also increase in Cau¬casian females
• CD147 is the primary receptor involved in the pathogenesis of ASCVD and LC long term may increase its incidence [30,31].
• The presence of CD147 receptors (but not those of ACE2) on platelets (and erythrocytes) creates platelet aggregates, further complicating the microcirculation (elevated mean platelet vol-ume (MPV)) [32].
• The presence of the CD147 epitope on the spike protein S por-tends dire consequences involving microvascular thrombosis in the short term for all exposed to the spike protein S.
• The incidence of PD in those with LC may increase, as LC increases risk for T2D, which predisposes to PD.
Discussion
Viral load and TGF-β/IFN-γ ratio determine Covid-19 symptoms (or not). This ratio decreases notably from the control group, passing through asymptomatic, up to symptomatic SARS-CoV-2 individuals [33]. But as IFN gamma secreting T cells are lost to the invading virus (see figure 1), this ratio inverts (increases). An depressed TGF beta/IFN gamma is affiliated with PD [34, 35]. An elevated ratio is affiliated with AD [25,26]. This bodes ill for those with LC with respect to fibrosis [36,37].
Figure 1: SARS CoV2 destroys IFN gamma producing cells [38].
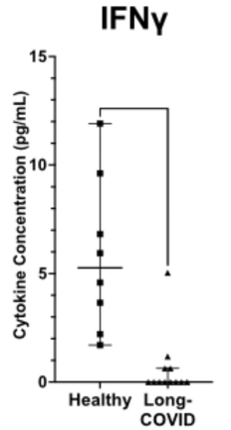
Figure 2: Low levels of IFN gamma persist into the LC phase [39].
The cytokines of LC provide insight to possible future compli-cations. TGF beta and IFN gamma are pleiotropic LC linked cy¬tokines that can work in either direction, i.e., anti- to proinflam¬matory, suppressing to promoting EMT or tumor for TGF beta and anti- to proliferative, pro-apoptotic to necrotic, antitumor to tumor for IFN gamma. However, it appears that the deleteri¬ous effects of the switch are more significant for TGF beta. IFN gamma appears to retain a net positive effect [40]. TGF beta func¬tions initially as an anti-inflammatory, keeping the inflammatory response of IFN gamma under control. Under chronic inflamma¬tory conditions TNF alpha is elevated and can upregulate TGF beta, which opposes IFN gamma.
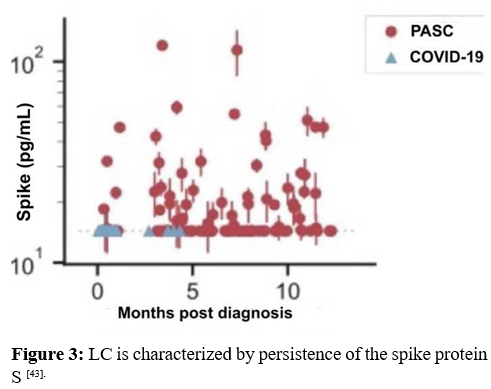
Chronic low doses of TGF beta when combined with chronic low doses of TNF alpha facilitate the switch of TGF beta from suppressing to promoting tumor23. This also appears to be the case for switching TGF-beta from wound healing fibrosis to en-dothelial or epithelial mesenchymal transition (EndMT or effect [40]. Therefore, it seems reasonable to assume that TNF alpha (chronic inflammation) might redirect TGF beta from anti-in-flammatory to pro-inflammatory and open the door to neurode¬generative disease. The constant stimulus to chronic inflamma¬tion posed by residual spike protein S could easily trigger this (see figure 3)
AD, PD, and LBD
The three primary forms of accelerated pre-senile dementia are AD, PD, and LBD. AD, the predominant form of pre-senile de-mentia, is more common in females while PD is more common in males. LBD is intermediate [44]. AD Is characterized by an ele-vated TGF beta/IFN gamma Onset of PD in females is later and less severe than that in men and might be due to benefits from premenopausal estrogen or hormone replacement therapy [45, 46]. Although protective against PD, estrogen possesses ACE inhibitor properties that elevate BKN. Lung and brain BKN induced leakage contributes to LC and AD. African American females appear to be less affected by LC but suffer the highest incidence of AD (by gender or race). At first this seems contradictory for two reasons. First, in African American but not in Caucasian fe-male’s estrogen levels decrease with increasing premenopausal age and BMI decreasing their risk of LC [47]. Secondly, the inci- dence of the ACE DD genotype (tighter endothelial junctions) is higher, also decreasing their risk of LC. However, according to a recent NHANES survey the incidence of obesity in African American females was 50% greater than that in Caucasian males, Caucasian females, or African-American males. This is presumed to be due to dietary factors, possibly monosodium glutamate (MSG) [48]. Perhaps escalating dietary MSG, induced by obesity and diabetes, overwhelms the protective properties of the ACE DD genotype, yielding more AD in African-American [49,50]. Obesity and diabetes also up regulate TGF beta, increasing the risk of AD [51]. Individuals with diabetes are up to four times more likely to develop LC [49, 51] Although homocysteine is elevated and contributes to the development of AD, LBD, and PD, PD is different. It is in some ways the opposite of AD, e.g., TGF beta/IFN gamma is depressed, not elevated. AD, LBD, and PD all feature extra cellular plaques - amyloid beta in AD, alpha-synuclein in LBD and PD. IFN gamma, elevated in PD, triggers microglial removal of amyloid [52,53]. PD also exhibits abnormal tryptophan metabolism due to increased IFN gamma and perhaps P5P deficiency [54] (see figure 4).

P5P is a required cofactor for aromatic amino acid decarboxylase (AADC), which produces dopamine and serotonin. Additionally, magnesium is a required cofactor for the synthesis of melatonin from serotonin. All three hormones are deficient in PD. CD4+ (superior producers of IFN gamma) and not CD4+ T cells ap¬pear to be responsible for the increased IFN gamma in PD [57,58]. SARS CoV2 preferentially attacks "HIV is tightly linked to hep¬atitis C virus (HCV) and hepatitis B virus (HBV). Viral hepatitis and alcohol induced hepatitis elicit elevated IFN-γ. On the other hand, SARS CoV2 and TGF-β have been linked to non-alcohol-ic steatohepatitis (NASH) aka non-alcoholic fatty liver disease (NAFLD). Liver resident CD8+ T cells appear to be responsible for the elevated IFN-γ [57,58]. CD8+ T cells produce the majority of IFN-γ [59]. CD8+ T cells increase in frequency in the aging brain and become a major source of IFNγ [60]. Loss of CD4+ T cells appears to potentiate CD8+ T cells [57]. IFN-γ causes blood-brain barrier leakage [61] and connects chronic alcoholism, viral hepatitis, and HIV to PD (depressed brain TGF-β/IFN-γ). SARS CoV2 preferentially attacks CD8+ T cells [62] (no ACE2 recep¬tors [63) restricting IFN-γ synthesis."
TGF and IFN
"HIV preferentially destroys CD4+ T cells [64], accentuating the IFN-γ response from cytopathic CD8+ T cells [57]. The HIV in-duced IFN-γ then increases the risk of viral hepatitis, PD [65] and autoimmune disease [66]. T2D also increases the risk of PD [67,68]. IFN-γ induces loss of dopamine neurons and nigrostriatal de-generation [69]. HIV increases the incidence of T2D [70]. The most recent research suggests that Parkinson’s is an autoimmune disease, which conforms to the well known linkage between IFN-γ and autoimmune disease. Abnormal tryptophan metabolism exhibited in Parkinson’s disease, due to an IFN-γ imbalance, is also seen in T2D and HIV [71].
In summary brain TGF-β is a key player in AD; likewise for brain IFN-γ in PD. IFN-γ is directly linked to a “leaky brain” via angiotensin II type one receptors (ATR1s) blocked by lo-sartan. TGF-β is indirectly linked through BKN and the KKS. TGF-β and IFN-γ are both pleiotropic and the direction of each appears to be determined by the chronic cytokine milieu, in-cluding pleiotropic IL-1β, pleiotropic TNF-α, and IL-6. Perhaps chronic inflammation (post viral infection that targets CD4+ T cells, CD8+ T cells, hepatocytes, or pericytes [72]) in those with marginal onboard antioxidants are predisposed to AD and PD."
TGF-β/Smad signaling pathway in renal, hepatic, pulmonary and cardiac fibrosis has been well documented [73]. Recent studies show this to include the brain as well [74-76]. Magnesium pos¬sesses the capacity to down regulate this SMAD pathway in the liver and the lungs [77,78]. IFN gamma also appears capable in this regard [79]. Exercise also helps by increasing IFN gamma [80]. This is because exercise upregulates angiotensin II and angiotensin II upregulates IFN gamma [81,82].
ACE and BKN
Angiotensin converting enzyme (ACE) produces angiotensin II and degrades BKN. Estrogen downregulates ACE, up regulat¬ing BKN. Bradykinin upregulates tyrosine hydroxylase, the rate limiting step in dopamine synthesis [83]. This might help explain the protective effects of estrogen in avoiding PD. The frequency of the ACE II genotype in AD is 1.4x higher than that in controls v 0.4x for the ACE DD genotype [84]. ACE levels are up to 70% higher in the DD genotype [85]. Endothelial cell junctions are tighter and less permeable in the DD ACE genotype [86]. However, after menopause tight junction permeability due to endothe¬lial dysfunction initiated by oxidative stress, microthrombosis (loss of RBC deformability), immune complexes (endothelial CD147 and perhaps ACE2 receptors linking to spike S epitopes), increases. The ACE DD genotype is protective against AD not only due to tighter endothelial junctions but also to increased ACE and less BKN [87,88]. BKN induced endothelial permeability not only produces perivascular angioedema but also leads to increased fibrosis [89]. Either mechanism may contribute to the brain fog, dyspnea, and post exertional malaise of LC.
Because ACE is higher and bradykinin levels are commensu- rately lower in those with the ACE DD genotype, African Amer¬ican females should be less likely to develop LC versus their Caucasian counterparts. Furthermore, in African American but not in Caucasian females estrogen levels decreased with increasing premenopausal age and BMI, theoretically minimizing their susceptibility to LC. Yet the incidence of AD is higher in African American females than that in African American men, Cauca¬sian men, or Caucasian females. A 2017-18 NHANES survey indicate that the incidence of obesity in African-American fe¬males was almost 50% greater than that in their Caucasian coun-terparts.
Perhaps the AD inducing properties of postmenopausal obe-sity (and diabetes) overwhelm the protective properties of the ACE DD genotype. Some studies purport to show a decreased incidence of cancer in those with Alzheimer’s disease. However, African-American females [90]. are 40% more likely to die of breast cancer than Caucasian females. African-Americans have the highest death rate and shortest survival of any racial/ethnic group in the US for most cancers and have a greater incidence of Alzheimer’s disease than any other racial group in America [91]. On the other hand, overall cancer risk is lower in people with PD, compared to the general population. This difference in cancer risk between AD and PD speaks to further linkage be-tween TGF beta in cancer causation and IFN gamma in cancer avoidance [92].
Estrogen and HRT for more than 10 years have been linked to a slight increase in cancer risk [93]. Estrogen downregulates ACE and is an ACE inhibitor of sorts. ACE inhibitors have been linked to an increase in lung cancer [94]. Not surprisingly, brady¬kinin has been linked to aggressive prostate cancer [95].
Homocysteine and B Vitamins
Homocysteine plays a prominent role in all forms of dementia [96]. Asians have a lower Ca:Mg diet and a lower incidence of AD [97]. P5P figures prominently in homocysteine recycling and is a required cofactor for aromatic amino acid decarboxylase (AADC)(see figure 5). PD patients are frequently B6 deficient [98]. and exhibit abnormalities in both dopamine and serotonin synthesis [99].
Dopamine synthesis shortfall in PD appears to be primarily driv¬en by IFN gamma and its effect on tryptophan metabolism (see figure 4).
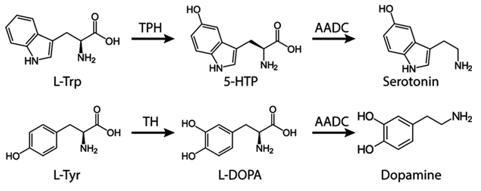
Figure 5: AADC requires the cofactor P5P
Figure 6: Vitamins B2, B3, B6, B9, and B12 are all heavily involved in both the folate cycle and the methionine cycle. B6 is also required as a cofactor to recycle homocysteine [100].
Homocysteine induces oxidative stress and is a marker for COVID-19 severity, LC, and demential [101-105]. As shown in fig-ure 6, vitamins B2,3,6,9,12 are prominent cofactors in both the folate and methionine cycles, which are integral in the metab-olism of homocysteine [106]. ATP and methylation both require Mg++. These B vitamin deficiencies are precisely those associ-ated with cognitive impairment and AD [107]. B complex supple-ments usually provide methylated B12, but pyridoxine (B6), and folate (B9) are not their active forms. Indeed, the active forms of B1,2,3,9,12 all require magnesium and some require activated B6 as a cofactor [100]. B1,3, and 6 must be phosphorylated (ATP and magnesium as chelate); B2,9,12 must be methylated magnesium as cofactor [108]. The active form of B6 requires ATP and chelated Mg (phosphorylation) and its activated form (P-5-P) as a cofactor, creating a catch 22 situation. Most B6 supplements contain pyridoxine (PNP), which in excess can lead to peripheral neuropathy [109]. This can be avoided by substituting pyri-doxal-5-phosphate. Rxn 2 (PNPO) in figure 7 mediated by P5P oxidase is the rate limiting step
Figure 7: Note the need for ATP (and Mg++) to convert pyridoxal (and PNP, PMP) to P5P. PNPO requires P-5-P as cofactor.
P-5-P is critical to its own synthesis, to that of the active forms of other B vitamins, and to the recycling of homocysteine to gluta-thione (see figure 8). Its critical role in cognition is well known. Magnesium is also critical to the synthesis of all endogenous and most exogenous antioxidants [110-112].
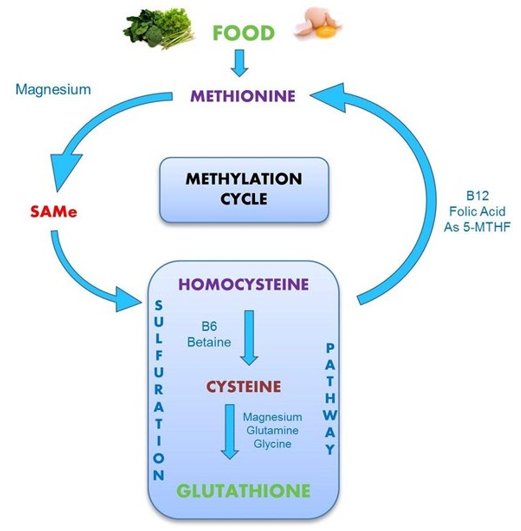
Figure 8: Magnesium and P5P are required to recycle homocysteine.
Vitamin B5 (pantothenate) deficiency is also associated with both AD band PD. In order for pantothenate to reach its active form, three phosphorylation’s must occur. Each requires ATP and magnesium [113-115].
Vitamin D and Ca:Mg
An elevated Ca++ and a depressed Mg++ (high Ca:Mg) are linked to AD [16,17]. and PD [18,19]. A recent 2023 article reported that vitamin D, folic acid and vitamin B12 could reverse the cognitive decline leading to AD. Total benefit exceeded that from any single supplement [120]. Adding magnesium and P5P to this regimen would improve the results immeasurably. As Ca:Mg increases, Vitamin D loses its efficacy for colorectal cancer, prostate can¬cer, esophageal cancer, cardiovascular disease, metabolic syn¬drome, total mortality, and cognitive function [121]. The Western diet is high in processed foods with high monosodium glutamate (MSG)/calcium and low fiber/magnesium. The typical Asian diet offers more magnesium but less calcium. The target Ca:Mg for both is 2.0. A fiber rich diet that includes fermented vegetables is inversely proportional to Covid-19 mortality in Europe [122,123]. as demonstrated in Germany (sauerkraut) and South Korea ((kimchi) Ionized serum Ca:Mg and 25(OH)D3 are measures of general health that reflect CRP and HRV [124-126]. Serum mag-nesium also reflects diversity of gut microbiota and gut health [127,128]. The correlation between the serum magnesium and de-mentia depends on the Ca:Mg. On a Western diet the healthy upper limit is 2.6. On an Eastern diet the lower limit is 1.7 [129].
APOE and Methylation
Reducing the Ca:Mg ratio reduces the risk of dementia [130]. Methylation of DNA for stability retards the onset of both neu-rodegenerative diseases, e.g., the APOE gene, and cancer, e.g., basigin (CD147) gene [131-133]. Hypomethylation increases with age and leads to an unstable genome, with activation of some tumor promoter genes [134,135]. Hypomethylation of CpG islands (cytosine-guanine pairs) promotes AD, LBD, and PD [136-139]. There are three alleles for APOE and APOE4 is the major risk factor for Alzheimer’s disease [140]. Hypomethylation of APOE4 is a major determinant in this.
15% of the US population is heterozygous for this and 5% are homozygous. Chris Hemsworth recently announced a pullback in his schedule to spend more time with family. It was also re-ported that while working on a nature film he learned that he was homozygous for APOE4. But as worrisome as that might seem, AD appears to be less genetic and more epigenetic. The native American Indian population presents plenty of APOE4 but very little Alzheimer’s disease [141]. The Paleolithic diet provides an excellent Ca:Mg balance. Dairy is not included (eggs 5.4, milk 10). Perhaps this dementia gene (APOE4) itself is not the prob¬lem but an elevated Ca:Mg, crowding out the Mg++. Ca++ and Mg++ share the same receptor - CaSR (Calcium sensing recep¬tor). DNA methylation occurs via SAMe and magnesium (see figure 8).
Treatment
1. Magnesium and vitamin D (50 ng/L target) are at the top of the list for both prevention and treatment. Magnesium is a critical mineral in the human body and is involved in ~80% of known metabolic functions [142]. Vitamin D possesses invaluable antioxidant and anti-inflammatory properties (see figure 9). Ap¬proximately 75% of human immune system functions depend on maintaining a healthy, physiological serum 25(OH)D con¬centration [143]
Figure 9. Vitamin D provides anti-inflammatory protection up¬stream and downstream of the AT1R [144].
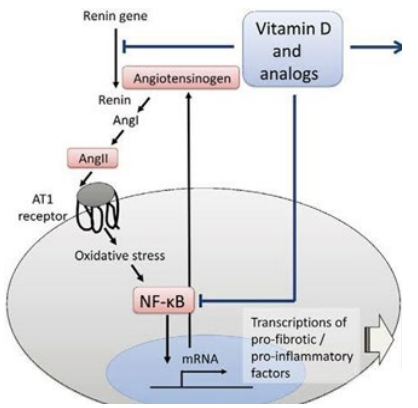
2. The target Ca:Mg is 2.0, but any ratio greater than 2.6 com¬promises the efficacy of vitamin D [121]. Covid-19 makes AD worse [145]. Ca++ dysregulation plays a prominent role in both AD and amyloid beta deposition [130].
3. Antioxidants are vital in the defense of COVID-19 infection. However, if the onboard supply is suboptimal, the vast numbers of ROS generated may overwhelm mitochondria and markedly compromise ATP production. Most endogenously produced and some exogenous antioxidants require ATP (and magnesium) to attain activated status. Vitamin C (water-soluble), vitamins A, D3, E, K (fat-soluble), Zn, D-ribose, selenium, and many others require no processing [112]. Furthermore, hydroxylation of C1 of 25(OH)D in the synthesis of active vitamin D occurs in the mito¬chondria and is suppressed by calcium [146]. Loss of mitochondria due to oxidative stress compromises vitamin D efficacy in addi¬tion to the elevated Ca:Mg.
4. P5P aka PLP is the active form of B6, which is required for activation of many of the B vitamins associated with homocys¬teine metabolism.
5. A sedentary lifestyle risks eventual obesity and diabetes. Ex¬ercise also facilitates a better IFN gamma:TGF beta balance by increasing IFN gamma levels [80].
6. Probiotics, especially after antibiotic therapy, improves the diversity of the gut microbiome [147-149].
7. Dehydration triggers the renal resorption of Na+ and water. This also means loss of Mg++ (and K+) to maintain electroneu-trality. Also, the thirst reflex diminishes with age. What good is increased dietary/supplemental Mg++ in the face of a magnesuric drain. Hydration maintenance, easily overlooked, potentially trumps all the other suggestions.
Conclusion
"The most recent research presented here suggests that AD, PD, LBD, and T2D might all be secondary to an imbalanced TGF-β/IFN-γ perhaps induced by some previous viral infection that preferentially targeted CD4+ and/or CD8+ T cells These T cells secrete IFN-γ with the lion’s share coming from CD8+ T cells. HIV preferentially attacks CD4+ T cells, leaving the heavy IFN-γ producing CD8+ T cells. HIV mimics PD in many ways, including susceptibility to de novo T2D. TGF-β/IFN-γ is elevat-ed in AD (and VZV), at least in the brain, and depressed in PD, LBD, HIV, and T2D."
Estrogen protects against and delays PD, but predisposes to LC and postmenopausal AD, especially if on HRT. Increased BKN mediates LC and AD. The ACE DD genotype is protective against LC and AD. Estrogen down regulates ACE, causing an increase in BKN. Obesity and diabetes predispose to AD, LBD, and PD. Magnesium and P5P, the active form of vitamin B6, are deficient in most with AD, LBD, and PD.
CD147 and BKN in the proposed model play vital roles both in the development of LC and in its long-term consequences. But an imbalance between TGF beta and IFN gamma due to SARS CoV2 induced lymphopenia supercharges the roles of CD147 and BKN. The implications of the proposed hypothesis with re¬spect to LC and its possible long-term consequences extend not only to the unvaccinated but also to the vaccinated, who might be even more susceptible to recurrent SARS CoV2 (see figure 10), unless preventative measures are taken.
Figure 10: Recurrent COVID-19 is directly related to the number of boosters [150].
Vaccine efficacy or not is irrelevant. ACE2 receptors are not present on immune cells (CD4+, CD8+ T cells, NK cells), the primary combatants against SARS CoV2. The presence of the ACE2 receptor on endothelial cells is controversial [152]. Indeed the pathogenesis of microvascular thrombosis, lymphopenia, and TGF beta predominance in Covid-19 cannot be explained without acknowledging the presence of the CD147 epitope on the spike protein S, first reported in a Chinese study [1]. This find-ing was quickly challenged, but those challenges were later de-bunked [2]. However, the worrisome implications of this model are not irreversible.
These preventative measures include serum Ca:Mg near 2.0, se¬rum 25(OH)D3 near 50 ng/L, and an abundance of micronutrient antioxidants, especially P-5-P [153]. The efficacy of vitamin D is compromised in the face of an elevated Ca:Mg [121]. But D3 may be even better for endothelial health [154]. Endothelial competence is not only at the center of LC, AD, LBD, PD, and many cancers but also in the progression of cardiovascular disease, arthritis, multiple sclerosis, and sepsis [154]. So, D3 may be of benefit even in those with an elevated Ca:Mg.
Vitamin D (1,25(OH)2D, the active form, is much more reliant on Mg++ for its efficacy. It requires Mg++ as a cofactor for three steps in its synthesis. Even parathormone synthesis requires Mg++. Ultimately exercise, diet including some supplementation, and hydration are the primary determinants of epigenetically de¬termined health over and above those associated with LC. HRV, the fifth vital sign, connects all the vital players - gut microbiome diversity155, dietary micronutrients, e.g., magnesium156, balanced Ca:Mg125, balanced TGF-beta157 and TNF-α158, and D3125. It’s all about balance[125,155-158].
References
- Wang, K. E., Chen, W., Zhang, Z., Deng, Y., Lian, J. Q., Du, P., ... & Chen, Z. N. (2020). CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal transduction and targeted therapy, 5(1), 283.
- Chambers, M. D. (2021). Basigin Binds Spike S on SARS-CoV-2. Open Access Library Journal.
- Behl, T., Kaur, I., Aleya, L., Sehgal, A., Singh, S., Sharma, N., & Bungau, S. (2022). CD147-spike protein interaction in COVID-19: Get the ball rolling with a novel receptor and therapeutic target. Science of the Total Environment, 808, 152072.
- Fenizia, C., Galbiati, S., Vanetti, C., Vago, R., Clerici, M., Tacchetti, C., & Daniele, T. (2021). SARS-CoV-2 entry: at the crossroads of CD147 and ACE2. Cells, 10(6), 1434.
- Song, X., Hu, W., Yu, H., Zhao, L., Zhao, Y., Zhao, X., & Zhao, Y. (2020). Little to no expression of angiotensin-con-verting enzyme-2 on most human peripheral blood immune cells but highly expressed on tissue macrophages. Cytom-etry Part A.
- Manne, B. K., Denorme, F., Middleton, E. A., Portier, I., Rowley, J. W., Stubben, C., & Campbell, R. A. (2020). Platelet gene expression and function in patients with COVID-19. Blood, 136(11), 1317-1329.
- Varghese, J., Sandmann, S., Ochs, K., Schrempf, I. M., Frömmel, C., Dugas, M., & Tepasse, P. R. (2021). Persistent symptoms and lab abnormalities in patients who recovered from COVID-19. Scientific Reports, 11(1), 1-8.
- Kim, M. H., Salloum, S., Wang, J. Y., Wong, L. P., Regan, J., Lefteri, K., & MGH COVID-19 Collection & Processing Team. (2021). Type I, II, and III interferon signatures correspond to coronavirus disease 2019 severity. The Journal of infectious diseases, 224(5), 777-782.
- Zahedi, K., Prada, A. E., & Davis 3rd, A. E. (1994). Transcriptional regulation of the C1 inhibitor gene by gamma-interferon. Journal of Biological Chemistry, 269(13), 9669-9674.
- Bossi, F., Peerschke, E. I., Ghebrehiwet, B., & Tedesco,F. (2011). Cross-talk between the complement and the kinin system in vascular permeability. Immunology letters, 140(1-2), 7-13.
- Gallagher, P. E., Li, P., Lenhart, J. R., Chappell, M. C., & Brosnihan, K. B. (1999). Estrogen regulation of angiotensin-converting enzyme mRNA. Hypertension, 33(1), 323-328.
- Wang, W., Zhou, Y., Wei, R., Jiang, G., Li, F., Chen, X., & Xi, L. (2019). Bradykinin promotes proliferation, migration, and invasion of cervical cancer cells through STAT3 signaling pathways. Oncology reports, 42(6), 2521-2527.
- Greco, S., Elia, M. G., Muscella, A., Romano, S., Storelli, C., & Marsigliante, S. (2005). Bradykinin stimulates cell proliferation through an extracellular-regulated kinase 1 and 2-dependent mechanism in breast cancer cells in primary culture. Journal of endocrinology, 186(2), 291-302.
- Sun, D. P., Lee, Y. W., Chen, J. T., Lin, Y. W., & Chen, R. M.(2020). The bradykinin-BDKRB1 axis regulates aquaporin 4 gene expression and consequential migration and invasion of malignant glioblastoma cells via a Ca2+-MEK1-ERK1/2-NF-κB mechanism. Cancers, 12(3), 667.
- Ichinose, M., & Barnes, P. J. (1990). Bradykinin-induced airway microvascular leakage and bronchoconstriction are mediated via a bradykinin B2 receptor. Am Rev Respir Dis,142(5), 1104-1107.
- Sarker, M. H., Hu, D. E., & Fraser, P. A. (2000). Acute effects of bradykinin on cerebral microvascular permeability in the anaesthetized rat. The Journal of physiology, 528(Pt 1), 177.
- Schultheiß, C., Willscher, E., Paschold, L., Gottschick, C., Klee, B., Henkes, S. S., & Binder, M. (2022). The IL-1β, IL-6, and TNF cytokine triad is associated with post-acute se-quelae of COVID-19. Cell Reports Medicine, 3(6), 100663.
- Freitas, F., Tibiriçá, E., Singh, M., Fraser, P. A., & Mann, G.E. (2020). Redox regulation of microvascular permeability: IL-1β potentiation of bradykinin-induced permeability is prevented by simvastatin. Antioxidants, 9(12), 1269.
- Delvig AA, Lee JJ, Chrzanowska-Lightowlers ZM, Robinson JH. (2002) TGF-beta1 and IFN-gamma cross-regulate antigen presentation to CD4 T cells by macrophages. J Leukoc Biol. 72(1):163-6.
- Sobral LM, Montan PF, Martelli-Junior H, Graner E, Co-letta RD. (2007) Opposite effects of TGF-β1 and IFN-γ on transdifferentiation of myofibroblast in human gingival cell cultures. Journal of Clinical Periodontology 34: 397-406.
- Yoshimatsu, Y., Kimuro, S., Pauty, J., Takagaki, K., Nomi-yama, S., Inagawa, A., & Watabe, T. (2020). TGF-beta and TNF-α cooperatively induce mesenchymal transition of lymphatic endothelial cells via activation of Activin signals. PLoS One, 15(5), e0232356.
- Masola, V., Carraro, A., Granata, S., Signorini, L., Bellin, G., Violi, P., ... & Zaza, G. (2019). In vitro effects of interleukin (IL)-1 beta inhibition on the epithelial-to-mesenchy-mal transition (EMT) of renal tubular and hepatic stellate cells. Journal of translational medicine, 17(1), 1-11.
- Liu, Z. W., Zhang, Y. M., Zhang, L. Y., Zhou, T., Li, Y. Y.,Zhou, G. C., & Liu, Y. Q. (2022). Duality of interactions between tgf-β and tnf-α during tumor formation. Frontiers in Immunology, 12, 5628.
- Rustenhoven, J., Aalderink, M., Scotter, E. L., Oldfield,R. L., Bergin, P. S., Mee, E. W., & Dragunow, M. (2016). TGF-beta1 regulates human brain pericyte inflammatory processes involved in neurovasculature function. Journal of neuroinflammation, 13(1), 1-15.
- Rota, E., Bellone, G., Rocca, P., Bergamasco, B., Emanuel-li, G., & Ferrero, P. (2006). Increased intrathecal TGF-β1, but not IL-12, IFN-γ and IL-10 levels in Alzheimer’s disease patients. Neurological Sciences, 27, 33-39.
- Taipa, R., das Neves, S. P., Sousa, A. L., Fernandes, J., Pin-to, C., Correia, A. P., ... & Sousa, N. (2019). Proinflammatory and anti-inflammatory cytokines in the CSF of patients with Alzheimer's disease and their correlation with cognitive decline. Neurobiology of aging, 76, 125-132.
- Zhang, X., Huang, W. J., & Chen, W. W. (2016). TGF-beta1 factor in the cerebrovascular diseases of Alzheimer's disease. Eur Rev Med Pharmacol Sci, 20(24), 5178-5185.
- Maki, P. M., Girard, L. M., & Manson, J. E. (2019). Meno-pausal hormone therapy and cognition. Bmj, 364.
- Savolainen-Peltonen, H., Rahkola-Soisalo, P., Hoti, F., Vat-tulainen, P., Gissler, M., Ylikorkala, O., & Mikkola, T. S. (2019). Use of postmenopausal hormone therapy and risk of Alzheimer’s disease in Finland: nationwide case-control study. bmj, 364.
- Chen, P. Y., Qin, L., Li, G., Wang, Z., Dahlman, J. E., Malag-on-Lopez, J., & Simons, M. (2019). Endothelial TGF-β signalling drives vascular inflammation and atherosclerosis. Nature metabolism, 1(9), 912-926.
- Wang, C., Jin, R., Zhu, X., Yan, J., & Li, G. (2015). Function of CD147 in atherosclerosis and atherothrombosis. Journal of cardiovascular translational research, 8, 59-66.
- Nishikawa, M., Kanno, H., Zhou, Y., Xiao, T. H., Suzuki, T., Ibayashi, Y., & Goda, K. (2021). Massive image-based single-cell profiling reveals high levels of circulating platelet aggregates in patients with COVID-19. Nature communications, 12(1), 7135.
- Villalba, M. C. M., Ramírez, O. V., Jiménez, M. M., Gar-cia, A., Alfonso, J. M., Baéz, G. G., ... & Tirado, M. G. G. (2020). Interferon gamma, TGF-β1 and RANTES expression in upper airway samples from SARS-CoV-2 infected patients. Clinical Immunology, 220, 108576.
- Tesseur, I., Nguyen, A., Chang, B., Li, L., Woodling, N. S., Wyss-Coray, T., & Luo, J. (2017). Deficiency in neuronal TGF-β signaling leads to nigrostriatal degeneration and activation of TGF-β signaling protects against MPTP neu- rotoxicity in mice. Journal of Neuroscience, 37(17), 4584- 4592.
- Mohamed, A., Maha, A., Hanan, H., & Samaa, T. (2014). Association of serum interferon gamma level with Parkinson's disease in Egyptian patients.
- Hu, Z. J., Xu, J., Yin, J. M., Li, L., Hou, W., Zhang, L. L., ...& Jin, R. H. (2020). Lower circulating interferon-gamma is a risk factor for lung fibrosis in COVID-19 patients. Frontiers in immunology, 11, 585647.
- Weng, H., Mertens, P. R., Gressner, A. M., & Dooley, S. (2007). IFN-γ abrogates profibrogenic TGF-β signaling in liver by targeting expression of inhibitory and receptor Smads. Journal of hepatology, 46(2), 295-303.
- Dhanda, A. D., Felmlee, D., Boeira, P., Moodley, P., Tan, H., Scalioni, L. D. P., & Cramp, M. E. (2022). Patients with moderate to severe COVID-19 have an impaired cytokine response with an exhausted and senescent immune phenotype. Immunobiology, 227(2), 152185.
- Williams, E. S., Martins, T. B., Hill, H. R., Coiras, M., Shah,K. S., Planelles, V., & Spivak, A. M. (2022). Plasma cytokine levels reveal deficiencies in IL-8 and gamma interferon in Long-COVID. medRxiv, 2022-10.
- Castro, F., Cardoso, A. P., Gonçalves, R. M., Serre, K., & Oliveira, M. J. (2018). Interferon-gamma at the crossroads of tumor immune surveillance or evasion. Frontiers in immunology, 9, 847.
- Yoshimatsu, Y., Wakabayashi, I., Kimuro, S., Takahashi, N., Takahashi, K., Kobayashi, M., & Watabe, T. (2020). TNF-α enhances TG-β-induced endothelial-to-ÂÂmesenchymal transition via TGF-β signal augmentation. Cancer science, 111(7), 2385-2399.
- Borthwick, L. A., Gardner, A., De Soyza, A., Mann, D. A., & Fisher, A. J. (2012). Transforming growth factor-β1 (TGF-β1) driven epithelial to mesenchymal transition (EMT) is accentuated by tumour necrosis factor α (TNFα) via crosstalk between the SMAD and NF-κB pathways.Cancer microenvironment, 5, 45-57.
- Swank, Z., Senussi, Y., Manickas-Hill, Z., Yu, X. G., Li,J. Z., Alter, G., & Walt, D. R. (2022). Persistent circulating severe acute respiratory syndrome coronavirus 2 spike is as-sociated with post-acute coronavirus disease 2019 se-quelae. Clinical Infectious Diseases.?
- Mouton, A., Blanc, F., Gros, A., Manera, V., Fabre, R., Sau-leau, E., & David, R. (2018). Sex ratio in dementia with Lewy bodies balanced between Alzheimer’s disease and Parkinson’s disease dementia: a cross-sectional study. Alzheimer's Research & Therapy, 10, 1-10.
- Reekes, T. H., Higginson, C. I., Ledbetter, C. R., Sathivad-ivel, N., Zweig, R. M., & Disbrow, E. A. (2020). Sex specific cognitive differences in Parkinson disease. npj Parkinson's Disease, 6(1), 7.
- de Souza HDN, Oliveira A, Ulrich H. (2017) Bradykinin reverts contralateral rotation and restores dopamine levels in an animal model of Parkinson disease. 11th World Congress on Neurology and Therapeutics March 27-29, Madrid, Spain
- Manson, J. M., Sammel, M. D., Freeman, E. W., & Gris-so, J. A. (2001). Racial differences in sex hormone levels in women approaching the transition to menopause. Fertility and sterility, 75(2), 297-304.
- Pepino, M. Y., Finkbeiner, S., Beauchamp, G. K., & Men-nella, J. A. (2010). Obese women have lower monosodium glutamate taste sensitivity and prefer higher concentrations than do normal-weight women. Obesity, 18(5), 959-965.
- Bautista, R. J. H., Mahmoud, A. M., Königsberg, M., & Guerrero, N. E. L. D. (2019). Obesity: Pathophysiology, monosodium glutamate-induced model and anti-obesity medicinal plants. Biomedicine & Pharmacotherapy, 111, 503-516.
- Bahadoran, Z., Mirmiran, P., & Ghasemi, A. (2019). Monosodium glutamate (MSG)-induced animal model of type 2 diabetes. Pre-Clinical Models: Techniques and Protocols, 49-65.
- Tan, C. K., Chong, H. C., Tan, E. H. P., & Tan, N. S. (2012).Getting ‘Smad’about obesity and diabetes. Nutrition & diabetes, 2(3), e29-e29.
- He, Z., Yang, Y., Xing, Z., Zuo, Z., Wang, R., Gu, H., &Yao, Z. (2020). Intraperitoneal injection of IFN-γ restores microglial autophagy, promotes amyloid-β clearance and improves cognition in APP/PS1 mice. Cell Death & Disease, 11(6), 440.
- Zheng, C., Zhou, X. W., & Wang, J. Z. (2016). The dual roles of cytokines in Alzheimer’s disease: update on inter-leukins, TNF-α, TGF-β and IFN-γ. Translational neurodegeneration, 5(1), 1-15.
- Shen, L. (2015). Associations between B vitamins and Par-kinson’s disease. Nutrients, 7(9), 7197-7208.
- Ma, H., Yan, J., Sun, W., Jiang, M., & Zhang, Y. (2022).Melatonin treatment for sleep disorders in parkinson's disease: A meta-analysis and systematic review. Frontiers in Aging Neuroscience, 44.
- Müller N, Myint A-M, Schwarz MJ. (2009) The impact ofneuroimmune dysregulation on neuroprotection and neuro- toxicity in psychiatric disorders - relation to drug treatment.Dialogues in Clinical Neuroscience 11(3):319-332.
- Luxenburger, H., & Neumann-Haefelin, C. (2023). Liver-resident CD8+ T cells in viral hepatitis: not always good guys. The Journal of Clinical Investigation, 133(1).
- Malagnino, V., Cerva, C., Teti, E., Campogiani, L., Com-pagno, M., Foroghi Biland, L., ... & Sarmati, L. (2021). Poor CD4/CD8 ratio recovery in HBcAb-positive HIV patients with worse immune status is associated with significantly higher CD8 cell numbers. Scientific Reports, 11(1), 1-9
- Ren, Y. L., Li, T. T., Cui, W., Zhao, L. M., Gao, N., Liao,H., ... & Pan, L. L. (2021). CD8+ T lymphocyte is a main source of interferon-gamma production in Takayasu’s arte-ritis. Scientific Reports, 11(1), 17111.
- Liston, A., & Yshii, L. (2023). T cells drive aging of the brain. Nature Immunology, 24(1), 12-13.
- Bonney, S., Seitz, S., Ryan, C. A., Jones, K. L., Clarke, P., Tyler, K. L., & Siegenthaler, J. A. (2019). Gamma interferon alters junctional integrity via rho kinase, resulting in blood-brain barrier leakage in experimental viral encephali-tis. MBio, 10(4), e01675-19.
- Urra, J. M., Cabrera, C. M., Porras, L., & Ródenas, I. (2020). Selective CD8 cell reduction by SARS-CoV-2 is associated with a worse prognosis and systemic inflammation in COVID-19 patients. Clinical immunology, 217, 108486Shen.
- Shen, X. R., Geng, R., Li, Q., Chen, Y., Li, S. F., Wang,Q., ... & Zhou, P. (2022). ACE2-independent infection of T lymphocytes by SARS-CoV-2. Signal transduction and targeted therapy, 7(1), 83.
- Gulzar, N., & Copeland, K. F. (2004). CD8+ T-cells: function and response to HIV infection. Current HIV research, 2(1), 23-37.
- Koutsilieri, E., Sopper, S., Scheller, C., Ter Meulen, V., & Riederer, P. (2002). Parkinsonism in HIV dementia. Journal of neural transmission, 109, 767-775.
- Lees, J. R. (2015). Interferon gamma in autoimmunity: A complicated player on a complex stage. Cytokine, 74(1), 18-26.
- Chohan, H., Senkevich, K., Patel, R. K., Bestwick, J. P., Jacobs, B. M., Bandres Ciga, S., ... & Noyce, A. J. (2021). Type 2 diabetes as a determinant of Parkinson's disease risk and progression. Movement disorders, 36(6), 1420-1429.
- De Pablo-Fernandez, E., Goldacre, R., Pakpoor, J., Noyce, J., & Warner, T. T. (2018). Association between diabetes and subsequent Parkinson disease: a record-linkage cohort study. Neurology, 91(2), e139-e142.
- Chakrabarty, P., Ceballos-Diaz, C., Lin, W. L., Beccard, A., Jansen-West, K., McFarland, N. R., ... & Golde, T. E. (2011). Interferon-γ induces progressive nigrostriatal degeneration and basal ganglia calcification. Nature neuroscience, 14(6), 694-696.
- Kalra, S., Kalra, B., Agrawal, N., & Unnikrishnan, A. G. (2011). Understanding diabetes in patients with HIV/AIDS. Diabetology & metabolic syndrome, 3, 1-7.
- Murray, M. F. (2003). Tryptophan depletion and HIV Tryptophan depletion and HIV infection: a metabolic link to pathogenesis. The Lancet infectious diseases, 3(10), 644-652.
- Butsabong, T., Felippe, M., Campagnolo, P., & Maringer,K. (2021). The emerging role of perivascular cells (pericytes) in viral pathogenesis. The Journal of General Virology, 102(8).
- Hu, H. H., Chen, D. Q., Wang, Y. N., Feng, Y. L., Cao,G., Vaziri, N. D., & Zhao, Y. Y. (2018). New insights into TGF-β/Smad signaling in tissue fibrosis. Chemico-biologi-cal interactions, 292, 76-83.
- Town, T., Laouar, Y., Pittenger, C., Mori, T., Szekely, C. A., Tan, J., & Flavell, R. A. (2008). Blocking TGF-β–Smad2/3 innate immune signaling mitigates Alzheimer-like pathology. Nature medicine, 14(6), 681-687.
- Yang, C., & Xu, P. (2022). The role of transforming growth factor β1/Smad pathway in Alzheimer’s disease inflammation pathology. Molecular Biology Reports, 1-12.
- Xu, L., Pan, C. L., Wu, X. H., Song, J. J., Meng, P., Li, L.,& Zhang, Z. Y. (2021). Inhibition of Smad3 in macrophages promotes Aβ efflux from the brain and thereby ameliorates Alzheimer's pathology. Brain, Behavior, and Immunity, 95, 154-167.
- Tee, J. K., Peng, F., Tan, Y. L., Yu, B., & Ho, H. K. (2018).Magnesium isoglycyrrhizinate ameliorates fibrosis and disrupts TGF-β-mediated SMAD pathway in activated hepatic stellate cell line LX2. Frontiers in Pharmacology, 9, 1018.
- Luo, X., Deng, Q., Xue, Y., Zhang, T., Wu, Z., Peng, H., & Pan, G. (2021). Anti-fibrosis effects of magnesium lithospermate B in experimental pulmonary fibrosis: By inhibiting TGF-βri/smad signaling. Molecules, 26(6), 1715.
- Ulloa L, Doody J, Massagué J. (1999) Inhibition of transforming growth factor-β/SMAD signalling by the interfer-on-γ/STAT pathway. Nature 397, 710–713.
- Vijayaraghava, A., & Radhika, K. (2014). Alteration of interferon gamma (IFN-γ) in human plasma with graded physical activity. Journal of clinical and diagnostic research: JCDR, 8(6), BC05.
- Li, X., & Wang, K. (2017). Effects of moderate-intensity endurance exercise on angiotensin II and angiotensin II type I receptors in the rat heart. Molecular Medicine Reports, 16(3), 2439-2444.
- FERNANDEZ-CASTELO, S. I. L. V. I. A., ARZT, E. S., PESCE, A., CRISCUOLO, M. E., DIAZ, A., FINKIEL-MAN, S., & NAHMOD, V. E. (1987). Angiotensin II regulates interferon-γ production. Journal of interferon research, 7(3), 261-268.
- Gebreyesus, K., Kilbourne, E. J., & Sabban, E. L. (1993). Bradykinin elevates tyrosine hydroxylase and dopamine β-hydroxylase mRNA levels in PC12 cells. Brain research, 608(2), 345-348.
- Hu, J., Igarashi, A., Kamata, M., & Nakagawa, H. (2001). Angiotensin-converting enzyme degrades Alzheimer amyloid β-peptide (Aβ); retards Aβ aggregation, deposition, fibril formation; and inhibits cytotoxicity. Journal of Biological Chemistry, 276(51), 47863-47868.
- Rigat, B., Hubert, C., Alhenc-Gelas, F., Cambien, F., Cor-vol, P., & Soubrier, F. (1990). An insertion/deletion polymorphism in the angiotensin I-converting enzyme gene accounting for half the variance of serum enzyme levels. The Journal of clinical investigation, 86(4), 1343-1346.
- Narain, Y., Yip, A., Murphy, T., Brayne, C., Easton, D., Evans, J. G., & Rubinsztein, D. C. (2000). The ACE gene and Alzheimer's disease susceptibility. Journal of Medical Genetics, 37(9), 695-697.
- Brown, N. J., Blais Jr, C., Gandhi, S. K., & Adam, A. (1998). ACE insertion/deletion genotype affects bradykinin metabolism. Journal of cardiovascular pharmacology, 32(3), 373-377.
- Singh, P. K., Chen, Z. L., Ghosh, D., Strickland, S., & Norris, E. H. (2020). Increased plasma bradykinin level is associated with cognitive impairment in Alzheimer's patients. Neurobiology of disease, 139, 104833.
- Douillet, C. D., Velarde, V., Christopher, J. T., Mayfield, R. K., Trojanowska, M. E., & Jaffa, A. A. (2000). Mechanisms by which bradykinin promotes fibrosis in vascular smooth muscle cells: role of TGF-β and MAPK. American Journal of Physiology-Heart and Circulatory Physiology, 279(6), H2829-H2837.
- Bandera, E. V., Qin, B., Lin, Y., Zeinomar, N., Xu, B., Cha-numolu, D., & Hong, C. C. (2021). Association of body mass index, central obesity, and body composition with mortality among black breast cancer survivors. JAMA oncology, 7(8), 1186-1195.
- Giaquinto AN, Miller KD, Tossas KY, Winn RA, Jemal A, Siegel RL. (2022) Cancer statistics for African Amer-ican/African-American People 2022. CA Cancer J Clin 72(3):202-229
- Jorgovanovic, D., Song, M., Wang, L., & Zhang, Y. (2020). Roles of IFN-γ in tumor progression and regression: a review. Biomarker research, 8, 1-16.
- Liang J, Shang Y. (2013) Estrogen and cancer. Annu Rev Physiol. 75:225-40.
- Hicks, B. M., Filion, K. B., Yin, H., Sakr, L., Udell, J. A., & Azoulay, L. (2018). Angiotensin converting enzyme inhibitors and risk of lung cancer: population based cohort study. bmj, 363.
- Yu, H. S., Lin, T. H., & Tang, C. H. (2013). Bradykinin en- hances cell migration in human prostate cancer cells through B2 receptor/PKCδ/c-Src dependent signaling pathway. The Prostate, 73(1), 89-100.
- Smith, A. D., Refsum, H., Bottiglieri, T., Fenech, M., Hooshmand, B., McCaddon, A., & Obeid, R. (2018). Homocysteine and dementia: an international consensus statement. Journal of Alzheimer's Disease, 62(2), 561-570.
- Jang, Y., Yoon, H., Park, N. S., Rhee, M. K., & Chiriboga,D. A. (2018). Asian Americans’ concerns and plans about Alzheimer's disease: The role of exposure, literacy and cultural beliefs. Health & social care in the community, 26(2), 199-206.
- Rojo-Sebastián, A., González-Robles, C., & de Yébenes, J.G. (2020). Vitamin B6 deficiency in patients with Parkinson disease treated with levodopa/carbidopa. Clinical Neuro-pharmacology, 43(5), 151-157.
- Politis, M., & Loane, C. (2011). Serotonergic dysfunction in Parkinson's disease and its relevance to disability. TheSci-entificWorldJournal, 11, 1726-1734.
- Kennedy, D. O. (2016). B vitamins and the brain: mecha-nisms, dose and efficacy—a review. Nutrients, 8(2), 68.
- Tyagi N, Sedoris KC, Steed M. (2005) Mechanisms of homocysteine-induced oxidative stress. American Journal of physiology. Heart and Circulatory Physiology 289(6):H2649-56.
- Keskin A, U Ustun G, Aci R, Duran U. (2022) Homocysteine as a marker for predicting disease severity in patients with COVID-19. Biomark Med 16(7):559-568.
- McCaddon, A., & Regland, B. (2021). COVID-19: A meth-yl-group assault?. Medical hypotheses, 149, 110543.
- Fan X, Zhang L, Li H, Chen G, Qi G, Ma X. (2020) Role of homocysteine in the development and progression of Parkinson's disease. Ann Clin Transl Neurol 7(11):2332-2338.
- Farkas M, Keskitalo S, Smith DE, Bain N, Semmler A, In-eichen B. (2013) Hyperhomocysteinemia in Alzheimer's disease: the hen and the egg? J Alzheimers Dis 33(4):1097-104.
- Gilfix BM. (2005) Vitamin B12 and homocysteine. CMAJ173 (11):1360
- Morris MC, Schneider JA, Tangney CC. (2006) Thoughts on B-vitamins and dementia. J Alzheimers 9(4):429-33.
- Lobos, P., & Regulska-Ilow, B. (2021). Link between methyl nutrients and the DNA methylation process in the course of selected diseases in adults. Roczniki Panstwowego Zakladu Higieny, 72(2), 123-136.
- Hadtstein, F., & Vrolijk, M. (2021). Vitamin B-6-induced neuropathy: exploring the mechanisms of pyridoxine toxicity. Advances in Nutrition, 12(5), 1911-1929.
- 1Noah, L., Dye, L., Bois De Fer, B., Mazur, A., Pickering, G., & Pouteau, E. (2021). Effect of magnesium and vitamin B6 supplementation on mental health and quality of life in stressed healthy adults: Post-hoc analysis of a randomised controlled trial. Stress and Health, 37(5), 1000-1009.
- Malouf R, Grimley Evans J. (2003) The effect of vitamin B6 on cognition. Cochrane Database Syst Rev. 2003;(4):CD004393.
- Chambers P. (2022) Antioxidants and Long Covid. Open Access Library Journal 9:e9414.
- Xu, J., Patassini, S., Begley, P., Church, S., Waldvogel, H. J., Faull, R. L., & Cooper, G. J. (2020). Cerebral deficiency of vitamin B5 (d-pantothenic acid; pantothenate) as a potentially-reversible cause of neurodegeneration and dementia in sporadic Alzheimer’s disease. Biochemical and biophysical research communications, 527(3), 676-681.
- Scholefield, M., Church, S. J., Xu, J., Patassini, S., Hooper,N. M., Unwin, R. D., & Cooper, G. J. (2021). Substantively lowered levels of pantothenic acid (vitamin b5) in several regions of the human brain in Parkinson’s disease dementia. Metabolites, 11(9), 569.
- Leonardi R, Jackowski S. (2007) Biosynthesis of Pantothenic Acid and Coenzyme A. EcoSal Plus 2(2):10.1128/ ecosalplus.3.6.3.4
- Alzheimer's Association Calcium Hypothesis Workgroup, & Khachaturian, Z. S. (2017). Calcium hypothesis of Alz- heimer's disease and brain aging: a framework for integrat- ing new evidence into a comprehensive theory of pathogen- esis. Alzheimer's & Dementia, 13(2), 178-182.
- Chui, D., Chen, Z., Yu, J., Zhang, H., Wang, W., Song, Y., & Zhou, L. (2011). Magnesium in Alzheimer’s disease. Mag-nesium in the Central Nervous System.
- Surmeier, D. J., Schumacker, P. T., Guzman, J. D., Ilijic, E., Yang, B., & Zampese, E. (2017). Calcium and Parkinson's disease. Biochemical and biophysical research communications, 483(4), 1013-1019.
- Miyake, Y., Tanaka, K., Fukushima, W., Sasaki, S., Kiyo-hara, C., Tsuboi, Y., & Fukuoka Kinki Parkinson's Disease Study Group. (2011). Dietary intake of metals and risk of Parkinson's disease: a case-control study in Japan. Journal of the Neurological Sciences, 306(1-2), 98-102.
- Wang, L., Zhou, C., Yu, H., Hao, L., Ju, M., Feng, W., &Xiao, R. (2022). Vitamin D, Folic Acid and Vitamin B12 Can Reverse Vitamin D Deficiency-Induced Learning and Memory Impairment by Altering 27-Hydroxycholesterol and S-Adenosylmethionine. Nutrients, 15(1), 132.
- Chambers, P. (2022). Ca: Mg+ D, the Shield that Interdicts the Crown Viruses and Vaccines. Open Access Library Journal, 9(9), 1-24.
- Fonseca, S. C., Rivas, I., Romaguera, D., Quijal, M., Czarlewski, W., Vidal, A., & Bousquet, J. (2020). Association between consumption of fermented vegetables and COVID-19 mortality at a country level in Europe. MedRx-iv, 2020-07.
- Wastyk, H. C., Fragiadakis, G. K., Perelman, D., Dahan, D., Merrill, B. D., Feiqiao, B. Y., & Sonnenburg, J. L. (2021). Gut-microbiota-targeted diets modulate human immune status. Cell, 184(16), 4137-4153.
- Vajargah, K. T., Zargarzadeh, N., Ebrahimzadeh, A., Mousavi, S. M., Mobasheran, P., Mokhtari, P., & Milajer-di, A.(2022). Association of fruits, vegetables, and fiber intake with COVID-severity and symptoms in hospitalized patients: A cross-sectional study. Nutraceuticals for the Recovery of COVID-19 Patients.
- Kim, Y. H., Jung, K. I., & Song, C. H. (2012). Effects of serum calcium and magnesium on heart rate variability in adult women. Biological trace element research, 150, 116-122.
- Haensel, A., Mills, P. J., Nelesen, R. A., Ziegler, M. G., & Dimsdale, J. E. (2008). The relationship between heart rate variability and inflammatory markers in cardiovascular diseases. Psychoneuroendocrinology, 33(10), 1305-1312.
- Schiopu, C., Stefanescu, G., Diaconescu, S., Balan, G. G., Gimiga, N., Rusu, E., & Stefanescu, C. (2022). Magnesium Orotate and the Microbiome–Gut–Brain Axis Modulation: New Approaches in Psychological Comorbidities of Gastrointestinal Functional Disorders. Nutrients, 14(8), 1567.
- Winther, G., Jørgensen, B. M. P., Elfving, B., Nielsen,D. S., Kihl, P., Lund, S., & Wegener, G. (2015). Dietary mag- nesium deficiency alters gut microbiota and leads to depres- sive-like behaviour. Acta neuropsychiatrica, 27(3), 168-176.
- Luo, J., Zhang, C., Zhao, Q., Wu, W., Liang, X., Xiao, Z., & Ding, D. (2022). Dietary calcium and magnesium intake and risk for incident dementia: The Shanghai Aging Study. Alzheimer's & Dementia: Translational Research & Clinical Interventions, 8(1), e12362.
- Zhu, X., Borenstein, A. R., Zheng, Y., Zhang, W., Seidner,D. L., Ness, R., & Dai, Q. (2020). Ca: Mg ratio, APOE cy-tosine modifications, and cognitive function: results from a randomized trial. Journal of Alzheimer's disease, 75(1), 85-98.
- Kong, L. M., Liao, C. G., Chen, L., Yang, H. S., Zhang, S. H., Zhang, Z., & Chen, Z. N. (2011). Promoter hypomethylation up-regulates CD147 expression through increasing Sp1 binding and associates with poor prognosis in human hepatocellular carcinoma. Journal of cellular and molecular medicine, 15(6), 1415-1428.
- Liao, C. G., Liang, X. H., Ke, Y., Yao, L., Liu, M., Liu, Z.K., & Kong, L. M. (2022). Active demethylation upregulates CD147 expression promoting non-small cell lung cancer invasion and metastasis. Oncogene, 41(12), 1780-1794.
- Ehrlich M. (2009) DNA hypomethylation in cancer cells. Epigenomics 1(2):239-59.
- Kulis M, Esteller M. (2010) DNA methylation and cancer.Adv Genet 70:27-56.
- Johnson, A. A., Akman, K., Calimport, S. R., Wuttke, D., Stolzing, A., & De Magalhaes, J. P. (2012). The role of DNA methylation in aging, rejuvenation, and age-related disease. Rejuvenation research, 15(5), 483-494.
- Song, H., Yang, J., & Yu, W. (2022). Promoter Hypomethylation of TGFBR3 as a Risk Factor of Alzheimer’s disease: An Integrated Epigenomic-Transcriptomic Analysis. Frontiers in Cell and Developmental Biology, 9, 3944.
- Chouliaras, L., Kumar, G. S., Thomas, A. J., Lunnon, K., Chinnery, P. F., & O’Brien, J. T. (2020). Epigenetic regulation in the pathophysiology of Lewy body dementia. Progress in Neurobiology, 192, 101822.
- Tulloch, J., Leong, L., Chen, S., Keene, C. D., Millard, S. P., Shutes-David, A., & Tsuang, D. (2018). APOE DNA methylation is altered in Lewy body dementia. Alzheimer's & Dementia, 14(7), 889-894.
- Harhangi, B. S., De Rijk, M. C., Van Duijn, C. M., Van Bro-eckhoven, C., Hofman, A., & Breteler, M. M. B. (2000). APOE and the risk of PD with or without dementia in a population-based study. Neurology, 54(6), 1272-1276.
- Foraker J, Millard SP, Leong L, Thomson Z, Chen S, Keene CD, Bekris LM, Yu CE. (2015) The APOE Gene is Differentially Methylated in Alzheimer's Disease. J Alzheimers Dis 48(3):745-55.
- Suchy-Dicey, A., Howard, B., Longstreth Jr, W. T., Reiman,E. M., & Buchwald, D. (2022). APOE genotype, hippocampus, and cognitive markers of Alzheimer's disease in American Indians: Data from the Strong Heart Study. Alzheimer's & Dementia.
- Workinger, J. L., Doyle, R. P., & Bortz, J. (2018). Challenges in the diagnosis of magnesium status. Nutrients, 10(9), 1202.
- Nakayama, Y., Ueda, S., & Okuda, S. (2014). Molecular-mechanism underlying the renoprotective action of vitaminD. Circulation Journal, 78(3), 599-600.
- Wimalawansa, S. J. (2022). Rapidly Increasing Serum 25 (OH) D Boosts the Immune System, against Infections— Sepsis and COVID-19. Nutrients, 14(14), 2997.
- Xian X, Wang Y, Zheng J. (2021) COVID-19 and Alzhei-mer’s disease: how one crisis worsens the other. Transl Neu-rodegener 10:15.
- Bikle, D. D. (2014). Vitamin D metabolism, mechanism of action, and clinical applications. Chemistry & biology, 21(3), 319-329.
- Thye, A. Y. K., Tan, L. T. H., Law, J. W. F., Pusparajah, P., & Letchumanan, V. (2022). Long COVID-19: Psychological symptoms in COVID-19 and probiotics as an adjunct therapy. Progress In Microbes & Molecular Biology, 5(1).
- Gutiérrez-Castrellón, P., Gandara-Martí, T., Abreu Y Abreu, T., Nieto-Rufino, C. D., López-Orduña, E., Jiménez-Es- co-bar, I., & Espadaler-Mazo, J. (2022). Probiotic improves symptomatic and viral clearance in Covid19 outpatients: a randomized, quadruple-blinded, placebo-controlled trial. Gut Microbes, 14(1), 2018899.
- Yong, S. J., Tong, T., Chew, J., & Lim, W. L. (2020). An-ti-depressive mechanisms of probiotics and their therapeutic potential. Frontiers in neuroscience, 13, 1361.
- Shrestha, N. K., Burke, P. C., Nowacki, A. S., Simon, J. F., Hagen, A., & Gordon, S. M. (2022). Effectiveness of the Coronavirus Disease 2019 (COVID-19) Bivalent Vaccine. medRxiv, 2022-12.
- McCracken, I. R., Saginc, G., He, L., Huseynov, A., Daniels, A., Fletcher, S., & Randi, A. M. (2021). Lack of evidence of angiotensin-converting enzyme 2 expression and replicative infection by SARS-CoV-2 in human endothelial cells. Circulation, 143(8), 865-868.
- Ganier, C., Du-Harpur, X., Harun, N., Wan, B., Arthurs, C., Luscombe, N. M., & Lynch, M. D. (2020). CD147 (BSG)but not ACE2 expression is detectable in vascular endothe-lial cells within single cell RNA sequencing datasets derived from multiple tissues in healthy individuals. BioRxiv, 2020-05.
- Hsu, C. C., Cheng, C. H., Hsu, C. L., Lee, W. J., Huang,S. C., & Huang, Y. C. (2015). Role of vitamin B6 status on antioxidant defenses, glutathione, and related enzyme activities in mice with homocysteine-induced oxidative stress. Food & Nutrition Research, 59(1), 25702.
- Gibson, C. C., Davis, C. T., Zhu, W., Bowman-Kirigin, J. A., Walker, A. E., Tai, Z., & Li, D. Y. (2015). Dietary vitamin D and its metabolites non-genomically stabilize the endothelium. PloS one, 10(10), e0140370.
- Tsubokawa, M., Nishimura, M., Mikami, T., Ishida, M., Hisada, T., & Tamada, Y. (2022). Association of Gut Microbial Genera with Heart Rate Variability in the General Japanese Population: The Iwaki Cross-Sectional Research Study. Metabolites, 12(8), 730.
- Brilla, L., Teichler, L., Hahn, T., Freeman, J., & Li, Y. (2010). Effect of magnesium on heart rate variability in healthy subjects. The FASEB Journal, 24, 917-3.
- Wyller, V. B., Nguyen, C. B., Ludviksen, J. A., & Mollnes,T. E. (2017). Transforming growth factor beta (TGF-β) in adolescent chronic fatigue syndrome. Journal of Translational Medicine, 15(1), 1-11.
- Thanou, A., Stavrakis, S., Dyer, J. W., Munroe, M. E., James, J. A., & Merrill, J. T. (2016). Impact of heart rate variability, a marker for cardiac health, on lupus disease activity. Arthritis Research & Therapy, 18, 1-10.




