Research Article - (2023) Volume 2, Issue 1
Lipoma; A Common Neoplasm
Received Date: Apr 10, 2023 / Accepted Date: May 08, 2023 / Published Date: Aug 17, 2023
Copyright: ©©2023 Seyed Amirhossein Dormiani Tabatabaei. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Tabatabaei, S. A. D. (2023). Lipoma; A Common Neoplasm. J Surg Care, 2(1), 35-42.
Abstract
Objective: In this essay, the manifestations, possible etiologies, diagnostic approachs, treatment methods and complications of lipomas are reviewed.
Methods: A thorough review of the literature is conducted, a series of 12 patients are outlined and briefly examined.
Results: Lipomas may present as asymptomatic tumors or produce distressing signs and symptoms such as pain, swelling and overgrowth. Some lipomas may be identified by physical examination alone. However, magnetic resonance imaging (MRI) is the conclusive method of evaluation especially in deeper lesions.
Conclusions: In the absence of concerning indications or cosmetic concerns, observation stays the standard of care. When pain, itching, compression symptoms or disfigurement impress the patient, surgical removal and pathology assessment are typically recommended. Malignant transformation rarely occurs.
Keywords
Neoplasms, Lipoma, Pathology.Introduction
Lipomas are determined as a common subcutaneous neoplasms composed primarily of mature adipose (fat) cells That present as painless palpable masses [1]. In fact, these painless, slow-grow-ing masses are the most common tumors in the body encoun¬tered by clinicians [2]. In 2% to 3% of affected patients, multiple lipomas can occasionally be associated with specific disorders such as multiple hereditary lipomatosis, which has an autosomal dominant mode of inheritance; Dercum disease (adiposis dolo¬rosa), which is typically seen in obese, postmenopausal women in whom multiple painful lipomas occur mostly around the hips and thighs with a simple dominant pattern of inheritance [3,4]. There are also rare congenital syndromes that include: Proteus syndrome, Multiple endocrine neoplasia (MEN) type 1, Gardner syndrome, Cowden syndrome, Benign symmetric lipomatosis (Madelung disease) and Bannayan-Riley-Ruvalcaba syndrome (BRRS) [5-7].
Anatomical Pathology
Histologically, lipomas are soft, yellow to orange lobulat¬ed mass enclosed in a fine layer of fibrous tissue, uniform and well-cir- cumscribed. These benign tumors weighing only a few grams and usually range from 1 to >10 cm in size distributed through- out the body expected to affect the trunk and upper ex¬tremity [8]; however, occasionally can be found elsewhere ad¬ipocytes are normally present such as deeper locations of the body featuring the following: intermuscular and intramuscular lipoma, periosteal and parosteal lipoma, synovial lipomatosis and Intracranial lipoma [9].
Clinical Pathology
Although these often benign soft tissue lesions are rarely distinguishable from normal adipose tissue (Figure1), lipomas drive from mesenchymal preadipocytes instead of mature adipocytes [10, 11].
Figure 1: Histopathological examination of the specimen (haematoxylin and eosin).(A) A view of the lipoma showing adipose cells. (B) A view with mature adipose cells without atypia.
Some related benign variants of lipomas include a heteroge-neous combination of other mesenchymally derived tissues such as: angiolipoma, which composed of vast narrow vascular ruts the mature adipocytes; fibrolipoma which contain plentiful fi-brous tissue; chondroid lipoma, which contain cartilaginous elements among adipocytes; ossifying lipomas, which contains mature metaplastic bone; myolipoma which include adipocytes inter mixed with hematopoietic tissue; myxolipomas, which contain abundant mucoid mesenchymal tissues; lipoblastoma, a rapidly growing neoplasm composed of embryonal white fat with variable degrees of myxoid changes Occurs predominantly in infancy; spindle cell lipoma, which get their name from the spindle cells they contain [9, 12, 13].
Epidemiology
Lipomas represent the most common noncancerous soft-tissue tumor in humans and have a prevalence of about 1 out of every 100 persons worldwide [13]. The prevalence of multiple lipomas has been cited as 5% of patients [8]. Lipomas have a negligibly higher incidence in males compared to females [14]. Although they can arise at any age, they are typically noted in middle-aged adults especially those of the fourth to sixth decades of age and develops in all races equally [15-17].
Etiology
The exact cause of lipomas is unknown [13]. A genetic associ-ation has been demonstrated in a minority of patients, a gene association has been described on chromosome 12 in some sol-itary lipomas; In a subgroup of lipomas, spontaneous mutation in the HMGA2-LPP (located on 12q14.3) was involved in tu¬mor pathogenesis [18]. The following chromosomal aberrations have been linked to proliferation of adipocytes: 13q portion loss, 12q13-15 region, 6p21-23 region, and Other loci anomalies [19]. The incidence of lipomas is also boosted in patients with obesity, dyslipidemia, mitochondrial dysfunction and endocr-inopathies such as nodular goiter, diabetes mellitus and Cush-ing’s syndrome [20-24].
Considerable reports of long-term immunosuppression (especially corticosteroid therapy) association with the development of lipomas have been stated [25]. A massive lipoma evolution in a diabetic patient treated with chlorpropamide has also been reported [26]. Following statements postulates the mechanisms responsible for the genesis of lipomatous neoplasms following antecedent traumatic injury. A conceivable link between trauma and lipoma development which have been dubbed posttraumatic lipomas (PTLs) has been proposed, hypothesizing implicated trauma leads to the penetration of normal deep adipose tissue through a dissociated overlying septum named Scarpa's fascia at the level of a perforating vessel to shape "pseudolymphoma" [27].
It consists of typical adipose tissue in an abnormal site, and is not supposed as a true lipoma because it is not encapsulated [28, 29]. Alternatively, the local blunt trauma to the respective area also causes subsequent haematoma, lymphatic effusion, local inflammation and necrosis leading to cytokine release trigger¬ing mesenchymal precursors (preadipocytes) differentiation and development to mature adipocytes and the formation of lipomas [9,30-32]. It is also speculated that chronically minor traumas especially to the areas, in which fat tissue is found between the bone and the firm skin, may activate the development and enlargement of lipomas [33]. Metabolic regulation of adipose tissue have complex controlling aspects. Local or circulating growth factors (GH) or insulin-like growth factor (IGF), cy¬tokines, or products of degradating haematoma could initiate these differentiations [34].
A specific example of this clinical condition is the ‘lipoma ar-borescens’. This entity can be illustrated as subsynovial villous proliferation of mature adipocytes due to coexisting chronic in¬flammation of the synovium [34, 35]. Several criteria have been cited to clarify a cause-and-effect relationship: The severity of the injury to initiate reparative multiplication of cells; prior in¬tegrity of the affected part; tumor arising within the site of the injury; histologic combination of tumor consistent with underly¬ing tissue; and reasonable latent period [36-38].
Diagnosis
History and physical examination by a medical doctor are the basis of diagnosis. Lipomas oftentimes present as a soft non-ten-der mass as illustrated in Figure 2.
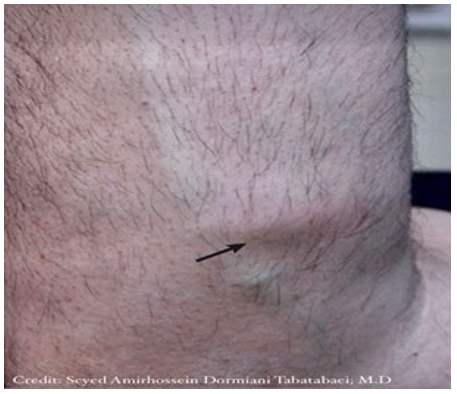
Figure 2: A 39-year-old man presented with a subcutaneous lipoma on the lateral side of the left forearm (the arrow). The mass, was mobile with respect to the over- lying skin but fixed to underlying structures.
Subcutaneous lipoma has a characteristic “doughy” feel on palpation [39]. Occasionally radiologic study may be prudent in cases to confirm the adipose nature of the lesion, include the following:
Location in deep tissues, the thigh, or retroperitoneal space, fixation to underlying tissues, pain, rapid growth and giant size. Plain radiograph shows an area of typical radiolucency known as a “water-clear density” [40]. Using ultrasound examination, a homogeneous and circumscribed hyperechoic area may be observed as shown in Figure 3 [41,42].
Figure 3: (A) A lipomas sonogram of a 40-year old man with a 4-year history of a right buttack mass. (B) lipoma sonogram of a 60-year old man with superficial abdominal mass. The arrow shows a lipoma’s hyperechoic capsule.
computed tomography (CT), exhibit non-enhancing, low atten-uation lesions with smooth edges and distinct margins [43-46]. In magnetic resonance (MR) imaging, lipomas appear as a ho-mogeneous, high-intensity signal similar to subcutaneous fat [47,48]. However, histologic examination after complete exci-sional biopsy remains definite diagnostic method [49]. It is cited that a clinicopathological correlation between the cytogenetic and the morphological subtypes of lipomatous neoplasms could serve as a significant diagnostic standards in controversial cases [35].
Treatment
Lipomas do not regressess spontaneously. Unchanging lesions often are observed without intervention. The majority of the patients desire treatment for lipomas due to cosmetic reasons [50]. However, the decision to employ surgical excision relies on considerable factors, including size of lesion (especially in cases with the size of greater than 10cm), rapid growth, location, patient comorbidities and symptoms such as pain or itching. li¬posuction is an alternative removing strategy for large lipomas [51]. Complete surgical excision of the capsule and lobules is recommended for a better outcome (Figure 4) [52].
Figure 4: The exisional biopsy of a subcutaneous lipoma located on the back of a 41-year old woman. The patient complained of swelling and itching over the upper trunk around the site of mass. Intraoperatively, the lobulated tumor was found to resect com¬pletely. Eight months after resection, there was no recurrence.
Liposuction and lipolytic injection therapies are used in the management of lipomas [53]. Employing injections of Predniso-lone (a corticosteroid agent) and isoproterenol (a β-2 adrenergic agonist agent) in combination is also a modality of treatment that induces the lipolysis and decreases superficial subcutaneous lipoma’s volume by 50% over 4 weeks in addition to the ease of post-injection surgical removal [54]. Furthermore, intralesional phosphatidylcholine and deoxycholate have been employed to shrink tiny lipomas [55, 56].
Complications
Lipomas seldom cause complications. However, the mass effect is reported in some cases, Operative complications include neu-rovascular injury, hematoma, and hemorrhage [52].
Prognosis
The prognosis is remarkable for benign lipomas and carry no risk for malignant transformation [2]. Recurrence is not expect-ed if the anatomical excision was complete [52,57]. However a 3-year follow-up is recommended. In patients with complex an¬atomical infiltration, follow-up MR imaging to monitor for local recurrence is reasonable if symptoms develop [53].
Malignant Variation
Liposarcomas Like lipomas, usually present as a small painless mass, although high-grade lesions may develop rapidly [59]. The etiology of a liposarcoma is unknown, malignant transfor- mation of lipomas is rare. MR imaging shows thick irregular septa are enhanced with gadolinium contrast [60]. Treatment of liposarcomas requires wide local excision even amputation, (neo)adjuvant chemotherapy or radiotherapy are administered [61]. The most common complications of liposarcomas are re¬currence and metastasis [62].
Clinical Case Series
To demonstrate the clinical manifestation and surveillance of lipomas, a series of 12 patients is briefly reviewed (Table 1). 8 male and 4 female patients presented at an average age of 49 years (range, 30-68). The mean tumor size was 2.3 cm × 1.4 cm × .9 cm. A miscellany of presentations was inspected that includ¬ed a lesion on the body surface. The most common presenting symptoms were the explanation of a bulk, discomfort, swelling and decreased function. Two patients complained of itching over the mass location.
Surgical incision and approach were the same. Some lipomas involved surrounding tissues such as nerves, vasculature, mus-culature, tendons, and skin, which demonstrated the possibility of these neoplasms to interfere with functions of the limbs. As a result, half of them needed significant dissection from adherent structures to provide complete excision. Ten of 12 patients returned for follow-up between 6 and 36 months. There were no recurrences.
|
Patient No. |
Age |
Sex |
Location |
Presenting symptoms |
Tissue involv-ment |
Tumor size |
Treatment approach |
Follow-up |
Recurrence |
Note |
|
1 |
60 |
M |
Lumbo-sacral |
Painless mass × 4 years |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
5.1×2.9×2 cm3 |
Surgery Longitudinal incision, simple dissection. |
36 months |
No |
- |
|
2 |
39 |
M |
Anterior ulnar aspect of forearm |
Painless mass × 4 years |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
2×1.1×.5 cm3 |
Surgery Longitudinal incision, simple dissection. |
N/A |
N/A |
Figure 2 |
|
3 |
66 |
M |
Posterior aspect of the left thigh |
Painless mass × 12 months, Itching |
Superfi-cial,Fixed To Over-lying/un-derlying Tissue |
3.5×1.7×1.1 cm3 |
Surgery Longitudinal incision, simple dissection. |
12 months |
No |
- |
|
4 |
41 |
F |
Left shoulder |
Painless mass × 16 Months |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
1.8×1×.9 cm3 |
Surgery Longitudinal incision, simple dissection. |
30 months |
No |
- |
|
5 |
40 |
M |
Left lower quadrant of abdomen |
Painless mass × 4 years |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
2.2×1.8×1.1 cm3 |
Surgery Longitudinal incision, simple dissection. |
30 months |
No |
Figure 3 (B) |
|
6 |
68 |
M |
Left Infra-scap-ular |
Painless mass × 14 months |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
2.6×2×1.1 cm3 |
Observation |
N/A |
N/A |
- |
|
7 |
41 |
F |
Posterior upper trunk |
Swelling mass × 3 Years, ItchinG |
Superfi-cial,Fixed To Over-lying/un-derlying Tissue |
3×1.6×1.3 cm3 |
Surgery Longitudinal incision, simple dissection. |
8 months |
No |
Figure 4 |
|
8 |
54 |
M |
dorsal radial aspect of forearm |
Painless mass ×14months, itching |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
1.4×1.1×.5 cm3 |
Surgery Longitudinal incision, simple dissection. |
N/A |
N/A |
- |
|
9 |
56 |
M |
Multiple masses |
Painless mass × 6 years |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
8 masses observed, the largest one Was 2.6×1.7×.5 cm3 |
Observation |
26 months |
No changes was noticed |
Unremarkable family history |
|
10 |
33 |
F |
Anterior aspect of left thigh |
Painless mass × over 10 years |
Superfi-cial,Fixed To underlying Tissue, fixed to overlying skin |
3.4×2.4×1 cm3 |
Surgery Longitudinal incision, simple dissection. |
6 months |
No |
- |
|
11 |
54 |
M |
Medial aspects of right and left arms |
Painless mass × 2 years |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
1.1×.9×.5 cm3 And 2.1×1.3×.5 cm3 |
Observation |
10 months |
No |
- |
|
12 |
30 |
F |
Lateral aspect of left flank |
Painless mass × 6 Months |
Superfi-cial,Fixed To underlying Tissue, not fixed to overlying skin |
.9×.5×.- cm3 |
Observation, Changed to surgery |
18 months |
Changed to1.4×.9×.6 cm3 and another 1×.5×.- cm3 Lesion detected |
Pathological assessment Reported benign lipomas |
*F, female; M, male; L, left; R, right; N/A, not availabale
Table 1: Clinical Manifestation and Surveillance of Lipomas
Summary
This article summarizes the present understanding of the patho-physiology, modalities of diagnosis/treatment, and complica¬tions of lipomatous neoplasms. Lipomas stay a common benign tumor of the body. Superficial lipomas may be diagnosed by physical examination unaided. For more complex situations, Magnetic resonance imaging (MRI) is most beneficial for diag¬nosis and treatment planning. Most lipomas are approached by surgical excision with few complications or reproduction. Ma¬lignant permutation seems rare.
Declaration
The author would like to thank all patients who participated in this study. Conceptualization, investigation, methodolo- gy, project administration, resources, supervision, validation, visu¬alization, and writing-review and editing were done by the cor¬responding author.
Ethical Approval
This study was registered and approved by the local Ethics Committee of Islamic azad university, Najafabad branch and all participants gave their informed consent.
Conflicts of Interests
Author certifies that has no affiliations with or involvement in any organization or entity with any fi- nancial interest or non-fi-nancial interest in the subject matter or materials discussed in this manuscript.
Funding
None.
References
- Kosztyuova T, Shim TN. Rapidly enlarging lipoma. BMJ Case Rep. 2017 Sep 23;2017.
- Johnson CN, Ha AS, Chen E, Davidson D. Lipomatous Soft-tissue Tumors. J Am Acad Orthop Surg. 2018 Nov 15;26(22):779-788.
- McFarland GB, Jr, Hoffer MM. Paralysis of the intrinsic muscles of the hand secondary to lipoma in Guyon’s tunnel. J Bone Joint Surg Am. 1971;53(2):375-6.
- Brodovsky S, Westreich M, Leibowitz A, Schwartz Y. Adi-posis dolorosa (Dercum’s disease): 10-year follow-up. Ann Plast Surg. 1994;33(6):664-8.
- Vásquez Elera L, Guzman Rojas P, Sánchez Herrera M, Prado V, García Encinas C, Bravo Paredes EA, Bussalleu A. [Familiar adenomatous polyposis: report of 2 cases]. Rev Gastroenterol Peru. 2018 Jan-Mar;38(1):78-81.
- Baldino ME, Koth VS, Silva DN, Figueiredo MA, Salum FG, Cherubini K. Gardner syndrome with maxillofacial manifestation: A case report. Spec Care Dentist. 2019 Jan;39(1):65-71.
- Barisella M, Giannini L, Piazza C. From head and neck lipoma to liposarcoma: a wide spectrum of differential diagnoses and their therapeutic implications. Curr Opin Otolar-yngol Head Neck Surg. 2020 Apr;28(2):136-143.
- Creytens D. A contemporary review of myxoid adipocytic tumors. Semin Diagn Pathol. 2019 Mar;36(2):129-141.
- Charifa A, Azmat CE, Badri T. Lipoma Pathology. In: Stat-Pearls. Treasure Island (FL): StatPearls Publishing; December 5, 2022.
- Weiss SW, Goldblum JR. Benign Lipomatous Tumors. In: Enzinger FM, Weiss SW, eds. Soft Tissue Tumors. 3rd ed. St Louis, Mo: Mosby; 1995:381-430.
- Phalen GS, Kendrick JI, Rodriguez JM. Lipomas of the upper extremity: a series of fifteen tumors in the hand and wrist and six tumors causing nerve compression. Am J Surg. 1971;121(3):298-306.
- Nadar MM, Bartoli CR, Kasdan ML. Lipomas of the hand: a review and 13 patient case series. Eplasty. 2010;10:e66.
- Ameloot E, Cordier F, Van Dorpe J, Creytens D. Update of Pediatric Lipomatous Lesions: A Clinicopathological, Immunohistochemical and Molecular Overview. J Clin Med. 2022;11(7):1938.
- Rydholm A, Berg NO (December 1983). "Size, site and clinical incidence of lipoma. Factors in the differential diagnosis of lipoma and sarcoma". Acta Orthop Scand. 54 (6): 929–34.
- Salam GA (March 2002). "Lipoma excision". Am Fam Physician. 65(5): 901–4.
- Froimson AI. Benign solid tumors. Hand Clin. 1987;3(2):213-7.
- Phalen GS, Kendrick JI, Rodriguez JM. Lipomas of the upper extremity: a series of fifteen tumors in the hand and wrist and six tumors causing nerve compression. Am J Surg. 1971;121(3):298-306.
- Panagopoulos I, Gorunova L, Agostini A, Lobmaier I, Bjerkehagen B, Heim S. Fusion of the HMGA2 and C9orf92 genes in myolipoma with t(9;12)(p22;q14). Diagn Pathol.2016 Feb 09;11:22.
- Chrisinger JSA. Update on Lipomatous Tumors with Emphasis on Emerging Entities, Unusual Anatomic Sites, and Variant Histologic Patterns. Surg Pathol Clin. 2019 Mar;12(1):21-33.
- Chrisinger JSA. Update on Lipomatous Tumors with Emphasis on Emerging Entities, Unusual Anatomic Sites, and Variant Histologic Patterns. Surg Pathol Clin. 2019 Mar;12(1):21-33.
- Berkovic SF, Andermann F, Shoubridge EA, et al. Mitochondrial dysfunction in multiple symmetrical lipomatosis. Ann Neurol. 1991;29(5):566-9.
- Leiva SF, Navachia D, Nigro N, Ibarra R, Cresto JC. Lipoma in the thyroid? J Pediatr Endocrinol Me-tab.2004;17(7):1013-5.
- Weinberg T, Feldman M, Sr. Lipomas of the gastrointestinal tract. Am J Clin Pathol. 1955;25(3):272-81.
- Miyake O, Hara T, Matsumiya K, Oka T, Takaha M, KurataA. [Adrenal myelolipoma associated with Cushing’s syn-drome: a case report]. Hinyokika Kiyo. 1992;38(6):4-681.
- Clayman E, King K, Harrington MA. Corticosteroid-Asso-ciated Angiolipomatosis. Eplasty. 2017;17:ic9. Published 2017 Apr 6.
- Garfinkel HA. A massive lipoma in a patient receiving chlorpropamide therapy. Postgrad Med J. 1971;47(544):137-8
- Brooke RI, MacGregor AJ. Traumatic pseudolipoma of the buccal mucosa. Oral Surg Oral Med Oral Pathol. 1969;28(2):223-225.
- Meggitt BF, Wilson JN: The battered buttock syndrome: fat fractures: a report on a group of traumatic lipomata. Br J Surg. 1972, 59: 165-169.
- Herbert DC, DeGeus J: Post-traumatic lipomas of the abdominal wall. Br J Plast Surg. 1975, 28: 303-306.
- Zilefac B, Kharim L, Achumbom H, Fai K. Post-Traumatic Scalp Lipoma: a Case Report. Health Sci. Dis. [Internet]. 2023 Mar. 30; 24(4).
- Aust MC, Spies M, Kall S, Jokuszies A, Gohritz A, Vogt P. Posttraumatic lipoma: fact or fiction? Skinmed. 2007 Nov-Dec;6(6):266-70.
- Signorini M, Campiglio GL: Posttraumatic lipomas: where do they really come from?. Plast Reconstr Surg. 1998, 101: 699-705.
- Copcu E. Sport-induced lipoma. Int J Sports Med. 2004;25(3):182-185.
- Copcu E. Can intramuscular lipoma have a postâ?traumatic origin. British Journal of Dermatology. 2003;149(5): 1084–1085
- Tiao WM, Yeh LR, Liang HL et al. Lipoma arborescens of the knee: a case report. J Formos Med Assoc 2001; 100: 412–15.
- Cohen S, Ad-El D, Benjaminov O, Gutman H. Post-trau-matic soft tissue tumors: case report and review of the literature a propos a post-traumatic paraspinal desmoid tumor. World J Surg Oncol. 2008;6:28. Published 2008 Feb 29.
- Ewing J: Buckley lecture: Modern attitude toward traumatic cancer. Arch Pathol. 1935, 19: 690.
- Warren S: Minimal criteria required to improve causation of traumatic or occupational neoplasms. Ann Surg. 1943, 117: 585-10.
- Posch JL. Tumors of the hand. J Bone Joint Surg Am. 1956;38-A(3):517-39; discussion, 539–540; passim.
- Leffert RD. Lipomas of the upper extremity. J Bone Joint Surg Am. 1972;54(6):1262-6.
- James JJ, Robin A, Wilson M, Evans AJ. The Breast. In: Adam A, Dixon AK, eds. Grainger & Allison’s Diagnostic Radiology. 5th ed. Philadelphia, Pa: Elsevier; 2008.
- Pant R, Poh AC, Hwang SG. An unusual case of an intramuscular lipoma of the pectoralis major muscle simulating a malignant breast mass. Ann Acad Med Singapore. 2005;34(3):275-6.
- Weekes RG, McLeod RA, Reiman HM, Pritchard DJ. CT of soft-tissue neoplasms. AJR Am J Roentgenol. 1985;144(2):355-60.
- Egund N, Ekelund L, Sako M, Persson B. CT of soft-tissuetumors. AJR Am J Roentgenol. 1981;137(4):725- 9.
- Halldorsdottir A, Ekelund L, Rydholm A. CT-diagnosis of lipomatous tumors of the soft tissues. Arch Orthop Trauma Surg. 1982;100(4):211-6.
- Dooms GC, Hricak H, Sollitto RA, Higgins CB. Lipomatous tumors and tumors with fatty component: MR imaging potential and comparison of MR and CT results. Radiology. 1985;157(2):479-83.
- Lee YH, Jung JM, Baek GH, Chung MS. Intramuscular lipoma in thenar or hypothenar muscles. Hand Surg. 2004;9(1):49-54.
- Capelastegui A, Astigarraga E, Fernandez-Canton G, Sara-legui I, Larena JA, Merino A. Masses and pseudomasses of the hand and wrist: MR findings in 134 cases. Skeletal Radiol. 1999;28(9):498-507.
- Lichon S, Khachemoune A. Clinical presentation, diagnostic approach, and treatment of hand lipomas: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Sep;27(3):137-139.
- Lichon S, Khachemoune A. Clinical presentation, diagnostic approach, and treatment of hand lipomas: a review. Acta Dermatovenerol Alp Pannonica Adriat. 2018 Sep;27(3):137-139.
- Datir A, James SL, Ali K, Lee J, Ahmad M, Saifuddin A. MRI of soft-tissue masses: the relationship between lesion size, depth, and diagnosis. Clin Radiol. 2008 Apr;63(4):373-8; discussion 379-80.
- Bancroft LW, Kransdorf MJ, Peterson JJ, O’Connor MI. Benign fatty tumors: classification, clinical course, imaging appearance, and treatment. Skeletal Radiol. 2006;35(10):719-33.
- Amber KT, Ovadia S, Camacho I. Injection therapy for the management of superficial subcutaneous lipomas. J Clin Aesthet Dermatol. 2014;7(6):46-48.
- Redman LM, Moro C, Dobak J, Yu Y, Guillot TS, Greenway FL. Association of β-2 adrenergic agonist and corticosteroid injection in the treatment of lipomas. Diabetes Obes Metab. 2011;13(6):517-522.
- Kopera D, Binder B, Toplak H. Intralesional lipolysis with phosphatidylcholine for the treatment of lipomas: pilot study. Arch Dermatol. 2006;142(3):395-6.
- Rotunda AM, Ablon G, Kolodney MS. Lipomas treated with subcutaneous deoxycholate injections. J AmAcad Dermatol. 2005;53(6):973-8.
- Drylewicz MR, Lubner MG, Pickhardt PJ, Menias CO, Mellnick VM. Fatty masses of the abdomen and pelvis and their complications. Abdom Radiol (NY). 2019 Apr;44(4):1535-1553.
- Sawhney KK, McDonald JM, Jaffe HW. Liposarcoma of the hand. Am Surg. 1975;41(2):117-20.
- Sampson CC, Saunders EH, Green WE, Laurey JR. Liposarcoma developing in a lipoma. Arch Pathol. 1960;69:506-10.
- MJ, Naqvi AH, Cohen HE, Damron TA. Predictive value of gadolinium enhancement in differentiating ALT/WD liposarcomas from benign fatty tumors. Skeletal Radiol. 2005;34(5):272-8.
- Mack LA, Crowe PJ, Yang JL, et al. Preoperative chemora-diotherapy (modified Eilber protocol) provides maximum local control and minimal morbidity in patients with soft tissue sarcoma. Ann Surg Oncol.2005;12(8):646-53.
- Loubignac F, Bourtoul C, Chapel F. Myxoid liposarcoma: a rare soft-tissue tumor with a misleading benign appearance. World J Surg Oncol. 2009;7:42.