Research Article - (2024) Volume 9, Issue 2
Is Sentinel Lymph Node Tumor Burden a Prognostic Factor for Disease-Free Survival in Patients with Melanoma of the Trunk and Extremities?
2Specialist in Oncological Pathology, Functional Unit of Pathology and Molecular Biology, Instituto Nacional de Cancerologia, Bogota, Colombia
3Medical Surgeon Specialist in Psychiatry, MA in Clinical Epidemiology, and Specialist in Statistics, Universidad Nacional de Colombia and Instituto Nacional de Cancerologia, Bogota, Colombia
4Specialist in Oncological Surgery, Functional Unit for Breast and Soft Tissue Tumors, Instituto Nacional de Cancerologia, Bogota, Colombia
Received Date: Mar 07, 2024 / Accepted Date: Apr 19, 2024 / Published Date: Apr 30, 2024
Copyright: ©Ã?©2024 Raul Suarez RodriÂguez, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Lehmann-Mosquera, C., Suarez-Rodriguez, R., Lopez-Correa, P., Sanchez, R., Morales, S. D., et al. (2024). Is Sentinel Lymph Node Tumor Burden a Prognostic Factor for Disease-Free Survival in Patients with Melanoma of the Trunk and Extremities. Int J Cancer Res Ther, 9(1), 01-10.
Abstract
Introduction Sentinel lymph node (SLN) involvement in patients with melanoma is the most important prognostic factor and is decisive for the indication of adjuvant systemic therapy. Scientific evidence shows that SLN tumor burden is a prognostic factor for disease- free survival.
Methods A descriptive observational historical cohort study was conducted, which included patients diagnosed with melanoma of the trunk and extremities who underwent SLN biopsy in the Functional Unit for Breast and Soft Tissue Tumors at the Instituto Nacional de Cancerología (Bogotá, Colombia). The clinical characteristics were described, with a review by pathology of the size of lymph node metastasis and extracapsular extension in the histological plate of each included patient. The analyses were performed with the statistical program Stata 16®.
Results The study identified 195 patients diagnosed with melanoma who underwent SLN biopsy; 71 of them (36%) had metastatic involvement, and 62% (n=44) were acral lentiginous melanoma (ALM). When applying the Rotterdam criteria, no statistically significant difference was found in overall survival (Log-rank test, χ 2(2) = 2.35; p=0.3), but there was a marginal difference in disease-free survival (Log-rank test, χ 2(2) =5.97; p=0.0505) for the group with the highest tumor burden.
Conclusions Metastatic SLN involvement greater than 1 mm is associated with lower diseasefree survival. SLN tumor burden could be used as a clinically relevant prognostic factor for patients with a medical decision to skip lymphadenectomy, given that it predicts additional nodal involvement and the indication for adjuvant immunotherapy.
Keywords
Melanoma, Tumor Burden, Sentinel Lymph Node, Lymph Node Dissections
Introduction
Melanoma is a malignant tumor originating in melanocytes. It accounts for only 4% of all malignant skin tumors but responsible for 80% of all skin cancer deaths. In recent decades, its incidence has considerably increased globally and become a public health problem in places like Australia. According to the World Health Organization Globocan 2020 database, the highest incidence rate is in Australia and New Zealand, with 35.8 cases per 100,000 inhabitants, compared to 3.4 cases per 100,000 inhabitants worldwide. South America ranks tenth with 2.9 cases per 100,000 inhabitants, equivalent to 16,000 new cases, with an estimated mortality of more than 4,000 cases per year [1]. In Colombia, melanoma accounts for 1.6% of all tumors, with 1,805 new cases by 2020 and more than 450 deaths from this cause. Epidemiological information on the incidence of melanoma in the country is limited; however, according to data from the statistical yearbook of the Instituto Nacional de Cancerologia (INC) (Bogota, Colombia), 28 deaths from cutaneous melanoma were recorded in 2020 [3].
Sentinel lymph node (SLN) involvement in patients with melanoma is the most important prognostic factor and is decisive for the indication of adjuvant systemic therapy [4]. The Rotterdam criteria use the linear measurement of the maximum diameter of the largest focus of micrometastatic disease in the SLN, with categorical cut-off points of 0.1 mm and 1.0 mm. With these cut-off points, in 2008, Van der Ploeg et al identified three groups of patients with lymph node involvement who, according to tumor burden, had different prognoses in terms of disease-specific survival [2].
Some clinical trials have already implemented microscopic SLN tumor burden as an inclusion criterion. The European Organization for Research and Treatment of Cancer (EORTC) Study 18071 used adjuvant ipilimumab in stage III melanoma, whereas the COMBI-AD trial used dabrafenib plus trametinib in stage III melanoma. In these two trials, patients with a single positive SLN and a microscopic tumor burden >1 mm in diameter had a worse prognosis compared to those with <1 mm [9]. Based on currently available evidence, the American Joint Committee on Cancer (AJCC) Melanoma Expert Panel recommends that, at a minimum, the largest dimension (measured in millimeters using an ocular micrometer) of the largest tumor focus should be recorded in pathology reports according to the Rotterdam criteria [5]. Pivotal adjuvant studies with nivolumab, pembrolizumab, and dabrafenib/ trametinib included only patients who had undergone SLN biopsy; if this was positive, patients were submitted to complete lymphadenectomy. However, the DeCOG-SLT and MSLT-II clinical trials demonstrated that complete lymphadenectomy in SLN-positive patients had no impact on melanoma-specific survival [6,13].
Scientific evidence suggests that SLN tumor burden is an independent prognostic factor for overall survival, diseasefree recurrence, and the presence of nonsentinel lymph node metastases; nevertheless, the microscopic classifications used in different study groups are variable, as are the outcomes reported [7].
In Latin America, there are no reports on studies evaluating the clinical characteristics of SNL tumor burden according to the Rotterdam criteria in the prognosis of patients with melanoma of the trunk and extremities. The European and North American series in which these prognostic variables are evaluated have very little representation of patients with acral lentiginous melanoma (ALM), given their low incidence [8]. In Colombia, ALM is the most frequently diagnosed histological type; for this reason, this study may help better understand its biology and the prognostic significance of SLN tumor burden as a predictor for therapeutic decision-making in patients with melanoma.
Methodology
A descriptive observational historical cohort study was conducted, including patients diagnosed with melanoma of the trunk and extremities who underwent SLN biopsy, with surgical management of the primary tumor and follow-up in the Functional Unit for Breast and Soft Tissue Tumors at the INC between January 2011 and December 2018, whose pathology report showed metastatic SLN involvement. A search of the medical records of patients with melanoma was performed in the institutional SIAI database and the sentinel lymph node database of the Functional Unit of Nuclear Medicine, finding 1,144 patients who underwent SLN biopsy at the INC; 949 (82.9%) of them corresponded to patients diagnosed with breast cancer, squamous cell carcinoma, and gynecological and urological oncological pathologies, due to which they were excluded from the study. A total of 195 patients diagnosed with melanoma were found who underwent SLN biopsy. Of this group, 123 (62%) had a pathological anatomy report without metastatic SLN involvement, so they were excluded from the study. Finally, 71 patients (36%) had pathology reports with metastatic SLN involvement, meeting this inclusion criterion (Figure 1).
Data relevant to the study were recorded in the Research Electronic Data Capture (REDCap) platform. Information related to sociodemographic, clinical, and histological variables, treatments administered, oncologic outcomes for recurrence, and survival was collected.
To determine SLN tumor burden, the Functional Unit of Pathology at the INC performed a new review of the histological plate of the SLN to measure the largest diameter of SLN metastasis and evaluate the presence or absence of extracapsular extension.
Statistical analyses were performed using the statistical program Stata 16®. The study incorporated an analytical component that considered disease-free survival (DFS), melanoma-specific survival (MSS), and overall survival (OS) as outcomes. DFS was defined as the time between surgery and the date of diagnosis of relapse, MSS was defined as the time from confirmed diagnosis to death from the disease, and OS was defined as the time from confirmed melanoma diagnosis to death from any cause.
Figure 1: Selection of Study Patients
Results
Of the 71 patients included in the study, 45.1% (n=32) were male. The mean age at diagnosis was 62.2 years (SD 18.95). Most tumors were acral lentiginous melanoma (ALM) (61.9%, n=44);47.8% (n=34) of the patients had melanomas with ulceration, 34%(n=24) corresponded to T3 tumors. Most patients were in clinical stage IIB before SLN biopsy (28.1%, n=20), and the most frequent tumor location was on the foot (43.6%, n=31) (Table 1).
|
Characteristics |
n (%) |
|
Age |
62.2 |
|
Sex |
|
|
Men |
32 (45.1) |
|
Women |
39 (54.9) |
|
Histological type |
|
|
Acral lentiginous |
44 (61.9) |
|
Surface extension |
11 (15.4) |
|
Nodular |
9 (12.6) |
|
No data |
7 (9.8) |
|
Tumor location |
|
|
Trunk |
11 (15.4) |
|
Arm |
4 (5.6) |
|
Forearm |
1 (1.4) |
|
Fingers |
6 (8.4) |
|
Leg |
2 (2.8) |
|
Foot |
31 (43.6) |
|
Toes |
16 (22.5) |
|
Breslow |
|
|
<1 mm |
6 (8.4) |
|
1-2 mm |
21 (29.5) |
|
2.1-4 mm |
24 (33.8) |
|
>4 mm |
10 (14.1) |
|
No data |
10 (14.1) |
|
Clark's Level |
|
|
II |
2 (3) |
|
III |
7 (9.8) |
|
IV |
43 (60.5) |
|
V |
7 (9.8) |
|
No data |
12 (16.9) |
|
Ulceration |
|
|
Yes |
34 (47.8) |
|
No |
24 (33.8) |
|
No data |
13 (18.3) |
|
Number of mitoses (per mm2) |
|
|
1-3 |
47 (66.1) |
|
>4 |
10 (14.1) |
|
No data |
14 (19.7) |
|
Clinical stage pre-SLN biopsy |
|
|
AI |
6 (8.4) |
|
IB |
18 (25.3) |
|
IIA |
15 (21.1) |
|
IIB |
20 (28.1) |
|
IIC |
12 (16.9) |
Table 1: Clinicopathological Characteristics of Patients in the Cohort
The most frequently performed surgical procedure was wide local resection in 67.6% (n=48) of cases, and the marking technique used for SLN identification was lymphoscintigraphy in 100% (n=71) of patients. An average of 1.8 SLNs were resected. Pathology reports showed that the majority of patients (81.6%, n=58) had metastatic SLN involvement >1 mm, and extracapsular extension was evidenced only in 2.8% (n=2) of cases.
Surgical management of the involved lymph node chain was performed in 91.5% (n=65) of the patients, with the most frequent surgery being inguinal-iliac lymphadenectomy. On average, 23 lymph nodes were resected in the lymphadenectomy, with a mean of 2.4 positive nodes; 42.2% (n=30) of patients received adjuvant systemic therapy, with interferon being the most used drug (39.4%, n=28) given the time of inclusion of patients in the study (2011-2018). Only 14.1% (n=10) of the patients received adjuvant radiation therapy on the involved nodal chain. Disease progression was documented in 50.7% (n=36) of patients, most with distant metastases (88%, n=32), with lung involvement being the most frequent in 78% (n=25) of the cases, followed by liver involvement in 16% (n=5) and distant lymph node involvement in 12% (n=4) (Table 2).
|
Type of treatment |
n (%) |
|
Surgical treatment of the primary tumor |
|
|
Wide local resection + SLN |
48 (67.6) |
|
Minor amputation + SLN |
23 (32.3) |
|
Sentinel lymph node technique Lymphoscintigraphy |
71 (100) |
|
Resection borders of the primary tumor |
|
|
Negative |
68 (95.7) |
|
Positive |
2 (2.8) |
|
No data |
1 (1.4) |
|
Size of SLN metastasis |
|
|
<0.1 mm |
2 (2.8) |
|
0.1-1 mm |
11 (15.5) |
|
>1 mm |
58 (81.6) |
|
Number of positive SLNs |
|
|
1 |
36 (50.7) |
|
2-3 |
29 (40.8) |
|
>4 |
6 (8.4) |
|
Extracapsular extension |
|
|
Yes |
2 (2.8) |
|
No |
69 (97.1) |
|
Surgical treatment of lymph node chains Axillary |
|
|
lymphadenectomy |
19 (26.7) |
|
Inguinal-femoral lymphadenectomy |
12 (16.9) |
|
Inguinal-iliac lymphadenectomy |
30 (42.2) |
|
Popliteal lymphadenectomy |
4 (5.6) |
|
No data |
3 (4.2) |
|
Number of lymph nodes resected in lymphadenectomy 1 2-3 >4 |
1 (1.4) |
|
1 (1.4) |
|
|
63 (88.7) |
|
|
Number of positive lymph nodes in |
|
|
lymphadenectomy |
35 (49.2) |
|
Negative |
8 (11.2) |
|
1 |
7 (9.8) |
|
2-3 |
16 (22.5) |
|
>4 |
3 (4.2) |
|
No data |
|
|
Adjuvant systemic therapy |
|
|
Interferon |
28 (39.4) |
|
Immunotherapy Ipilimumab |
|
|
|
2 (2.8) |
|
Adjuvant radiation therapy to the lymph node chain |
|
|
Yes |
|
|
No |
10 (14.1) |
|
No data |
55 (77.4) |
|
|
6 (8.4) |
Table 2: Type of Treatment Administered to Patients and Final Anatomic Pathology Report of Patients in the Cohort
The histological characteristics were analyzed in relation to SLN tumor burden according to the Rotterdam criteria without finding an association with Clark's level (Pearson chi2 (6) = 9.6243, Pr=0.141), Breslow level (Pearson chi2 (6) =5.7166, Pr=0.456), number of mitoses (Pearson chi2 (4) = 4.5841, Pr=0.333) and with the presence or absence of ulceration (Pearson chi2 (2) = 3.3236, Pr=0.190).
Regarding the number of positive lymph nodes in lymphadenectomy, no association was found with SLN tumor burden according to Rotterdam criteria (Kruskal-Wallis test, X2=2.83; p=0.243). Nevertheless, a higher number of positive lymph nodes was evidenced when analyzing patients with tumor burden >1 mm (56%, n=29). The presence of extracapsular extension did not show an association with a higher number of positive lymph nodes in lymphadenectomy (Kruskal-Wallis test, X2=0.024; p=0.88).
Survival Analysis
Disease-free survival was 35.7 months, where 36 events were recorded, with an incidence rate of 1.8 relapses per 100 patients per month (CI95%: 1.3-2.5), which provided 1,991 months of follow-up with a median of 17.8 months (Figure 2A). There were 32 distant relapse events with a survival of 37.4 months and an incidence rate of 1.53 relapses per 100 patients per month (CI95%: 1.1-2.2).
Overall survival was 115.7 months with an incidence rate of 0.8 deaths per 100 patients per month (CI95%: 0.55-1.25), where 22 deaths were recorded in 71 patients, which contributed 2,672 months of follow-up with a median of 30.1 months (Figure 2B).
Melanoma-specific survival was 115 months, with an incidence rate of 0.75 deaths per 100 patients per month (CI95%: 0.48-1.16), with 20 events, where patients in the sample provided 2,670 months of follow-up with a median of 30.1 months (Figure 2C).
Based on SLN tumor burden according to the Rotterdam criteria, a DFS with marginal significance was reported (Log-rank test, X2 (2) = 5.97; p=0.0505), with an incidence rate of 1.2 events for the tumor burden group of <0.1 mm (CI95%: 0.17-8.6), 0.53 events in the 0.1 to 1 mm group (CI95%: 0.13-2.1), and 2 events per 100 patients per month for the >1 mm group (CI95%: 1.4 to 2.9) (Figure 3).
For OS by SLN tumor burden, no statistically significant difference was found (Log-rank test, X2 (2) = 2.35; p=0.3), with an incidence rate of 0.4 deaths (CI95%: 0.11-1.85) per 100 patients per month in the 0.1 to 1 mm group, 0.9 deaths (CI95%: 0.60-1.44) per 100 patients per month in the >1 mm group, and no events were reported in the <0.1 mm group (Figure 4).
Figure 2: Cancer Outcomes in Cohort Patients
Figure 3: Disease-Free Survival in Relation to SLN Tumor Burden (Rotterdam Criteria) in Patients of the Cohort
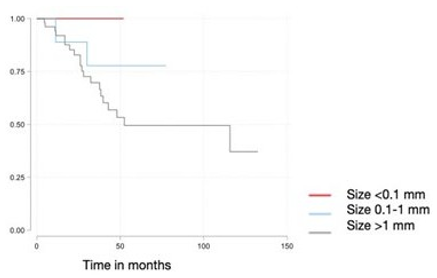
Figure 4: Overall Survival in Relation to SLN Tumor Burden (Rotterdam Criteria) in Patients of the Cohort
Discussion
Radical surgical treatment of lymph node chains in patients with melanoma has been recommended since the beginning of the last century, based on the hypothesis that greater local and regional surgical radicality improved the possibility of curing these patients. However, this radical nodal therapy was questioned due to high secondary morbidity and poor evidence of the impact on patient OS [6,13]. In the 1990s, with the implementation of the use of SLN in melanoma by Morton and later with the realization of several clinical trials in patients without clinical involvement of the lymph node chains with melanoma, SLN was positioned as the standard of management of these patients, reducing morbidity and providing relevant prognostic information without a decrease in overall or disease-specific survival [31]. Thus, SLN became the most important prognostic factor in patients with non-metastatic melanoma.
Preliminary results of the German Dermatologic Cooperative Oncology Group-SLT trial were recently published, with a median follow-up of 3 years [6]. Patients with melanoma who had positive SLN were assigned to lymphadenectomy vs. observation, finding no difference in OS between the two groups. Results from the prospective MSLT-II study also demonstrated that immediate lymphadenectomy did not increase melanoma-specific survival among patients with SLN metastasis, thus questioning the recommendation for lymphadenectomy after positive SLN [13]. An analysis of the Surveillance, Epidemiology, and End Results (SEER) database showed that, even before the publication of the MLST-II trial results, only 50% of SLN-positive patients underwent lymphadenectomy in the United States [22].
Acral lentiginous melanoma is the most frequent variant of melanoma in Asian, Afro-descendant, and Latin American populations, in contrast to the white population, where only 5-10% of all diagnosed cases correspond to this histological type [12]. Resulting from this distribution in pivotal SLN research, acral melanomas are a low percentage in these studies. In the MD Anderson Cancer Center series, ALM accounted for only 10% of patients, as the main histological type in this and other studies corresponds to superficial spreading melanoma (SSM), ranging from 60 to 70% [6,11,13]. ALM does not seem to be associated with the risk factors originally described for malignant melanoma, such as a history of atypical nevi (only less than 10% have been associated with this history), family history of melanoma, or chronic sun exposure. In the context of ALM, different factors have been related, such as biological conditions (race, age, sex, number of pregnancies), genetics, smoking, and history of trauma in the area of tumor appearance [23-25].
In the present cohort, ALM represented the majority of cases, with 62% (n=44) of the total, while SSM was diagnosed only in 15% (n=11) of patients. In the series reported in Latin America, ALM has a similar presentation in countries with mixedrace and/or Indo-American populations, with 44.1% of cutaneous melanoma cases in Mexico (16) and 61.2% in Peru (17).
Consistent with what has already been mentioned, in 2019, Namikawa et al. showed that patients with SLN tumor burden >1.0 mm had a worse DFS compared to patients with SLN tumor burden <1.0 mm (p=0.018) [11]. When MSS was analyzed, it was closer to statistical significance (p=0.08) and lower in patients with higher tumor burden. These results are comparable with the present study in terms of DFS, where a higher tumor burden demonstrated worse DFS. In addition, interferon was also the adjuvant treatment in most patients. In Europe, the EORTC 18991 study was conducted, which explored the use of pegylated interferon alpha for several years against observation, being the first immunomodulator with positive results for oncologic outcomes in melanoma [28]. However, the modest impact on OS, its high toxicity and cost, and the advent of targeted therapies and immunotherapy made it fall into disuse. For the data collection period for this cohort, the adjuvant therapy of choice was interferon. Currently, the adjuvant therapy options in Colombia are targeted therapy with dabrafenib plus trametinib in patients with BRAF V600 mutation and immunotherapy with nivolumab, which has been approved since 2016 for use in patients with melanoma [29].
Different studies have established the importance of SLN tumor burden parameters and how this provided an accurate prognostic stratification, regardless of the outcome of lymphadenectomy. In 2008, Eggermont et al. demonstrated that patients with micrometastases of 0.1 mm had the same prognosis as patients with negative SLN, making it safe to omit lymphadenectomy in this group [9]. Subsequently, in 2009, the EORTC melanoma Group ratified the safety of establishing tumor burden based on the Rotterdam criteria, which use the linear measurement of the maximum diameter of the largest focus of micrometastatic disease in SLN, using categorical cut-off points of 0.1 mm and 1.0 mm [20]. On the other hand, the Starz classification measures the depth of invasion from the lymph node capsule, thus stratifying patients at risk of death and non-sentinel lymph node metastasis. The Dewar criteria classify micrometastases according to their microanatomical location within the lymph node to determine the risk of non-sentinel lymph node metastasis. Given the heterogeneity of the classification systems, a comparison between them concluded that a single measurement of the maximum linear diameter of the largest focus of melanoma micrometastases in the SLN is the most consistent, and, in turn, it as a predictor of non-sentinel lymph node status, DSF, and OS [4].
Previous studies have shown that 70 to 80% of patients with positive SLN have only that one positive node [21]. In our cohort, in 49% (n=35) of patients, the only positive node was SLN, with no additional involvement of other lymph nodes in lymphadenectomy. Thus, according to the 8th edition of the AJCC, for this study, the most frequent stage was IIIA (51%, n=36) [5].
The initiation of adjuvant therapy is indicated with an SLN tumor burden >1 mm. Nevertheless, recently, a cut-off point with a lower tumor burden has been studied to define the initiation of adjuvant therapy. According to the cohort of Moncrieff et al, patients with AJCC IIIA melanoma and with SLN tumor deposits of >0.3 mm maximum dimension have a higher risk of disease progression and may benefit from adjuvant systemic therapy, finding 5-year MSS rates of 80.3% and 94.1% for patients with metastatic SLN tumor deposits >0.3 mm and <0.3 mm, respectively (p<0.0001) [30]. Similar findings were observed for OS and DFS.
Regarding the oncologic outcomes of patients with positive SLN in melanoma of the trunk and extremities, studies have been developed based on the Rotterdam criteria. Thus, in a multicenter study that included 1,085 patients, Van der Ploeg et al reported a 10-year MSS of 87% for patients with tumor burden <0.1 mm, 57% for the 0.1-1 mm group, and 48% for the >1mm group [2]. However, in this study, no statistically significant difference was found between the groups according to SLN tumor burden. The 10-year DFS was 83% for patients with Rotterdam criteria <0.1 mm, 49% in the 0.1 to 1.0 mm group, and 32% in the 1 mm group, which is consistent with the findings of this cohort, given that despite presenting marginal significance, worse DFS was evidenced in the group of patients with SLN tumor involvement >1 mm.
The low survival rates in our study can be explained by the higher proportion of patients with advanced disease stages at the time of diagnosis since the INC is a reference hospital [26]. The results of studies in different populations converge on the same point: the low survival of ALM is secondary to late diagnosis, that is, melanomas with greater thickness, ulceration, and worse prognostic factors. However, studies including molecular subtypes, immune profiles, DNA methylation, and gene expression of melanomas are lacking [27].
The findings of this study indicate that SLN tumor burden could be used as a clinically relevant prognostic factor for patients left under the observation of the lymph node chain because of its relationship with the number of positive lymph nodes found in lymphadenectomy.
Conflict of Interest
None of the authors have conflicts of interest.
References
- WHO. (2020). Globocan. Melanoma of skin.
- Van der Ploeg, A. P., Van Akkooi, A. C., Rutkowski, P., Nowecki, Z. I., Michej, W., Mitra, A., ... & Eggermont, A.M. (2011). Prognosis in patients with sentinel node-positive melanoma is accurately defined by the combined Rotterdam tumor load and Dewar topography criteria. J Clin Oncol, 29(16), 2206-14.
- Instituto Nacional de Cancerología (INC). (2020). Anuario estadístico. Bogota
- Egger, M. E., Bower, M. R., Czyszczon, I. A., Farghaly, H.,Noyes, R. D., Reintgen, D. S., ... & McMasters, K. M. (2014). Comparison of sentinel lymph node micrometastatic tumor burden measurements in melanoma. Journal of the American College of Surgeons, 218(4), 519-528.
- Gershenwald, J. E., Scolyer, R. A., Hess, K. R., Sondak, V. K., Long, G. V., Ross, M. I., ... & Sommariva, A. (2017). Melanoma staging: evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA: a cancer journal for clinicians, 67(6), 472-492.
- Leiter, U., Stadler, R., Mauch, C., Hohenberger, W., Brockmeyer, N. H., Berking, C., ... & German Dermatologic Cooperative Oncology Group. (2019). Final analysis of DeCOG-SLT trial: no survival benefit for complete lymph node dissection in patients with melanoma with positive sentinel node. Journal of Clinical Oncology, 37(32), 3000-3008.
- Kim, C., Economou, S., Amatruda, T. T., Martin, J. C., & Dudek, A. Z. (2015). Prognostic significance of microscopic tumor burden in sentinel lymph node in patients with cutaneous melanoma. Anticancer Research, 35(1), 301-309.
- Satzger, I., Leiter, U., Gräger, N., Keim, U., Garbe, C., & Gutzmer, R. (2019). Melanoma-specific survival in patients with positive sentinel lymph nodes: Relevance of sentinel tumor burden. European Journal of Cancer, 123, 83-91.
- Eggermont, A. M., Chiarion-Sileni, V., Grob, J. J., Dummer, R., Wolchok, J. D., Schmidt, H., ... & Testori, A. (2015). Adjuvant ipilimumab versus placebo after complete resection of high-risk stage III melanoma (EORTC 18071): a randomised, double-blind, phase 3 trial. The lancet oncology, 16(5), 522-530.
- Verver, D., van Klaveren, D., van Akkooi, A. C., Rutkowski, P., Powell, B. W., Robert, C., ... & Grünhagen, D. J. (2018). Risk stratification of sentinel node–positive melanoma patients defines surgical management and adjuvant therapy treatment considerations. European Journal of Cancer, 96, 25-33.
- Namikawa, K., Aung, P. P., Milton, D. R., Tetzlaff, M. T.,Torres-Cabala, C. A., Curry, J. L., ... & Prieto, V. G. (2019). Correlation of tumor burden in sentinel lymph nodes with tumor burden in nonsentinel lymph nodes and survival in cutaneous melanoma. Clinical Cancer Research, 25(24), 7585-7593.
- Chang, J. W. C. (2013). Acral melanoma: a unique disease in Asia. JAMA dermatology, 149(11), 1272-1273.
- Faries, M. B., Thompson, J. F., Cochran, A. J., Andtbacka, R. H., Mozzillo, N., Zager, J. S., ... & Elashoff, R. M. (2017). Completion dissection or observation for sentinel-node metastasis in melanoma. New England Journal of Medicine, 376(23), 2211-2222.
- Palve, J., Ylitalo, L., Luukkaala, T., Jernman, J., & Korhonen,N. (2020). Sentinel node tumor burden in prediction of prognosis in melanoma patients. Clinical & experimental metastasis, 37, 365-376.
- Borgognoni, L., Bellucci, F., Urso, C., Manneschi, G., Gerlini, G., Brandani, P., ... & Sestini, S. (2019). Enhancing the prognostic role of melanoma sentinel lymph nodes through microscopic tumour burden characterization: clinical usefulness in patients who do not undergo complete lymph node dissection. Melanoma research, 29(2), 163-171.
- Lino-Silva, L. S., Domínguez-Rodríguez, J. A., Aguilar-Romero, J. M., Martínez-Said, H., Salcedo-Hernández, R. A., García-Pérez, L., ... & Cuellar-Hubbe, M. (2016). Melanoma in Mexico: clinicopathologic features in a population with predominance of acral lentiginous subtype. Annals of surgical oncology, 23, 4189-4194.
- Castaneda, C. A., Torres-Cabala, C., Castillo, M., Villegas, V., Casavilca, S., Cano, L., ... & Abugattas, J. (2017). Tumor infiltrating lymphocytes in acral lentiginous melanoma: a study of a large cohort of cases from Latin America. Clinical and Translational Oncology, 19, 1478-1488.
- Vazquez, V. D. L., Silva, T. B., Vieira, M. D. A., De Oliveira, A.T. T., Lisboa, M. V., De Andrade, D. A. P., ... & Carneseca, E.C. (2015). Melanoma characteristics in Brazil: demographics, treatment, and survival analysis. BMC research notes, 8, 1-9.
- Loria, D. I., González, A., & Latorre, C. (2010). Epidemiología del melanoma cutáneo en Argentina: análisis del Registro Argentino de Melanoma Cutáneo. Dermatología Argentina, 16(1), 39-45.
- Van Akkooi, A. C., De Wilt, J. H., Verhoef, C., & Eggermont,A. M. (2008). The Rotterdam criteria for sentinel node tumor load: the simplest prognostic factor?. Journal of Clinical Oncology: Oficial Journal of the American Society of Clinical Oncology, 26(36), 6011-author.
- Bhutiani, N., Egger, M. E., Stromberg, A. J., Gershenwald,J. E., Ross, M. I., Philips, P., ... & McMasters, K. M. (2018). A model for predicting low probability of nonsentinel lymph node positivity in melanoma patients with a single positive sentinel lymph node. Journal of surgical oncology, 118(6), 922-927.
- Bilimoria, K. Y., Balch, C. M., Bentrem, D. J., Talamonti, M. S., Ko, C. Y., Lange, J. R., ... & Wayne, J. D. (2008). Complete lymph node dissection for sentinel node-positive melanoma: assessment of practice patterns in the United States. Annals of surgical oncology, 15, 1566-1576.
- Nova, J., Acosta, Á., Toquica, A., GU-Quiñones, S., Gutiérrez,L. D., & Montero, A. (2021). Acral lentiginous melanoma: Review of one of the most frequent melanomas in Latin America. Revista Colombiana de Cancerología, 25(3), 140-153.
- Amini, A., Rusthoven, C. G., Waxweiler, T. V., Jones, B. L., Fisher, C. M., Karam, S. D., & Raben, D. (2016). Association of health insurance with outcomes in adults ages 18 to 64 years with melanoma in the United States. Journal of the American Academy of Dermatology, 74(2), 309-316.
- Asgari, M. M., Shen, L., Sokil, M. M., Yeh, I., & Jorgenson,E. (2017). Prognostic factors and survival in acral lentiginous melanoma. British Journal of Dermatology, 177(2), 428-435.
- Duarte, C. A., Flórez, J. P., López, H. G., Meneses, M. X., &De Vries, E. (2017). Survival of acral lentiginous melanoma in the National Cancer Institute of Colombia. Journal of the European Academy of Dermatology and Venereology, 31(3), 438-442.
- Cust, A. E. (2018). Prognostic features for acral lentiginous melanoma. British Journal of Dermatology, 178(2), 311-312.
- Eggermont, A. M., Suciu, S., Testori, A., Santinami, M., Kruit, W. H., Marsden, J., ... & Keilholz, U. (2012). Longterm results of the randomized phase III trial EORTC 18991 of adjuvant therapy with pegylated interferon alfa-2b versus observation in resected stage III melanoma. J Clin Oncol, 30(31), 3810-3818.
- Medina, M. L., & Calidonio, C. L. (2020). Tratamiento adyuvante de melanoma cutáneo: indicaciones y alternativas.Revista Colombiana de Hematología y Oncología, 7(2), 56-63.
- Moncrieff, M. D., Lo, S. N., Scolyer, R. A., Heaton, M. J.,Nobes, J. P., Snelling, A. P., ... & Zager, J. S. (2022). Clinical outcomes and risk stratification of early-stage melanoma micrometastases from an international multicenter study: implications for the management of American Joint Committee on Cancer IIIA Disease. Journal of Clinical Oncology, 40(34), 3940-3951.
- Morton, D. L., Wen, D. R., Wong, J. H., Economou, J.S., Cagle, L. A., Storm, F. K., ... & Cochran, A. J. (1992). Technical details of intraoperative lymphatic mapping for early stage melanoma. Archives of surgery, 127(4), 392-399