Research Article - (2022) Volume 3, Issue 4
Invitro Activity of Tigecycline against Multidrug-Resistant Enterobacteriaceae from Blood Stream Infection in a Tertiary Care Hospital of Nepal
2Professor, Department of Microbiology, B.P. Koirala Institute of Health Sciences, Dharan, Nepal
3Additional Professor, Department of Microbiology, B.P. Koirala Institute of Health Sciences, Dharan, Nepal
Received Date: Sep 15, 2022 / Accepted Date: Sep 21, 2022 / Published Date: Oct 22, 2022
Copyright: ©Copyright: ©2022 Yadav, Khanal B. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Yadav, Khanal B, Bhattarai NR. (2022). Invitro Activity of Tigecycline against Multidrug-Resistant Enterobacteriaceae from Blood Stream Infection in a Tertiary Care Hospital of Nepal. J Vet Heal Sci, 3(4), 305-311.
Abstract
Background: Multidrug resistant (MDR) enterobacteriaceae like extended spectrum beta lactamase (ESBL) and metallo-beta lactamase (MBL) are being encountered as the causative agents of blood stream infection. To overcome this, accurate diagnosis of MDR pattern with appropriate antibiotics is required. Tigecycline is a broad spectrum antibiotic which exhibit strong activity against MDR enterobacteriaceae. This study is aimed to find out resistance pattern like ESBL and MBL with invitro activity of tigecycline against enterobacteriaceae.
Methods: A descriptive cross-sectional study was conducted in the Department of Microbiology, B.P. Koirala Institute of Health Sciences, from 1st September 2014 to 31st August 2015. Confirmation for ESBL was done as recommended by Clinical and Laboratory Standard Institute (CLSI) and MBL production was detected by double disk synergy test. Antibiotic sensitivity test against tigecycline was done by Kirby-Bauer disk diffusion method.
Results: 192 (1.70%) enterobacteriaceae were isolated throughout the study. Among them, 94 (49%) were ESBL, 51 (26.5%) were carbapenemase and 22 (11.5%) were MBL producers. A total of 64 (33.4%) isolates were found to be MDR. None of the isolates was resistant against tigecycline.
Conclusion: Tigecycline is found to have excellent invitro activity against MDR enterobacteriaceae from BSI
Keywords
Enterobacteriaceae, ESBL, MBL, MDR, tigecycline
Introduction
Blood stream infection (BSI) is potentially a life-threatening dis-ease as it can cause serious secondary infections like – infective endocarditis, osteomyelitis and may result in severe sepsis. BSI due to multidrug-resistant (MDR) organisms has been associated with multiple poor outcomes like duration of hospital stay, health care cost and a high morbidity and mortality rate [1–3].
Over recent years, problem of multidrug resistant Enterobacte-riaceae has increased dramatically and occupies the third most leading cause of BSI in most of the settings [4]. The escalating burden of multidrug resistance in Enterobacteriaceae is largely due to production of beta lactamase and subset of beta lactamase that are enzymes that bind, deactivate the different types of beta lactam antibiotics and confer broad resistance against them [5].
Tigecycline is a broad-spectrum of antibiotic, which falls under glycylcycline group. It acts as a protein synthesis inhibitor. It binds 30S ribosomal subunit of bacteria and thereby blocking the inter¬action of amino acyl tRNA with A site of the ribosome. It also overcomes the effect of efflux pumps thus remains unaffected by the typical mechanisms that render bacteria resistant to the tetra-cycline class [6]. Tigecycline is also not associated with cross-re-sistance to other antibiotics, conferring another advantage in its ac-tivity against several Extended Spectrum Beta-Lactamase (ESBL) and carbapenemase producing Enterobacteriaceae [6]. Tigecycline has become an alternative to face the challenges of many MDR organisms [7].
Use of tigecycline in bacteremia is controversy because of its low serum levels with standard dose [3]. A prospective study demon-strates that tigecycline along with standard dose imipenem showed good clinical efficacy on patients with bacteremia [8]. Therefore, we aimed to study the invitro activity of tigecycline against mul-tidrug-resistant enterobacteriaceae isolates from blood stream in-fection.
Methods
A descriptive cross-sectional study was conducted in the Depart-ment of Microbiology, B.P. Koirala Institute of Health Sciences, Dharan, from 1st September 2014 to 31st August 2015. Ethical ap¬proval was obtained from Institutional Review Committee (IRC), B.P. Koirala Institute of Health Sciences, Dharan, Nepal. All meth¬ods in this study was carried out in the hospital premises of BP¬KIHS in accordance with guidelines provided by “BPKIHS - code no IRC/424/014”.
A total of 11,264 blood samples were obtained in the Department of Microbiology for culture. Three ml blood was collected in Brain Heart Infusion (BHI) Broth in a dilution of 1:10. Blood cultures were processed in BD 9050 system (Becton and Dickinson, New York, USA). BHI Broths were aerobically incubated at 35°C for 5 days and observed for growth of microorganisms. If there is any sign of growth, sub-culture was done on Chocolate agar, Blood agar and MacConkey’s agar (Hi Media, Mumbai, India). Culture plates were incubated aerobically for overnight at 35 °C except for chocolate and blood agar, as they were incubated in 5% CO2 incubator [9]. Identification of isolates was performed following standard microbiological techniques which involved the morpho¬logical appearance of the colonies, Gram’s staining, motility test and a battery of biochemical tests which included oxidase test, sugar fermentation test, nitrate reduction test, decarboxylation test and other biochemical parameters [10].
Antibiotic susceptibility against enterobacteriaceae isolates was determined by Kirby–Bauer disk diffusion method. A suspension was made by inoculating 3-5 pure bacterial colonies in nutrient broth. Before inoculating the suspension on Muller Hinton Agar (MHA), turbidity was matched with 0.5 McFarland standards. MHA plate was allowed to come to room temperature before use. Antibiotics were placed on the surface of the inoculated 120 mm MH agar plate within 15 minutes of inoculation. Gentle pressure was applied on the disk to ensure complete contact with the agar surface. Edge to edge distance between two antimicrobial disks was not less than 15 mm. The plates were then covered with its lid, inverted and incubated overnight at 35oC aerobically in a non CO2 incubator [11].
Antibiotics used were several beta-lactam and tigecycline (TGC) (15µg). Beta-lactam antibiotics used were Cefotaxime (CTX) (30µg), Ceftazidime (CAZ) (30µg), Ceftriaxone (CTR) (30µg), Cefotaxime with clavulanic acid (CAC) (30/10µg), Ceftazidime with clavulanic acid (30/10µg), Ertapenem (ETP) (10µg), Ce¬foxitin (CX) (30µg), Cefepime (CPM) (30µg), Aztreonam (AO) (30µg), Imipenem (IPM) (10 µg). After 18-24 hours of incubation, each plate was examined for the zone of inhibition. Zone diameter of complete inhibition (as judged by unaided eye) was measured in a transmitted light with the help of scale or vernier caliper, held on the back of the petri plate. Interpretation of antibiotic susceptibility test results was made as per the guidelines provided by the Clini¬cal and Laboratory Standards Institute (CLSI) [12]. Isolates were further tested by performing sensitivity test against tigecycline by Kirby-Bauer disk diffusion method. As for interpretation of sus¬ceptibility pattern of tigecycline, isolates showing zone of inhi¬bition (ZOI) ≥ 18 mm were interpreted as sensitive, 15 to 17 mm interpreted as intermediate susceptible and ≤ 15 mm interpreted as resistant. Most of the studies have done E-test or agar dilution test for calculation of minimum inhibitory concentration (MIC) of tigecycline, but in this study Kirby-Bauer disk diffusion method is used as this method is cost effective for community perspec¬tive as compared to MIC [13, 14]. Escherichia coli ATCC 25922 was used as the control organisms for the antibiotic sensitivity test. Isolates showing resistant to at least one antibiotic in three or more antimicrobial classes were confirmed as multidrug-resistant (MDR) phenotype [9, 15].
Detection of ESBL
Isolate showing zone of inhibition (ZOI) of ≤ 27 mm for cefotax¬ime (CTX) (30 µg) and ≤ 22 mm for ceftazidime (CAZ) (30 µg) was confirmed by combine disk method. For this, antimicrobial disks (HI Media, Mumbai, India) used were CTX (30 µg), CAZ (30 µg), cefotaxime-clavulanic acid (CEC) (30 µg/10 µg) and ceftazidime-clavulanic acid (CAC) (30 µg/10 µg). Each disk was placed at a distance of at least 20 mm (center to center). Isolates resistant to CTX and CAZ but sensitive to CEC and CAC with enhanced ZOI ≥ 5 mm was confirmed as ESBL producers [12].
Detection of Carbapenemase
Isolates that showed resistant to ertapenem (ETP) (10 µg) (ZOI ≤ 22 mm) as well as other antibiotics but sensitive to imipenem (IPM) (10 µg) (ZOI ≥ 23 mm) were considered as carbapenemase producers [16].
Metallo β-lactamase (MBL)
All carbapenemase producers were tested for double disk synergy test and were confirmed by Modified Hodge Test (MHT).
Double Disk Synergy Test
Double disk synergy test was done by using anhydrous ethylene diamine tetra acetic acid (EDTA) disk of concentration 1.5 mg/ disk (0.5 mol.) and imipenem (IPM) (10 µg). EDTA-IPM disks were kept at a distance of 10 mm apart (edge to edge). Enhanced ZOI showing synergistic effect between the two disks was consid-ered as MBL producers [17, 18].
Modified Hodge Test (MHT)
For this, ATCC Escherichia coli was inoculated on MHA plate. With the help of sterile forcep, IPM disk was placed at the center of the plate. Isolated strain was inoculated perpendicular to IPM disk. After incubation for 18-24 hours at 35oC in an aerobic condi-tion, appearance of a clover leaf at the streaking line of the isolated strain was confirmed as MBL producers [16].
Data and statistical analysis
The data generated during the study period were analyzed by us-ing SPSS version 16.0 and were analyzed according to frequency distribution and percentage.
Results
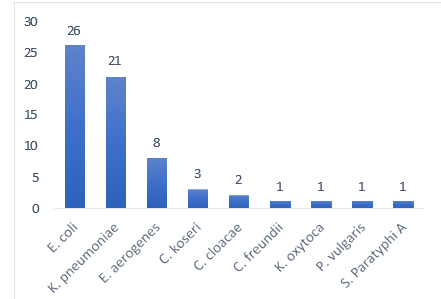
During the study period, a total of 11,264 blood samples were collected from the patients subjected to Blood Stream Infections. Of these isolates, 192 (1.70%) were Enterobacteriaceae compris-ing of Escherichia coli 95 (49.48%), Klebsiella pneumoniae 45 (23.44%), Enterobacter aerogenes 27 (14.06%), Citrobacter fre- undii 6 (3.13%), Citrobacter koseri 6 (3.13%), Proteus vulgaris 3 (1.56%), Salmonella Typhi 3 (1.56%), Enterobacter cloacae 2 (1.04%), Proteus mirabilis 2 (1.04%), Klebsiella oxytoca 1 (0.52%), Morganella morganii 1 (0.52%) and Salmonella Paraty- phi A 1 (0.52%). (Figure 1).
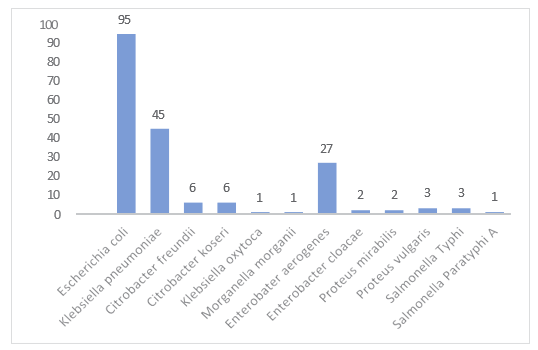
Figure 1: Frequency of Enterobacteriaceae Isolates (n=192)
Sex Wise Distribution of Studied Patients (n=192):
Of the total Enterobacteriaceae isolates, 113 (59%) were obtained from male patients and 79 (41%) from female patients. Figure 2.
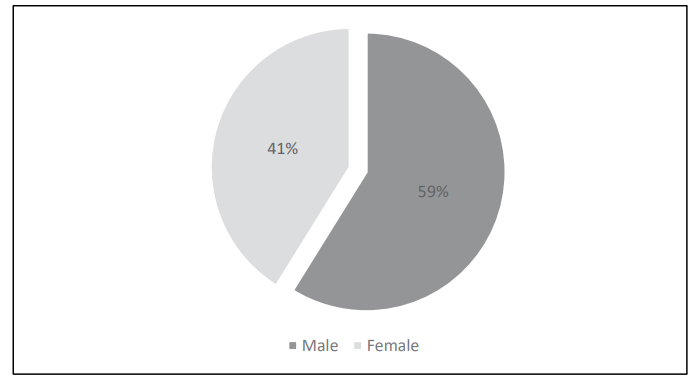
Figure 2: Sex Wise Distribution
Age Wise Distribution (n=192):
Enterobacteriaceae was isolated from blood in all aged groups. Among them, majority were from age group <10 years (71; 37%), followed by age groups 50-89 years (46; 24%) and age groups 20-29 years (25; 13%). The number of Enterobacteriaceae in other age groups were almost similar, i.e. 30- 39 years (17; 9%), 40-49 years (17; 9%) and 10-19 years (16; 8%). There were 15 neonates, 19 infants and 30 children (<12 Years). Figure 3
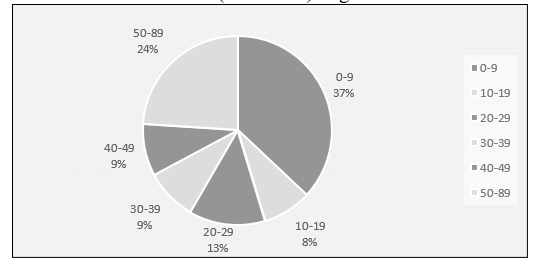
Figure 3: Age wise distribution (n=192)
Frequency of β-lactamases Producers Results of different types of β-lactamases produced by Entero¬bacteriaceae are given in figure 4. ESBL was the most common β-lactamase 94 (49%), followed by carbapenemase 51 (26.5%) and MBL 22 (11.5%).
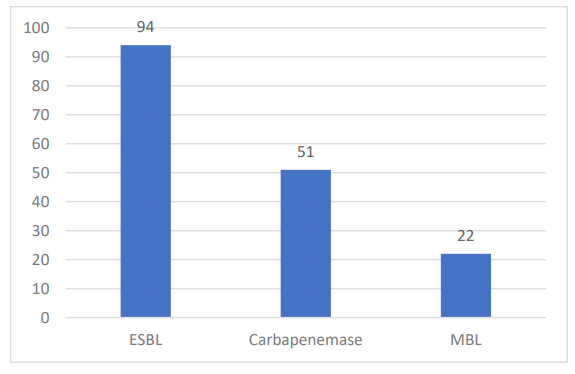
Figure 4: β-lactamase producers (n=192
Antibiotic Sensitivity Pattern of Enterobacteriaceae Most of the isolates were resistant to third generation cephalospo¬rins, cefotaxime (141; 73.40%), ceftazidime (139; 72.40%) and ceftriaxone (105; 54.70%). Similar sensitivity pattern for cefepime (113; 58.80%) was observed compared to third generation cepha¬losporins. Most of the isolates of E. aerogenes (17; 63%) and C. freundii (4; 67%) were sensitive to cefepime. All three isolates of S. Typhi were sensitive to cefepime. Regarding monobactam, 77 (40%) isolates were found sensitive and the number of resistant isolates were equal to the sensitive isolates. Among carbapenems, 22 (11.5%) isolates were resistant to imipenem and 51 (26.5%) isolates were found resistant to ertapenem. Most of the isolates (164; 85.4%) were sensitive to imipenem. Almost 99% of the iso¬lates were found sensitive to tigecycline (zone of inhibition ≥18 mm). Only two of the isolates (K. pneumoniae) were found to have intermediate susceptibility (zone of inhibition ≤16 mm) to tigecy¬cline. (Table 1)
Table 1: Antibiotic Sensitivity Pattern of the Enterobacteriaceae
|
Antibiotic used |
Sensitive (%) |
Resistance (%) |
Intermediate (%) |
|
Cefotaxime |
25.50 |
73.40 |
1.10 |
|
Ceftazidime |
23.96 |
72.40 |
3.64 |
|
Ceftriaxone |
42.70 |
54.70 |
2.60 |
|
Cefotaxime-clavulanic acid |
71.90 |
28.10 |
0.00 |
|
Ceftazidime-clavulanic acid |
68.30 |
31.70 |
0.00 |
|
Cefoxitin |
41.70 |
41.70 |
16.6 |
|
Cefepime |
39.60 |
58.9 |
1.50 |
|
Aztreonam |
40.10 |
56.25 |
3.65 |
|
Ertapenem |
66.63 |
26.56 |
7.81 |
|
Imipenem |
85.40 |
11.50 |
3.10 |
|
Tigecycline |
98.96 |
0.00 |
1.04 |
MDR Enterobacteriaceae
Discussion
In our study, the frequency of Enterobacteriaceae from BSI was found to be 1.7% (192) out of total blood culture positive 17.3% (1948). It is similar to the study conducted by Abdallah et al in 2015 in Egyptian patients and in Nepal by where the number of Enterobacteriaceae isolated found to be 94 and 96 respectively, which was less as compared to our study [19, 20].
β-lactamase production remains the most important mediator of β-lactam resistance in enterobacteriaceae [21]. In the present study, among 49% ESBL producers, 64% were Escherichia coli followed by K. pneumoniae 20%. Similar type of study conducted by Shres¬tha et al at BPKIHS in the year 2007 where the prevalence of ESBL among the clinical isolates of pyogenic infections were - E. coli (53%), K. pneumoniae (14.8%), P. mirabilis (12.9%), Entero- bacter species (5.5%) and Citrobacter species (5.5%) [22]. Emer¬gence of ESBL is probably due to widespread use of third-gener-ation cephalosporins which is believed to be the major cause of mutations in TEM and SHV enzymes [21].
In this study, Carbapenemase producers was found to be 51 (26.5%). In screening MBL, 22 (11.5%) isolates were imipenem resistant, whereas 51 (26.5%) were resistant to ertapenem. MBL producing Klebsiella species in the present study was found to be higher in number than that shown by [23]. In a study conducted by Vinod Kumar et al., 20% resistance to imipenem and 17% MBL production was reported. Similarly, reported 20% MBL produc¬tion in their study [24].
Antimicrobial susceptibility profile of Enterobacteriaceae showed a high degree of resistance to the antimicrobials. Resistance against cefotaxime and ceftazidime were highest (72–74%) as compared to the resistance pattern against antimicrobials. In BSIs, third-generation cephalosporins have been extensively used as a first-line antibiotic, because of which they are rendered useless. Our isolates showed least resistance for imipenem (11.5%) and ertapenem (26.5%). The rate of resistance to the various drugs was in concordance with other studies [21, 24 and 25]. In our study, 64 (33.3%) isolates were MDR. Various authors have reported high percentage of MDR in their study [21, 25 and 26].
Among the isolates, 190 (99%) were sensitive to tigecycline. Two K. pneumoniae were found to have intermediate susceptibility to tigecycline. In a study conducted by Mohanty and Mahapatra, 6.7% of the isolates (all K. pneumoniae) and seven K. pneumoniae (14.5%) was found resistant to tigecycline, which is different from our study [6]. Present study showed good activity of tigecycline (99%) against the isolates. Only two isolates were found to have intermediate susceptibility to tigecycline. Similar results were documented by Sader et al [27]. Tigecycline was very active and appears to be an excellent option for treatment of infections caused by these multidrug-resistant Enterobacteriaceae [27, 28]. Clinical efficacy of tigecycline in BSI has not yet been established. In vitro, evaluation of its efficacy in ESBL and MBL producing isolates in septicemia have been reported by Roy et al. in two different studies [29, 30].
Conclusion
In conclusion, tigecycline has showed excellent effect against MDR Enterobacteriaceae. The limited availability of suitable al-ternate therapeutic armamentarium necessitates the use of tigecy¬cline with a critical and urgent need to continuously monitor the emergence and spread of resistance. Present study has documented the increasing antimicrobial resistance among isolates from BSI which is a matter of concern for clinicians and microbiologists. This reflects the need for early detection and prevention of further spread of resistance to other bacteria.
Limitations
We are unable to perform minimum inhibitory concentration (MIC) for tigecycline as E-strip for tigecycline is expensive and fund was not available for this study. It would not be helpful for the community people for diagnostic purpose as it is not cost effective. Therefore, Kirby-Bauer disk diffusion test is done for determina¬tion of sensitivity pattern of tigecycline.
Abbreviations
AMR Antimicrobial Resistance
ATCC American Type Culture Collection
BSI Blood Stream Infection
CLSI Clinical and Laboratory Standards Institute
ESBL Extended Spectrum Beta-lactamase
MBL Metallo Beta-lactamase
MDR Multi Drug Resistant
ZOI Zone of Inhibition
Declarations
Acknowledgement
All members of the Department of Microbiology.
Funding
None.
Data availability
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
Author’s contributions
Conceptualization: AY, NRB, BK. Investigation: AY. Methodolo¬gy: AY, NRB, BK. Resources: AY, NRB, BK. Supervision: NRB, BK. Writing-original draft: AY. Writing-review and editing: NRB, BK. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Ethical approval for this study was obtained from Institution¬al Review Committee (IRC), BPKIHS, Dharan, Nepal (code no. IRC/424/014). Written informed consent was obtained from each patient before enrollment and where participants are children (<16 years old) from their parent or guardian.
Consent for publication
Not applicable.
Competing interest
We declare that we have no conflict of interest.
Declarations
Acknowledgement
All members of the Department of Microbiology.
Funding
None.
Data availability
The data generated and analyzed during the current study are available from the corresponding author on reasonable request.
Author’s contributions
Conceptualization: AY, NRB, BK. Investigation: AY. Methodolo¬gy: AY, NRB, BK. Resources: AY, NRB, BK. Supervision: NRB, BK. Writing-original draft: AY. Writing-review and editing: NRB, BK. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Ethical approval for this study was obtained from Institution¬al Review Committee (IRC), BPKIHS, Dharan, Nepal (code no. IRC/424/014). Written informed consent was obtained from each patient before enrollment and where participants are children (<16 years old) from their parent or guardian.
Consent for publication
Not applicable.
Competing interest
We declare that we have no conflict of interest.
References
- Edmond, M. B., Ober, J. F., Dawson, J. D., Weinbaum, D. L., & Wenzel, R. P. (1996). Vancomycin-resistant enterococcal bacteremia: natural history and attributable mortality. Clinical Infectious Diseases, 23(6), 1234-1239.
- Bearman, G. M., & Wenzel, R. P. (2005). Bacteremias: a leading cause of death. Archives of medical research, 36(6), 646-659.
- Wang, J., Pan, Y., Shen, J., & Xu, Y. (2017). The efficacy and safety of tigecycline for the treatment of bloodstream infections: a systematic review and meta-analysis. Annals of clinical microbiology and antimicrobials, 16(1), 1-10.
- Demirturk, N., & Demirdal, T. (2011). Causative agents of nosocomial bloodstream infections and their antimicrobial susceptibility patterns. Southeast Asian J Trop Med Public Health, 44, 1036-42.
- Ruppé, É., Woerther, P. L., & Barbier, F. (2015). Mechanisms of antimicrobial resistance in Gram-negative bacilli. Annals of intensive care, 5(1), 1-15.
- Mohanty S., Mahapatra A. (2021). In vitro activity of tigecycline against multidrug-resistant Enterobacteriaceae isolates from skin and soft tissue infections. Ann Med Surg. 62(De-cember 2020):228–30.
- Muralidharan, G., Micalizzi, M., Speth, J., Raible, D., & Troy,S. (2005). Pharmacokinetics of tigecycline after single and multiple doses in healthy subjects. Antimicrobial agents and chemotherapy, 49(1), 220-229.
- Jean, S. S., Hsieh, T. C., Hsu, C. W., Lee, W. S., Bai, K. J., &Lam, C. (2016). Comparison of the clinical efficacy between tigecycline plus extended-infusion imipenem and sulbactam plus imipenem against ventilator-associated pneumonia with pneumonic extensively drug-resistant Acinetobacter bauman-nii bacteremia, and correlation of clinical efficacy with in vitro synergy tests. Journal of Microbiology, Immunology and Infection, 49(6), 924-933.
- Parajuli, N. P., Acharya, S. P., Mishra, S. K., Parajuli, K., Rijal,B. P., & Pokhrel, B. M. (2017). High burden of antimicrobial resistance among gram negative bacteria causing healthcare associated infections in a critical care unit of Nepal. Antimicrobial Resistance & Infection Control, 6(1), 1-9.
- Garcia, L. S. (2007). Calibration of microscope with an ocular micrometer. Clinical Microbiology procedures handbook. 2nd ed. update, ASM Press, Washington DC.
- Clinical Laboratory Standard Institute. Performance standard for antimicrobial susceptibility test. 9th ed.
- Clinical and Laboratory Standards Institute. (2014). Wayne, PA. Clinical and Laboratory Standards Institute. Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Susceptibility Testing. 24 th Informational Supplement. CLSI document M100-S24.
- Hudzicki, J. (2009). Kirby-Bauer disk diffusion susceptibilitytest protocol. American society for microbiology, 15, 55-63.
- Antimicrobial Susceptibility Systems. (2010). HiCrome Mueller Hinton Agar.
- Magiorakos, A. P., Srinivasan, A., Carey, R. B., Carmeli, Y., Falagas, M. E., Giske, C. G., & Monnet, D. L. (2012). Multidrug-resistant, extensively drug-resistant and pandrug-resis-tant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clinical microbiology and infection, 18(3), 268-281.
- Anderson, K. F., Lonsway, D. R., Rasheed, J. K., Biddle, J., Jensen, B., McDougal, L. K., & Patel, J. B. (2007). Evaluation of methods to identify the Klebsiella pneumoniae carbapenemase in Enterobacteriaceae. Journal of clinical microbiology, 45(8), 2723-2725.
- Franklin, C., Liolios, L., & Peleg, A. Y. (2006). Phenotypic detection of carbapenem-susceptible metallo-β-lactamase-pro-ducing gram-negative bacilli in the clinical laboratory. Journal of clinical microbiology, 44(9), 3139-3144.
- Lee, K., Lim, Y. S., Yong, D., Yum, J. H., & Chong, Y. (2003). Evaluation of the Hodge test and the imipenem-ED-TA double-disk synergy test for differentiating metal-lo-β-lactamase-producing isolates of Pseudomonas spp. and Acinetobacter spp. Journal of clinical microbiology, 41(10), 4623-4629.
- Abdallah, H. M., Wintermans, B. B., Reuland, E. A., Koek,A., Al Naiemi, N., Ammar, A. M., & Vandenbroucke-Grauls,C. M. J. E. (2015). Extended-spectrum β-lactamase-and carbapenemase-producing Enterobacteriaceae isolated from Egyptian patients with suspected blood stream infection. PloS one, 10(5), e0128120.
- Easow, J. M., Joseph, N. M., Dhungel, B. A., Chapagain, B., & Shivananda, P. G. (2010). Blood stream infections among febrile patients attending a teaching hospital in Western Region of Nepal. Australasian Medical Journal (Online), 3(10), 633.
- Modi, D. J., Patel, B. V., Patel, M. H., Bhatt, S. S., Sood,N. K., & Vegad, M. M. (2012). A Study of Extended Spectrum β-Lactamase (ESBL) and AmpC β-Lactamase Producing Klebsiella Pneumoniae in Neonatal Intensive Care Unit at Tertiary Care Hospital, Ahmedabad. National Journal of Community Medicine, 3(03), 523-528.
- Shrestha, S., Amatya, R., & Dutta, R. (2011). Prevalence of extended spectrum beta lactamase (ESBL) production in gram negative isolates from pyogenic infection in tertiary care hospital of eastern Nepal. Nepal Medical College journal: NMCJ, 13(3), 186-189.
- Mishra, S. K., Shrestha, R., Rijal, B. P., & Pokhrel, B. M. (2015). The bad, the ugly and the demon: a tale of extensively drug-resistant, extended-spectrum-beta-lactamase-and metallo-beta-lactamase-producing superbugs associated with nosocomial pneumonia. Asian Pacific Journal of Tropical Disease, 5(1), 71-76.
- Gajul, S. V., Mohite, S. T., Mangalgi, S. S., Wavare, S. M., & Kakade, S. V. (2015). Klebsiella pneumoniae in septicemic neonates with special reference to extended spectrum β-lac-tamase, AmpC, metallo β-lactamase production and multiple drug resistance in tertiary care hospital. Journal of Laboratory Physicians, 7(01), 032-037.
- Chandel, D. S., Johnson, J. A., Chaudhry, R., Sharma, N., Shinkre, N., Parida, S., & Panigrahi, P. (2011). Extended-spec-trum β-lactamase-producing Gram-negative bacteria causing neonatal sepsis in India in rural and urban settings. Journal of medical microbiology, 60(Pt 4), 500.
- Vinod Kumar, C. S., Kalappanavar, N. K., Patil, U., & Basa-varajappa, K. G. (2011). Change in spectrum of microbial aetiology in relation to gestational age and birth weight and emergence of ESBL in tertiary neonatal intensive care units. Int J Biol Med Res, 2(3), 727-734.
- Castanheira, M., Sader, H. S., Deshpande, L. M., Fritsche, T. R., & Jones, R. N. (2008). Antimicrobial activities of tigecycline and other broad-spectrum antimicrobials tested against serine carbapenemase-and metallo-β-lactamase-producing Enterobacteriaceae: report from the SENTRY Antimicrobial Surveillance Program. Antimicrobial agents and chemotherapy, 52(2), 570-573.
- Bogdanovich, T., Adams-Haduch, J. M., Tian, G. B., Nguyen,M. H., Kwak, E. J., Muto, C. A., & Doi, Y. (2011). Colistin-re-sistant, Klebsiella pneumoniae carbapenemase (KPC)–pro-ducing Klebsiella pneumoniae belonging to the international epidemic clone ST258. Clinical infectious diseases, 53(4), 373-376.
- Roy, S., Datta, S., Viswanathan, R., Singh, A. K., & Basu, S. (2013). Tigecycline susceptibility in Klebsiella pneumoniae and Escherichia coli causing neonatal septicaemia (2007–10) and role of an efflux pump in tigecycline non-susceptibility.Journal of Antimicrobial Chemotherapy, 68(5), 1036-1042.
- Roy, S., Viswanathan, R., Singh, A. K., Das, P., & Basu, S. (2011). Sepsis in neonates due to imipenem-resistant Klebsiella pneumoniae producing NDM-1 in India. Journal of antimicrobial chemotherapy, 66(6), 1411-1413.


