Research Article - (2025) Volume 3, Issue 1
Investigating the behavioral and antioxidative effects of ethanolic extract from Schumanniophyton magnificum (K. Schum) in ketamine-induced psychosis in mice
2Department of Pharmacology, Faculty of Basic Clinical Sciences, University of Medical Sciences, Ondo, Ondo State, Nigeria
3Department of Biochemistry, Faculty of Basic Medical Sciences, University of Medical Sciences, Ondo, Ondo State, Nigeria
Received Date: Apr 05, 2025 / Accepted Date: May 27, 2025 / Published Date: Jun 05, 2025
Copyright: ©©2025 Abiola M. Asowata-Ayodele, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Olusola, A. G., Asowata- Ayodele, A. M., Olatuyi, O., Afolabi, F., Salemcity, A. J., Olatunji, B. P. (2025). Investigating the behavioral and antioxidative effects of ethanolic extract from Schumanniophyton magnificum (K. Schum) in ketamine-induced psychosis in mice. Int J Bot Hor Res, 3(1), 01-07.
Abstract
This study investigates the behavioral and antioxidative effects of ethanolic extract from Schumanniophyton magnificum (K. Schum) in a mouse model. Psychosis poses significant health risks globally, influencing various aspects of mental and physical well-being, such as impaired reasoning, depression, memory loss, and social isolation. Despite its relevance, there is a lack of research into the potential therapeutic effects of Schumanniophyton magnificum leaves in managing psychosis, underscoring the need for innovative treatment approaches. In this study, Schumanniophyton magnificum was administered orally to groups of ketamine-induced psychotic mice at dosages of 100, 200, and 400 mg/kg for 14 days, with a control group for comparison. Behavioral assessments were conducted using the Open Field Test to measure hyperlocomotion, the Y-Maze Test to evaluate learning and memory (cognition), and the Novel Object Recognition Test to assess exploration and environmental interaction. Following the behavioral tests, biochemical assays were performed. The results showed that after 14 days of treatment, Schumanniophyton magnificum significantly (P < 0.05) reduced hyperlocomotion induced by ketamine, a marker of positive symptoms of psychosis. Additionally, doses of 100 mg/kg and 400 mg/kg significantly improved spatial memory and prevented cognitive impairments associated with ketamine administration. The plant treatments also resulted in increased activities of superoxide dismutase (SOD) and catalase (CAT), while decreasing malondialdehyde (MDA) levels, indicating potential antipsychotic effects via the inhibition of oxidative stress. This study suggests that Schumanniophyton magnificum may offer a promising, cost-effective approach to managing psychosis and mitigating its associated health and socioeconomic impacts.
Keywords
Schumanniophyton magnificum, Cognition, Behavioural Studies, Hyperlocomotion
Key Points
Envenoming, Cognition, Mechanism of Identification, Medicinal PlantsIntroduction
The field of psychopharmacology explores the complex interactions between pharmacological agents and brain circuits, shedding light on how these substances influence behavior. This study examines the behavioral and antioxidative effects of ethanolic extract from Schumanniophyton magnificum (K. Schum) in mice. While various medications can enhance cognitive functions, they do not necessarily cure the underlying mental disorders. Research suggests that although medications may improve communication and cognitive abilities, these changes can lead to psychological shifts that extend beyond biological explanations [1]. This challenges the widely held view that mental disorders can be solely understood through a neurobiological framework, highlighting the interconnectedness of psychological processes and brain function [1]. This perspective calls for a reassessment of the relationship between pharmacological interventions and the psychological dimensions of mental illness, encouraging further research that bridges neuroscience with behavioral studies in psychopharmacology.
Psychosis is a debilitating condition that severely impacts individuals’ health and quality of life. Effective therapeutic strategies to prevent or manage psychosis are essential for global health, as mental illness manifests through behaviors such as impaired reasoning, depression, memory deficits, and social withdrawal [1,2]. However, there is limited knowledge about the potential of Schumanniophyton magnificum leaves in treating or preventing psychosis, as no studies have investigated its effects on this condition. This highlights the need for further exploration into the plant's therapeutic potential. Schumanniophyton magnificum has been reported to be effective in managing various ailments, including epilepsy, snake envenomation, viral infections, depression, and fertility issues [3]. Despite its traditional uses, there is a lack of research into its role in psychosis treatment, making this investigation particularly significant. The focus of this study is to evaluate the neurobehavioral effects of Schumanniophyton magnificum ethanolic extract in a mouse model of ketamine-induced psychosis, with the aim of contributing valuable insights into new therapeutic strategies. By investigating the plant’s potential, this research seeks to offer cost-effective solutions for addressing both the health and socioeconomic challenges associated with psychosis.
Schumanniophyton magnificum, a small tree from the Rubiaceae family, is commonly known as the magnificent shrub or "mgba mmiri" in the Igbo language of Nigeria. It thrives in the tropical lowland rainforests of West and Central Africa, particularly in regions such as Ghana, Cameroon, Sierra Leone, and southeastern Nigeria (Calabar and Igbogodo) [3]. This study aims to explore the behavioral profile of Schumanniophyton magnificum ethanolic extract in a ketamine-induced psychosis model in mice.
Methodology
Study area
Collection of plant samples: Fresh leaves of Schumanniophyton magnificum were collected from a private farm in Okitipupa, Ondo state and authenticated by Mrs. Olofinlade Jumoke, the herbarium curator with herbarium number UNIMED P.B.T.H Number 0075 in the Department of Biosciences and Biotechnology, University of Medical Sciences, Ondo state. Voucher specimen of each plant sample was thereafter deposited in the herbarium of the same Department.
Plant extraction
The leaves of Schumanniophyton magnificum were rinsed properly with water and air dried; the dried leaves were ground into a powdered form with a blender. Powdered leaves (100g) was soaked into a reagent bottle containing 1000ml of 100% ethanol for 48 hours [4]. The mixture was filtered using a mesh sieve cloth, a funnel and a clean container, the filtrate is then poured into a conical flask and then connected to the rotary evaporator to obtain a pure extract without the ethanol. The extract (10g) was obtained and put in a sterile sample bottle. The plant’s extraction was done at Department of Biosciences and Biotechnology, University of Medical Sciences.
Experimental Animals
Thirty-six (36) mice weighing between 18g -22g were procured from the Animal House of the University of Medical Sciences, Ondo State. The animals were maintained 12/12- hour light/dark cycle at room temperature (25 ± 1â??) at the same animal house. The animals had access to standard pellet diet and clean water at liberation. The experimental protocol has been approved by the Animal Ethics Committee of the University of Medical Sciences, Ondo. (Attached with this work is the ethical approval certified to carry out this work by the ethics committee of the University).
Drug Preparation and Treatment
Doses of Schumanniophyton magnificum extract (100 mg/kg, 200 mg/kg and 400 mg/kg) were chosen for this study based on the findings from preliminary studies of the toxicity test conducted. Schumanniophyton magnificum extract and risperidone were dissolved in distilled water and administered orally, and ketamine was diluted with distilled water and administered intraperitioneally (i.p).
Experimental Design
According to earlier report by with a little modification, this experiment assessed the effect of ethanolic extract of Schumanniophyton magnificum on ketamine-induced schizophrenia-like behaviors and neurochemical alterations in mice [5,6]. Mice were randomly sorted into 6 groups: test (n = 6). Group 1 was kept as Normal control (vehicle or d.H2O), Group 2 was kept as negative control group (ketamine 10 mg/kg, i.p.), Group 3-5 were kept as treatment and were given ethanolic extract of Schumanniophyton magnificum (100, 200, and 400 mg/kg) together plus ketamine (10 mg/kg, i. p.) each, and Group 6 was kept as the positive control group (Risperidone 0.5 mg/kg) plus ketamine (10 mg/kg, i.p.) respectively for 14 days. After day 14th, each group were be assessed for behavioral test (hyperlocomotion, Y-maze and NORT) and biochemical assays.
Behavioral Assessment
Open Field Test (OFT): Open-field observation box (dimensions: 25 cm × 25 cm × 30 cm) made of transparent Perspex and behavioral events recorded using a camcorder and tracked with Behavior Tracker® software. The base of the maze had 16 squares (6.5 cm × 6.5 cm) demarcated with a non-toxic permanent marker. Thirty minutes after the treatments, the animals were placed individually into the open-field observational box and their behavior recorded for 5 min using a camcorder (Everio™ model, GZ-MG 130 U, JVC, Tokyo, Japan) suspended above the maze with the aid of a stand. Novelty-induced rearing was counted as the number of times the mouse stood on its hind limbs with its forelimbs against the wall of the observation cage (supported rearing) or in free air (unsupported rearing). The number of rearing (both supported and unsupported) was tracked for 5 min. Also, the number of line crossing was counted as a representation of locomotor activity. After each session, the observation chamber was cleaned with 70% ethanol to remove residual odor [7].
Y-Maze Test
Individual mice were gently placed in a Y-maze apparatus comprised of three arms labeled A, B, and C that is, all the same length = 21 cm, breadth = 7 cm, and height = 15.5 cm, and each arm symmetrically separated from the other by one hundred and twenty degree. Each mouse spent 8 min in the apparatus, and its exploration was automatically recorded and saved on a computer by a camera set above the apparatus. By allowing the mouse to explore all three arms of the labyrinth, spontaneous alternation (which is a predictor of spatial working memory) was measured using Y-maze software and this parameter is driven by mice’s intrinsic desire to explore previously unexplored arms. Based on the software settings, spontaneous alternation was calculated as the total number of correct alternations (ABC, BCA, or CAB but not ABA or BAB) / (total arm entries – 2) [7,8]. To remove the bias caused by the previous mouse’s odor cues, the apparatus was cleaned properly with 70% ethanol and allowed to dry.
NORT (Novel Object Recognition Test)
The effect of ethanolic extract of Schumanniophyton magnificum on ketamine-induced cognitive impairment, as an index for the spatial cognitive dysfunction associated with schizophrenia was evaluated by [7]. Basically, in the NORT there are no positive or negative re-enforcers and this methodology assesses the natural preference for novel object displayed by rodents. The task consists of three phases. The habituation phase in which the animal was allowed freely exploring the field arena in the absence of object, familiarization phase, a single animal was placed in the open field arena containing two identical sample object (A + A), for a few minutes. The experimental context is not drastically different during the familiarization and the test phase. After a retention interval during the test phase, the animal is returned to the open field arena with two objects, one is identical to the sample and the other was novel (A + B) [8]. During both familiarization and test phase, objects are located in opposite and symmetrical corners of the arena and location of novel versus familiar object was counterbalanced [8]. Normal mice spend more time exploring the novel object during the first few minutes of the test phase, and when this bias was observed, the animal could remember the sample object and can discriminate the sample from a novel object after delays of several minutes [9].
Biochemical Assays
Immediately after the behavioral tests, the animals were sacrificed by cervical dislocation and the brains were immediately removed and kept in the refrigerator with ice block for 20 minutes. Thereafter, the whole brain was weighed and homogenized with 5 ml of 10% w/v phosphate butter (0.1M, PH 7.4). Each brain tissue homogenates were centrifuged at 10,000 g for 10 minutes at 40oC, the pellet was discarded and the supernatant was immediately separated into various portions for the different biochemical assays.
Antioxidant Enzymes Assay
Estimation of Superoxide Dismutase Activity
SOD activity was measured by the method of [10]. The reaction mixture consists of 2.875 ml Tris-HCL buffer (50 mM, pH 8.5), pyrogallol (24 mM in 10 mM Hcl) and 100µl PMS in a total volume of 3ml. The enzyme activity was measured at 420 nm and was expressed as Units/mg protein. One unit of enzyme is defined as the enzyme activity that inhibits auto-oxidation of pyrogallol by 50%.
Estimation of reduced glutathione (GSH) level
GSH content of the tissue will be determined by the method of [10]. Briefly 1 ml of PMS (10%) is mixed with 1.0 ml of sulphosalicylic acid (4%). The samples incubated at 4oC for 1 h and then centrifuge at 1200 Xg for 15 min at 4oC. The assay mixture (3ml) consists of 0.4 ml supernatant, 2.2 ml phosphate buffer (0.1 M, pH 7.4) and 0.4 ml DTNB (4 mg /1 ml). The yellow color developed read immediately at 412 nm. The GSH concentration was calculated as nmol DTNB conjugate formed/gm tissue.
Estimation of Catalase Activity
Catalase activity was assayed by the method of [10]. Briefly the reaction mixture consists of 1.95 ml phosphate buffer (0.1M, pH 7.4), 1.0 ml hydrogen peroxide (0.019M) and 0.05 ml PMS in a final volume of 3 ml. Changes in absorbance was recorded at 240 nm every minute for 5 minutes. Catalase activity was calculated as nmol H2O2 consumed/min/mg protein.
Estimation of Glutathione Peroxidase Activity
Glutathione peroxidase (GPx) activity was measured according to the procedure of with some modifications [10]. Briefly the reaction mixture consists of sodium azide (10mM), hydrogen peroxide (2.5mM), trichloroacetic acid (10%), reduced glutathione (4mM), dipotassium hydrogen orthophosphate (0.3M), Ellman’s reagent (DTNB) and phosphate buffer (0.1M, pH 7.4). To 0.5 ml of phosphate buffer in a test tube was added 0.1 ml of NaN3, 0.2 ml of GSH, 0.1 ml of H2O2 and 0.5 ml of sample (added last). The reaction mixture was incubated for 3 min at 37°C after which 0.5 ml of TCA was added and the final mixture centrifuged at 3000 rpm for 5 min. To 1 ml of the supernatants, 2 ml of K2HPO4 and 1 ml of DTNB were added and the absorbance read against a reagent blank of 1 ml distilled water, 2 ml of K2HPO4 and 1 ml of DTNB at 412 nm.
Estimation of Glutathione S-Transferase (GST) Activity
Glutathione S-transferase activity was determined. Briefly the reaction mixture consists of phosphate buffer (0.1M, pH 6.5), reduced glutathione (0.1M) and 1-Chloro-2,4-dinitrobenzene (20mM). The medium for the estimation was prepared by adding 150µl of CDNB in a test tube to which 30µl of reduced glutathione, 2.79 ml and 30µl of sample was added. The reaction was allowed to run for 3 min with readings taken every 60 seconds against the blank at 340 nm [10].
Estimation of Myeloperoxidase Activity
Peroxidase activity with 3,3’,5,5’- Tetramethylbenzidine (TMB, Sigma) was measured. Briefly, 10 µl sample was combined with 80ul 0.75 nM H2O2 (sigma) and 110 µl TMB solution (2.9 mM TMB in 14.5% DMSO (sigma) and 150 mN sodium phosphate butter at pH 5.4),and the plate was incubated at 370c for 5 min. The reaction was stopped by adding 50 µl 2 M H2SO4 (Sigma) and absorption was measured at 450 nm to estimate MPO activity [10].
Statistical Analysis
The data obtained were expressed as mean ± S.E.M. (standard error of mean). The data were analyzed using one-way analysis of variance (ANOVA) followed by post-hoc test (Student Newman-Keuls) for multiple comparisons where appropriate using Graph-pad Prism software version 7.0. A level of P<0.05 was considered as statistically significant for all test.
Results
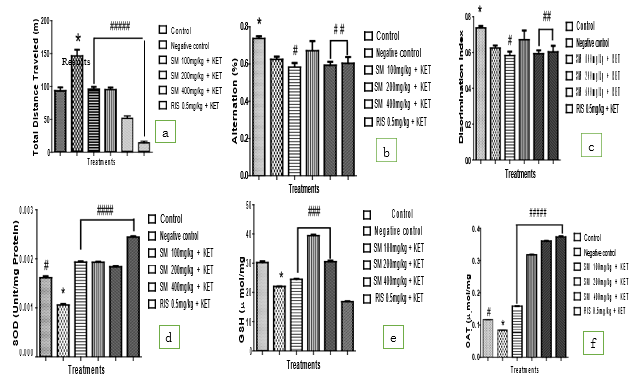
Effect of Schumanniophyton Magnificum on Ketamine-Induced Hyper-Locomotion
The effect of ethanolic extract of Schumanniophyton magnificum on ketamine-induced hyper-locomotion in mice based on the total distance travelled in open-field test (OFT) was shown in Figure 1a. The administration of ketamine (10 mg/kg, i.p.) significantly (P< 0.05) induced hyper locomotion compared with the group treated with vehicle (10mL/kg o.p), as shown by the increase in total distance travelled in the OFT compared with standard treatment. Schumanniophyton magnificum treatment across all groups show significant (P < 0.05) having decreased in the total distance travelled in the OFT compared with the standard treatment. Similarly, treatment with RIS (0.5 mg/kg, o.p) also significantly (P < 0.05) prevented the hyper-locomotion compared with the treated group [11].
Effect of Schumanniophyton Magnificumon Ketamine-Induced Spatial Memory Alteration in YMT
Figure 1b shows the effects of Schumanniophyton magnificum on ketamine-induced spatial working memory deficit based on the percentage (%) alternations in the Y-maze test (YMT) in mice. Administration of ketamine (10 mg/kg i.p) daily for 14 days, produced no significant (P < 0.05) % alternation behaviour of the animals in the YMT compared to control group, suggesting non-spatial working memory impairment. Schumanniophyton magnificum treatments of (100 and 400 mg/kg o.p) with ketamine administration for 14 days produced a significant (P < 0.05) decreased in the (%) level of cognitive alternation performance of the animals when compared with the control group. Similarly, treatments with RIS (0.5 mg/kg, o.p) produced a significant (P < 0.05) decreased in the (%) level of cognitive alternation performance of the animals when compared with the control group.
Effect of Schumanniophyton Magnificum on Ketamine-Induced Spatial Memory Alteration in NORT
Figure 1c shows the effect of Schumanniophyton magnificum on ketamine-induced spatial working memory deficit based on the discrimination index (NORT) in mice. Administration of ketamine (10 mg/kg i.p) daily for 14 days, produced a no significant (P < 0.05) decrease in discrimination index behaviour of the animals in the NORT compared to control group, suggesting of non-spatial working memory impairment. Schumanniophyton magnificum treatments of (100 and 400 mg/kg o.p) with ketamine administration for 14 days produced a significant (P < 0.05) decreased in the (%) level of cognitive alternation performance of the animals when compared with the control group. Similarly, treatments with RIS (0.5 mg/kg, o.p) produced a significant (P < 0.05) decreased in the (%) level of cognitive alternation performance of the animals when compared with the control group.
Effect of Schumanniophyton Magnificum on Superoxide Dismutase (SOD) Activity in Mice Brain
The effect of ketamine administered alone or in combination with Schumanniophyton magnificum on the activities of SOD in the brain of mice was shown in Figure 1d. Administration of Schumanniophyton magnificum (100, 200 and 40 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days significantly (P < 0.05) increased brain SOD activity compared with the group treated with the standard. Similarly, Risperidone (0.5 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) groups for 14 days produced a significantly (P < 0.05) increased brain SOD activity compared with the ketamine-treated group.
Effect of Schumanniophyton Magnificum on Glutathione (GSH) Concentration in Mice Brain
The effect of ketamine administered alone or in combination with Schumanniophyton magnificumon the concentration of GSH in the brain of mice was shown in Figure 1e. Administration of Schumanniophyton magnificum (100 and 400 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days has no significantly (P < 0.05) increased in brain GSH concentration compared with the ketamine-treated group. Treatment with standard drugs risperidone (0.5 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) groups does not show any significant (P < 0.05) increase in GSH concentration compared with the ketamine-treated group.
Effect of Schumanniophyton Magnificum on Catalase (CAT) Activity in Mice Brain
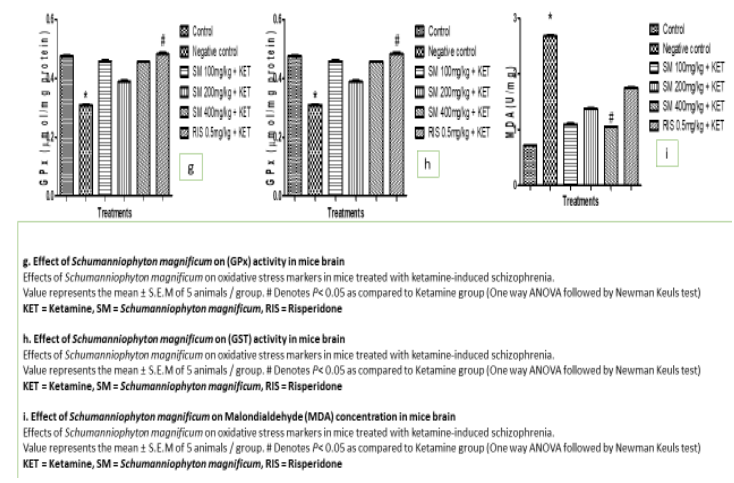
The effect of ketamine administered alone or in combination with Schumanniophyton magnificum on the activities of CAT in the brain of mice was shown in Figure 1 f. Administration of Schumanniophyton magnificum (100, 200, and 400 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days significantly (P < 0.05) increased brain CAT activity compared with the ketamine-treated group. Also (atypical antipsychotic drug) Risperidone (0.5 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days significantly (P < 0.05) increased brain CAT activity compared with the ketamine-treated group. 5.7 Effect of Schumanniophyton Magnificum on GPx Concen¬tration in Mice Brain The effect of ketamine administered alone or in combination with Schumanniophyton magnificum on the concentration of GPx in the brain of mice is shown in Figure 1g. Administration of Schumanniophyton magnificum (100, 200 and 400mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days has no significantly (P < 0.05) increased in brain GPx concentration compared with the ketamine-treated group. Treatment with standard drugs Risperidone (0.5 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) groups only show a significant (P < 0.05) increase in GPx concentration compared with the ketamine-treated group.
Effect of Schumanniophyton Magnificum on GST Concen-tration in Mice Brain
The effect of ketamine administered alone or in combination with Schumanniophyton magnificum on the concentration of GST in the brain of mice is shown in Figure 1h. Administration of Schumanniophyton magnificum (100mg/kg, o.p) with ketamine (10 mg/kg, i.p.) daily for 14 days only has significantly (P < 0.05) increased in brain GST concentration compared with the ketamine-treated group, while treatment of Schumanniophyton magnificum (200 and 400mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days has no significantly (P < 0.05) increased in brain GST concentration compared with the ketamine-treated group. Also treatment with standard drugs Risperidone (0.5 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) groups with no significant (P < 0.05) increase in GST concentration compared with the ketamine-treated group.
Effect of Schumanniophyton Magnificum on Malondialde¬hyde (MDA) Concentration in Mice Brain
The effect of Schumanniophyton magnificum of different doses (100, 200 and 400 mg/kg, o.p.), antipsychotic standard drugs with ketamine (10 mg/kg. i.p.) and ketamine alone on lipid peroxidase based on MDA content in the brain of mice was shown in Figure 1. Administration of Schumanniophyton magnificum (400 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) daily for 14 days significantly (P < 0.05) decreased lipid peroxidation as evident in the MDA content compared with the ketamine-treated group. Treatment with (200 and 400 mg/kg, o.p.) and standard drugs Risperidone (0.5 mg/kg, o.p.) with ketamine (10 mg/kg, i.p.) groups for 14 days does not significantly (P < 0.05) decreased lipid peroxidation as evident in the MDA content compared with the ketamine-treated group.

Discussion
The investigation of cognitive dysfunctions and behavioral despondency induced by pharmacological substances, especially ketamine, has gained considerable attention in recent times. This research delves into the complex interactions between cognitive performance and behavioral responses in animal models, with a particular focus on the opposing effects of ketamine and the possible therapeutic benefits of Schumanniophyton magnificum and risperidone.
Cognitive deficits are often assessed using the spontaneous alternation test, which is commonly employed to evaluate hippocampal damage in transgenic mice and the impact of drugs on cognitive abilities. Meanwhile, the recognition memory test helps to assess memory functions in mice [7,8,12]. Mice exhibiting a lack of recollection for recently visited arms show a decrease in spontaneous alternation, a potential indicator of memory loss. Memory loss is a hallmark of psychosis, and as the condition worsens, it impairs other cognitive functions, such as tool use. Several brain regions, including the hippocampus, septum, basal forebrain, and prefrontal cortex, are involved in these tasks. A study by Wolf et al. outlines a comprehensive set of behavioral tests aimed at evaluating multiple cognitive domains, including working memory, object discrimination, spatial memory, learning, and episodic-like memory in two dementia mouse models on a 129S6 background [12]. The results showed that although the 129S6/Tg2576 mice had significant impairments in spatial memory and episodic-like learning, they did not exhibit substantial deficits in novel object recognition or the spontaneous alternation task. This raises concerns about the applicability of specific cognitive assessments across different mouse models, indicating that the 129S6 background strain may require tailored testing methods. These findings have significant implications for understanding cognitive deficits linked to hippocampal damage, highlighting the need for customized approaches that accurately reflect the cognitive profiles of diverse transgenic mouse strains. A more precise assessment of cognitive impairments in animal models can greatly aid in evaluating the effects of pharmacological interventions and understanding the progression of memory disorders [13].
The exploration of the behavioral and anti-oxidative properties of Schumanniophyton magnificum's ethanolic extract provides an essential foundation for understanding its pharmacological potential. By administering the extract orally to mice, the study seeks to identify the mechanisms that underpin its therapeutic effects. This investigation not only underscores the importance of empirical research in validating traditional plant-based treatments but also emphasizes the necessity for rigorous scientific evaluation of the plant's phytochemical components and their respective biological actions.
Building upon this research, the study reveals that Schumanniophyton magnificum demonstrates promising effects in reversing cognitive deficits caused by ketamine administration. Treatment with Schumanniophyton magnificum, at doses of 100 and 400 mg/ kg, notably improves cognitive impairments observed in Y-maze tests, showing effects comparable to risperidone at a dose of 0.5 mg/kg. This comparison highlights the potential of Schumanniophyton magnificum as a therapeutic agent, similar to risperidone, in mitigating the cognitive alterations induced by ketamine. In synthesizing these findings, this study examines the neuro-behavioral dynamics between ketamine, Schumanniophyton magnificum, and risperidone, assessing how these substances influence cognitive performance and behavior.
The research by Adase et al. offers a platform for more in-depth investigations into the pharmacological profile of Schumanniophyton magnificum, contributing to the broader discussion on the significance of ethnobotanical knowledge in modern therapeutic practices [13,14]. Additionally, research on (R)-ketamine emphasizes its ability to cause cognitive impairments and behavioral despair during a 14-day treatment period. The observed decline in performance on Y-maze tests, a key measure of non-spatial working memory deficits, is central to understanding the adverse cognitive effects of ketamine and sets the stage for exploring potential therapeutic solutions [7,8,15]. The growing body of literature on Schumanniophyton magnificum offers compelling insights into its diverse roles in health management while calling for further research to unlock its full therapeutic potential in contemporary medicine [3,16].
Conclusion
In summary, this research reveals a compelling insights surrounding the neuroprotective properties of Schumanniophyton magnificum, particularly in relation to its ability to avert oxidative stress and neuronal damage [15]. The insights provided serve as a foundation for further exploration of this promising compound, suggesting that it may not only reduce the symptoms of psychosis but also enhance cognitive function in affected individuals. This emerging evidence positions Schumanniophyton magnificum as a significant area of interest for future research and therapeutic development. Statement and Declaration:
Competing interests: Authors de-clared and disclosed no financial or non-financial competing in-terests.
Funding Declaration
There was no funding to be declared, the work was funded by the authors.
References
- Tsapakis, E. M., Diakaki, K., Miliaras, A., & Fountoulakis,K. N. (2023). Novel compounds in the treatment of schizophrenia—a selective review. Brain Sciences, 13(8), 1193.
- Moghaddam, A. H., Estalkhi, F. M., Jelodar, S. K., Hasan,T. A., Farhadi-Pahnedari, S., & Karimian, M. (2024). Neuroprotective effects of alpha-pinene against behavioral deficits in ketamine-induced mice model of schizophrenia: Focusing on oxidative stress status. IBRO Neuroscience Reports, 16, 182-189.
- Joshua, P. E., Anosike, C. J., Asomadu, R. O., Ekpo, D.E., Uhuo, E. N., & Nwodo, O. F. C. (2020). Bioassay-guided fractionation, phospholipase A2-inhibitory activity and structure elucidation of compounds from leaves of Schumanniophyton magnificum. Pharmaceutical biology, 58(1), 1078-1085.
- Hague, J. (2022). Neuropsychopharmacology: An Overview. 5 (2013), 4583.
- Ardila, A. (2007). Normal aging increases cognitive heterogeneity: analysis of dispersion in WAIS-III scores across age. Archives of Clinical Neuropsychology, 22(8), 1003-1011.
- 6 Aminoff, E. M., Clewett, D., Freeman, S., Frithsen, A., Tipper, C., Johnson, A., ... & Miller, M. B. (2012). Individual differences in shifting decision criterion: A recognition memory study. Memory & Cognition, 40, 1016-1030.
- Mumby, D. G., Gaskin, S., Glenn, M. J., Schramek, T. E., & Lehmann, H. (2002). Hippocampal damage and exploratory preferences in rats: memory for objects, places, and contexts. Learning & memory, 9(2), 49-57.
- Foyet, H. S., Hritcu, L., Ciobica, A., Stefan, M., Kamtchouing, P., & Cojocaru, D. (2011). Methanolic extract of Hibiscus asper leaves improves spatial memory deficits in the 6-hydroxydopamine-lesion rodent model of Parkinson's disease. Journal of ethnopharmacology, 133(2), 773-779.
- Shahamat, Z., Abbasi-Maleki, S., & Motamed, S. M. (2016). Evaluation of antidepressant-like effects of aqueous and ethanolic extracts of Pimpinella anisum fruit in mice. Avicenna journal of phytomedicine, 6(3), 322.
- Chatterjee, M., Ganguly, S., Srivastava, M., & Palit, G. (2011). Effect of ‘chronic’versus ‘acute’ketamine administration and its ‘withdrawal’effect on behavioural alterations in mice: implications for experimental psychosis. Behavioural brain research, 216(1), 247-254.
- Ennaceur, A. (2010). One-trial object recognition in rats and mice: methodological and theoretical issues. Behavioural brain research, 215(2), 244-254.
- Wolf, A., Bauer, B., Abner, E. L., Ashkenazy-Frolinger, T., & Hartz, A. M. (2016). A comprehensive behavioral test battery to assess learning and memory in 129S6/Tg2576 mice. PloS one, 11(1), e0147733.
- Zhang, S., Pu, Y., Liu, J., Li, L., An, C., Wu, Y., ... & Yan,W. (2024). Exploring the multifaceted potential of (R)-ketamine beyond antidepressant applications. Frontiers in Pharmacology, 15, 1337749.
- Adase, E., Ankutse, P., Kumadoh, D., Archer, M. A., Kyene,M. O., Yeboah, G. N., & Asamoah Agyare, D. O. (2022). A Review of Parquetina nigrescens (Afzel.) Bullock, A plant for traditional medicine: phytochemical and pharmacological properties. Evidenceâ?Based Complementary and Alternative Medicine, 2022(1), 6076707.
- Rotruck, J. T., Pope, A. L., Ganther, H. E., Swanson, A. B., Hafeman, D. G., & Hoekstra, W. (1973). Selenium: biochemical role as a component of glutathione peroxidase. Science, 179(4073), 588-590.
- Iwu, M. M. (1993). African medicinal plants. CRC Press, Maryland. Jamkhande, PG, Ajgunde, BR, & Jadge, DR (2017). Annona cherimola Mill.(Custard apple): A review on its plant profile, nutritional values, traditional claims and ethnomedicinal properties. Oriental Pharmacy and Experimental Medicine, 17(3), 189-201.



