Case Report - (2024) Volume 3, Issue 1
Intrapulmonary Lymph Nodes Positive for Metastasis, In A Typical Multifocal Neuroendocrine Tumor: Case Report
2Thoracic Surgeon, Pablo Tobon Uribe Hospital, Colombia
Received Date: Jun 03, 2023 / Accepted Date: Jan 18, 2024 / Published Date: Jan 26, 2024
Copyright: ©©2024 Habib Mantilla Gaviria, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Gaviria, H. M., Ruiz, W. C. (2024). Intrapulmonary Lymph Nodes Positive for Metastasis, In A Typical Multifocal Neuroendocrine Tumor: Case Report. J Surg Care, 3(1), 01-03.
Abstract
Background: Multifocal neuroendocrine tumors of the lung are rare, usually having low nodal involvement in early stages. We bring to the literature a case of a typical multifocal carcinoid associated to tumors with intrapulmonary lymph node (IPLN) involvement.
Case Presentation: 59-year-old female asymptomatic patient with multiple nodules in the right lower lobe, PET-CT with hypermetabolic nodule, adequate pulmonary function, for which a right lower lobectomy was performed plus intrapulmonary lymph nodes dissection in the fresh surgical specimen. The lymph nodes of stations 11, 12, 13 and 14 were sent to pathology. The pathology laboratory report showed that stations 10, 11, 12 and 13 were negative, but station 14 was positive. Thus, changing the stage from Ia2 to IIb.
Conclusion: This patient had an upstaging because of IPLN were positive for metastasis. The search for a more accurate oncologic staging through intrapulmonary lymph node may allow a more accurate staging and therefore a better treatment for the patient regardless type histology.
Keywords
Pulmonary Neuroendocrine Tumor, Intrapulmonary Lymph Nodes, Lymphadenectomy, VATSList Of Abbreviations
IPLN: Intrapulmonary lymph node
NETs: Neuroendocrine tumors
TC: Typical carcinoids
AC: Atypical carcinoids
OS: Overall survival
CT: Computed tomography
RLL: Right lower lobe
VATS: Video-assisted thoracoscopic surgery
Background
Neuroendocrine tumors (NETs) of the lung have an incidence of 0.35 to 1.62 per 100,000 inhabitants. The most frequent age is between 48 and 70 years, and there is no relationship with smoking [1].
NETs can be typical carcinoids (TC), atypical carcinoids (AC) and neuroendocrine carcinomas such as small and large cell car-cinomas. Nodal metastases occur up to 17% in N1 stations in TC and 37.5% in AC. This defines the risk of locoregional recur- rence and overall survival (OS). Current reports do not specify the N1 stations involved, especially 12, 13 and 14 [2].
Current general practice does not include routine IPLN screen-ing in pathology laboratories, creating a group of patients with understaging if only stations 12, 13, or 14 are positive. We present the first case in the literature of multifocal NET with IPLN dissection in search of metastatic lymph nodes, which determined N1 positive and understaging if they had not been explored. This makes it possible to group patients into more pre-cise stages and therefore offer them more appropriate treatment.
Case Presentation
A 59-year-old female asymptomatic patient with a history of ar¬terial hypertension, grade I obesity, prediabetes with HbA1c of 6.1%, CHILD A liver cirrhosis and thrombocytopenic purpura. During controls with hepatology, a thoracic computed tomogra¬phy (CT) scan was performed, and 3 pulmonary nodules were found in the right lower lobe (RLL) (Figure 1).
Figure 1: Chest CT: 13mm Pulmonary Nodule (green arrow).
One of 13mm with central position, and others of 9- and 6-mm peripherals, without mediastinal adenopathy’s. PET-CT hypermeta- bolic lesion with SUV: 3.88 corresponding to the 13mm nodule (figure 2).
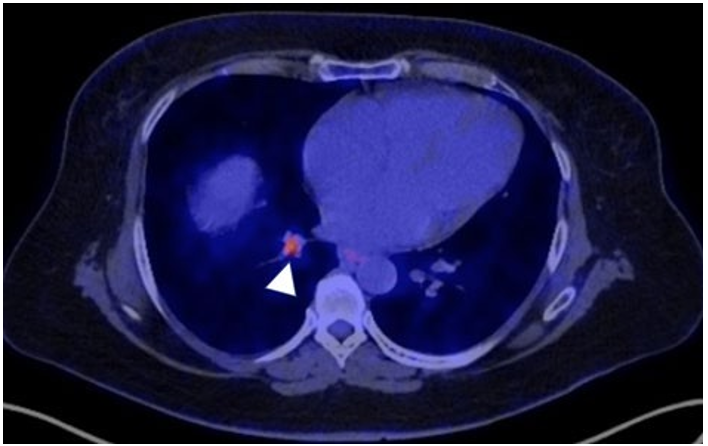
Figure 2: PET-CT: 13mm Pulmonary Nodule with SUV 3.88 (white arrowhead)
Spirometry with FEV1 126% and DLCO 124%. The patient is considered a candidate for right lower lobectomy by Video-as-sisted thoracoscopic surgery (VATS). VATS wedge resection sin-gle-port technique of the 9mm nodule was performed and sent to freezing, reporting a typical carcinoid tumor. Considering that the hypermetabolic lesion is central and the high probability of malignancy, the right lower lobectomy is completed with medi-astinal lymph node dissection and intrapulmonary node dissec-tion (stations 12, 13 and 14) on the fresh specimen, immediately after completing the procedure.
Intrapulmonary Lymph Node Dissection Technique
Starting from the lobar bronchus distally, following the peri bronchial route, obtaining the lymph nodes of stations 12, 13 and 14. Which are then sent to pathology duly labeled.
Pathology Report
Typical multifocal neuroendocrine tumors without lympho-vas-cular or perineural invasion of 9mm in upper segment (sample sent to freezing), another one of 14 mm and a tumor of 4 mm. 29 mediastinal nodes without tumor involvement and 1 of 6 nodes involved by tumor (metastasis of 2mm) in N1, being the nodal station 14R.
Analyzed in the chest staff, concluding a stage IIB (T1bN1M0 multifocal) with complete lobar resection and follow-up is de¬fined. The patient gives informed consent for publication of her case.
Conclusion
Nodal involvement of NET is infrequent. Our case has a multi¬focal NET and IPLN (station 14) metastatic component. There is no report in the literature of this type of presentation in multi¬focal NET. Most of the studies include only adenocarcinomas or squamous cell carcinoma for this reason we consider it import¬ant to contribute our case to the current literature [3].
It has been described that pathology reports do not inform in-trapulmonary nodal status up to 90%, this may leave a gap in oncologic staging, that is why it is important to investigate the behavior of IPLN [4].
Dr. Osarogiagbon R. describe the IPLN dissection with the lung lobe in formaldehyde and following the peribronchial pathway. In our case we decided to perform the dissection in the RLL in the operating room once the specimen has been extracted, considering that the histopathological evaluation of these lymph nodes is not routinely performed in pathology laboratories in our country [5].
Different authors have demonstrated “upstaging” when analyz¬ing IPLN histology. Lei be et al. published a study in 2021 with 234 patients, identifying lymph node metastases in stations 12, 13 and 14 between 6.8% and 12% [6].
If we had not dissected the IPLN, the patient's staging would be lower. In 2021 Pathipati MP et al, published a study with 98 patients with 16.6% of metastases in N1 and showed a higher percentage of recurrence (29% vs 6%; p:0.01%) associated with lymph node metastases, being higher in NER/CT (11/NER, 8/ CT and 3/CA) [7].
These studies do not specify which stations were positive. Wang et al.(25) conducted a study with 435 patients (170 study group in which stations 10-14 were routinely examined vs 265 in the control group in which only stations 10-12 were examined), found that the study group had better OS and DFS (5-year OS rate, 89 ± 3% vs. 77 ± 4%, P= 0.027; 5-year DFS rate, 81 ± 4% vs. 67 ± 4%, P= 0.021), in its multivariate analysis, the collec-tion of IPLN 13 and 14 had a significant impact on OS [hazard ratio (HR), 0.518; 95% confidence interval (CI), 0.298–0.898] and DFS (HR, 0.590; 95% CI, 0.387–0.901). This is just one case report on the metastatic involvement of IPLN in a multifo¬cal NET and it is interesting to perform the dissection of stations 12, 13 and 14 because they have shown an important role in staging and OS. This is a reminder to readers about the presence of IPLNs and that their metastatic involvement may be present. Due to de lack of information in NET and IPLN we have to ex¬plored more this area.
Patient Perspective
The patient did not want to give us an opinion.
References
- Dasari, A., Shen, C., Halperin, D., Zhao, B., Zhou, S., Xu, Y., ... & Yao, J. C. (2017). Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA oncology, 3(10), 1335-1342.
- Mineo, T. C., Guggino, G., Mineo, D., Vanni, G., & Am-brogi, V. (2005). Relevance of lymph node micrometastases in radically resected endobronchial carcinoid tumors. The Annals of thoracic surgery, 80(2), 428-432.
- Zhang, D., Chen, X., Zhu, D., Qin, C., Dong, J., Qiu, X., ... & Tang, X. (2019). Intrapulmonary lymph node metastasis is common in clinically staged IA adenocarcinoma of the lung. Thoracic Cancer, 10(2), 123-127.
- Ramirez, R. A., Wang, C. G., Miller, L. E., Adair, C. A.,Berry, A., Yu, X., ... & Osarogiagbon, R. U. (2012). Incomplete intrapulmonary lymph node retrieval after routine pathologic examination of resected lung cancer. Journal of clinical oncology, 30(23), 2823-2828.
- Osarogiagbon, R. U., Eke, R., Sareen, S., Leary, C., Coleman, L., Faris, N., ... & Spencer, D. (2014). The impact of a novel lung gross dissection protocol on intrapulmonary lymph node retrieval from lung cancer resection specimens. Annals of diagnostic pathology, 18(4), 220-226.
- Bi, L., Zhang, H., Ge, M., Lv, Z., Deng, Y., Rong, T., & Liu,C. (2021). Intrapulmonary lymph node (stations 13 and 14) metastasis in peripheral non-small cell lung cancer. Medicine, 100(27).
- Pathipati, M. P., Yohannan, T. K., Tian, L., Hornbacker, K., Benson, J. A., Berry, G. J., ... & Padda, S. K. (2021). Examination of factors associated with lymph node metastases in lung carcinoids: Results from a single institution retrospective cohort study. Lung Cancer, 154, 186-194.