Research Article - (2020) Volume 5, Issue 1
Innovative Approach to Vaginal Drug Delivery
2School of Medicine, University of California, San Diego, USA
Received Date: Feb 02, 2020 / Accepted Date: Feb 18, 2020 / Published Date: Feb 26, 2020
Copyright: ©Shihata Alfred A. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: The vagina is an ideal site for drug delivery, particularly for those drugs associated with women’s reproductive health. For years traditional applicators have been the primary method used to deliver therapeutic creams into the vagina. The applicator’s lack of efficacy is mainly attributed to the vagina’s rhythmic muscular contractions, which naturally flush out any substances from its canal. As a self-cleaning organ; the vagina not only expels cervical secretions and menstrual blood, but also any therapeutic creams, which will render them less effective.
Objectives: To provide women with an alternative to vaginal applicators, which will increase the time of contact between the therapeutic vaginal preparations and the cervix. Explore the possibility of topical treatment of Bacterial Vaginosis and Candida infections using a new cervical barrier called FemCap; where it is used as a delivery system for antibacterial and antifungal preparations.
Methodology: To prove this concept we recruited 30 women to insert a stained gel (Gentian Violet dye) with a vaginal applicator and another 30 women to insert the same gel with the FemCap. We then compared the retention and distribution of the stained gel over the cervix and we photographed the cervices at 12 and 24 hours.
Results: None of the participants reported any side effects using the stained gel with either device. Women reported leakage while using the vaginal applicator and did not have any with the FemCap. Women who used the applicator had no visible stained gel over the cervix after 12 hours of insertion. Cervical photographs have shown that the FemCap participants retained the stained gel on the cervix more than 24 hours after insertion.
Conclusion: This pilot study has proved the concept that FemCap is more efficient than the vaginal applicator in delivering vaginal preparations to cervix and vagina. The FemCap can shield the cervix from sperm penetration and potentially from STI organism’s invasion. Further studies should be conducted to explore the possibility of treating Bacterial Vaginosis, Candida topically and some STIs.
Keywords
Bacterial Vaginosis, Candida, FemCap Delivery System, Showerhead Applicator
Introduction
The vagina is ideal for drug delivery, however physiologically the vagina provides several obstacles. The main obstacle is the natural, physiologic, rhythmic contraction of the vagina that flushes out menstrual fluid, cervical secretions and any therapeutic creams to keep the vagina clean and, empty. Many women misdiagnose and treat any vaginal discharge as a yeast infection with an over the counter antifungal cream. This can delay the proper diagnosis, waste a lot of money and pollute the environment.
The advantages of localized topical treatment for vaginal and cervical infections are as follows:
• Use of a smaller dose for a shorter period, which would be more effective than a larger systemic dose and have less side effects
• Avoid oral digestive enzymes that may degrade the medication or bypass the liver’s metabolism
• Under the control of the user
• Does not interfere with intercourse
Materials
To overcome the expulsion problem of the traditional applicator (Figure 1), we set out on a quest for better retention and distribution of therapeutic cream. We then developed the showerhead applicator. This applicator has a rounded tip with multiple perforations (Figure 2a & 2b). Unfortunately, the vagina could still expel the bulk of the cream inserted by the showerhead applicator.

Figure 1: Traditional Applicator Figure 2a: Showerhead Applicator Figure 2b: Showerhead Applicator Demo
The FemCap is a new contraceptive device (Figure 3, 3a, 3b) and is designed with a unique delivery system for any vaginal preparations such as gels or creams. These preparations can be applied on the cervical and vaginal sides of the FemCap (Figure 3b). The goal of this design is to allow for better adherence, coverage, distribution and retention of any preparation for up to 24 hours.
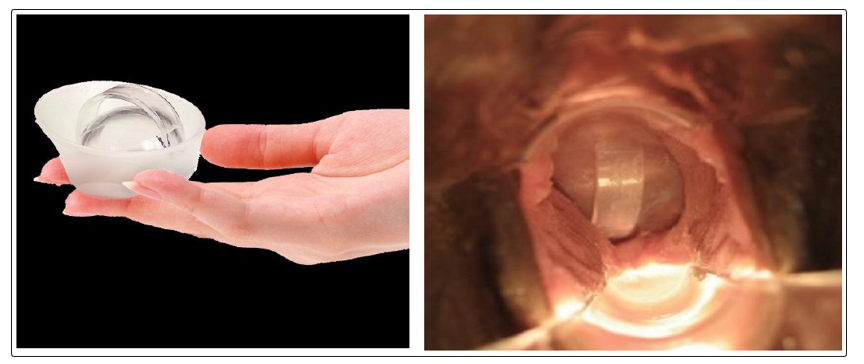
Figure 3: The FemCap Figure 3a: FemCap Covering

Figure 3b: The FemCap Vaginal & Cervical Side
Methods
Participants selected for this study were ages 18 to 40, were not pregnant and did not desire to be pregnant during the 4-week study duration. All subjects were free from sexually transmitted infections and reported being sexually active. Forty-five of the participants (75% of whom were sterilized and 25% were on hormonal contraception) signed an informed consent, underwent a complete physical and pelvic exam, as well as a pap smear and wet mount. Women who have had an STI, vaginitis or an abnormal pap smear were excluded.
Thirty women (Group A) were randomly assigned to use the vaginal applicator (Figure 1) and the other 30 women (Group B) were assigned to use the FemCap (Figure 3) to deliver a high viscosity vaginal lubricant stained with Gentian violet dye to enhance visibility. After one week of wash over group (A) then crossed over and used the FemCap and group (B) used the vaginal applicator for two additional weeks. Participants were instructed to record and report the use of the vaginal applicator and the FemCap and report any side effects on a calendar.
This facilitated a comparison between and compliance with the vaginal applicator to deliver a vaginal lubricant stained with Gentian violet and with the FemCap to deliver the same stained lubricant. We provided participants with hygienic pads to monitor the expulsion of the stained gel and instructed them to bring the pads to the clinic for inspection.
We swabbed each participant’s vagina and introitus for the presence or absence of the stained lubricant at 12 and 24 hours after insertion using both devices. In addition, we photographed the cervix at 12 and 24 hours. Finally, the investigators conducted in-depth interviews with each of these 60 women. This communication will help us gain insight to better treat vaginitis and possibly STIs topically in the future.
Results
None of the participants reported any significant side effects using either device. Naked eye examination did not show any significant lesions over the cervix or the vagina. Women did not report any leakage while wearing the FemCap and the photograph of their cervix shows the presence of the stain over the cervix 24 hours after insertion (Figure 4). Women who used the vaginal applicator reported increased leakage of the stained lubricant and the photograph shows the absence of the stain 12 hours after insertion (Figure 5).
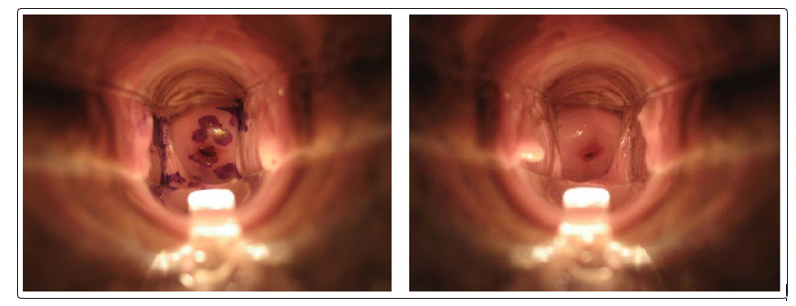
Figure 4: FemCap 24hr after insertion Figure 5: Vaginal Applicator 12hr after insertion
Table 1: Comparison between distribution and retention of the stained vaginal lubricant
|
After Application |
FemCap Delivery System Stained vaginal lubricant with Gentian Violet applied with FemCap |
The Vaginal Applicator Stained vaginal lubricant with Gentian violet applied with the vaginal applicator |
|
Cervix |
Stain present at 12 & 24 hours (Figure 4) |
Absent at 12 & 24 hours (Figure 5) |
|
Upper Vagina |
Stain present with many bare spots at 12 & 24 hours (Figure 4) |
Absent at 12 & 24 hours (Figure 5) |
|
Introitus |
Absent at 24 hours |
Absent at 12 & 24 hours Conclusion |
Conclusion
This pilot study did prove the concept that the FemCap, when used as a delivery system for therapeutic vaginal gels or creams, would be much more efficient than the traditional vaginal applicator. This is due to the better distribution and retention of the vaginal creams as shown by the photograph of the cervix 24 hours after insertion. This study raised the possibility of topical treatment for common vaginitis such as Bacterial Vaginosis and Candidiasis. The use of the FemCap can also prevent pregnancy and thus will enhance compliance. Further studies are warranted, to ensure safety, effectiveness and acceptability of the FemCap delivery system.
Acknowledgement
We are indebted to the women who consented to participate in this research study, and to Dr. Steven Brody who co-authored and edited this manuscript. No funding was requested or received from any institution. The authors are responsible for the content of this article.
References
- Dobaria, R Mashru, NH Vadia (2007) Vaginal drug delivery systems: A Review of Current Status. East and Central African Journal of Pharmaceutical Sciences 10: 3-13.
- Chinmaya Keshari Sahoo, Prakash Kumar Nayak, Deepak Kumar Sarangi, Tanmaya Keshari Sahoo (2013) Intra Vaginal Drug Delivery System: An Overview. American Journal of Advanced Drug Delivery 8522: 43-55.
- Janet G Vail, Jessica A Cohen, Kimberly L Kelly (2004) Improving Topical Microbicide Applicators for Use in Resource-Poor Settings. American Journal of Public Health 94: 1089-1092.
- Pudney J, Quayle AJ, Anderson DJ (2005) Immunological microenvironments in the human vagina and cervix: mediators of cellular immunity are concentrated in the cervical transformation zone. Biol Reprod 73: 1253-1263.
- Carcio H, Clarke Secor M, Koeniger-Donohue R (2010) Advanced Health Assessment of Women: Clinical Skills and Procedures Chapter 15 The FemCap. Springer Publishing Company 271-278.
- Koeniger-Donohue R (2006) The FemCap a Non-Hormonal Contraceptive. Women’s Health Care NPWH 5: 79-91.
- Shihata A (1998) The FemCap™, a new contraceptive choice.Eur J Contracept and Reprod Health Care 3: 160-166.
- Shoupe D, Kjos S (2006) The Handbook of Contraception, Barrier Contraceptives Chapter 10 Humana Press 147-177.
- Shihata A (2007) Microbicides Delivery System. 5th European Congress on Tropical Medicine and International Health. Proceedings Amsterdam May 24-28. 131-134.
- Shihata A (2007) Novel Delivery System for Microbicides. Presented in 2007 National HIV Prevention Conference 378.
- Shihata A (2004) New FDA approved woman-controlled, latex-free barrier contraceptive device “FemCap™”. Fertil Steril. International Congress Series 1271. 103: 303-330.
- Shihata A (1992) Gollub E Acceptability of a new intravaginal barrier contraceptive device FemCap. Contraception 46: 511-519.
- Ariane van der Straten, Nuriye Sahin-Hodoglugil, Kate Clouse, Sibongile Mtetwa, Mike Z Chirenje (2010) Feasibility and potential acceptability of three cervical barriers among vulnerable young women in Zimbabwe. J Fam Plann Reprod Health Care 36: 13-19.
- Shihata A, Brody S (2010) HIV Prevention by Enhancing Compliance of Tenofovir Microbicide. Using a Novel Delivery System. HIV &AIDS Review 9: 105-108.


