Research Article - (2019) Volume 4, Issue 1
Hypertension and Takayasu Disease
2Professor of Vascular Surgery, Oran, Algeria
Received Date: Dec 03, 2018 / Accepted Date: Feb 01, 2019 / Published Date: Feb 13, 2019
Copyright: ©BENALLAL Souad. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Takayasu’s disease (TD) is a primary, inflammatory, rare, segmental and multifocal arteritis of the aorta and its branches, of unknown etiopathogenesis. Hypertension is a major complication, sometimes revealing the disease, whose mechanisms are multiple, dominated by the lesion of the aorta and renal arteries.
Patients and methods: We report a series of 63 cases treated for Takayasu’s disease resulting from a monocentric, retrospective study from April 2oo6- July 2014, with an average age of 30 years with a female predominance of 83%.
Result: In our series: 51 patients with hypertension, including 29 (57%) resistant hypertension (under tri see quadritherapy). In 87% of cases lesion of the supra aortic trunk, 29% with aortic lesions and renal arteries, 7.93% with isolated renal arteries and 7% with aortic coarctation cases, valvular heart disease in 7% of cases. Association with tuberculosis in 7.93% of cases and a dysthyroidism in 11.1% of cases. That 49% benefited from surgical treatment.
Discussion: Involvement of the aorta and renal arteries seen in half of patients with TD is the leading cause of hypertension. In general, hypertension is severe, not or poorly controlled by medical treatment and is accompanied by cardiac and renal repercussions.
Conclusion: Hypertension is common during TD; it is a poor prognostic factor, more serious than the cause is renovascular. Early diagnosis and appropriate therapy will help to prevent a handicap, which is sometimes major for these patients, most of whom are young. Surgical treatment of renal and aortic lesions of TD is indicated when there is severe hypertension, not or poorly controlled by heavy medical treatment, and the indications should be weighed well. The spontaneous evolution in these patients is burdened with a not insignificant mortality. Management and therapeutic decisions must be multidisciplinary.
Keywords
Takayasu disease, hypertension, renal angioplasty, renovascular hypertension, inflammatory arteritis, non-specific aorto-arteritis.
Abbreviations
EIA: external iliac artery; CIA: common iliac artery; AT: arteritis of Takayasu; TLA: transluminal angioplasty; Hypertension: Hypertension; IDR: intradermal reaction; NIPH: National Institute of Public Health; TD: Takayasu disease; BP: blood pressure; TBC: tuberculosis; SAT: supra-aortic trunk; MI: mitral insufficiency; AOI: aortic insufficiency.
Introduction
Described in 1908, Takayasu’s disease is an idiopathic systemic vasculitis affecting large vessels, the aorta and its main branches (supra-aortic trunks, carotid arteries and subclavian arteries, renal arteries, but also coronary and pulmonary arteries) [1]. The injury may be segmental or diffuse to the entire thoracic and abdominal aorta [2].
It is an inflammatory arteriopathy of the young subject, most often occurring between the ages of 20 and 40, predominantly female, hence its definition of the disease of women without pulse [3].
It is a medio-adventitious giant cell granulomatosis [4].
Clinical manifestations are most often related to vascular involvement, either related to arterial inflammation or related to consequences such as stenos is or thrombosis. The mechanisms of vascular obstruction are different from atherosclerosis
Hypertension is most often related to the involvement of one or more renal arteries.
A classification into account the arterial locations are the types I to V [5]:
• Type I: involvement of the aortic arch,
• Type IIa: involvement of the Ascending aorta, aortic arch and its branches,
• Type IIb: involvement of the ascending aorta, aortic arch and its branches, descending thoracic aorta,
• Type III: involvement of the Descending thoracic aorta, abdominal aorta and / or renal arteries,
• Type IV: involvement of the Abdominal aorta and / or renal arteries,
• Type V: IIb and IV.
The aim of our study is to investigate the reno-aortic involvement of takayasu disease, to evaluate the prevalence of hypertension during TD, to assess the signs of gravity, the evolution of the disease over time and prognosis as well as the results of surgical and medical treatment.
Materials and Methods
A retrospective nonrandomized study in a single center was conducted from April 2006 to July 2014; sixty three (63) Patients were treated for Takayasu disease. These patients were admitted to vascular surgery department at Oran University Hospital November 1st 1954; validate the criteria of the American College of Rheumatology (ACR) [8]. Table 1
Table 1: American College of Rheumatology (ACR), Criteria for Classification of Takayasuâ??s Arteritis
|
1. Age at disease onset ≤ 40 years. |
|
2. Claudication of extremities. |
|
3. Decreased brachial artery pulse. |
|
4. Blood pressure difference > 10 mm Hg. |
|
5. Bruit over subclavian arteries or aorta. |
|
6. Arteriogram abnormality. |
|
N.B : A diagnosis of Takayasu’s arteritis requires that at least 3 of the 6 criterias are met. |
We received patients referred by internal medicine or cardiology department for the management of hypertension resistant to medical treatment with vascular lesions.
Para clinical exploration were done in all our patients: Chest X Ray, duplex ultrasound of supra aortic trunk, renal arteries, lower and upper limbs, computed tomography, electrocardiogram, cardiac echography and a complete laboratory tests including erythrocyte sedimentation rate (ESR) at the time of diagnosis and its fluctuations, white blood cell (WBC), rheumatoid factor (RF), C-reactive protein (CRP) and tuberculin intradermal reaction was performed in 10 patients.
The classification of Lupi Herrera (Table 2) allowed to divide our patients according to the topography of their lesions
Table 2: Lupi-Herrera classification (1977)
|
Type |
Vessel involvement |
|
Type I |
isolated involvement of the aortic arch and its branches |
|
Type II |
involvement of the abdominal aorta and its branches |
|
Type III |
Type I and II lesions |
|
Type IV |
Lesions of pulmonary arteries added to such lesions I, II or III |
The statistical analysis was performed with IBM SPSS Statistics software 20 (IBM SPSS Inc., Chicago, IL).
Results
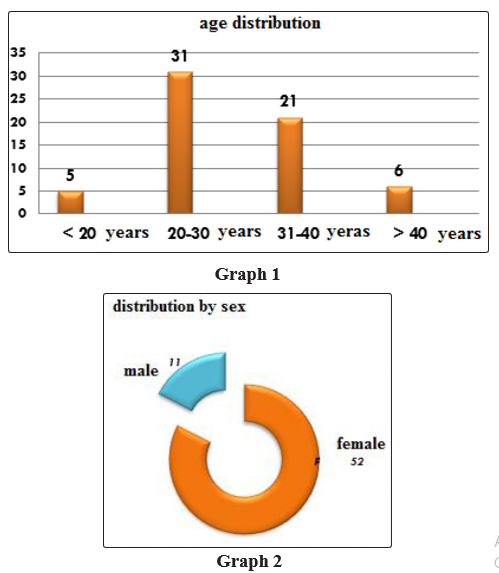
In our series, the mean age was 30 years with extremes from 12 to 64 years (graph No.1); with a female predominance sex ratio 5F/1H (graph No.2).
51 patients had hypertension, of which 29 (57%) were resistant (under/ tri or quadritherapy). The incidence of involvement of supra aortic trunk was found in 87% of cases (55 cases including 43 cases of bilateral lesions), the associated lesions of the aorta and the renal arteries in 29% of cases, isolated attack of the renal arteries in 7.93% of cases. , aorta coarctation in 8% of cases, involvement of the digestive arteries 02 cases, valvular heart disease in 7% of cases (04 cases of mitral insufficiency and 01 cases of aortic insufficiency). The lesional distribution of the disease is summarized in Tables 3. an association with tuberculosis in 5 cases (01cas intestinal TBC, 03 cases of pulmonary TBC and 01 cases of tuberculous adenitis), a Dysthyroidism in 7 cases, 01 cases of ankylosing spondylitis and 01 cases of systemic lupus erythematosus.
Table 3: Localization and type of lesions
|
Localization of lesions |
Nombers of cases |
Type of lesions |
|
Carotids |
19 |
05 bilateral stenosis 02 occlusions 05 stenosis<60% 07 unilateral stenosis |
|
Vertebrals |
22 |
Stenosis (bilaterale in 17 cases) |
|
Sub Clavian |
12 |
Stenosis/short occlusion |
|
Axillary |
02 |
Stenosis |
|
Thoracic aorta |
05 |
04 aortic Coarctation 01 aneurysm |
|
abdominal aorta |
02 |
01 lengthly stenosis 01 infrarenal aortic aneurysm |
|
Renal arteries |
23 |
08 isolated stenosis 17 associated with aortic lesions |
|
Digestive arteries |
02 |
Stenosis |
|
Iliac arteries |
02 |
01 stenosis 01Occlusion CIA (TASC A) |
|
Valvular Heart disease |
05 |
04 MI 01 AOI |
Corticosteroid therapy was instituted in all our patients at a dose of 1 mg/kg/day, immunosuppressants in 05 patients and antihypertensives. A dual anti-platelet agent in all patients benefited from endovascular treatment.
Only 31 patients (49%) of whom underwent surgical treatment, 9 patients responded well to medical treatment, while the others either lost sight of them or refused the procedure. The revascularization procedures are summarized in Table 4.
Table 4: revascularization procedures
|
TLA-STENT artères rénales |
05cas |
|
TLA Sous clavières |
07cas |
|
TLA Carotides |
01cas |
|
Thoracic aorta Endoprothesis |
02 cases for aortic coarctation |
|
Abdominal aortic Endoprosthesis for false aneurysm |
01 cases for aneurysm |
|
TLA-Stent CIA |
01 case |
|
Resection-anastomosis for aortic coarctation |
02cases |
|
Ventral aortic bypass |
02cases |
|
Aorto- carotid bypass |
02cases |
|
Aorto-renal bypass |
02cases |
|
Carotid endarterectomy |
02cases |
|
nephrectomy |
02cases |
In the follow up, we deplore 01 death on the first day secondary to hemorrhagic syndrome after ventral aortic bypass for “middle aortic syndrome” in a young woman of 23 years; 02 conversions after failure of angioplasty and 04 patients have degraded their renal function. Regarding hypertension, the immediate postoperative evolution was towards the total normalization for 07 (8.77%) patients and an improvement in blood pressure in 18 patients, for whom the antihypertensive treatment has been lightened. Finally persistence of hypertension in 02 patients.
Discussion
TD is an inflammatory, rare, segmental and multifocal primary arteritis of the aorta and its main branches. It represents 5% of vasculitis. It is more common in Asia, India, South America (Mexico, Latin America). In Europe, its incidence was estimated at 2.6 cases/ million/year [6]. In the Maghreb, the incidence is imprecise but a study conducted in 2009 collected 378 cases [7]. In Algeria, there have been no epidemiological studies, however several cases have been reported, revealed in particular by hypertension in the young subject (more than 279 cases of TD) [8].
Female dominance was in the majority in all studies, with a variable F/M ratio of 8/1 in Japan, 4.8/1 in France, 1.2/1 in India, 5.9/1 in Mexico [9]. In our series it was 5F/1M. The third decade of life is the period when the incidence of onset is highest, age <40 years is also a major criterion, but there may be about 20% of older patients at diagnosis. We had 06cas diagnosed> 40 years old, including 02> 60 years old.
The pathogenesis of Takayasu’s disease is still unclear. Nevertheless, several hypotheses are advanced. The infectious origin has been incriminated, especially its relations with tuberculosis (tbc).
In most series there is a high prevalence of confirmed tuberculosis or positive intradermal reactions (IDR), (05 cases of tbc in our series), but this is not the only cause, because association of dysimmunitary pathology with TD, gives it the character of autoimmune disease [[10- 12]. In our study we reported 01 cases of ankylosing spondylitis, 01 case of systemic lupus erythematosus and 07 cases of Dysthyroidism.
The disease evolves classically in two phases. The first, called “pre-occlusive”, is characterized by general signs and a biological inflammatory syndrome. The second, known as “occlusive”, results in the occurrence of ischemic clinical manifestations [9].
Hypertension is a serious public health problem, noted that worldwide approximately one billion, 978 million people with high blood pressure according to WHO “Lancet 2011,” first death risk factor.
In Algeria, its prevalence is more than 35% or 7 million Algerians including 21% deaths from cardiovascular diseases in 2010 (NIPH) [13].
In Takayasu’s disease, high blood pressure (hypertension) is a major complication, frequent, often revealing of the disease, dominated by the involvement of the renal arteries 30%, the thoracic or abdominal aorta above renal arteries (coarctation) and supra-aortic trunks inducing carotid bulb baroreceptor dysfunction and decreased cerebral blood flow [14, 15].
In general, hypertension is severe, not or poorly controlled by medical treatment, and is associated with cardiac and renal repercussions.
In our study, the disease was discovered during an HTA in 51 patients, of whom 27 cases had a malignant or resistant hypertension (under three /quadritherapy and poorly controlled). According to several studies, hypertension is considered one of the main factors of poor prognosis during TD [16-18].
In Algeria, the study of Mammeri et al. hypertension was severe in 54% of patients and responsible for 70% of deaths [8].
Mirault et al. reported a survival in Takayasu disease of 95% at 5 years and 91% at 10 years in France [19].
The main causes of death are heart failure (46%), the occurrence of stroke (10%) or renal impairment (11%) and postoperative complications (6.4%). Ruptures of arterial aneurysm were also responsible for deaths.
Scores were recently established to assess cardiovascular involvement during Takayasu’s disease. The “disease extend index in Takayasu arterirtis” (DEI-TAK) developed on the basis of the Birmingham Vasculitis Activity Score (BVAS) [20].
The diagnosis of this disease is based on a set of radio-clinical arguments (Table 2), including clinical status, arterial topography (Table.3), its character (stenosis, occlusion or aneurysm) and the association with cutaneous, cardiac and visceral lesions [21].
In our study we had associations of aortic aneurysm and stenosis of subclavian arteries (fig.1), multiple localizations of arterial stenosis (fig.2), as well as aortic stenosis associated with renal artery stenosis in 8 case (fig.3)
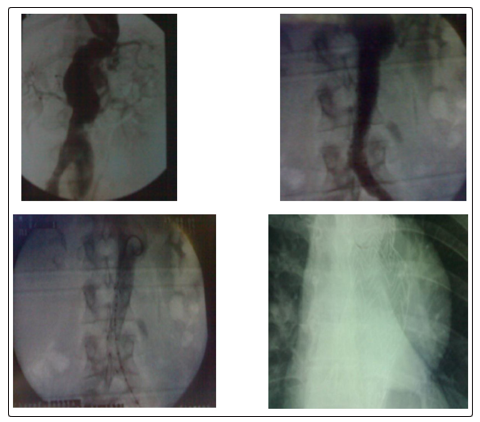
Figure 1: female, 35 years, married, Hypertension under tritherapy uncontrolled, with antecedents of tuberculous adenitis.
Lesions: Fusiform aneurysm of the descending thoracic aorta + sacciform aneurysm of the infrarenal abdominal aorta + stenosis of the left sub clavian artery.
Procedure: 1st time: Medical treatment: Corticotherapy + Anti- tuberculosis treatment
2nd time: Treatment of thoracic aneurysm with aortic endoprosthesis (Gore 40/19) dropped flush with the sub Clavian artery.
3rd time: Treatment of abdominal aortic false aneurysm
Follow up: good evolution, with improvement of blood pressure under dual therapy.
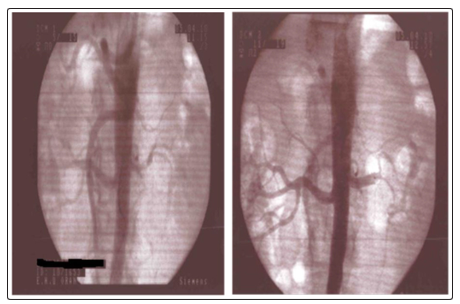
Figure 2: Female, 64 years old, with a medical history of primary infertility treated, (1 abortion). Hypertension for 10 years under triple therapy.
Lesions: Stenosis of the right Sub Clavian artery.
-Tight stenosis of the left renal artery.
- tight stenosis of the right EIA + stenosis of the hypogastric artery.
Gesture: Medical Treatment
-TLA of Sub clavian artery + TLA of renal artery + TLA of EIA.
Evolution: Good with normalization of blood pressure.
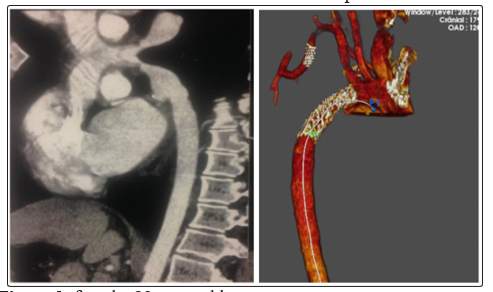
Figure 3: female, 23 years old, Hypertension under tritherapy + claudication of the right upper limb.
Lesion: aortic coarctation + tight stenosis of the sub clavian post vertebral artery.
Procedure: Thoracic endoprosthesis + TLA -Stent of sub clavian artery
Evolution: good, improvement of the blood pressure under monotherapy with a good right radial pulse.
New imaging tools such as computed tomography or magnetic resonance angiography, positron fludeoxy glucose computed tomography emission tomography and recently high contrast ultrasound is frequently used diagnostic and to evaluate vascular inflammation [22]. Biology has no specific test for MT and its only interest is to show an inflammatory syndrome. In our study 39 patients had a biological inflammatory syndrome [23].
The treatment of Takayasu’s disease is medical, according to data in the literature, corticosteroid therapy is the first-line treatment and if failure, the addition of methotrexate may stabilize the disease [24].
All our patients received corticosteroid therapy at a dose of 1 mg / kg / day, immunosuppressants, and more antihypertensive, as well as anti-tuberculosis treatment in 05 patients.
Biological agents such as anti-tumor, necrosis factor, tocilizumab and rituximab can be used effectively in refractory cases [25]. Surgical treatment of hypertension during Takayasu arteritis is indicated in case of failure of medical treatment.
Revascularization may be made by endovascular or open surgery. The latest 2009 EULAR recommendations on the treatment of vasculitis of large vessels reflect the low level of evidence (level 3) series of open surgery in the reported cases, hence the low grade (C) recommendations [26].
However, revascularization during the active phase may increase the risk of procedural complications [27], including restenosis, dissection, and anastomotic pseudoaneurysms; for this a medical treatment first is warranted before any surgical procedure or angioplasty (ATL). This was the case in all our patients.
(The criteria for activity and remission of the disease are shown in Table 5) [2].
Table 5: Disease Activity Criteria (NIH)
|
Recent appearance or aggravation of at least 2 of the following criteria: |
|
Signs of ischemia or vascular inflammation: limb claudication, decrease or abolition of a pulse, breath or vascular pain, asymmetric blood pressure |
|
Typical angiographic abnormalities |
|
Systemic symptoms such as fever, arthromyalgia |
|
Increase in sedimentation rate |
|
|
|
The criteria for remission are: |
|
Complete resolution or stabilization of all clinical signs |
|
Stable nature of vascular lesions |
Endovascular treatment of occlusive or aneurysmal lesions during MT is a minimally invasive, feasible, safe and effective procedure, and may be indicated as first-line. According to the literature excellent results have been reported ranging from 81% to 100% and low morbidity and mortality. [28]
Nevertheless, the success and results of balloon angioplasty alone are variable depending on the degree of stenosis and the length of the lesion, since the rate of restenosis could reach up to 80% [29] due to thickening of the wall and the parietal fibrosis, hence the implantation of a stent. Some studies have reported improved results in stenting [30, 31].
In our series, we have performed 04 TLA-stent of supra-aortic trunks, 03 TLA-STENT of subclavian, Endoprosthesis for aneurysm of the thoracic aorta and the abdominal aorta (Fig1.) 05 TLA -renal arteries (Fig.2), 02 ATL-Stent of iliac arteries, 01 Endoprosthesis for coarctation of the aorta (Fig.3) and TLA of the abdominal aorta and right renal artery for aortic hypoplasia, surgically taken after failure of the angioplasty (fig.4).
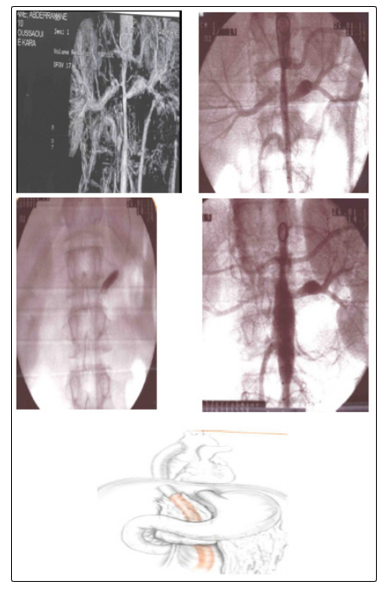
Figure 4: young male, 16 years old,
Hypertension uncontrolled under triple therapy.
Lesion: length tubular stenosis of the abdominal aorta and tight stenosis of the right renal artery.
Procedure: Medical Treatment. 1st time: abdominal endoprosthesis +angioplasty of the right renal artery
Evolution: failure of the endovascular treatment.
2nd time: conversion with aorto bi-iliac bypass from the ascending aorta
Evolution: good with stabilization of the blood pressure. The surgical treatment of our patients was in all cases motivated by the severity of hypertension that was resistant to medical treatment and the failure of angioplasty, either by difficulty of catheterization, dissection or restenosis.
We performed 02 bypass by ventral aorta, 02 aorto-renal bypass, 02 aorto-carotid bypass, 02 carotid endarterectomy, 02 resection- anastomosis for aortic coarctation and 02 nephrectomies.
Chaudhry suggests renal revascularization in all cases of renal artery stenosis ≥ 70% with or without renovascular hypertension and according to Kim et al. surgery is an effective method in patients with lesions of supra aortic trunk. This would avoid major complications such as stroke [32, 33].
Other therapeutic modalities are under study for the treatment of Takayasu arteritis, such as TNF inhibitors and active angioplasty by eluting drug balloons, but they have not yet proven effective. [34, 15]
The evolution and prognosis of the disease are related to complications. Park et al examined the influence of severe complications on survival rates [16]. The presence of more serious complications increases the mortality rate in five years (69.9%) and 10 years (36.7%).
Ishikawa and Maetini found that survival varies not only depending on the time of diagnosis, but also to the presence of major complications that included valvular heart disease, stroke, heart failure, retinopathy and renovascular hypertension [17].
In addition, ischemia and bleeding are also common causes of death in the TD [35]. The right time for surgery requires further investigation.
Overall, surgical results have been improved over time, aided by advances in surgical and endovascular techniques, as well as optimization of pre, peri and postoperative care. Early postoperative death has become rare in many centers.
Some cases are illustrated (Figures.1-2-3-4).
Conclusion
Hypertension is common in the TD. It is a factor of poor prognosis all the more serious that the cause is renovascular.
Early diagnosis and appropriate treatment will help to avoid certain handicaps in the future, sometimes major for these patients, mostly young.
Surgical treatment of renal aortic lesions is indicated when there is severe hypertension not controlled by heavy medical treatment.
The spontaneous evolution in these patients in fact burdened with a significant mortality, the same for operated patients whose disease can progress either locally or remotely, which may require multiple and successive interventions, for this reason the surgical indications must be carefully weighed.
The multidisciplinary medical-radiological-surgical approach for the diagnosis and management of patients with Takayasu arteritis is essential to obtain satisfactory results.
References
- Numano F, Kakatu T (1996) Takayasu arteritis –five doctors in the history of Takayasu arteritis. Int J Cardiol 54: S1-S10.
- Kerr GS, Hallahan CW, Giordano J, Leavitt RY, Fauci AS, et al. (1994) Takayasu arteritis. Annals of internal medicine 120:919-29.
- Fiessinger JN (1991) Aorto-artérite non spécifique (maladie de Takayasu). In: Kahn MF, Peltier AP, Meyer O, Piette JC, eds. Les maladiessystémiques. Paris: Médecine-Sciences Flammarion 713-726.
- Fiessinger JN, Camilleri JP, Chousterman M, Cormier JM, Housset E (1978) Maladie de Horton et maladie de Takayasu: critères anatomopathologiques. Deux observations. Nouv Presse Med 7: 639-42.
- Moriwaki R, Noda M, Yajima M, Sharma BK, Numano F (1997) Clinical manifestations of Takayasu arteritis in India and Japan--new classification of angiographic findings. Angiology 48: 369-379
- Mekinian A, Soussan M, Saadoun D, Gomez L, Comarmond C, et al. (2017) Management of Takayasu arteritis. Revue du rhumatisme monographies, Elsevier.
- Kechaou M, Frigui M, Ben Hmida M, Bahloul Z (2009) Maladie de Takayasu au sud tunisien : étude de 29 cas. Presse Med 38: 1410–1414.
- Mammeri A, Guermaz b, Hatri A, Kessal F, Zekri S, et al. (2017) Lâ??hypertension artérielle au cours de la maladie de Takayasu : à propos dâ??une série monocentrique de 279 patients. Annales de Cardiologie et d’Angéiologie 66: 154-158
- Arnaud L, Haroche J, Limal N, Toledano D, Gambotti L, et al. (2010) Takayasu arteritis in France. Medicine (Baltimore) 89: 1-17.
- Soto ME, Espinola N, Flores-Suarez LF, Reyes PA (2008) Takayasu arteritis: clinical features in 110 MexicanMestizo patients and cardiovascular impact on survival and prognosis. Clin Exp Rheumatol 26: S9-15.
- Nakao K, Ideda M, Nahtani H (1967) Takayasu„s arteritis. Clinical report on eighty-four cases and immunological studies in seven cases. Circulation 35: 141-155.
- Kinare SG, Gandhi MA, Deshpande JR (1998) Non-specific aortoarteritis (Pathology and Radiology) Mumbai: Quest Publications.
- Étude épidémiologique de lâ??hypertension, en Afrique du Nord: ETHNA, N°18 - Mai 2013 Santé-MAG 2. 11
- Lupi-Herrera E, Sanchez-Torres G, Marcushamer J, Mispireta J, Horwitz S, et al. (1977) Takayasuâ??s arteritis. Clinical study of 107 cases. Am Heart J 93: 94-103.
- Vanoli M, Daina E, Salvarani C, Sabbadini MG, Rossi C, et al. (2005) Takayasuâ??s arteritis: a study of 104 Italian patients. Arthritis Rheum 53: 100-107.
- Park MC, Lee SW, Park YB, Chung NS, Lee SK (2005) Clinical characteristics and outcomes of Takayasuâ??s arteritis: analysis of 108 patients using standardized criteria for diagnosis, activity assessment, and angiographic classification. Scand J Rheumatol 34: 284-292.
- Ishikawa K, Maetani S (1994) Long-term outcome for 120 Japanese patients with Takayasuâ??s disease. Clinical and statistical analyses of related prognostic factors. Circulation 90: 1855-1860.
- Ben Dhaou B, Aydi Z, Boussema F, Ben Dahmen F, Baili L et al. (1012) La maladie de Takayasu en Tunisie : Etude mono- centrique de 11 cas. La tunisie Medicale 90: 867–872.
- Mirault T, Messas E ((2016)) La maladie de Takayasu. Rev Med Interne 37: 223-229.
- Aydin SZ, Yilmaz N, Akar S, Aksu K, Kamali S, et al. (2010) Assessment of disease activity and progression in Takayasuâ??s arteritis with Disease Extent Index-Takayasu. Rheumatology 49: 1889–1893.
- Jacq F, Emmerich J (1998) Maladie des vaisseaux. Ed Doin Paris. pp 187-193.
- Gaye M, Sawadogo A, Dieng PA, Sow NF, Diatta S, et al. (2017) Diagnosis and indications for revascularization in Takayasuâ??s Arteritis: Report of two cases and literature review. Int J Vasc Surg Med 3: 036-039.
- Hoffman GS, Ahmed AE (1998) Surrogate markers of disease activity in patients with takayasu arteritis. A preliminary report from the international network for the study of the systemic vasculitides (inssys). Int J Cardiol 66: S191–S194.
- Bouzerda A, khatouri A (2016) Manifestations cardiaques de la maladie de Takayasu: A propos dâ??une observation et revue de la littérature. Pan Afr Med J 24: 82.
- Seyahi E (2017) Takayasu arteritis. An update Curr Opin Rheumatol 29: 51-56.
- Mukhtyar C, Guillevin L, Cid MC, Dasgupta B, de Groot K, et al. (2009) EULAR recommendations for the management of large vessel vasculitis. Ann Rheum Dis 68: 318–323.
- Keser G, Direskeneli H, Aksu K (2014) Management of Takayasu arteritis: a systematic review. Rheumatology 53: 793-801.
- Jeong HS, Jung JH, Song GG, Choi SJ, Hong SJ (2017) Endovascular balloon angioplasty versus stenting in patients with Takayasu arteritis: A meta-analysis. Medicine 96: 29.
- Qureshi MA, Martin Z, Greenberg RK (2011) Endovascular management of patients with Takayasu arteritis: stents versus stent grafts. Semin Vasc Surg 24: 44–52.
- Lee BB, Laredo J, Neville R, Villavicencio JL (2009) Endovascular management of Takayasu arteritis: is it a durable option? Vascular 17: 138–46.
- Isobe M (2013) Takayasu arteritis revisited: current diagnosis and treatment. Int J Cardiol 2013 168: 3–10.
- Chaudhry MA, Latif F (2013) Takayasuâ??s arteritis and its role in causing renal artery stenosis. Am. J Med Sci 346: 314-318.
- Kim YW, Kim DI, Park YJ, Yang SS, Lee GY, et al. (2012) Surgical bypass vs endovascular treatment for patients with supra-aortic arterial occlusive disease due to Takayasu arteritis. J Vasc Surg 55: 693-700.
- Andrews J, Mason JC (2007) Takayasuâ??s arteritis-recent advances in imaging offer promise. Rheumatology 46: 6-15.
- Li J, Zhu M, Li M, Zheng W, Zhao J, et al. (2016) Cause of death in Chinese Takayasu arteritis patients. Medicine 95: 27 (e4069).




