Research Article - (2024) Volume 2, Issue 2
HRP2 and PLDH Performance Comparison as Rapid Diagnostic Tools for Symptomatic Malaria Patients at Rugazi HC IV in Rubirizi District
2Malaria Control Programme, Ministry of Health, Uganda
Received Date: Jan 02, 2024 / Accepted Date: Jan 22, 2024 / Published Date: Feb 13, 2024
Copyright: ©Â©2024 Nakavuma Anitah, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Anitah, N., Afrika, M. I., Hassan, L., Rose, A. P., Evalyne, A. et al. (2024). HRP2 and PLDH Performance Comparison as Rapid Diagnostic Tools for Symptomatic Malaria Patients at Rugazi HC IV in Rubirizi District. Int J Med Net, 2(2),01-14.
Abstract
Background: Rapid and precise laboratory diagnosis is essential for effectively diagnosing and managing malaria patients. Malaria rapid diagnostic tests (mRDTs) play a critical role in malaria diagnosis in resource-constrained settings with limited access to labs and trained staff. The efficiency of two mRDTs (HRP2 and pLDH) was compared against microscopy, the gold standard for malaria diagnosis, in this study.
Methods: In a cross-sectional study, 308 participants were tested for malaria parasites using two mRDTs and blood smear microscopy as the gold standard. STATA software version 17 was used for data analysis.
Results: Out of the 308 participants, 82 tested positive for HRP2 mRDT (26.62% (95% CI: 21.97-31.86) positivity rate, 73.38% (95% CI: 68.14-78.83) negativity rate), and 99 tested positive for pLDH mRDT (32.14% (95% CI: 27.14-37.59) positivity rate, 67.86% (95% CI: 62.41-72.86) negativity rate). Using microscopy as the gold standard, the sensitivity, specificity, PPV, and NPV were determined. For HRP2 mRDT, sensitivity was 77.78% (95%CI: 73.13-82.42), specificity 97.61% (95%CI: 95.90 - 99.31), PPV 93.90% (95%CI: 91.23 – 96.56), and NPV 90.27% (95%CI: 86.96-93.58). For pLDH mRDT, the sensitivity was 94.95% (95% CI: 92.50-97.40), specificity was 97.61% (95% CI: 95.90-99.31), PPV was 94.95% (95% CI: 92.50 – 97.40), and NPV was 97.61% (95% CI: 95.90 - 99.31). The dominant malaria species was Plasmodium falciparum (74.7%), followed by Plasmodium vivax (12.1%) and Plasmodium ovale (1%). Mixed infections of plasmodium falciparum and plasmodium vivax (7.1%) and plasmodium falciparum and plasmodium ovale (5.1%) were also observed.
Conclusion: There was no significant difference in the positivity rate, negativity rate, specificity or PPV in both mRDTs. However, a significant difference in the NPV and sensitivity of HRP2 and pLDH mRDTs was observed where HRP2’s sensitivity was below the WHO recommendation of ≥95% in this study population; thus, there is a need to improve the sensitivity of this mRDT kit. In our study, most malaria infections were caused by Plasmodium falciparum, followed by P. vivax and P. ovale.
We therefore recommend the use of pLDH mRDTs over HRP2 mRDTs based on the results of our study.
Keywords
Malaria Rapid Diagnostic Tests, Malaria Microscopy, Symptomatic Infections.
Abbreviations and Acronyms
DHO: District Health Officer.
HCIV: Health Centre Four.
HMIS: Health Management Information System.
HRP2: Histidine Rich Protein 2.
mRDTs: Malaria Rapid Diagnostic Tests.
MUST: Mbarara University of Science and Technology.
NPV: Negative Predictive Value
OPD: Out Patient Department.
P.f: Plasmodium falciparum.
PCR: Polymerase Chain Reaction.
pLDH: Plasmodium Lactate Dehydrogenase.
PPV: Positive Predictive Value
RBM: Roll Back Malaria
SOPs: Standard Operating Procedures.
WHO: World Health Organization.
Background
In Uganda, malaria is a serious public health issue with a high prevalence rate. Malaria is the primary cause of morbidity and mortality, according to the World Health Organization (WHO), accounting for 30% of all outpatient visits and 20% of all deaths [1]. Children under the age of five and pregnant women are extremely affected by the condition [2].
Malaria rapid diagnostic tests (mRDTs) are important tools for the diagnosis of malaria in environments where resources are limited and where laboratory facilities and trained staff are scarce [3,4]. Effective treatment and management of malaria patients depend on a quick and precise laboratory diagnosis. These mRDTs are inexpensive and simple to use, and they produce results quickly, making them ideal for use in remote or underserved areas [5]. Histidine-rich protein 2 (HRP2) and plasmodium lactate dehydrogenase (pLDH) tests are the two main common mRDT kits used for malaria diagnosis [6].
Histidine-rich protein 2 (HRP2) mRDTs detect the antigen histidine-rich protein 2, which is released by only the P. falciparum species that causes malaria [7]. HRP2 is released into blood as a byproduct of the parasite’s life cycle, specifically during the asexual blood stage when the parasite replicates within red blood cells. This parasite species is the utmost cause of malaria in sub- Saharan Africa. Therefore, detection of this protein only confirms Plasmodium falciparum species [8].
On the other hand, pLDH mRDTs detect plasmodium lactate dehydrogenase (pLDH), which is an enzyme produced by all species of plasmodium. pLDH is released into the blood as a byproduct of parasite metabolism during the intraerythrocytic stage when the parasite replicates within red blood cells [9-11]. The presence of pLDH in blood is detected using mRDTs that look for pLDH antigen in the patient’s blood. The pLDH antigen is released into the blood throughout the course of malaria infection, with increasing levels as the infection progresses, and can be detected in blood as early as a few days after infection, where levels of pLDH tend to peak around the time of symptom onset. The pLDH antigen levels gradually decline after treatment and can remain detectable in blood for a few days after successful treatment of infection; hence, this test may be more helpful for tracking patients' healing after treatment and preventing needless malaria treatment repetition [14-16].
However, microscopic examination and polymerase chain reaction (PCR) tests remain the gold standard methods by which malaria infection is diagnosed. The PCR test is a very sensitive replacement for microscopy; however, the method is not commonly used because it is very expensive to establish in a rural setting [17,18].
The pitfalls of most mRDTs are their low specificity and sensitivity in detecting malaria parasite antigens compared to gold standard tests. A recent study conducted in northern Uganda on asymptomatic patients showed that the sensitivity and specificity for HRP2 mRDTs were lower than those for P. falciparum/pLDH mRDTs (95.2% and 90%, respectively) [19]. Therefore, there is a need to study the sensitivity and specificity of mRDTs among symptomatic patients in a malaria-endemic area of southwestern Uganda, which will enable us to determine the most appropriate mRDT to be used for malaria diagnosis in Rugazi HC IV, Rubirizi District.
Methodology
Study Site
Rugazi Health Centre IV is located in Kasungu Village within the Parish of Kasarara Ward in the subcounty of Rubirizi Town Council, Rubirizi District western Uganda (0°15’43” south, 30°6’12” east), with a population of 144,100 according to 2020 estimates. The health center serves as a referral for the district and hence covers 90% of the population. The district’s main physical features include natural rainforests, Kirinzu, Maramagambo and Kasyoha-Kitomi, a large concentration of crater lakes, over 30 in number. The main economic activities include fishing, brick laying, and farming. All of these factors provide favorable habitats for mosquitoes that transmit the plasmodium species.
Study Design
We used a cross-sectional study design.
Study Population
All patients presented with malaria-like symptoms, such as fever, flu-like illness, shaking chills, headache, muscle aches, tiredness, nausea, malaise, vomiting, diarrhoea, joint pain, anaemia and jaundice (yellow colouring of the skin and eyes), at Rugazi Health Centre IV.
Sample Size Determination
This was calculated using the diagnostic accuracy test’s formula for two proportions (Akoglu, 2022):
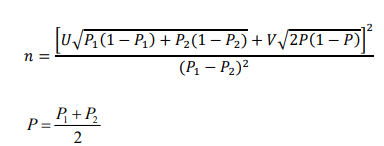
u- represents the desired power (typically 0.84 for 80% power).
v- Represents the desired level of statistical significance (typically 1.96 for 5% alpha).
P- Average proportion. P1 and P2- Expected sensitivity or specificity of index tests 1 and 2. From a recent meta-analysis study regarding the precision of HRP2 and pLDH rapid diagnostic tests in diagnosing malaria, the combined sensitivity and specificity were 0.92 (95% CI 0.83-0.96) and 0.92 (0.86–0.95), the pLDH-based assays were 0.96 (0.85– 0.98) and 0.93 (0.86–0.95), and the HRP2-based assays were 0.94 (0.84–0.98) and 0.86 (0.77–0.91), respectively (Hu et al., 2021).
Using the specificities of the two tests, P1 and P2 are 0.93 and 0.86, respectively:
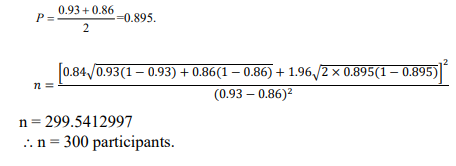
Therefore, 300 participants were recruited for the study, and data collection was conducted from June to July 2023.
Sampling Technique
It was a nonprobability sampling where all patients who presented with malaria-like symptoms at the outpatient department of Rugazi Health Centre IV were conveniently selected.
Study Selection Criteria
Inclusion Criteria
All patients presented with malaria-like symptoms, including fever, flu-like illness, shaking chills, headache, muscle aches, tiredness, nausea, malaise, vomiting, diarrhoea, joint pain, anaemia and jaundice (yellow colouring of the skin and eyes).
Exclusion Criteria
Patients who were on anti-malarial drugs and those who had just finished their dosage of antimalarials.
Data Processing and Laboratory Processing
The data collection tools were the patients’ clinical notes, and tests were used. Upon consenting or assenting to the study, samples were collected from the patients by performing a venipuncture on the ideal and best locations of the body.
Each patient was tested for malaria using both HRP2 and pLDH mRDTs. The standard procedure per the manufacturer’s instructions was followed, and the results were read and confirmed by a second person.
Malaria RDTs
These are lateral flow immuno-chromatographic antigen-detection tests, which rely on the capture of dye-labelled antibodies to produce a visible band on a strip of nitrocellulose, often encased in plastic housing, referred to as cassettes.
Generally, after labelling the mRDT with the patient’s unique identification number, a drop of blood was added to the mRDT through the sample well, followed by four drops of buffer through a buffer well. Blood was carried by buffer along the length of the mRDT. With malaria RDTs, the dye-labelled antibody first bound to a parasite antigen, and the resultant complex was captured on the strip by a band of bound antibody, forming a visible line (T -test line) in the results window for positive patients. The control line (C- control line) provided information on the integrity of the antibody-dye conjugate but did not confirm the ability to detect parasite antigen.
Figure 1: An mRDT Cassette
Malaria Microscopy
Microscopic examination was used as the “gold standard” for laboratory confirmation of malaria. A blood specimen collected from the patient was spread on a slide labelled with a unique laboratory number for a thick and thin blood smear. Then, the cells were stained with a Romanowsky stain (Giemsa was used) and examined with a 100X oil immersion objective. Visual criteria were used to detect malaria parasites and to differentiate the various species.
Examination of the slides was performed independently by two researchers and an experienced laboratory technologist/ technician, and for varying results, another experienced laboratory technologist/technician re-examined the slides, and the majority decision was taken to be the final result. For each positive, the corresponding thin film was examined for species identification.
Quality Assurance
The SOPs in place at the Rugazi Health Centre IV OPD laboratory were strictly followed for each step of the testing procedure for different batches of mRDTs.
We had a one-day training on malaria microscopy before the start of the data collection.
Records including patient information, test results, quality control data and training records were kept safely.
We also tracked and monitored the expiration dates of the mRDT kits to ensure that they did not expire and were within their shelf life.
Quality Control
Internal quality control of mRDTs involved the visualization of the control line in the result window of the test card. This indicated that the active ingredients of the strip were functional and that migration was successful but was not an assurance that the specimen had been applied properly; it was not a positive specimen control.
Both positive and negative control samples were included with each batch of the mRDTs to ensure accuracy, and the controls were tested along with the patients’ samples to verify that the test results were correct and reliable.
Prepared reagents or stains were quality controlled with known positive and negative samples from a confirmed symptomatic patient with malaria, and one without malaria was pretested with the reagents to check and confirm their quality.
Positive and negative control slides were used to validate the performance of the microscopist. We compared the identified plasmodium species with those on the bench aid-colored charts and stained microscopic glass slides.
Positive samples were cross-checked by a certified malaria microscopist.
We took corrective actions for some quality control deviations, and this included replacing expired reagents.
Data Quality Control Management
Data were collected based on the patients’ requested clinical notes and tests. The results were recorded and checked for completeness and consistency, and the data were coded, double entered into an Excel spreadsheet to ensure the accuracy of the data and then cleaned before analysis to ensure quality. Raw data were later stored in the box files for retrieval.
Data Analysis
The raw data were double entered into a Microsoft Excel spreadsheet and then transferred to STATA software version 17 for analysis. The sociodemographic characteristics were compared between people who had malaria and those without malaria. Gender was summarized using frequencies and proportions that were expressed as percentages. A chi-square test was used to compare the distribution of gender by malaria status. A P value less than 0.05 was considered to be significant. Age was not normally distributed according to the Shapiroâ??Wilk normality test (p value<0.001) and therefore was summarized using the median and interquartile range. The age distribution between those with malaria and those without malaria was compared using Student’s t test, which compared the mean age in the two categories of malaria status.
The positivity or negativity rates of each of the mRDTs together with their 95% confidence intervals were expressed as a proportion by dividing the total number of patients who tested positive or negative for that particular mRDT by the total number of patients tested and then multiplying by 100%.
The sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) with their 95% confidence intervals were estimated using their respective formulas:
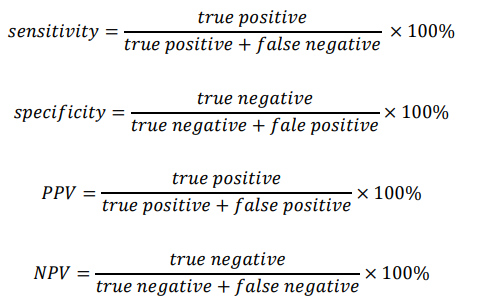
The most common plasmodium species was calculated by dividing the total number of a specific plasmodium species by the total number of plasmodium species counted by microscopy. This is also a proportion, and was expressed as a percentage.
Ethical Considerations
Ethical authorization was acquired from the Faculty of Medicine Research Ethics Committee (Appendix VI) before conducting the study. Administrative approval (Appendix VII) from the DHO of Rubirizi district and the medical superintendent of Rugazi Health Centre IV was sought. The study sought informed consent and ascent (Appendix IV and Appendix V) from the study participants, and patient confidentiality was ensured by giving unique identification numbers to patients. All study data were kept securely and accessible only by the supervisor and research team.
Results
Sociodemographic Characteristics of the Participants
Out of the 308 participants, 189 (61.36%) were females and 119 (36.64%) were males. The median age was 15 years, with an interquartile range of 8 to 27 years. Ninety-nine of the 308 study participants were positive for malaria, giving an overall proportion of 32.14% (95% CI: 27.14-37.59%). There was no significant difference in the distribution of sex between people who had malaria and people who did not have malaria. There was also no significant difference between the mean age of study participants positive for malaria and those negative for malaria, as indicated in Table 1.
|
Variable |
Total
308(100%) |
Malaria Status |
P- Value |
||
|
Present
99(32.14%) |
Absent
209(67.86%) |
||||
|
Gender: |
Male |
119(38.64%) |
45(45.45%) |
74(35.41%) |
0.091 |
|
Female |
189(61.36%) |
54(54.55%) |
135(64.59%) |
||
|
Age: (Median)(IQR)
(Mean)(SD) |
15(8-27) |
17.60(16.67) |
21.55(18.35) |
0.070 |
|
Table 1: Sociodemographic Characteristics of the Study Participants Stratified by Malaria Status
Positivity and Negativity Rates of HRP2 and pLDH mRDTs.
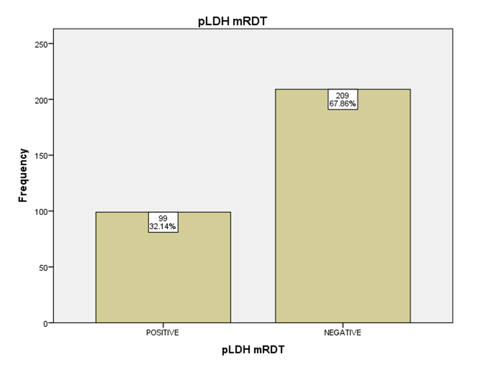
Out of the 308 participants, 82 participants were reactive for HRP2 mRDT, giving an overall positivity rate of 26.62% (95% CI: 21.97- 31.86). However, 99 participants were reactive for pLDH mRDT, giving an overall positivity rate of 32.14% (95% CI: 27.14-37.59). As indicated in Table 2 and Figure 1 below.
There was no significant difference between the positivity rate of HRP2 mRDT and the positivity rate of pLDH mRDT (p value=0.1327, proportion test).
|
Rapid
Diagnostic Test |
Results |
Frequency |
Percentage(95% CI) |
|
HRP2 mRDT |
Negative |
226 |
73.38%(68.14 78.83) |
|
Positive |
82 |
26.62%(21.97-31.86) |
|
|
pLDH mRDT |
Negative |
209 |
67.86%(62.41-72.86) |
|
Positive |
99 |
32.14%(27.14-37.59) |
Table 2: Proportion of Study Participants Positive for HRP2 mRDT and pLDH mRDT Tests
Figure 2: The Positivity and Negativity Rates of HRP2 mRDT
Figure 3 Positivity and Negativity Rates of pLDH mRDT
The Sensitivity, Specificity, and Positive and Negative Predictive Values of HRP2 and pLDH mRDTs
The sensitivity of pLDH mRDT [94.95% (95% CI: 92.50-97.40)] was observed to be significantly higher in this study than the sensitivity of HRP2 mRDT [77.78% (95% CI: 73.13-82.42)]. However, no significant difference was observed between the specificity of the pLDH mRDT (97.61%, 95% CI: 95.90-99.31) and the specificity of the HRP2 mRDT [97.61% (95% CI: 95.9-99.31)].
The sensitivity and specificity of HRP2 and pLDH mRDTs are shown in table 3 and table 4, respectively:
|
|
Microscopy (gold standard) |
|
||
|
MPs seen |
No MPs seen |
Total |
||
|
HRP2 mRDT |
Positive |
77 |
5 |
82 |
|
Negative |
22 |
204 |
226 |
|
|
|
Total |
99 |
209 |
308 |
Table 3: Sensitivity, Specificity, and Positive and Negative Predictive Values of HRP2 mRDT.
Sensitivity = 77.78% (95% CI: 73.13 - 82.42)
Specificity = 97.61% (95% CI: 95.90 - 99.31)
Positive predictive value = 93.90% (95% CI: 91.23 – 96.56)
Negative predictive value = 90.27% (95% CI: 86.96-93.58)
|
|
Microscopy (gold standard) |
|
||
|
MPs seen |
No MPs seen |
Total |
||
|
pLDH mRDT |
Positive |
94 |
5 |
99 |
|
Negative |
5 |
204 |
209 |
|
|
|
Total |
99 |
209 |
308 |
Table 4: Sensitivity, Specificity, and Positive and Negative Predictive Values of the pLDH mRDT.
Sensitivity 94.95% (95% CI: 92.50 - 97.40)
Specificity 97.61% (95% CI: 95.90 - 99.31)
Positive predictive value = 94.95% (95% CI: 92.50 – 97.40)
Negative predictive value = 97.61% (95% CI: 95.90 - 99.31)
The Most Common Plasmodium Species
Plasmodium falciparum (74, 74.75%) was the most common plasmodium species among the study participants, followed by plasmodium vivax (12, 12.1%) and plasmodium ovale (1, 1.0%). Mixed infection of plasmodium falciparum and plasmodium vivax 7 (7.1%) was also observed to be greater than mixed infection of plasmodium falciparum and plasmodium ovale 5 (5.1%), as shown in Table 5 below.
|
Plasmodium species |
Frequency |
Percentage (%) |
|
Plasmodium falciparum |
74 |
74.70 |
|
Plasmodium vivax |
12 |
12.10 |
|
Plasmodium ovale |
1 |
1.00 |
|
P.falciparum and P.vivax |
7 |
7.10 |
|
P.falciparum and P.ovale |
5 |
5.10 |
|
Total |
99 |
100.00 |
Table 5: Percentage Frequencies of the Different Plasmodium Species
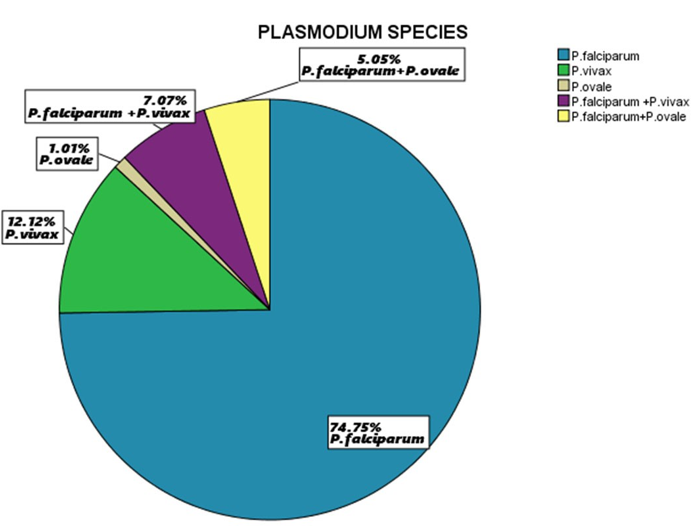
Figure 4: Percentage Distribution of the Plasmodium Species
Discussion
Positivity and Negativity Rates of HRP2 and pLDH mRDTs.
A similar study conducted in Kenya had positivity and negativity rates of 45.65%[95%CI(39.0-52.0)] and 54.35%[95%CI(48.0- 61.0)], 46.09%[95%CI(40.0-53.0)] and 53.9%[95%CI(47.0-60.0)] for HRP2 and PLDH mRDTs, respectively [20]. These results are similar to those reported from Nigeria and Cameroon, which turned out to be significantly higher than what we found in our study [21,22]. This is because of the small sample sizes in their studies compared to our study, making the positivity rate more susceptible to specific positive or negative cases. In other words, even though the prevalence of the disease in the community as a whole is low, a small number of positive instances can result in a significantly higher positivity rate. As a result, the computed positive rate may be more variable when sample sizes are small. Another reason is the high endemicity of malaria in their study site compared to our study.
The results from some studies agree with our study results [19,23- 25]. These studies also concentrated on using mRDTs to identify malaria. There are various reasons for the parallelism in the positive and negative rates between these studies and ours. We all had comparable demographics, geographic areas, and malaria frequencies and were directed towards similar people. Consistent frequencies of positive cases discovered by both types of mRDTs result from the diagnostic sensitivity and specificity for both HRP2 and pLDH markers being similar across investigations. These studies all used the same testing procedures and protocols, and the mRDTs' similar test quality, production standards, and storage conditions also contributed to the consistency that was seen [26,27].
We performed a proportion test to assess whether there was a significant difference between the positivity rates of HRP2 and pLDH mRDTs. The p value obtained from the proportion test was 0.1327, which indicated that the difference in positivity rates between HRP2 and pLDH mRDTs was not statistically significant; thus, both tests are comparable in terms of their ability to detect malaria infections among the study population.
Sensitivity, Specificity, and Positive and Negative Predictive Values of HRP2 and pLDH mRDTs
Expert blood smear microscopy was utilized as the gold standard in this study to compare the performance of two distinct mRDTs. The pLDH mRDTs demonstrated a higher sensitivity of 94.95% (95% CI: 92.50-97.40) compared to HRP2 mRDTs, which gave 77.78% (95% CI: 73.13-82.42) in this study population, which is lower than the WHO recommendation of ≥95% and the manufacturer’s sensitivity of 99.7% (95% CI: 98.5-100). The specificities were the same for both mRDTs [97.61% (95% CI: 95.90 - 99.31)] because they were able to detect the same number of true negatives [20].
A similar study in Kenya showed that HRP2 mRDTs had a higher sensitivity of 99.4% compared to our study, whereas the sensitivity of pLDH mRDTs (94.4%) was not significantly different from our study results. Their specificities (85.71% and 85.00%) for HRP2 and pLDH mRDTs, respectively, were lower in identifying patients without malaria compared to our study results [28]. Other studies in agreement with this study include [24,29,30]. This could have resulted from the difference in antigen expression brought about by the genetic diversity of malaria parasites, which impairs the test’s capacity to detect the parasites accurately. Variations in sample collection, storage and laboratory techniques may also have an impact on test outcomes. Changes in the local prevalence of several malaria species may be important [31-34].
Additionally, the sensitivity of HRP2 (77.78%) is also compromised by the ability to identify the remaining HRP2 antigen after treatment and parasite clearance. HRP2 mRDTs are currently the most widely used mRDTs for malaria diagnosis in Uganda and other sub-Saharan African countries where P. falciparum is the main parasite species [35,36]. However, several factors can impair the usefulness of mRDTs as malaria diagnostic instruments, necessitating regular monitoring [37-39].
The high PPV and NPV of pLDH mRDTs are in agreement with a study conducted in Nanoro, Burkina Faso, which reported PPV and NPV values of 85.2 and 97.6, respectively, for pLDH, whereas the PPV and NPV of HRP2 mRDTs were lower [40]. Studies also agree with these findings [41-43]. This could be attributed to the higher specificity and lower susceptibility to false positives of pLDH mRDTs. HRP2 mRDTs are sensitive to P. falciparum but can produce false positives due to antigen persistence [22], whereas pLDH mRDTs are more specific and efficient in areas with mixed infections or nonfalciparum species [44].
The Most Common Plasmodium Species
Speciation of Plasmodium species is an important malaria epidemiological metric for assessing a country's parasite population, which eventually guides public health management actions for malaria. The study identified three plasmodium species among the study participants, with Plasmodium falciparum being the most common, followed by Plasmodium vivax and Plasmodium ovale, and there were also mixed infections of Plasmodium falciparum with Plasmodium vivax and Plasmodium falciparum with Plasmodium ovale.
A study conducted in four contagious villages in Mulanda subcounty in Tororo District, Eastern Uganda, showed that infection due to Plasmodium falciparum was 94.0%, which was also the most common species identified; Plasmodium malariae was 6.0%, and there were no mixed infections seen [45]. Similar studies conducted in selected health care facilities across Ghana and Kisoro Hospital in southwestern Uganda showed results that were not significantly different from our study, with the exception of P. malariae, which was not identified in our study [24,46]. Studies in the same region of Uganda show results that are not significantly different from our study results [47-49]. This is due to the geographical distribution.
Plasmodium falciparum is the most predominant plasmodium species because it is well adapted to the female anopheles that are abundant in Sub-Saharan Africa, leading to higher transmission rates than the other plasmodium species, which have a lower level of compatibility with the dominant mosquito species in the region, leading to reduced transmission opportunities [50]. Plasmodium falciparum is the most dangerous pathogenic strain of malaria, causing severe malaria; hence, the implementation of its control measures should be revised [51]. Because the present HRP2 tests only detect P. falciparum monoinfection, the presence of other species has an impact on the type of mRDTs to be used in this situation [7]. In situations where P. falciparum-only mRDTs are utilized exclusively, non-P. falciparum can cause misleading negative mRDT results. Furthermore, the existence of mixed infections of P. falciparum + P. vivax, as well as P. falciparum + P. ovale, may have consequences for the training and development of laboratory employees in this environment to be able to report all species [52,53].
Conclusion and Recommendation
There was no significant difference in the positivity rate, negativity rate or specificity in both mRDTs. However, a significant difference in the sensitivity of HRP2 and pLDH mRDTs was observed where HRP2’s sensitivity was below the WHO recommendation of ≥95% in this study population; thus, there is a need to improve the sensitivity of this mRDT kit. In our study, most malaria infections were caused by Plasmodium falciparum, followed by P. vivax and P. ovale. Mixed infections were also observed, which may have implications for the choice and deployment of diagnostic tools.
We therefore recommend the use of pLDH mRDTs over HRP2 mRDTs basing on the results of our study.
Limitations of the Study
The study had limited geographical coverage; therefore, generalizability may not be possible. The sample size of 308 participants may be relatively small, and the study might not have captured the full heterogeneity of malaria cases in the population [54].
Although blood smear was used as a gold standard as recommended by WHO, the use of molecular tools such as PCR could have been more precise in detecting and speciation of the plasmodium parasites.
Among the uncooperative patients, some failed to consent, and others withdrew from the study after realizing there was no reward for their participation in the study, whereas others withdrew because of a phobia for needles.
Acknowledgements
We are extremely appreciative to our dear parents and guardians for the financial support during the course of the study. We acknowledge the District Health Officer (DHO) of Rubirizi District, the Medical Superintendent of Rugazi Health Centre IV, laboratory team and the custodian of MUST hostels in Rugazi health Centre IV for welcoming and supporting us during the period of data collection.
Funding
The Authors did not receive any grant for the study. The expenses for the study were funded by five authors (NA, MIA, LH, APR, and AE).
Authors Contribution
NA, MIA, LH, APR, AE, SF, KR, SPR, AB and BNC designed the study. NA, MIA, LH, APR and AE collected data. CNB and MIA performed data cleaning, data analysis and results interpretation. MIA drafted the manuscript. NA, MIA, LH, APR, AE, AB, SPR, OB, ME all reviewed the manuscript. All authors read and approved the final manuscript.
Declarations
Ethical Considerations
Ethical authorization was acquired from the Faculty of Medicine Research Ethics Committee before conducting the study. Administrative approval from the DHO of Rubirizi district and the medical superintendent of Rugazi Health Centre IV was sought. The study sought informed consent and ascent from the study participants and patient confidentiality was ensured by giving unique identification numbers to patients. All study data was kept securely and only accessible by the supervisor and research team.
Competing Interests
All authors declare that they have no competing interests.
Supplementary Information
The detailed datasets analysed are uploaded, follow the DOI: https://doi.org/10.6084/m9.figshare.24624768.v1
References
- Zalwango, J. F., Nankabirwa, J. I., Kitutu, F. E., Akunzirwe, R., Buhuguru, R., Rokani, J. B., ... & Kalyango, J. N. (2022). Malaria diagnostic and treatment practices for febrile children under 5 years at two general hospitals in Karamoja, a high transmission setting in Uganda. Malaria Journal, 21(1), 312.
- Imboumy-Limoukou, R. K., Maghendji-Nzondo, S., Sir- Ondo-Enguier, P. N., Niemczura De Carvalho, J., Tsafack- Tegomo, N. P., Buekens, J., ... & Lekana-Douki, J. B. (2020). Malaria in children and women of childbearing age: infection prevalence, knowledge and use of malaria prevention tools in the province of Nyanga, Gabon. Malaria journal, 19, 1-8.
- Ling, X. X., Jin, J. J., Zhu, G. D., Wang, W. M., Cao, Y. Y.,Yang, M. M., ... & Huang, J. Y. (2019). Cost-effectiveness analysis of malaria rapid diagnostic tests: a systematic review. Infectious Diseases of Poverty, 8(06), 12-27.
- Hansen, K. S., Clarke, S. E., Lal, S., Magnussen, P., & Mbonye,K. (2017). Cost-effectiveness analysis of introducing malaria diagnostic testing in drug shops: a cluster-randomised trial in Uganda. PLoS One, 12(12), e0189758.
- Ndyomugyenyi, R., Magnussen, P., Lal, S., Hansen, K., & Clarke, S. E. (2016). Appropriate targeting of artemisininâ? based combination therapy by community health workers using malaria rapid diagnostic tests: findings from randomized trials in two contrasting areas of high and low malaria transmission in southâ?ÂÂwestern Uganda. Tropical medicine & international health, 21(9), 1157-1170.
- Kyabayinze, D. J., Zongo, I., Cunningham, J., Gatton, M., Angutoko, P., Ategeka, J., ... & Bell, D. (2016). HRP2 and pLDH-based rapid diagnostic tests, expert microscopy, and PCR for detection of malaria infection during pregnancy and at delivery in areas of varied transmission: a prospective cohort study in Burkina Faso and Uganda. PLoS One, 11(7), e0156954.
- Poti, K. E., Sullivan, D. J., Dondorp, A. M., & Woodrow, C.J. (2020). HRP2: transforming malaria diagnosis, but with caveats. Trends in parasitology, 36(2), 112-126.
- Akotet, M. K. B., Mawili-Mboumba, D. P., Madoungou, B., & Kombila, M. (2013). Performances of malaria Pf/Pan rapid test device Acon®(Pf HRP2/pan aldolase) and malaria Pf rapid test device Acon®(Pf HRP2) for the diagnosis of malaria in adults and children living in Gabon, Central Africa. Diagnostic Microbiology and Infectious Disease, 77(1), 58- 63.
- Peng, X., Wang, N., Zhu, A., Xu, H., Li, J., Zhou, Y., ...& Deng, D. (2021). Structural characterization of the Plasmodium falciparum lactate transporter PfFNT alone and in complex with antimalarial compound MMV007839 reveals its inhibition mechanism. PLoS Biology, 19(9), e3001386.
- Possemiers, H., Vandermosten, L., & Van den Steen, P. E. (2021). Etiology of lactic acidosis in malaria. PLoS pathogens, 17(1), e1009122.
- Tewari, S. G., Swift, R. P., Reifman, J., Prigge, S. T., & Wallqvist, A. (2020). Metabolic alterations in the erythrocyte during blood-stage development of the malaria parasite. Malaria journal, 19(1), 1-18.
- Barney, R., Velasco, M., Cooper, C. A., Rashid, A., Kyle,D. E., Moon, R. W., ... & Jang, I. K. (2022). Diagnostic characteristics of lactate dehydrogenase on a multiplex assay for malaria detection including the zoonotic parasite Plasmodium knowlesi. The American journal of tropical medicine and hygiene, 106(1), 275.
- Krampa, F. D., Aniweh, Y., Kanyong, P., & Awandare, G. A. (2020). Recent advances in the development of biosensors for malaria diagnosis. Sensors, 20(3), 799.
- Mbabazi, P., Hopkins, H., Osilo, E., Kalungu, M., Byakika- Kibwika, P., & Kamya, M. R. (2015). Accuracy of two malaria rapid diagnostic tests (RDTS) for initial diagnosis and treatment monitoring in a high transmission setting in Uganda. The American journal of tropical medicine and hygiene, 92(3), 530.
- Park, S. H., Jegal, S., Ahn, S. K., Jung, H., Lee, J., Na, B.K., ... & Kim, T. S. (2020). Diagnostic performance of three rapid diagnostic test kits for malaria parasite Plasmodium falciparum. The Korean Journal of Parasitology, 58(2), 147.
- Grandesso, F., Nabasumba, C., Nyehangane, D., Page,L., Bastard, M., De Smet, M., ... & Etard, J. F. (2016). Performance and time to become negative after treatment of three malaria rapid diagnostic tests in low and high malaria transmission settings. Malaria journal, 15(1), 1-12.
- Ghai, R. R., Thurber, M. I., El Bakry, A., Chapman, C. A., & Goldberg, T. L. (2016). Multi-method assessment of patients with febrile illness reveals over-diagnosis of malaria in rural Uganda. Malaria Journal, 15(1), 1-6.
- Kang, J. M., Cho, P. Y., Moe, M., Lee, J., Jun, H., Lee, H.W., ... & Na, B. K. (2017). Comparison of the diagnostic performance of microscopic examination with nested polymerase chain reaction for optimum malaria diagnosis in Upper Myanmar. Malaria journal, 16(1), 1-8.
- Agaba, B. B., Rugera, S. P., Mpirirwe, R., Atekat, M., Okubal, S., Masereka, K., ... & Muwanguzi, E. (2022). Asymptomaticmalaria infection, associated factors and accuracy of diagnostic tests in a historically high transmission setting in Northern Uganda. Malaria Journal, 21(1), 1-12.
- Okuta, C., Ogolla, H., Mambo, F., & Wanjala, C. (2020). Comparative Study between Rapid Diagnostic Tests and Microscopy for Diagnosis of Malaria in Seme, Kisumu County, Kenya.
- Muhammad, I., Abubakar, B., Mohammed, M. T., Abdullahi, A., Usman, A. M., & Abubakar, S. (2022). Determination of malaria rapid diagnostic test effectiveness compared to microscopy (Gold standard). DYSONA-Life Science, 3(2), 49- 56.
- Nana, R. R. D., Makoge, V., Ngum, N. L., Amvongo-Adjia, N., Singh, V., & Somo, R. M. (2021). Evaluating the dual reactivity on SD bioline malaria rapid diagnosis tests as a potential indicator of high parasitemia due to Plasmodium falciparum. Pathogens and Global Health, 115(7-8), 487-495.
- Apinjoh, T. O., Ntasin, V. N., Tataw, P. C. C., Ntui, V. N.,Njimoh, D. L., Cho-Ngwa, F., & Achidi, E. A. (2021). Comparison of conventional and non-invasive diagnostic tools for detecting Plasmodium falciparum infection in southwestern Cameroon: a cross-sectional study. Infectious Diseases of Poverty, 10(1), 75.
- Abuaku, B., Amoah, L. E., Peprah, N. Y., Asamoah, A.,Amoako, E. O., Donu, D., ... & Malm, K. L. (2021). Malaria parasitaemia and mRDT diagnostic performances among symptomatic individuals in selected health care facilities across Ghana. BMC public health, 21(1), 1-11.
- Bwire, G. M., Ngasala, B., Kilonzi, M., Mikomangwa, W. P., Felician, F. F., & Kamuhabwa, A. A. (2019). Diagnostic performance of CareStart™ malaria HRP2/pLDH test in comparison with standard microscopy for detection of uncomplicated malaria infection among symptomatic patients, Eastern Coast of Tanzania. Malaria Journal, 18, 1-7.
- Hamid, A. W. M., Suonyir, R. Y., Frimpomaa, S., Agyeman- Duah, K., Kissiedu, K. Y., Cudjoe, E. E., ... & Owusu-Agyei,S. (2023). Clinical Utility of Mono-Biomarker based Malaria Rapid Diagnostic Test Kits at a Military Medical Centre in Ghana: A Prospective Pilot Study. International Journal of Health Policy Planning, 2(2), 103-111.
- Yoboue, A. C. (2023). Molecular epidemiology of Mansonella perstans on Bioko Island: identification of risk factors, co- infection with malaria and Loa loa and impact in pregnant women (Doctoral dissertation, University_of_Basel).
- Okuta, C. A. (2021). EVALUATION OF DIAGNOSTIC PERFORMANCE AND FACTORS AFFECTING MALARIA RAPID TEST KITS IN SEME, KISUMU COUNTY (Doctoraldissertation, MMUST).
- Odeniyi, O. M., Agomo, C. O., Okangba, C. C., Olaniyan,K. O., & Oyibo, W. A. (2020). Comparison of performance characteristics of two malaria rapid diagnostic tests in detecting malaria infection among febrile patients in Urban Area of Lagos, Nigeria. Ife Journal of Science, 22(2), 191- 199.
- Ndewa, K., Kihamia, C. M., Mubi, M., & Semango, G. (2016). Performance Characteristics of Malaria Rapid Diagnostic Test and Its Utilization in Management of Febrile Patients in Korogwe District (Tanga) Tanzania. Universal Journal of Medical Science, 4(3), 81-87.
- SzöllÃÂ??si, E., Tóth, Z., Mahr, K., Hoi, H., & Lendvai, Á. Z. (2020). Extremely low malaria prevalence in a wetland specialist passerine. Parasitology, 147(1), 87-95.
- Neto, J. M., Pérez-Rodríguez, A., Haase, M., Flade, M., & Bensch, S. (2015). Prevalence and diversity of Plasmodium and Haemoproteus parasites in the globally-threatened Aquatic Warbler Acrocephalus paludicola. Parasitology, 142(9), 1183-1189.
- Olsson-Pons, S., Clark, N. J., Ishtiaq, F., & Clegg, S.M. (2015). Differences in host species relationships and biogeographic influences produce contrasting patterns of prevalence, community composition and genetic structure in two genera of avian malaria parasites in southern Melanesia. Journal of Animal Ecology, 84(4), 985-998.
- Carter, R. (2022). On the genetics of malaria parasites, a memoir: Part II, the diversity and phylogeny of the rodent malaria parasites. Parasitology International, 91, 102650.
- Monroe, A., Williams, N. A., Ogoma, S., Karema, C., & Okumu, F. (2022). Reflections on the 2021 World Malaria Report and the future of malaria control.
- MALARIA, O. MALARIA REPORT.
- Lin, K., Li, M., Wang, D., Luo, F., Lu, S., Michael, M. G.,... & Zhou, X. N. (2022). Evaluation of Malaria Standard Microscopy and Rapid Diagnostic Tests for Screening— Southern Tanzania, 2018–2019. China CDC Weekly, 4(28), 605.
- Mwangonela, Z. E., Ye, Y., Rachel, Q., Msuya, H. M.,Mwamlima, T. G., Mswata, S. S., ... & Mwangoka, G. W. (2023). Field evaluation of the novel One Step Malaria Pf and Pf/Pv rapid diagnostic tests and the proportion of HRP-2 gene deletion identified on samples collected in the Pwani region, Tanzania. Bulletin of the National Research Centre, 47(1), 17.
- Izere, C., Niyigena, J., de Dieu Tuyishime, J., Nshimiyimana, A., Habyarimana, T., Ndishimye, P., ... & Niyonzima, F. N. (2022). Correlation of Malaria Rapid Test and Peripheral Blood Smear Microscopy among Patients attending Byumba Health Centre. The East African Health Research Journal, 6(2), 203.
- Kiemde, F., Bonko, M. D. A., Tahita, M. C., Mens, P. F., Tinto, H., Schallig, H. D., & van Hensbroek, M. B. (2019). Algorithms for sequential interpretation of a malaria rapid diagnostic test detecting two different targets of Plasmodium species to improve diagnostic accuracy in a rural setting (Nanoro, Burkina Faso). Plos one, 14(2), e0211801.
- Ali, I. M., Bigoga, J. D., Forsah, D. A., Cho-Ngwa, F., Tchinda, V., Moor, V. A., ... & Leke, R. G. (2016). Field evaluation of the 22 rapid diagnostic tests for community management of malaria with artemisinin combination therapy in Cameroon. Malaria Journal, 15, 1-7.
- Awosolu, O. B., Yahaya, Z. S., Farah Haziqah, M. T., &Olusi, T. A. (2022). Performance Evaluation of Nested Polymerase Chain Reaction (Nested PCR), Light Microscopy, and Plasmodium falciparum Histidine-Rich Protein 2 Rapid Diagnostic Test (PfHRP2 RDT) in the Detection of Falciparum Malaria in a High-Transmission Setting in Southwestern Nigeria. Pathogens, 11(11), 1312.
- Abuaku, B., Amoah, L. E., Peprah, N. Y., Asamoah, A.,Amoako, E. O., Donu, D., ... & Malm, K. L. (2021). Malaria parasitaemia and mRDT diagnostic performances among symptomatic individuals in selected health care facilities across Ghana. BMC public health, 21(1), 1-11.
- Oboh, M. A., Ndiath, M., Ajibola, O., Oyebola, K., & Amambua-Ngwa, A. (2021). Increased Trends of P. vivax in Sub-Saharan Africa: What Does it Mean for Malaria Elimination?. In Current Topics and Emerging Issues in Malaria Elimination. IntechOpen.
- Pullan, R. L., Bukirwa, H., Staedke, S. G., Snow, R. W., & Brooker, S. (2010). Plasmodium infection and its risk factors in eastern Uganda. Malaria journal, 9, 1-11.
- Manirakiza, G., Kassaza, K., Taremwa, I. M., Bazira, J., & Byarugaba, F. (2022). Molecular identification and anti- malarial drug resistance profile of Plasmodium falciparum from patients attending Kisoro Hospital, southwestern Uganda. Malaria Journal, 21(1), 1-10.
- Asua, V., Tukwasibwe, S., Conrad, M., Walakira, A., Nankabirwa, J. I., Mugenyi, L., ... & Rosenthal, P. J. (2017). Plasmodium species infecting children presenting with malaria in Uganda. The American Journal of Tropical Medicine and Hygiene, 97(3), 753.
- Peyerlâ?ÂÂHoffmann, G., Jelinek, T., Kilian, A., Kabagambe, G., Metzger, W. G., & Von Sonnenburg, F. (2001). Genetic diversity of Plasmodium falciparum and its relationship to parasite density in an area with different malaria endemicities in West Uganda. Tropical medicine & international health, 6(8), 607-613.
- Roh, M. E., Oyet, C., Orikiriza, P., Wade, M., Kiwanuka, G.N., Mwanga-Amumpaire, J., ... & Yap Boum, I. I. (2016). Asymptomatic Plasmodium infections in children in low malaria transmission setting, southwestern Uganda. Emerging infectious diseases, 22(8), 1494.
- Molina-Cruz, A., Canepa, G. E., Kamath, N., Pavlovic, N. V., Mu, J., Ramphul, U. N., ... & Barillas-Mury, C. (2015). Plasmodium evasion of mosquito immunity and global malaria transmission: The lock-and-key theory. Proceedings of the National Academy of Sciences, 112(49), 15178-15183.
- Rasti, N., Wahlgren, M., & Chen, Q. (2004). Molecular aspects of malaria pathogenesis. FEMS Immunology & Medical Microbiology, 41(1), 9-26.
- Waltmann, A., Darcy, A. W., Harris, I., Koepfli, C., Lodo, J., Vahi, V., ... & Mueller, I. (2015). High rates of asymptomatic, sub-microscopic Plasmodium vivax infection and disappearing Plasmodium falciparum malaria in an area of low transmission in Solomon Islands. PLoS neglected tropical diseases, 9(5), e0003758.
- Bailey, J. W., Williams, J., Bain, B. J., Parkerâ?ÂÂWilliams, J.,Chiodini, P. L., & General Haematology Task Force of the British Committee for Standards in Haematology. (2013). Guideline: the laboratory diagnosis of malaria. British journal of haematology, 163(5), 573-580.
- Esayas, E., Tufa, A., Massebo, F., Ahemed, A., Ibrahim, I., Dillu, D., ... & Deribe, K. (2020). Malaria epidemiology and stratification of incidence in the malaria elimination setting in Harari Region, Eastern Ethiopia. Infectious Diseases of Poverty, 9(1), 1-12.
- Akoglu, H. (2022). User's guide to sample size estimation in diagnostic accuracy studies. Turkish Journal of Emergency Medicine, 22(4), 177.
- CYNTHIA E. PACUTHO, P. C., MALARIA PARTNERSUGANDA 2022. Responding to the malaria situation in Uganda. Malaria partners international.
- Dalrymple, U., Arambepola, R., Gething, P. W., & Cameron,E. (2018). How long do rapid diagnostic tests remain positive after anti-malarial treatment?. Malaria journal, 17(1), 1-13.
- DIALLO, P. M. T. B. A. D. A. 2020. RBM Partnership Strategic Plan 2021-2025.
- Hu, J., Zhang, C., Wang, S., Chen, X., Hu, J., & Liu, X. (2021). Accuracy of rapid diagnostic test to diagnose malaria in children under 5 years of age, a meta-analysis. Diagnostic Microbiology and Infectious Disease, 100(2), 115351.




