Research Article - (2025) Volume 10, Issue 1
High-Performance Liquid Chromatography Screening of Flavonoids in Monodora Myristica which Prevent Oxidation of Palm Fruit Emulsion
2Pharmacology Department, Faculty of Basic Cinical Sciences, Niger Delta University, Wilberforce Island, Nigeria
3Medical Biochemistry, Faculty of Basic Medical Sciences, Bayelsa Medical University, Imgbi Road, Nigeria
4Biochemistry, Faculty of Sciences, Federal University Otuoke, Nigeria
Received Date: Feb 06, 2025 / Accepted Date: Feb 27, 2025 / Published Date: Mar 11, 2025
Copyright: ©Ã?©2025Abraham Sisein Eboh, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Eboh, A. S., Frank-Oputu, A., Wodu, E., Chukuma, A. S., Sogoi, B. P., Toby, T. D. (2025). High-Performance Liquid Chromatography Screening of Flavonoids in Myristica Monodora which Prevent Oxidation of Palm Fruit Emulsion. Adv Nutr Food Sci, 10(1), 01-08.
Abstract
The oil stabilizing property of Monodora myristica extract was evaluated to ascertain its potential in inhibiting palm fruit oil emulsion. Monodora myristica was extracted with absolute methanol. Crude palm fruit emulsion was also prepared according to standard procedures. Oxidation of palm fruit emulsion in the presence or absence of 0.5 % Monodora myristica extract was carried out. Vitamin E served as reference antioxidant. The phytochemical analysis of Monodora myristica extract showed 2.49± 0.77mg GAE/g extract of phenol and 0.95 ± 0.34 mg QE/g extract of flavonoid. The high-performance liquid chromatography of flavonoid revealed many flavonoids, flavan-3-ol, catechin, gallocatechin and kaempferol. The peroxide value of Monodora myristica at 0 days and 7th day was 9.42± 0.17 meq O2 /kg oil and 13.50 ± 1.0 meq O2 /kg oil. In the oxidation experiment the value of thiobarbitanic acid expressed as malondialdehyde was higher at 0 day and 7th day which was 27.4 ± 1.71mg MDA/kg and 38.16 ±1.92 mg MDA/kg respectively. Conjugated diene and triene values increases in oxidation group but decreases in the Monodora myristica and vitamin E stabilizing groups as depicted. The GC analysis of fatty acid profile also revealed both saturated and unsaturated fatty acids. In conclusion the extract of Monodora myristica can be used in stabilizing oil during processing.
Keywords
Palm Fruit, Oxidation, Peroxide Value, GC-MS, HPLC, Monodora myristica
Introduction
Nowadays consumers of oil and related products are increasingly aware of the link between food and health. Therefore most consumers are interested in functional foods. These functional foods are also therapies that can fight sedentary related diseases [1]. Palm fruit is essential for man and animals’ diet. Man can process it into palm oil, it can also be processed and used for frying, cooking and for preserving other food products [2]. Crude palm fruits extract or palm fruit emulsion is obtained from the fruits of elaeis guinensis It has orange red colour, it contains saturated and unsaturated fatty acid like: Palmitic acid, oleic acid, stearic acid and linoleic acid [3,4]. It is also a common ingredient for the production of margarine, also used in the cosmetic industry like soap, cream lotions and shampoos [3]. Palm fruit emulsion can be oxidized to produce ketones, aldehydes, alcohol. These products can therefore affect the appearance of the emulsion, quality, palatability and texture [5]. Oxidation can be prevented by anti-oxidant compounds like butylated hydroxyl anisole and butylated hydroxyl toluene which are all synthetic anti-oxidants. They have the ability to extend the shelf life of Pharmaceutical and food products [6].
There is a large awareness for the use of natural anti-oxidant especially from plants or animals due to certain toxicity posed by synthetic anti-oxidant [7]. Also some synthetic antioxidants are banned in some lands, due to there health related risks Monodora myristica belongs to the Annonaceae family, it is commonly known as African nutmeg or calabash nutmeg, it flourishes in Africa and the Caribbean areas [8-10]. It is used in the repulsion of insects [11]. It is a spice for African cooking, enhancement of flavor, aroma to many foods’ preparation [9,12].The fruits and seeds of Monodora myristica are effective as stimulants and the management of certain African ailments like sores, pains and eye disorders [13]. The pleasant aroma from Monodora myristica makes it important in cooking [14,15]. Many scientific studies on Monodora myristica showed that extract from seeds are antibacterial, antioxidant, antifungal [10,16-18]. The therapeutic potential of Monodora myristica is due to the presence of bioactives [19]. Monodora mrystica possesses anti-inflammatory and anti-microbial properties. It also contain Eugenol and myristica which act as anti-oxidant by scavenging free radicals into [20,21]. Monodora myristica seeds are highly medicinal and are also used in African dishes. Several plant active compounds, such as phenols, saponin, alkaloids, glycosides, flavonoids are abundant in Monodora myristica seeds [22,23]. These active substances are secondary metabolites, and scientific studies has proven them to havemany positive health effects, such as antioxidant, antimicrobial, antidiabetic, hepatoprotective, antibacterial, antifungal, diuretic, antispasmodic, and antihypertensive properties [24,25]. Therefore, the aim of this study was to improve the stability and shelf life of palm fruit oil extract using Monodora myristica seed extracts as natural antioxidants.
Materials and Methods
Chemicals
Methanol, cyclohexane, chloroform, xylenol orange, iron(ii) chloride, iron(iii) chloride, dimethyl sulfoxide (DMSO), thiobarbituric acid, trichloroacetic acid, hydrochloric acid, potassium acetate, aluminium chloride, quercetin, gallic acid, sodium carbonate, Folin-Ciocalteau reagent.
Palm Fruit and Monodora myristica
Both palm fruits and Monodora myristica were obtained in sufficient quantities at Tombia market, Yenagoa Bayelsa state. The palm fruit as well as Monodora myristica were identified in Pharmacognosy Department, Faculty of Pharmacy, Niger Delta University, Bayelsa state. Voucher numbers were obtained.
Preparation of Monodora myristica Extract
Monodora myristica was grounded into powder afterwards 200 g of Monodora myristica powder was soaked in 0.5 L of methanol with occasional shaking for 3 days. The extract was filtered through Whatman No. 4 filter paper and was later concentrated into a light brownish paste. This was then stored at 40 C for future use.
Total Flavonoid Content
The extract of Monodora myristica was subjected to flavonoid content analysis spectrophotometrically according to the method of Ahmed et al., [26]. Extract (1mg/mg) and quercetin solutions 10 – 200 µg/ml were mixed with 10% Aluminum chloride 200µl and 100µl of 1M potassium acetate in tubes. All tubes were kept at 37°C for 30 minutes, Absorbance was determined at 520nm. Flavonoid content was expressed as mg QE/g of extract.
Total Phenol Content
Monodora myristica extract was determined according to the method reported by Ahmed et al., [25]. Monodora myristica dissolved in methanol (1mg/ml) and 10-20 µg/ml solutions of gallic acid as reference phenolic compound are mixed with 1ml of Folin- Ciocalteu reagent and were incubated for 5 min. Thereafter 10ml of sodium bicarbonate 7% was added and the volume raised to 25ml using distilled water. Solutions were incubated for 1.5 h at 37°C. Absorbance was read at 725nm. Total phenol in Monodora myristica was expressed as mg GAE/g extract.
High Performance Liquid Chromatography of Flavonoids in Monodora Myristica
Flavonoids from Monodora myristica were extracted with methanol, hexane hot water according to standard procedure. Flavonoid analysis was performed on a BUCK M910 HPLC equipped with a RESTEK 15 meter MXT-1 column (15m x 250um x 0.15um) was used. The injector temperature was 280o C with splitless injection of 2µL of sample and velocity of 30cms-1, methanol 5.0pas was the carrier gas with a flow rate of 40mlmin-1. The oven operated initially at 2000C was heated to 3300 C at a rate of 30 C min-1 and was kept at this temperature for 5min. the detector operated at a temperature of 3200C. Bioactive compound were determined by the ratio between the area and mass of internal standard and the area of the identified (Dolchy Analyticals).
Extraction of Palm Fruits
Exactly 100g of palm fruit were added to 500ml of boiling water. Cooking was carried out for 45minutes. The crude palm fruit oil was extracted by pounding the fruits until the oil and water emulsion comes out. The palm kernels and chaff were discarded but the oil and water was used for oxidation [3].
Preparation of Stock Solution of Monodora Myristica and Vitamin E
Exactly 0.5g of Monodora myristica or vitamin E was weighed and dissolved in 50ml of DMSO, to make a 1% stock solution. From the stock 0.5% of vitamin E or Monodora myristica was made.
Oxidation of Palm Oil Emulsion
Oxidation of crude palm fruit oil emulsion was heated in the absent, presence of vitamin E or Monodora myristica extract at 0.5 % concentration. The crude palm fruit oil emulsion was heated to 600 C in a water bath except the control before addition of 0.5 % vitamin E or Monodora myristica for 7 days. (1)The control: no heating (2) oxidation: heated to 600 C without antioxidant (3) oxidation + 0.5 % Monodora myristica and (4) oxidation + 0.5% vitamin E based on the method of Azman et al., [27]
Assay of Peroxide Value A
weighed amount of control, oxidation, oxidation + Monodora myristica and oxidation + Vitamin E were dissolved in 9.9ml of chloroform in methanol in the ratio 7:3 volume/volume. Thereafter methanol orange 50 µL was added to 50 µL of iron (ii) chloride solutions. The mixed solution was incubated at room temperature for 10 mins. The mixture was later centrifuged and the absorbance of the supernatant red at 560nm. A standard curve of iron (iii) chloride was constructed at concentration of 5µg/ml - 20µg/ml. Peroxide value was expressed as milliequivalent active oxygen per kg [28].
Conjugated Diene/Triene
Conjugated diene and triene was determined in control, oxidation, oxidation + Monodora myristca and oxidation + vitamin E at 0 days and 7 days according to the method of Abdalla and Roozen, [29]. Fifty milligram of samples were weighed and mixed with 5ml of cyclohexane and the CD and CT was determined using a UV spectrophotometer at 234 and 270nm respectively.
Measurements of Thiobarbituric Acid Reactive Substance
The method of Buege and Aust was applied [30]. The control, oxidation, oxidation + Monodora myristica and oxidation + vitamin E, one ml each was mixed with 5ml of TBA reagent. TBA reagen
Results
contained 0.375% thiobarbituric acid, 15% of trichloroacetic acid and 0.24 M HCl. The solutions were vortexed and heated in water bath at 95°C for 10 min. It was later cooled and centrifuged at 3000rpm for 20mims. The absorbance was measured at 532nm. The values were calculated and expressed as mg MDA/kg. 2.13 Gas Chromatography Analysis of Fatty Acid Profile in Palm Fruit Emulsion Ten milliliters of palm oil emulsion was mixed with sodium sulphate in a 250 ml beaker, fatty acids were then extracted with 100 ml of n-hexane for 24 hrs. The dried crude extract 5 mg was transferred into 10 ml centrifuge tube then 2 ml of water was added, 2 ml of 5% (w/v) methanolic sodium methoxide solution and 2 ml of hexane and neutralizing solution. The tubes were kept for 3 min before centrifuging at 1,750 rpm for 5 mins. About 200 µL of supernatant was transferred into 10 ml flask and then diluted to the mark with hexane, thereafter 1 µL was injected into the gas chromatography column [31]. 2.14 Statistical Analysis The results are mean ± SEM, n = 3. The data was processed using SPSS version 17.0 New York, USA. Significant values are
considered at p < 0.05.
Figure 1: Total Phenolic and Flavonoid Content in Methanolic Extract of Monodora Myristica
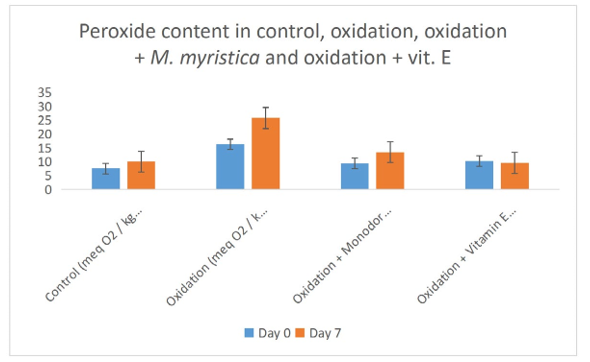
Figure 2: Effect of Monodora Myristica on Peroxide Content in Control, Oxidation, Oxidation + Monodora Myristica and Oxidation + Vitamin E of Palm Oil Emulsion
Figure 3: Effect of Monodora Myristica on Thiobarbituric Acid Content in Control, Oxidation, Oxidation + Monodora Myristica and Oxidation + Vitamin E of Palm Oil Emulsion
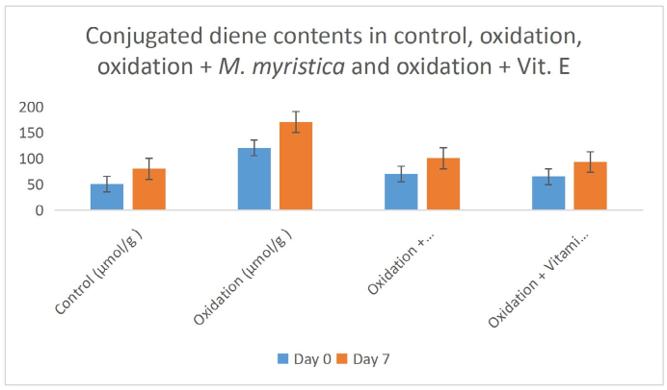
Figure 4: Effect of Monodora Myristica on Conjugated Diene Content in Control, Oxidation, Oxidation + Monodora Myristica and Oxidation + Vitamin E in Palm Fruit Emulsion.
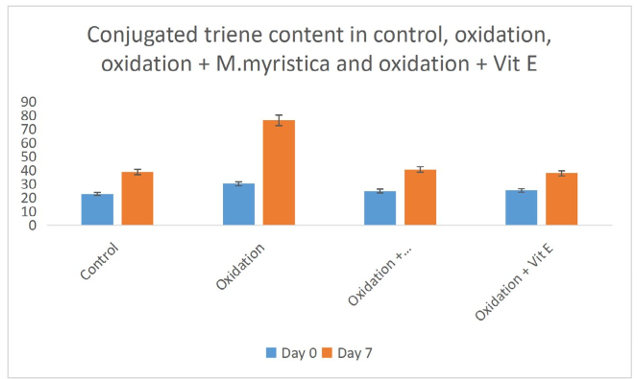
Figure 5: Effect of Monodora MYristica on Conjugated Triene Content in Control, Oxidation, Oxidation + Monodora Myristica and Oxidation + Vitamin E in Palm Fruit Emulsion
Figure 6: Showing the HPLC Chromatogram of Flavonoids in Monodora Myristica Extract
|
Component |
RT |
Area |
Height |
Concentration |
|
Resveratrol |
1.88 |
10310.46 |
79.00 |
11.38 µg/ml |
|
Catechin |
4.77 |
7981.31 |
148.70 |
22.53 µg/ml |
|
Flavonones |
15.69 |
15628.31 |
330.43 |
33.44 µg/ml |
|
Flavan-3-ol |
18.26 |
10767.58 |
226.43 |
30.48 µg/ml |
|
Flavone |
24.10 |
4624.65 |
97.99 |
12.65 µg/ml |
|
Gallocatechin |
32.34 |
13533.58 |
279.18 |
48.57 µg/ml |
|
Isoflavonoids |
39.24 |
5364.37 |
113.52 |
7.23 µg/ml |
|
Kaempferol |
45.27 |
5218.41 |
149.34 |
27.96 µg/ml |
Table 1: Showing the Retention Time, Height, Area and Concentration of Flavonoids in Monodora Myristica Extract
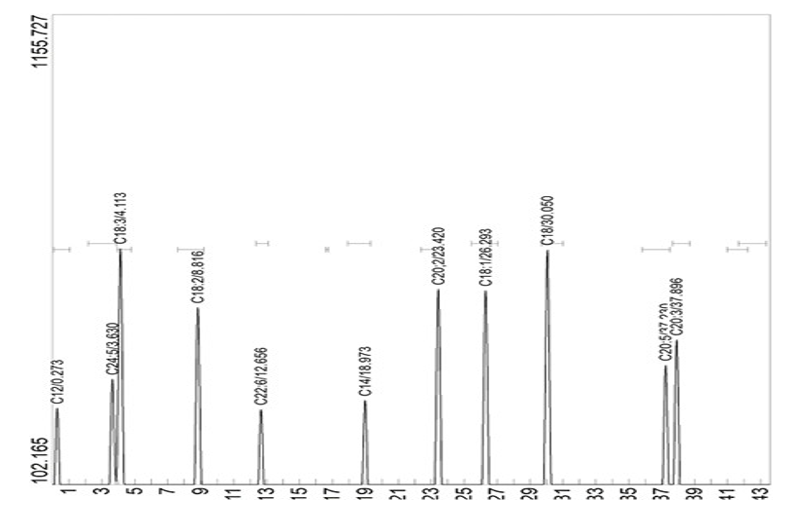
Figure 7: Showing the GC Chromatogram of Fatty Acids in Palm Fruit Emulsion
|
FFA |
RT |
Area |
Height |
Concentration |
|
C12 |
0.273 |
4636.95 |
273.84 |
19.32 µg/ml |
|
C24:5 |
3.630 |
5896.91 |
339.14 |
1.29 µg/ml |
|
C18:3 |
4.113 |
11295.95 |
632.31 |
52.06 µg/ml |
|
C18:2 |
8.816 |
8838.26 |
500.54 |
17.29 µg/ml |
|
C22:6 |
12.656 |
4803.14 |
272.73 |
1.05 µg/ml |
|
C14 |
18.973 |
5160.56 |
293.14 |
0.83 µg/ml |
|
C20:22 |
23.420 |
9581.19 |
542.12 |
2.10 µg/ml |
|
C18:1 |
26.293 |
9544.97 |
539.00 |
10.85 µg/ml |
|
C18 |
30.050 |
11160.01 |
630.66 |
13.48 µg/ml |
|
C20:5 |
37.230 |
6529.93 |
371.00 |
5.59 µg/ml |
|
C20:3 |
37.896 |
7548.70 |
427.83 |
1.11 µg/ml |
|
RT: retention time; FFA: free fatty acid |
||||
Table 2: Showing the Retention Time, Height, Area and Concentration of Fatty Acids in Palm Fruit Emulsion
Statistics
All the experimental results were analyzed statistically and were expressed as mean _+ 5.0 (n= 3). The results were also subjected to ANOVA using SPSS version, 6.0.
Discussion
Synthetic antioxidant like BHA and BHT were widely used especially in the past but of recent it is shown that they possess adverse health effects in this regard Monodora myristica extract was experimented as antioxidant that can prevent, improve or mitigate the deleterious effect of free radicals and improve the shelf life of palm fruit emulsion [3,7]. Flavonoids are medicinal bioactive that possess antioxidant activity. Studies abound that showed the importance of flavonoids capable of mitigating oxidative stress and lipid peroxidation link diseases [32]. Free radical, metal ion and enzymes that are contaminants in the food industry can lead to food deterioration especially those that are lipid based during processing and storage [33]. These deterioration reactions by radical, metals and enzymes produce offensive flavours and odors there by decreasing the shelf life of the food and the market value of the food [3]. The total phenols and flavonoids in Monodora myristica seed extract were depicted in figure 1 the reports of phytochemical values are similar to the findings of Afolabi et al., [34].
The high performance liquid chromatography of Monodora myristica seeds revealed the presence of resveratrol, catechin, flavones, flavan-3-ol, flavone, gallocatechin, isoflavonoids and kaempferol. The highest concentrated flavonoid was gallocatechin 48.57 µg/ml, these flavonoids are responsible for the preventive mechanism involved in the extract of Monodora myristica in preventing oxidation of palm oil emulsion, this report is similar to the findings of Fadul et al., [35]. The fatty acid composition of the palm oil indicated that there are more of unsaturated fatty acids than saturated as shown in the GC-MS analysis results this is similar to the reports of Li et al., [4]. They also reported more unsaturated fatty acids in Chinese palm fruit than the palm kernel. The unsaturated fatty acids in this report incuded linolenic acid, arachidonic acid, oleic acid etc. the unsaturated fatty acid of highest concentration was linolenic acid (52.06 µg/ml).
Lipid peroxidation give rise to offensive flavour, colour, texture, and loss of nutritional unsaturated lipids, health risk is on the increase due to the formation of secondary toxic compounds [36]. In the present study the levels of lipid peroxidation increased in the oxidation control of palm fruit emulsion at zero day and day seven, but these levels were decreased when Monodora myristica extract was added during oxidation and also when the standard antioxidant vitamin E was added the result is depicted in fig. the report of this findings is in agreement with the work of Kamkara 2010 who also reported the antioxidant effect of Mentha pulegium extracts in sunflower oil.
Peroxide value determination measures the amount of peroxides and hydroperoxides in lipid samples. Peroxide determination is a nice indicator of oxidation of oils. In the present study the peroxide values of oxidation of palm fruit oil increases from day zero and day 7 due to the increase in the formation of both hydroperoxide and peroxide. The peroxide value decreases in the experiments that Monodora myristica extract was added or vitamin E was added, this suggested that both Monodora myristica or vitamin E prevented the oil from oxidation due to their antioxidant and anti- radical effects. Our study is in line with the works of Womeni et al., and Alizadeh et al., Conjugated diene and triene are produced when double and single bond rearranges to form dienes and trienes during the preliminary stages of peroxidation [37,38]. These can be detected through the maxima absorption utilizing a UV spectrophotometer. The rate of CD and CT formation decreased significantly (p < 0.05) in the palm fruit emulsion treated with Monodora myristica or vitamin E (Figures 5 and 6).
This report is in tandem with the report of Siriwardhana et al., who also reported the preventive ability of Hizikia Fusiformis against fish oil and linoleic acid [38]. Oxidized palm oils produced unstable molecules that give rise to: alkens, alcohols, aldehydes, ketones, and acids, which are responsible for unwanted flavour and odour.
Malondialdehyde produced as part of these unwanted molecules was determined in the present study. The results revealed that at day zero and seven the amount of MDA significantly increased as compared to control experiment without oxidation. Therefore, the addition of Monodora myristica or vitamin E decreases the amount of MDA to the levels found in the control experiments. This report is in agreement with the works of kamkara et al., and Siriwardhana et al., In conclusion, the experiment has shown Monodora myristica can be additive to edible oils [39,40]. Therefore Monodora myristica can be a suitable alternative for many syntehetic antioxidants.
References
- Akusu, M. O., Achinewhu, S. C., & Mitchell, J. (2000). Quality attributes and storage stability of locally and mechanically extracted crude palm oils in selected communities in Rivers and Bayelsa States, Nigeria. Plant Foods for Human Nutrition, 55, 119-126.
- Matthäus, B. (2007). Use of palm oil for frying in comparison with other highâ?ÂÂstability oils. European Journal of Lipid Science and Technology, 109(4), 400-409.
- Tang, M., Xia, Q., Holland, B. J., Wang, H., Zhang, Y., Li, R., & Cao, H. (2017). Effects of different pretreatments to fresh fruit on chemical and thermal characteristics of crude palm oil. Journal of food science, 82(12), 2857-2863.
- Li, R., Xia, Q., Tang, M., Zhao, S., Chen, W., Lei, X., & Bai,X. (2012). Chemical composition of Chinese palm fruit and chemical properties of the oil extracts. African Journal of Biotechnology, 11(39), 9377-9382.
- Aleman, M., Nuchi, C. D., Bou, R., Tres, A., Polo, J., Guardiola, F., & Codony, R. (2010). Effectiveness of antioxidants in preventing oxidation of palm oil enriched with heme iron: A model for iron fortification in baked products. European journal of lipid science and technology, 112(7), 761-769.
- Webb, G. P. (2006). Dietary supplements and functional foods. Blackwell Publishing.
- Iqbal, S., Bhanger, M. I., & Anwar, F. (2005). Antioxidant properties and components of some commercially available varieties of rice bran in Pakistan. Food chemistry, 93(2), 265- 272.
- Esfarjani, F., Khoshtinat, K., Zargaraan, A., Mohammadiâ? Nasrabadi, F., Salmani, Y., Saghafi, Z., ... & Bahmaei, M. (2019). Evaluating the rancidity and quality of discarded oils in fast food restaurants. Food science & nutrition, 7(7), 2302- 2311.
- Adewole, E., Ajiboye, B. O., Idris, O. O., Ojo, O. A., Onikan, A., Ogunmodede, O. T., & Adewumi, D. F. (2013). Phytochemical, Antimicrobial and Gc-Ms of African Nutmeg (Monodora Myristica)., 2(5), 1-8.
- Okechukwu, Q. N., Ugwuona, F. U., Ofoedu, C. E., Juchniewicz, S., & Okpala, C. O. R. (2022). Chemical composition, antibacterial efficacy, and antioxidant capacity of essential oil and oleoresin from Monodora myristica and Tetrapleura tetraptera in Southeast Nigeria. Scientific Reports, 12(1), 19861.
- Tsila, H. G., Ntonga, P. A., Magnouet, S. C. L., & Tchuinkam,T. (2022). Efficacy and persistence of essential oil of Monodora myristica against Anopheles gambiae, the main vector of malaria in sub-Saharan Africa. Investigational Medicinal Chemistry & Pharmacology, 5(1), 57.
- Onyenibe, N. S., Fowokemi, K. T., & Emmanuel, O. B. (2015). African nutmeg (Monodora myristica) lowers cholesterol and modulates lipid peroxidation in experimentally induced hypercholesterolemic male Wistar rats. International Journal of Biomedical Science: IJBS, 11(2), 86.
- Akise, O. G., Fasakin, E. A., & Adeparusi, E. O. (2020). Chemical composition and in-vitro antimicrobial activity of essential oil of African nutmeg (Monodora myristica (Gaertn) Dunal on microorganisms isolated from smoke-dried catfish (Clarias gariepinus). African Journal of Microbiology Research, 14(4), 136-147.
- George, B. O., & Osioma, E. (2011). Phenolic content and total antioxidant capacity of local spices in Nigeria. African Journal of Food Science, 5(13), 741-746.
- Ezeuko, A. S., Bamgboye, O. A., Jonathan, H., & Uchenna, D. (2017). Extraction, physicochemical, phytochemical analysis and identification of some important compounds of Monodora myristica (African nutmeg) seed oil. International Journal of Innovative Research and Advanced Studies, 4, 406-410.
- Feyisayo, A. K., & Oluokun, O. O. (2013). Evaluation of antioxidant potentials of Monodora myristica (Gaertn) dunel seeds. African Journal of Food Science, 7(9), 317-324.
- Okpoghono, J., George, B. O., Achuba, F. I., Igue, U. B.,Okom, S. U., Seigha, A. A., ... & Ayogoi, K. I. (2021). Impact of crude petroleum oil and Monodora myristica on membrane bound ATPases and erythrocyte osmotic fragility in-vivo. Uniport J Eng Sci Res, 6(1), 65-77.
- Whiliki, O. O., Dowe, E., & Otue, E. I. (2023). Phytochemical screening and in –vitro antimicrobial activity of Monodora myristica seed extract on selected human pathogens. Nigerian Journal of Microbiology, 37(1), 6599–6608.
- Omar, J., Olivares, M., Alonso, I., Vallejo, A., Aizpurua Olaizola, O., & Etxebarria, N. (2016). Quantitative analysis of bioactive compounds from aromatic plants by means of dynamic headspace extraction and multiple headspace extraction gas chromatography-mass spectrometry. Journal of Food Science, 81(4), C867-C873.
- Rahman, N. A. A., Fazilah, A., & Effarizah, M. E. (2015). Toxicity of nutmeg (myristicin): a review. Int. J. Adv. Sci. Eng. Inf. Technol, 5(3), 61-64.
- Gupta, A. D., Bansal, V. K., Babu, V., & Maithil, N. (2013). Chemistry, antioxidant and antimicrobial potential of nutmeg (Myristica fragrans Houtt). Journal of Genetic engineering and Biotechnology, 11(1), 25-31.
- Afolabi, K. D., Eko, P. M., & Etukudoh, E. N. (2024). The Nutritional Value of African Nutmeg (Monodora myristica) Seed Meal. Asian Journal of Research in Biosciences, 6(1), 71-78.
- Okpoghono, J., George, B. O., & Achuba, F. I. (2018). Assessment of antioxidant indices after incorporating crude oil contaminated catfish and African nutmeg (Monodora myrstica) extracts into rat diet. Journal of Applied Sciencesand Environmental Management, 22(2), 197-202.
- Agiriga, A. N., & Siwela, M. (2018). Effects of thermal processing on the nutritional, antinutrient, and in vitro antioxidant profile of Monodora myristica (Gaertn.) dunal seeds. Preventive nutrition and food science, 23(3), 235.
- Okaiyeto, K., Adeoye, R. I., & Oguntibeju, O. O. (2021). Some common West African spices with antidiabetic potential: A review. Journal of King Saud University-Science, 33(6), 101548.
- Ahmed, S., Saeed-Ul-Hassan, S., Islam, M., Qureshi, F., Waheed, I., Munawar, I., ... & Peerzada, S. (2017). Anti-oxidant activity of Pistachia khinjuk supported by phytochemical investigation. Acta Pol Pharm, 74(1), 173-178.
- Mohd Azman, N. A., Segovia, F., Martínez-Farré, X., Gil, E., & Almajano, M. P. (2014). Screening of antioxidant activity of Gentian lutea root and its application in oil-in-water emulsions. Antioxidants, 3(2), 455-471.
- Pegg, R. B. (2001). Measurement of primary lipid oxidation products. Current protocols in food analytical chemistry, (1), D2-1.
- Abdalla, A. E., & Roozen, J. P. (1999). Effect of plant extracts on the oxidative stability of sunflower oil and emulsion. Food chemistry, 64(3), 323-329.
- AOAC Official Method 2012.13. Determination of Labeled Fatty Acids Content in food Products and Infant Formula. AOAC International 2012.
- Song, X., Wang, Y., & Gao, L. (2020). Mechanism of antioxidant properties of quercetin and quercetin-DNA complex. Journal of Molecular Modeling, 26, 1-8.
- Aruoma, O. I. (1998). Free radicals, oxidative stress, and antioxidants in human health and disease. Journal of the American oil chemists' society, 75(2), 199-212.
- Afolabi, O. B., Oloyede, O. I., Aluko, B. T., & Johnson, J. A. (2021). Biosynthesis of magnesium hydroxide nanomaterials using Monodora myristica, antioxidative activities and effect on disrupted glucose metabolism in streptozotocin-induced diabetic rat. Food Bioscience, 41, 101023.
- Fadul, S. E. K. M., Ahmed, Y. M., & Mariod, A. A. (2025).Oxidation of Sunflower Oil Using Annona Squamosa and Vangueria Madagascariensis Methanolic Extracts. Adv Nutr Food Sci, 10(1), 01-08.
- Kwon, H., Ko, J. H., & Shin, H. S. (2015). Evaluation of antioxidant activity and oxidative stability of spice-added mayonnaise. Food science and biotechnology, 24, 1285-1292.
- Womeni, H. M., Djikeng, F. T., Tiencheu, B., & Linder, M. (2013). Antioxidant potential of methanolic extracts and powders of some Cameroonian spices during accelerated storage of soybean oil. Advances in Biological Chemistry, 3(3), 304-313.
- Alizadeh, L., Abdolmaleki, K., Nayebzadeh, K., & Shahin,R. (2019). Effects of tocopherol, rosemary essential oil and Ferulago angulata extract on oxidative stability of mayonnaise during its shelf life: A comparative study. Food chemistry, 285, 46-52.
- Siriwardhana, N., Lee, K. W., Kim, S. H., Ha, J. H., Park,G. T., & Jeon, Y. J. (2004). Lipid peroxidation inhibitory effects of Hizikia fusiformis methanolic extract on fish oil and linoleic acid. Food Science and Technology International, 10(2), 65-72.
- Kamkar, A., Javan, A. J., Asadi, F., & Kamalinejad, M. (2010). The antioxidative effect of Iranian Mentha pulegium extracts and essential oil in sunflower oil. Food and Chemical Toxicology, 48(7), 1796-1800.
- Shahid, M. Z., Saima, H., Yasmin, A., Nadeem, M. T., Imran, M., & Afzaal, M. (2018). Antioxidant capacity of cinnamon extract for palm oil stability. Lipids in health and disease, 17, 1-8.



