Research Article - (2026) Volume 7, Issue 1
Hepatoprotective and Anticancer Potential of Ethyl Acetate Fraction of Buchanania Lanzan Evidence from Hepg2 Cell Studies and Afb1-Induced Hepatocellular Carcinoma in Rats
2Department of Pharmacology, NBRI (CSIR), Ranapratap Marg, Lucknow-221001 U.P, India
3Department of Pharmacology, MCOPS, Manipal, Karnataka, India
Received Date: Dec 15, 2025 / Accepted Date: Jan 12, 2026 / Published Date: Jan 29, 2026
Copyright: ©2026 G. Dayanand Reddy, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Reddy, G. D., Karthik, R., Varalakshmi, T., Rao, C. H. V., Unnikrishnan, M., Pushpangadan, P. (2026). Hepatoprotective and Anticancer Potential of Ethyl Acetate Fraction of Buchanania Lanzan Evidence from Hepg2 Cell Studies and Afb1-Induced Hepatocellular Carcinoma in Rats. J Vet Heal Sci, 7(1), 01-10.
Abstract
A common medicinal plant in traditional systems, Buchanania lanzan (B. lanzan) has a variety of bioactive compounds that may have therapeutic benefits. The current work uses both in-vitro (HepG2 cell line) and in-vivo (aflatoxin b1 -induced hepatocellular carcinoma in rats) models to examine the hepatoprotective and anticancer properties of the ethyl acetate fraction of B.lanzan. Strong anticancer efficacy was indicated by the ethyl acetate fraction's dose- dependent, considerable inhibition of HepG2 cell proliferation, as shown by in vitro cytotoxicity tests. Its capacity to cause programmed cell death was further validated by morphological alterations and apoptosis indicators. Rats exposed to aflatoxin b1 (AFb1) in the in-vivo investigation showedsigns of hepatocellular carcinoma, including tissue damage, oxidative stress, and increased liver enzymes.The ethyl acetate fraction treatment significantly improved the histological architecture of the liver, decreased lipid peroxidation, raised antioxidant enzyme levels, and restored liver function biomarkers. Overall, the results show that B.lanzanethyl acetate fraction has strong hepatoprotective and anticancer properties, indicating its potential as a natural treatment option for liver conditions such hepatocellular carcinoma.
Keywords
B. lanzan, AFb1, Ethyl Acetate, Anticancer Activity, Hepatocellular Carcinoma
Introduction
B. lanzan Spreng is a member of the Anacardiaceae family and is also referred to as char, achar, and chironji.The tree grows naturally in the tropical deciduous forests of Northern, Western, and Central India, primarily in the districts of Varanasi and Mirzapur in Uttar Pradesh and the states of Chhattisgarh, Jharkhand, and Madhya Pradesh. In addition to India, the plant can be found in Australia, Pacific islands, and other tropical Asian nations [1]. This plant has wound healing activity,antimicrobial,anti-biofilm,antioxidant, antidiabetic, and anticholinesterase activities.Tensile strength and wound contraction analysis demonstrated the extract's ability to heal wounds (excision and incision models). Additionally, the extract's antibacterial qualities were tested against various Gram positive and Gramnegative bacterial strains. The effects of B. lanzanon the formation of biofilms and the disruption of preexisting biofilms were also investigated [2,3]. HHCC was thought to be a serious health issue in Asia and Africa, but it was less common in Europe and America [4].Systemic therapies, typically sorafenib or lenvatinib in the first line and regorafenib, cabozantinib, or ramucirumab in the second, are administered to about half of patients with HCC [5]. About 90% of instances of liver cancer are hepatocellular carcinoma (HCC), which is the most prevalent kind. The primary risk factors for the development of HCC are hepatitis B and hepatitis C virus infection, while non-alcoholic steatohepatitis linked to metabolic syndrome or diabetes mellitus is increasingly common in the West [6]. HCC increased awareness of its molecular pathogenesis as well as issues with treatment and epidemiology [7]. HepG2 is a popular hepatic cell line.In order to determine whether the HepG2 cell line's degree of heterogeneity permits in vitro biomedical research as a model with assured production and quality, we set out to gather current data on the cell line [8]. However, under two-dimensional (2D) cultivation conditions the expression of xenobiotic-metabolizing enzymes and typical liver markers such as albumin is very low. HepG2 cells' metabolic competence has been shown to significantly increase after 21 days of cultivation in a three-dimensional (3D) Matrigel culture system [9]. Rat liver has oxidative stress- inflammatory system activation that is linked to AFb1-induced hepatocarcinogenesis [10]. AFb1 is a genotoxic hepatocarcinogen that is thought to induce cancer by causing DNA adducts that alter the target liver cells' genetic makeup.Cytochrome-P450 enzymes convert AFb1 to the reactive intermediate AFb1-8, 9 epoxide (AFBO), which attaches to the DNA of liver cells to form DNA adducts Consumption of AFb1 may take place in low-exposure regions, eventually building up in the liver and potentially causing liver cancer [11,12]. AFb1, which is mostly present in food crops and their processed foods, has the potential to cause mutagenesis, cancer, and teratogenesis. Although AFb1 exposure can result in either acute or chronic liver toxicity, little research has been done on the long-term effects of acute AFb1 exposure on the liver [13]. Due to their high liposolubility, aflatoxins are easily absorbed from the site of exposure and enter the bloodstream through the respiratory and digestive systems. There are two main types of aflatoxicosis: acute and chronic. Aflatoxin consumption at low to moderate levels causes chronic primary aflatoxicosis [14]. The low resting levels of the hepatic drug-metabolizing enzymes and the inability of the enzyme levels in the livers of deficient rats to react to repeated doses of AFb1 may account for the dietary influence [15]. Antioxidant, antitumour and Heptoprotective Activity of plants were co-related with the phenolics and flavonoids content [16].
Material and Method
Plant Material
Preparation of Plant Extract:
Bark of B. lanzan plant material was collected from the Botanical Garden of the National Botanical Research Institute in Lucknow, India (NBRI). Dr. Sayeeda Khatoon, a taxonomist, confirmed the plant material, and the voucher specimens (NAB2B04293, 200495, and 200492) were archived in the departmental herbarium and the Institutional Museum for future reference.
B. lanzan plant material was rinsed with distilled water to remove dirt and soil. B. lanzan powder (100g) was air-dried (30±20c) and extracted overnight with 10 ml of Ethyl Acectate. It was then centrifuged at 10000 rev/min on a Rota evaporator (Bouchi, USA) and dried in a lyophilizer (Labeonco, USA) under reduced pressure. The extract was then subjected to phytochemical and pharmacological testing.
In Vitro Analysis
HPTLC Analysis
Reflux 5 g of finely powdered medication with 25 ml of Ethyl Acetate over a water bath for 25 minutes three times in a row, then filter and extract the solvent under low pressure. Dissolve 25 milligrammes of extractive in 20 millilitres of Ethyl Acetate. Using an automator applicator (CAMAG Linomat IV), apply 10 microlitres of extract on Merck percoated silica gel 60 F254 plates with a 0.2mm thickness. The plates were then run using a fresh solvent system (ethyl acetate). Chloroform: Methanol (8.5:1.5) ratio for B. lanzan in a CAMAG twin through chamber up to a distance of approximately 9 cm, dry, and scan. UV 254 and visible light were used to view the plates. If necessary, spray the plate with anisaldehyde-sulphuric acid.If necessary, spray the plate with anisaldehyde-sulphuric acid and heat at 110°C for 10 minutes. Record the Rf values and colour of the resolved bands, then use the Desaga video documentation unit to document the movie. The plates were densitometrically scanned with a CAMAG TLC scanner at the relevant wavelength [17].
Cell Viability Assay
The MTT test was used to assess cell viability in control and ethyl acetate extract of B. lanzan treated HepG2 cells following a 48-hour exposure. HepG2 cells were cultivated at 37 °C in a humidified environment with 5% CO2 in Dulbecco's Modified Eagle Medium supplemented with 10% fetal bovine serum and antibiotics. After being seeded into 96-well plates, the cells were left to adhere for the entire night. B. lanzanethyl acetate extract was dissolved in DMSO and given to the cells at certain concentrations; untreated cells were used as controls, while cells treated with DMSO alone were used as solvent controls. and the percentage of cell viability in relation to the control cells was computed. Each well was filled with MTT solution after 48 hours of incubation, and the plates were then left to develop formazan crystals. After removing the medium, the crystals were dissolved with DMSO. Cell viability was computed as a percentage in relation to the control cells by measuring absorbance at 570 nm using a microplate reader [18].
In Vivo Analysis
Animals
Studies are carried out on rats weighing between 140 and 160 g. They were obtained from the toxicity control animal house at the Central Drug Research Institute in Lochnow, as well as cattle raised in the departmental animal facility. The rats were housed in polyacrylic cages with no more than 6 animals per cage and kept under typical laboratory conditions (temperature 25±20C, 12h dark/light cycle). They have free access to a standard dry pellet meal (amrut, India) and unrestricted tap water. The institutional committee for the ethical use of animals reviewed and approved(Reg. No.: 221/2000/CPCSEA) all of the disclosed procedures.
Administration of Drug
The rats were randomly assigned to the experimental and control groups (n=6). Group 1 rats were given 0.9% normal saline. Group II rats with chemically induced HCC (AFb1) were given a single oral administration of AFb1 (200mg/kg b.w.). Group III rats received 100 mg/kg b.w.i.p. of B. lanzan in Ethyl Acetate. Groups IV and V were given i.p injections of 200 and 400 mg/kg/b.w, respectively, whereas Group VI rats received 6 mg/kg b.w of cisplatin.
Laboratory Investigations
Body weights were measured on the day of receipt, before randomisation, on the day of dosing, and weekly thereafter for the treatment and recovery groups, while food and drink consumption were documented daily and reported weekly. All animals had their blood drawn for haematology and clinical biochemistry. Animals were placed in metabolic cages and fasted overnight before blood sample, but they had unlimited access to water. Blood was drawn from the retroorbital plexus using a micro-hematocrit heparinised glass capillary tube.
Potassium EDTA was employed as an anticoagulant during haematology tests. Blood samples were stored in serum tubes at room temperature for around 30 minutes before being aliquoted. After clotting, the blood tubes were centrifuged at 3000 rpm for 15 minutes. The supernatants were decanted and stored at -70°C for subsequent analysis.
Biochemical Marker Estimation
The biochemical markers like SGOT(U/l), SGPT(U/l), SALP(U/l), Bilirubin level (U/l) and Gamma glutamyl transpeptidase, GGT (U/l) were determined for both control and treated groups by using standard biochemical method [19].
Estimation of Free Radical Generation
The liver homogenate (5%) in ice-cold phosphate buffer was centrifuged at 800X g for 10 minutes, followed by centrifugation of the supernatant at 12,000X g for 15 minutes to obtain the mitochondrial fractions, which were used to measure lipid peroxidation (LPO), superoxide dismutase activity (SOD), catalase activity (CAT), reduced glutathione (GSH), and glutathione peroxidase (GPX) [20].
Hematological Estimation
Red blood cell counts, White blood cell counts and hemoglobin were estimated with the help of hematology analyzer (Medonic CA620, Boule, Sweden) [21].
Results
HPTLC Analysis of Ethyl Acetate Extract of B. Lanzan:
The HPTLC fingerprint analysis revealed thirteen different peaks with Rf values ranging from 0.22 to 1.20, indicating the presence of several phytoconstituents in the test sample. Peak 9 (Rf 0.78-0.91) had the biggest area percentage (35.84%), indicating that it was the main component, followed by Peak 13 (31.63%). Other peaks had area percentages ranging from 10.72% to 19.50%, indicating minor to moderate ingredients, corroborating the extract's chemical complexity and fingerprint profile. (Figure 1; Table 1).
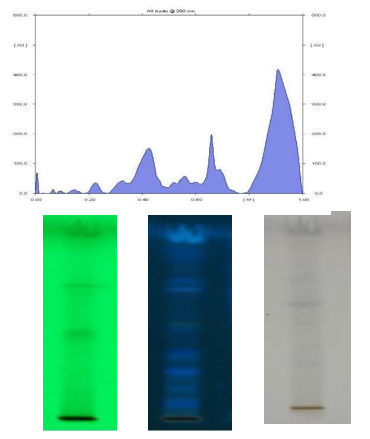
Figure 1: HPTLC Fingerprint of Chloroform Extract of Bark B. lanzan. Solvent: Chloroform: Methanol (8.5:1.5)
|
Peak |
Start Rf |
Height % |
End Rf |
Area % |
|
1 |
0.22 |
17.13 |
0.24 |
12.103 |
|
2 |
0.25 |
23.109 |
0.26 |
14.58 |
|
3 |
0.26 |
13.51 |
0.30 |
11.57 |
|
4 |
0.33 |
11.77 |
0.40 |
11.62 |
|
5 |
0.43 |
11.36 |
0.46 |
10.72 |
|
6 |
0.46 |
11.49 |
0.50 |
11.15 |
|
7 |
0.60 |
12.108 |
0.67 |
13.40 |
|
8 |
0.67 |
17.94 |
0.78 |
13.54 |
|
9 |
0.78 |
24.29 |
0.91 |
35.84 |
|
10 |
0.92 |
18.15 |
0.97 |
15.59 |
|
11 |
0.97 |
18.31 |
0.105 |
19.14 |
|
12 |
0.106 |
18.83 |
0.113 |
19.50 |
|
13 |
0.108 |
32.36 |
1.20 |
31.63 |
Table 1: HPTLC Values
Cell Viability in Control and Ethyl Acetate Extract of B. Lanzan treated Hep G2 Cell Safter 48 h of Exposure
Cell viability of HepG2 cells treated with the ethyl acetate extract of B.lanzan was assessed after 48 h of exposure. The control and DMSO-treated cells showed 100% viability, indicating that the solvent had no cytotoxic effect. Similarly, cells treated with 20 µg/ ml of the extract also exhibited 100% viability. A concentration- dependent decrease in cell viability was observed at higher concentrations. Cell viability reduced to 68.68 ± 1.30% at 40 µg/ ml, followed by a further decrease to 37.58 ± 1.39% and 20.46 ± 1.42% at 60 and 80 µg/ml, respectively. The lowest cell viability of 3.42 ± 1.41% was recorded at 100 µg/ml as shown in (Table 2). These findings indicate that the ethyl acetate extract of B.lanzan significantly reduced HepG2 cell viability in a dose-dependent manner.
|
Treatment |
Concentration |
Percentage of inhibition |
|
Control |
- |
0 |
|
DMSO |
1%(v/v) |
0 |
|
Buchanania lanzan |
20(µg/ml) |
0 |
|
Buchanania lanzan |
40(µg/ml) |
31.32±1.30a |
|
Buchanania lanzan |
60(µg/ml) |
62.42±1.39a |
|
Buchanania lanzan |
80(µg/ml) |
79.54±1.42a |
|
Buchanania lanzan |
100(µg/ml) |
96.58±1.41a |
Values are mean ± SEM;n=6aP<0.001comparedwithcontrolgroup
Table 2: Cell Viability in Control and Ethyl Acetate Extract of B. lanzan
Effect of Ethyl Acetate Extract of B. Lanzan on Body Weight, Liver Weight and Average Liver Weight in (AFb1) Induced HCC Rat
Ethyl acetate extracts of B. lanzan, at a dose of 100, 200 mg and 400 mg once daily for 28 days and standard Cisplatin at a dose of 6 mg/kg were subjected for studying the body weight, liver weight and average liver weight in HCC rats. The study showed that the liver weights were significantly increased from 19.3,15.33 to 22.8,15.78 in AFb1 group to 21.1,15.36 to 19.8, 10.66 in B. lanzan treated animal. whereas, standard drug Cisplatin 6 mg/kg showed significant reduction in liver weight compared to AFb1 group figure 2.
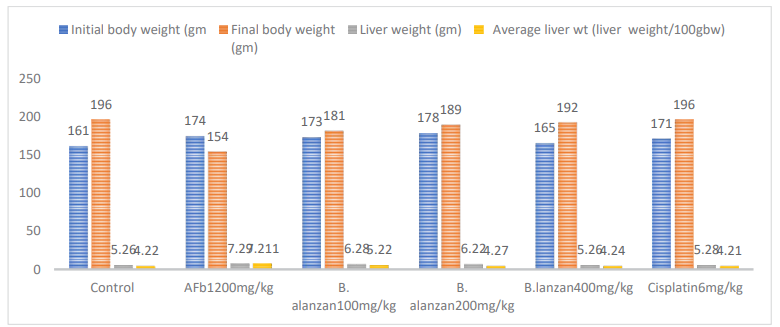
Figure 2: Effect of Ethyl Acetate Extract of B. Lanzan on Body Weight, Liver Weight And Average Liver Weight in (Afb1) Induced HCC Rat Values are mean ±SEM of 6 rats in each group
Effect of Ethyl Acetate of B. Lanzan on SGOT, SGPT, SALP, BL and GGT Against Afb1 Induced HCC
In the AFb1 group, the level of SGOT (221.22-392.21, P<0.001), SGPT (121.25-408.51, P<0.001), SALP (261.11-467.38, P<0.01), BL (0.106-1.62, P<0.001) and GGT (50.18-188.9, P<0.001). in contrast, the groups treated with B. lanzanextract at dose of (100- 400 mg/kg) once daily for 28 days prevented the incidence of cancer of in a dose related manner. The ranges of protection in the serum marker were found to be SGOT (324.14-248.84, P<0.05 to p<0.001), SGPT (355.33-154.03, P<0.05 to p<0.001), SALP (378.22-273.91, P<0.01 to p<0.001), BL (1.48-0.111, p<0.001), and GGT (172.1-104.9, P<0.001) respectively. The protection of cisplatin ranged for SGOT (392.21-226.38, p<0.001), SGPT (408.51-124.38, p<0.001), SALP (467.38-268.34, p<0.001), BL (1.62-0.99, p<0.01) and GGT (188.9-58.9, p<0.001) respectively as shown in (Table 3).
|
Groups |
Treatment |
Dose |
SGOT |
SGPT |
SALP |
BL |
GGT |
|
I |
Control |
--- |
221.22±3.19 |
121.25±1.58 |
261.11±11.31 |
0.106±0.03 |
50.18±5.1 |
|
II |
AFb1 |
200mg/kg AFb1 |
392.21±22.32z |
408.51±29.78z |
467.38±28.33z |
1.62±0.06z |
188.9±8.2z |
|
III |
Buchananialanzan |
100mg/kg |
324.14±19.51a |
355.33±27.24 |
408.22±24.38a |
1.48±0.05 |
172.1±6.8 |
|
IV |
Buchananialanzan |
200mg/kg |
283.48±18.36b |
248.11±26.21b |
324.21±23.33b |
0.119±0.13a |
154.8±5.6b |
|
V |
Buchananialanzan |
400mg/kg |
248.84±18.90c |
154.03±22.31c |
283.91±21.83c |
0.111±0.12b |
104.9±4.2c |
|
VI |
Cisplatin |
6mg/kg |
226.38±19.02c |
124.38±19.44c |
278.34±26.62c |
0.109±0.14b |
68.9±6.1c |
P values: a<0.05, b<0.01, c<0.001compared with group II AFb1
Values are mean ±S.E.M. of 6 rats in each group
P values: z<0.001 compared with respective control group
Table 3: Effect of The Ethyl Acetate Extract of B. Lanzanon SGOT(U/L), SGPT(U/L), SALP(U/L), Bilirubin Level (U/L) and Gamma Glutamyltranspeptidase, GGT (U/L) in Serum of Rat
Effect of Ethyl Acetate extract of B. lanzanon LPO, SOD, CAT, GPX, GST and GSH against AFb1 induced HCC
Administration of AFb1 led to increase in the levels of LPO (0.76- 6.24, p<0.001), and decrease in SOD (136.12-57.87, p<0.001), CAT (46.02-9.38), p<0.001), GPX (4.78-1.60), p<0.001), GST (1.34- 0.69, p<0.001) and GSH (3.68-3.26, P<0.001) levels in the 5% w/v liver homogenate. Treatment of rats with Ethyl Acetateextract of B. lanzan at dose of (100-400 mg/kg b.w) markedly prevented theAFb1induced alterations of various parameters LPO (4.109- 1.39, p<0.05 to p<0.01), SOD (82.18-102.50, p<0.05 to p<0.001), CAT (31.67-38.89, p<0.05 to p<0.001), GPX (2.87- 3.32, p<0.001), GST (0.96-0.118, p<0.001) and GSH (3.39-3.51, p<0.001) respectively. The protection of Cisplatin ranged for LPO (57.87-126.68, p<0.05 to p<0.01), SOD (57.87-126.68, p<0.05 to p<0.001), CAT (9.38-39.02, p<0.05 to p<0.001), GPX (1.60- 3.49, p<0.001), GST (0.69-1.24, p<0.001) and GSH (3.26-3.54, p<0.001) respectively as shown in (Table 4.)
|
Groups |
Treatment |
Dose |
SOD |
CAT |
LPO |
GPx |
GST |
GSH |
|
I |
Control |
--- |
136.12±8.4 |
46.02±1.60 |
0.76±0.01 |
4.78±0.02 |
1.34±0.16 |
3.68±0.34 |
|
II |
AFb1 |
200mg/kg AFb1 |
57.87±8.12z |
9.38±0.61z |
6.24±1.24z |
1.60±0.01z |
0.69±0.02z |
3.26±0.32 |
|
III |
Buchananialanzan |
100mg/kg |
82.18±9.56 |
31.67±1.40a |
4.109±0.43 |
2.87±0.02c |
0.96±0.04c |
3.39±0.34 |
|
IV |
Buchananialanzan |
200mg/kg |
101.29± 12.49a |
34.38±1.32c |
2.44±0.54a |
2.102±0.03c |
0.104±0.02c |
3.41±0.35 |
|
V |
Buchananialanzan |
400mg/kg |
102.50±9.21c |
38.89±0.89c |
1.39±0.82b |
3.32±0.02c |
0.118±0.05c |
3.51±0.32 |
|
VI |
Cisplatin |
6mg/kg |
126.68±6.28c |
39.02±1.10c |
0.108±0.62b |
3.49±0.02c |
1.24±0.09c |
3.54±0.33 |
Values are mean ±S.E.M. of6ratsineachgroup
P values: z<0.001 compared with respective control group
P values: a<0.05, b<0.01, c<0.001 compared with group II AFb1
Table 4: Effect of Ethyl Acetate Extract of B. alanzan on LPO, SOD, CAT, GPX, GST and GSH Against AFb1 Induced HCC
Effect of Ethyl Acetate Extract of B. Lanzanon Haematological Parameters (RBC, WBC and Hb) of Control and Afb1 Induced HCC
The figure 3shows the level of Hb, RBC counts, all of which were significantly decreased (10.101-8.54, p<0.001) and (8.52-5.118, p<0.05) and with simultaneous increase in WBC (6.76-9.47, p<0.01) with respect to control. In contrast, the groups treated with B. lanzan extract at dose of (100-400 mg/kg b.w) once daily for 28 days prevented the cancer in a dose related manner. The range of protection in the Hb, RBC and WBC shows (8.87-9.98, p<0.05), (6.54-7.9) and (8.65- 7.32, p<0.01). the protection of Cisplatin ranged for Hb (8.54-10.32, p<0.01), RBC (5.118-8.32, p<0.05) and WBC (9.47-10.32, P<0.01) respectively.
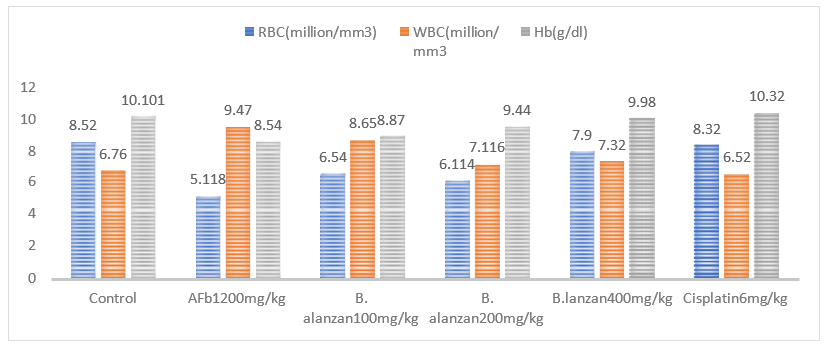
Figure 3: Effects of B. Alanzan on Haematological Parameter (RBC, WBC and Hb) of Control and (Afb1) Induced HCC in Rat
Effect of Ethyl Acetate Extract of B. Lanzanon The Level of Deoxyribonucleic Acid, DNA (Mg/G Wet Tissue) and Ribonucleic Acid, RNA (Mg/G Wet Tissue) and Protein (G/Dl)
Aflatoxin Bâ? (200 mg/kg) significantly increased DNA (7.72 ± 0.28 mg/g) and RNA (8.11 ± 0.56 mg/g) levels and reduced protein content (6.77 ± 0.82 g/dL) compared to control rats (DNA: 5.58 ± 0.39 mg/g; RNA: 7.49 ± 0.26 mg/g; protein: 7.12 ± 1.06 g/dL). Treatment with ethyl acetate extract of B.lanzan at 100, 200, and 400 mg/kg dose-dependently normalized these alterations, with the 400 mg/kg dose showing near-normal DNA (5.62 ± 0.38 mg/g), RNA (7.62 ± 0.38 mg/g), and improved protein levels (7.77 ± 1.55 g/dL). Cisplatin (6 mg/kg) produced comparable protective effects shown in Table 5.
|
Groups |
Treatment |
Dose |
DNA |
RNA |
Protein |
|
I |
Control |
--- |
5.58±0.39 |
7.49±0.26 |
7.117±1.06 |
|
II |
AFb1 |
200mg/kg AFb1 |
7.72±0.28z |
8.114±0.56z |
6.77±0.82x |
|
III |
Buchananialanzanon |
100mg/kg |
6.47±0.32a |
8.54±0.44 |
6.105±1.12 |
|
IV |
Buchananialanzanon |
200mg/kg |
5.79±0.34b |
7.118±0.45 |
7.49±1.14 |
|
V |
Buchananialanzanon |
400mg/kg |
5.62±0.38b |
7.62±0.38 |
7.77±1.55 |
|
VI |
Cisplatin |
6mg/kg |
5.60±0.35c |
7.52±0.30a |
7.101±1.26 |
Values are mean ± S.E.M. of 6 rats in each group
P values:x<0.05, z<0.001 compared with respective control group
P values:a<0.05, b<0.01, c<0.001 compared with group II AFb1
Table 5: Effect of Ethyl Acetate Extract of B. Lanzan on The Level of Deoxyribonucleic Acid,DNA(Mg/G Wettissue)and Ribonucleic Acid, RNA(Mg/G Wet Tissue) and Protein (G/Dl)
Estimation of Ethyl Acetate Extract of B. lanzan on RNA, DNA, Protein
The effect of the ethyl acetate extract of B. lanzan on cellular macromolecules was assessed by estimating DNA, RNA, and protein content using standard biochemical methods. HepG2 cells or liver tissue samples were treated with different concentrations of the ethyl acetate extract for 24–48 hours, while untreated samples served as controls. After treatment, cells or tissues were harvested and homogenized under cold conditions. DNA content was estimated using the diphenylamine method, RNA content by the orcinol method, and protein concentration by Lowry’s method with bovine serum albumin as the standard. The intensity of the colordeveloped in each assay was measured using a UV–visible spectrophotometer at their respective wavelengths. Concentrations were calculated from standard calibration curves and expressed per milligram of tissue or per 10ⶠcells. All experiments were conducted in triplicate, and the data were expressed as mean ± SEM and statistically analyzed using one-way ANOVA [22].
Histopathological Analysis
The livers were removed immediately upon autopsy for histological examination, and the tissues were stored in 10% formalin for at least 24 hours. The paraffin sections were then treated (Automatic Tissue Processor, Lipshaw) and sliced into 5 um-thick sections using a rotary microtome. The sections were then stained with haematoxilin-eosin dye (Merck) and mounted on Canada balasam. The histology slides were examined and photographed under a picture zoom microscope (3.2X10 and 10X10) [23].
Statistical Analysis
Data are expressed as mean +SEM (standard error of mean). The difference among means has been analyzed by unpaired students t-test.
Histopathological Analysis
Histopathological analysis of liver tissues was carried out to assess the hepatoprotective and anticancer potential of the ethyl acetate fraction of B.lanzan in AFbâ?ÂÂ-induced hepatocellular carcinoma in rats. Liver sections from the normal control group showed well- preserved hepatic architecture with normal hepatocytes arranged around the central vein and intact sinusoidal spaces. In contrast, AFbâ?ÂÂ-treated rats exhibited marked histopathological alterations, including disruption of hepatic architecture, hepatocellular degeneration, cytoplasmic vacuolization, nuclear abnormalities, inflammatory cell infiltration, and tumor-like nodular formations, confirming hepatocellular carcinoma induction. Treatment with the ethyl acetate fraction of B.lanzan resulted in significant improvement in liver histology, with reduced cellular damage, decreased inflammation, and gradual restoration of normal hepatic architecture in a dose-dependent manner. At higher doses, liver sections showed near-normal hepatocyte arrangement with minimal pathological changes, indicating strong hepatoprotective and anticancer effects of the extract against AFbâ?ÂÂ-induced liver damage.
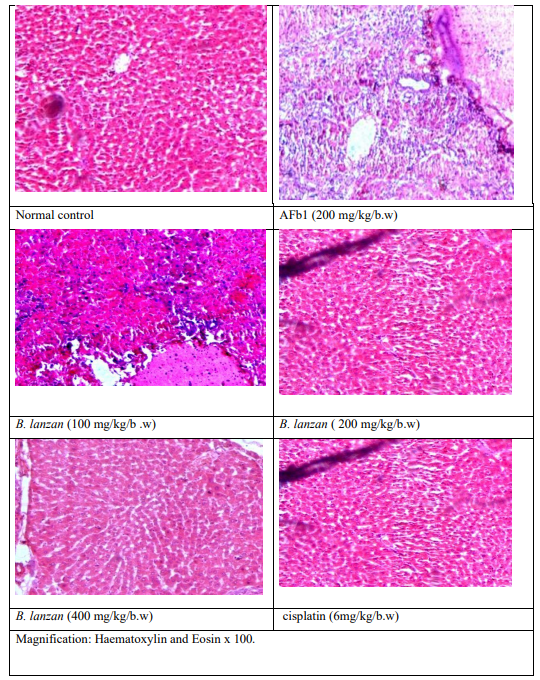
Figure 4: Histopathological Analysis of B. lanzan Against AFb1 Induced HCC
Discussion
While triterpenoids, flavonoids, and calotropins are found in lesser concentrations than dichloromethane and other extract fractions, the ethyl acetate fraction has the highest concentrations of cardiac glycosides and phenolics [24]. The ethyl acetate fraction of Dianthus superbus has been shown to have comparable anticancer efficacy in HepG2 cells, exhibiting dose-dependent activation of apoptosis and inhibition of anti-apoptotic proteins in human hepatoma HepG2 cells [25]. The ethyl acetate fraction extract (EAFE) of Glechoma hederacea L. was demonstrated to reduce HepG2 cell growth, promote apoptosis, cause S phase arrest, and activate caspase-dependent pathways, further demonstrating the antiproliferative efficacy of ethyl acetate fractions in liver cancer models [26]. Pre-treatment of rats with herbal extracts significantly reduced AFbâ?-induced elevations in liver enzyme markers and oxidative stress, indicating hepatoprotective potential. These studies are consistent with the protective effect against AFbâ? induced liver damage in rats [27]. Basic actions caused by AFb1 include intricate molecular processes. Over the past few decades, researchers have shown that AFb1 can cause oxidative stress, lipid peroxidation, apoptosis, mitochondrial dysfunction, autophagy, necrosis, immune repression, and inflammatory response, which can ultimately lead to a number of acute or chronic diseases in humans and animals, such as cancer, hepatitis, abnormalities in mutations, and problems with reproduction [28]. The aforementioned natural compounds may have a chemopreventive and therapeutic effect in HCC because of their strong anti-inflammatory and antioxidant qualities and their capacity to alter various signaling pathways linked to the carcinogenesis process [29]. Since flavonoids are naturally occurring compounds that have been found in foods and drinks since ancient times, they do not have the same harmful side effects as manmade anti-cancer medications. Numerous studies have demonstrated their potent beneficial effects on immune response modulation, inflammation reduction, and cell support and restoration [30]. Hepatocellular degeneration/regeneration, inflammation, necrosis, small-cell dysplasia, and progressive, morphologically different preneoplastic lesions/alterations can all be seen in HCC [31].
Conclusion
The study shows that B. lanzan ethyl acetate fraction has considerable hepatoprotective and anticancer potential. The fraction demonstrated its capacity to target hepatocellular carcinoma cells by successfully inhibiting cell proliferation and inducing apoptosis in HepG2 cell tests. Treatment with the ethyl acetate fraction reduced oxidative stress, improved histological architecture, restored serum liver enzymes, and strengthened antioxidant defences in rats with AFb1-induced hepatocellular cancer. These results imply that apoptosis-regulating, anti-inflammatory, and antioxidant pathways mediate the hepatoprotective and anticancer effects. Overall, the results show that B. lanzan ethyl acetate fraction has therapeutic potential as a natural treatment for hepatocellular carcinoma and liver toxicity, which calls for more research to identify bioactive chemicals and clarify molecular mechanisms.
Acknowledgments
We thank to all Ethno Pharmacology and Administrative staffs from NBRI (CSIR), Lucknow for their support and timely completion of studies.
Conflict of interest
There are no conflicts of interest declare by authors in this manuscript.
References
- Siddiqui, M. Z., Chowdhury, A. R., Prasad, N., & Thomas, M. (2014). Buchanania lanzan: a species of enormous potentials. World journal of pharmaceutical sciences, 374-379.
- Pattnaik, A., Sarkar, R., Sharma, A., Yadav, K. K., Kumar, A., Roy, P., ... & Sen, T. (2013). Pharmacological studies on Buchanania lanzan Spreng.-A focus on wound healing with particular reference to anti-biofilm properties. Asian Pacific journal of tropical biomedicine, 3(12), 967-974.
- Meher, N., Kisan, B., Swain, S. K., & Sahoo, A. K. (2024). Industrial importance seed kernel of Buchanania lanzan in contributing antioxidant, antidiabetic, and anticholinesterase activities. Food Bioscience, 59, 104135.
- Bruix, J., Boix, L., Sala, M., & Llovet, J. M. (2004). Focus onhepatocellular carcinoma. Cancer cell, 5(3), 215-219.
- Llovet, J. M., Castet, F., Heikenwalder, M., Maini, M. K., Mazzaferro, V., Pinato, D. J., ... & Finn, R. S. (2022). Immunotherapies for hepatocellular carcinoma. Nature reviews Clinical oncology, 19(3), 151-172.
- Llovet, J. M., Kelley, R. K., Villanueva, A., Singal, A. G.,Pikarsky, E., Roayaie, S., ... & Finn, R. S. (2024). Author correction: hepatocellular carcinoma. Nature Reviews. Disease Primers, 10(1), 10.
- Bruix, J., Boix, L., Sala, M., & Llovet, J. M. (2004). Focus onhepatocellular carcinoma. Cancer cell, 5(3), 215-219.
- Arzumanian, V. A., Kiseleva, O. I., & Poverennaya, E. V. (2021). The curious case of the HepG2 cell line: 40 years of expertise. International journal of molecular sciences, 22(23), 13135.
- Luckert, C., Schulz, C., Lehmann, N., Thomas, M., Hofmann, U., Hammad, S., ... & Hessel, S. (2017). Comparative analysis of 3D culture methods on human HepG2 cells. Archives of toxicology, 91(1), 393-406.
- Maurya, B. K., & Trigun, S. K. (2016). Fisetin modulates antioxidant enzymes and inflammatory factors to inhibit aflatoxinâ?B1 induced hepatocellular carcinoma in rats. Oxidative medicine and cellular longevity, 2016(1), 1972793.
- Hamid, A. S., Tesfamariam, I. G., Zhang, Y., & Zhang, Z. G. (2013). Aflatoxin B1-induced hepatocellular carcinoma in developing countries: Geographical distribution, mechanism of action and prevention. Oncology letters, 5(4), 1087-1092.
- Gramantieri, L., Gnudi, F., Vasuri, F., Mandrioli, D., Fornari, F., Tovoli, F., ... & Giovannini, C. (2022). Aflatoxin B1 DNA- adducts in hepatocellular carcinoma from a low exposure area. Nutrients, 14(8), 1652.
- Yan, J., Chen, L., Zhang, L., Zhang, Z., Zhao, Y., Wang, Y., & Ou, J. (2022). New insights into the persistent effects of acute exposure to AFB1 on rat liver. Frontiers in Microbiology, 13, 911757.
- Fetaih, H. A., Dessouki, A. A., Hassanin, A. A., & Tahan,A. S. (2014). Toxopathological and cytogenetic effects of aflatoxin B1 (AFB1) on pregnant rats. Pathology-Research and Practice, 210(12), 1079-1089.
- Pereira, C., Calhelha, R. C., Barros, L., & Ferreira, I. C. (2013). Antioxidant properties, anti-hepatocellular carcinoma activity and hepatotoxicity of artichoke, milk thistle and borututu. Industrial Crops and Products, 49, 61-65.
- Xu, F., Li, Y., Cao, Z., Zhang, J., & Huang, W. (2021).AFB1-induced mice liver injury involves mitochondrial dysfunction mediated by mitochondrial biogenesis inhibition.Ecotoxicology and environmental safety, 216, 112213.
- Bhargava, A., Shrivastava, P., & Tilwari, A. (2021). HPTLC analysis of Fumaria parviflora (Lam.) methanolic extract of whole plant. Future Journal of Pharmaceutical Sciences, 7(1), 1.
- Akter, S., Addepalli, R., Netzel, M. E., Tinggi, U., Fletcher, M. T., Sultanbawa, Y., & Osborne, S. A. (2019). Antioxidant-rich extracts of Terminalia ferdinandiana interfere with estimation of cell viability. Antioxidants, 8(6), 191.
- Hossain, M. A., & Nagooru, M. R. (2011). Biochemical profiling and total flavonoids contents of leaves crude extract of endemic medicinal plant Corydyline terminalis L. Kunth. Pharmacognosy Journal, 3(24), 25-30.
- Akinmoladun, A. C., Obuotor, E. M., & Farombi, E. O. (2010). Evaluation of antioxidant and free radical scavenging capacities of some Nigerian indigenous medicinal plants. Journal of Medicinal food, 13(2), 444-451.
- Ofem, O. E., Ani, E. J., & Eno, A. E. (2012). Effect of aqueous leaves extract of Ocimum gratissimum on hematological parameters in rats. International Journal of Applied and Basic Medical Research, 2(1), 38-42.
- Abubakar, B. M., Izham, N. H. M., Salleh, F. M., Omar, M.S. S., & Wagiran, A. (2018). Comparison of different DNA extraction methods from leaves and roots of Eurycoma longifolia plant. Advanced Science Letters, 24(5), 3641-3645.
- Gopal, V., Mandal, V., Tangjang, S., & Mandal, S. C. (2014). Serum biochemical, histopathology and SEM analyses of the effects of the Indian traditional herb Wattakaka volubilis leaf extract on wistar male rats. Journal of Pharmacopuncture, 17(1), 13.
- Chaisupasakul, P., Pekthong, D., Wangteeraprasert, A.,Kaewkong, W., Somran, J., Kaewpaeng, N., ... & Srisawang, P. (2024). Combination of ethyl acetate fraction from Calotropis gigantea stem bark and sorafenib induces apoptosis in HepG2 cells. Plos one, 19(3), e0300051.
- Yu, J. Q., Yin, Y., Lei, J. C., Zhang, X. Q., Chen, W., Ding, C.L., ... & Zou, G. L. (2012). Activation of apoptosis by ethyl acetate fraction of ethanol extract of Dianthus superbus in HepG2 cell line. Cancer Epidemiology, 36(1), e40-e45.
- Chao, W. W., Liou, Y. J., Ma, H. T., Chen, Y. H., Chou, S.T.(2021) Phytochemical composition and bioactive effects of ethyl acetate fraction extract (EAFE) of Glechoma hederaceaL. Journal of Food Biochemistry, 45(7): e13815.
- Shyamal, S., Latha, P. G., Suja, S. R., Shine, V. J., Anuja, G. I., Sini, S., ... & Rajasekharan, S. (2010). Hepatoprotective effect of three herbal extracts on aflatoxin B1-intoxicated rat liver. Singapore medical journal, 51(4), 326.
- Dai, C., Tian, E., Hao, Z., Tang, S., Wang, Z., Sharma, G., ... & Shen, J. (2022). Aflatoxin B1 toxicity and protective effects of curcumin: Molecular mechanisms and clinical implications. Antioxidants, 11(10), 2031.
- Alnajjar, A. M., & Elsiesy, H. A. (2015). Natural products and hepatocellular carcinoma: a review. Hepatoma Research, 1, 119-124.
- Kopustinskiene, D. M., Jakstas, V., Savickas, A., & Bernatoniene, J. (2020). Flavonoids as anticancer agents. Nutrients, 12(2), 457.
- Erkekoglu, P., Oral, D., Chao, M. W., & Kocer-Gumusel,B. (2017). Hepatocellular carcinoma and possible chemical and biological causes: a review. Journal of Environmental Pathology, Toxicology and Oncology, 36(2).


