Research Article - (2025) Volume 8, Issue 1
Health Risk Assessment of Some Heavy Metals of Saccostrea Cucullata Soft Tissue in Northern Shores of the Gulf of Oman
Received Date: Jan 24, 2025 / Accepted Date: Feb 19, 2025 / Published Date: Feb 23, 2025
Copyright: ©©2025 Mehran Loghmani, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Firouzkouhi, Y., Loghmani, M. (2025). Health Risk Assessment of Some Heavy Metals of Saccostrea Cucullata Soft Tissue in Northern Shores of the Gulf of Oman. J Mari Scie Res Ocean, 8(1), 01-11.
Abstract
Contamination with adverse effects on food quality and safety is a severe threat to human health. That is why food security assessment is a high-priority issue in the world. This study aimed to determine the concentration of heavy metals and estimate their consumption risk in the of Saccostrea cucullata soft tissue oyster collected (80 samples) from the eastern shores of Chabahar Bay (Gowatr, Beris, Ramin, and Tis) seasons of autumn (2020) and spring (2021). Heavy metal concentrations of the collected samples were measured after bioassay and preparation of soft tissue using an atomic absorption spectrometer. The consumption risk of THQ, estimated daily intake (EDI), and estimated weekly intake (EWI) were also assessed. According to the results, the overall mean concentrations of heavy metals copper, nickel, and cadmium were 145.25, 7.6, and 0.078 μg/g.dry weight, respectively. A significant difference was observed between the seasons in the study of metal concentrations (p<0.05). Estimation of daily and weekly metal intake in adults and children showed that the highest uptake was related to copper and the lowest uptake to cadmium. The consumption risk of THQ for adults and children is less than one, indicating that consumption of S.cucullata in the study area does not pose a health hazard. A comparison of heavy metals with world standards (WHO, FAO, and FDA) indicates high nickel concentrations.
Keywords
Heavy Metals, Hazard Index, Saccostrea Cucullate, Chabahar BayIntroduction
In recent years, environmental pollution has increased due to the accelerating industrialization of the world the increasing energy demand, and the excessive exploitation of natural resources. Heavy metal pollution has become an important and worrying environmental issue worldwide due to its toxicity, persistence, and bioaccumulation in the environment and its non-degradability [1]. Given the effects of bioaccumulation and biomagnification on various aquatic organs in the aquatic environment, even at deficient concentrations, they threaten human health through food chains such as oysters and other contaminated edible aquatic animals [2,3].
Seafood, after grains and legumes, is the most important human food that provides about 15% of the protein intake in the world and has long been an important part of the human diet [4]. Seafood, especially Bivalvia, is a valuable source of omega-3These compounds have been shown to have beneficial effects on human health. Therefore, the consumption of fish and oysters can be useful and valuable food in the diet and human health [5]. Risk assessment involves identifying and describing the risk and assessing the exposure. This leads to deciding whether legal action should be taken to prevent damage to this food or raw material [2].
Bivalve mollusks can store large amounts of heavy metals in their bodies because they have filtering properties. For this reason, they cause more accumulation of metals in their tissues [6]. Oysters are one of the major seafood in the human diet and are one of the main components of the biochemical cycle in the marine system as a filter. Saccostrea cuculata has been reported to be one of the most important types of environmental pollution index due to their high tolerance to various environmental conditions and long-term inactivity, high accumulation ability of various pollutants (heavy metals) [7].
Nourozifard et al., investigated the concentrations of Cu, Pb, Zn, Cd, Ni, and Cr in the shell and soft tissue of rocky oysters (Saccostrea cucullata), water, and sediments on Qeshm Island [8]. Their results showed that essential metals such as Zn and Cu accumulate in the crust due to their biological role in soft tissue and Pb and Cd are caused by their non-essential metals and tendency to accumulate in calcareous tissues and bind to organic matter [8]. Therefore, S. cucullata crust can be used as biomonitoring to evaluate Cu and Zn in water and Cu in sediments, and soft tissue can be used to monitor Cu and Zn in sediments. Also, in their research, Cu and Ni in the water exceeded all relevant standards, and Cu in sediments exceeded the effects range low (ERL), Threshold effect level (TEL), and United States Environmental Protection Agency (USEPA) standards. This could be a serious warning to living species of these ecosystems, including oysters. Heavy metals in oral tissues were most bivalve samples below the safety limits set by national and international regulations. Therefore, in the long term, monitoring and controlling the consumption of bivalves will be necessary. However, the current accumulation of bivalves is safe. Nevertheless, continuous and lifelong use for more than 70 years may increase cancer risk.
The Oman Sea is a vital transportation route for oil-producing countries in the Persian Gulf. Chabahar Bay, located on the coastline of Makoran, southeastern Iran, is a port and free industrial zone on the shores of the Oman Sea [9]. The particular importance of Chabahar Bay is due to its Ω shape and limited water circulation. Chabahar Bay has a humid climate, hot summers, and mild winters. Fishing and maritime trade are the main activities of this bay [9]. In recent years, petrochemical industries have started their activities in Chabahar Bay. Therefore, understanding the level of pollution in the region is an important step toward its protection and sustainable development. Nowadays, the bay is exposed to high pollution risks such as sewage discharge from vessels, boats, and water-balancing vessels [10].
This study aimed to determine the contamination of heavy metals and the risk of their consumption in S. cucullata due to its high distribution on the shores of the Oman Sea and the important export species in this area.
Materials and Methods
Preparation of Samples: In this study, oyster samples were collected from four stations on the east coast of Chabahar (very important commercial, shipping, and fishing areas in the Oman Sea) including, Gowatr, Beris, Ramin, and Tis in the two seasons of autumn (2020) and spring (2021)(Figure 1).
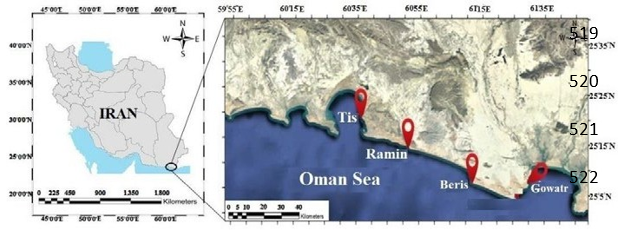
Figure 1: Sampling Location in Oman Sea (Makran Sea).
Saccostrea cuculata has become widespread in the sampling areas, both on natural rocky shores and in the hard parts of the piers. In the present study, to measure heavy metals from each station and in each season, 20 specimens of S. cuculata of almost the same size were collected at full tide using a hammer. Oyster shell sediments were washed with seawater. The samples were placed in heat-insulated polystyrene bags and inside Styrofoam. In the laboratory, the samples were first washed with distilled water and the length and weight of the samples were measured with a caliper with an accuracy of 1 mm and a digital scale with an accuracy of 0.01 g. The equipment needed to test and extract the soft tissue of the oyster was sterilized with 5% nitric acid. Using a steel spatula and a hammer, the hard part of the open bivalve and soft tissue were removed. Samples were kept at -20°C until the concentration of metals was measured [11].
The soft tissues were placed in an oven at 70°C for 24 hours and dried and finally pulverized. To carry out the acid digestion process, 1g of the tissue sample was pulverized and poured into glass tubes with 10 ml of nitric acid. They were then placed on a hot plate for one hour at 40°C and three hours at 140°C for complete digestion. After acid digestion, the samples were filtered and made up to 25 ml with deionized water [12]. Finally, after preparing the samples, the metals were read using an atomic absorption spectrometer (Varian AA220).
Converting Unit of the Dry Weight of Heavy Metal Concentration to A Unit of Wet Weight
The metal concentrations were calculated in terms of dry weight. However, to investigate the ecological risk of consumption in all cases, we must convert the dry weight to a wet weight by multiplying 0.17 (based on the average percentage of soft tissue moisture in the present study) by the heavy metal concentration of the present study. To calculate the percentage of soft tissue moisture, we used the equation of MC = (w-d)*100/w, MC is the moisture content (%), W is the weight while wet, and d is the weight while dry.
Consumption Risk Assessment
Estimated daily intake (EDI) (μg/kg body weight per day) was calculated using the equation of EDI = Cm*FIRd / BW, where Cm represents the mean concentration of heavy metals in tissue in terms of fresh weight (μg/g), FIRd is the oyster intake rate per day (g/day) (36.4 g per day) and BW is the average body weight (kg) (adults 70 and children 16 kg) [13,14].
The uptake rate of heavy metals in organisms based on nutrition is directly related to the content of metals in food and the amount of food consumed by organisms. The method and values provided by FAO and WHO in 2004 were used to estimate the risk of weekly uptake. To estimate the weekly intake of heavy metals into the body of consumers, the obtained average was used for the accumulation of metals in the studied oyster. Calculation of weekly intake of metals: Maximum allowable weekly intake × Body weight (Kg)/Concentration of metals obtained in the present study using the equation of EWI = Cm*FIRw / BW, where EWI: Estimated weekly intake, FIRw: Maximum allowed weekly intake (daily intake multiplied by 7), Cm: The concentration obtained for the target metal in wet weight, and W: body weight (70 kg for adults and 16 kg for children) [14].
Health risk assessments have been evaluated to assess the chemical quality of living organisms and to assess the potential risks associated with their consumption. The potential non-cancerous and carcinogenic hazards of each heavy metal are expressed as non-carcinogenic risk factors (Target Hazard Quotients = THQ) and carcinogenic risk (CR), respectively. The THQ formula is THQ = (EF*ED*FIR*C)/RfD*BW*AT) * 10-3, where EF represents the number of cycles (365 days per year) and ED denotes the duration (70 years in adults and 6 years in children) of exposure (USEPA, 2000). FIR is the food intake rate, C is the concentration of heavy metals in seafood (mg/kg.ww), RfD is the oral reference dose of metals (0.001 for cadmium, 0.04 for copper, and 0.02 for nickel) (mg/kg/day), and BW is the average body weight (70 kg for adults and 16 kg for children) and AT is the mean exposure time (day/ year × ED = 365 × 70 = 25/550 days).
According to the Fisheries Organization of Iran (1400), per capita, aquatic consumption for adults is estimated at 36.4 grams per day. In the study conducted by Khoshnood et al. (2015), the consumption of aquatic organisms in adults to children is 39%. Therefore, the daily intake for children will be 14.19 grams per day. According to Wang et al. (2018), THQ is classified into five categories: No significant risk: THQ ≤1, Low risk: 1< THQ < 9.9, Moderate risk:10 < THQ <19.9, High risk: 20 < THQ < 99, and Serious risk: ≥ 100 [15]. Humans are often exposed to more than one heavy metal and suffer from combined or interactive effects. As a result, total THQ (TTHQ) was used to calculate the overall health risks, which is the sum of the THQ values of a single metal. If the TTHQ value is less than one, there is no significant risk. If the TTHQ is more than one, there is a possibility of side effects likely to increase with increasing value [15].
Statistical Analysis
Data analysis was performed using SPSS software version 22. First, the normality of the data was examined using the Kolmograph-Smirnov test. The correlation between the data was examined by Pearson test and linear regression and finally, the graphs were drawn using Excel 2015. The results of Kolmogorov-Smirnov test showed that all data were normal in both autumn and spring. Therefore, to investigate the statistically significant differences between the data between the two seasons, the studied stations used a T-test, and to investigate the accumulation of heavy metals between sampling stations in two separate seasons, one-way analysis of variance (ANOVA) and Tukey post-test were used.
Results
According to the results, the crust length, and the total weight of S. cuculata samples (Table 1), the maximum and minimum lengths were recorded at Beris station in autumn and Tis station in autumn, respectively. The highest and lowest weights were found in specimens of the Gowatr station in autumn and Tis station in autumn, respectively. The length of samples between the two seasons showed there is not significant difference (p>0.05) and their weight showed a significant difference (p<0.05).
|
Spring |
Autumn |
|
||
|
Weight(g) |
length(cm) |
Weight(g) |
length(cm) |
|
|
39.61±16.9 b |
5.52±1.3 a |
77.9±22.2 a |
7.16±0.2 a |
Gowatr |
|
56.9±8.7 b |
5.84±0.4 a |
64.1±5.2 a |
7.4±1.3 a |
Bris |
|
32.8±7.7 b |
5.66±0.7 a |
46.9±17.3 a |
4.9±1.02 a |
Ramin |
|
30.3±6.5 b |
5.34±0.6 a |
24.5±8.13 a |
4.7±0.9 a |
Tis |
* Statistical comparison between length in two seasons and weight in two seasons. Matched letters indicate insignificance and heterogeneous letters indicate significance
Table 1: Biometric Factor of S. Cucullata of Sampling Areas in Autumn and Spring
Accumulation of Heavy Metals in Two Seasons
The results showed that the concentrations of Cd and Cd were higher in autumn (Table 2). The results showed that Cd in Gowatr station had no significant difference between the two seasons (p>0.05). But the Cu and Ni had a significant difference between the two seasons at this station (p<0.05).
At Beris station, the accumulation of Cu and Ni metals except Cd was higher in autumn than in spring (Table 2). The results of t-test showed no significance of metals between the two seasons in the Beris station (p> 0.05).
At Ramin station, the accumulation of Cd and Ni metals except Cu was higher in autumn than in spring (Table 2). T-test showed that nickel metal in the Ramin station had no statistically significant difference between the two seasons (P>0.05). However, Cd and Cu metals had a statistically significant difference between the two stations (P <0.05).
Concentrations of Ni and Cd except Tis station Cu were higher in autumn than in spring (Table 2). The results of the t-test showed Cd metal in Tis station between the two seasons had a statistically significant difference (P<0.05), but the two metals Cu and Ni contents did not have a statistically significant difference between the two seasons at this station (P> 0.05).
|
|
Cu |
Ni |
Cd |
|
||
|
148.8±47.7b |
17.71±6.2b |
0.04±0.006a |
Spring |
|
||
|
61.4a |
7.02±2.3a |
0.09±0.02a |
Autumn |
Gowatr |
||
|
|
265.5± |
|
|
|
||
|
60.43±6.2 a |
6.51±3.52 a |
0.04±0.005 a |
Spring |
|
||
|
64.1±28.8 a |
7.53±3.2 a |
0.03±0.007 a |
Bris |
|||
|
Autumn |
||||||
|
229.8±49.2 b |
5.08±3.5 b |
0.02±0.003a |
Spring |
|
||
|
216.6±72.8 a |
9.06±0.75 a |
0.12±0.02 a |
Ramin |
|||
|
Autumn |
||||||
|
89.4±31.9 a |
3.8±2.5 a |
0.09±0.02 b |
Spring |
|
||
|
84.3±12.51 a |
7.08±1.4 a |
0.18±0.09 a |
Tis |
|||
|
Autumn |
|
|||||
* Identical letters indicate insignificance and dissimilar letters indicate significance.
Table 2: Accumulation of Heavy Metals in S.Cuculata Soft Tissue (μg/g.ww) between Two Seasons
Accumulation in Different Stations
In autumn, the accumulation of Cd metal had the highest at Tis station and the lowest at Beris station (Figure2). The results of the ANOVA test (one-way analysis of variance) showed a significant difference in terms of Cd metal content between the studied stations, which was the difference between the Beris station and the Tis and the Ramin station (P <0.05).
Ni accumulation was highest in the Ramin station in autumn and lowest in Gowatr station (Figure. 2). The results showed no significant difference in terms of Ni content between the studied stations (P> 0.05).
The accumulation of Cu metal was highest in Gowatr station in autumn and lowest in the Beris station (Figure. 3). The results of ANOVA test (one-way analysis of variance) showed the significant difference in copper metal content between the studied stations, which was observed between Beris and Tis station and Gowatr and Ramin stations (P <0.05).
In spring, the highest accumulation of cadmium metal was at Tis station and the lowest at Ramin station (Figure. 2). ANOVA test showed that cadmium metal had a statistically significant difference between stations (P <0.05). Tukey’s test showed that Tis, Gowatr, Beris and Ramin stations had no statistically significant differences from each other (P> 0.05).
The mean of nickel metal concentration in spring was the highest in Gowatr station and the lowest in the Tis station (3.8-2.5) (Figure 3). ANOVA test showed a statistically significant difference between stations for nickel metal (P<0.05). Tukey’s post-test showed that three stations of the Beris, Tis and Ramin had no statistically significant difference (P> 0.05).
The highest and lowest accumulation of copper metal in spring was in Ramin station and Beris station, respectively (Figure 2). ANOVA test showed that copper metal had a statistically significant difference between the studied stations (P <0.05). Tukey’s post-test of copper showed that all stations had a statistically significant difference from each other (P <0.05).
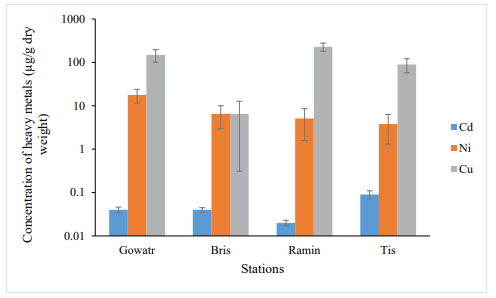
Figure 2: Concentration of Heavy Metals of Soft Tissue of S.Cucullata in the Studied Stations in Spring
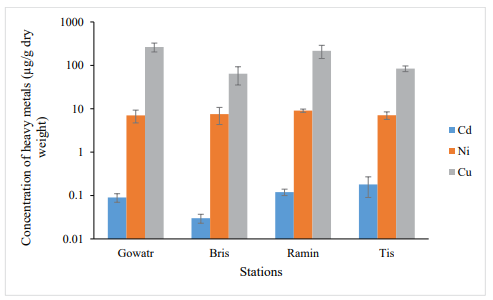
Figure 3: Concentration of Heavy Metals of Soft Tissue of S.Cucullata in the Studied Stations in Autumn
Examination of the Daily and Weekly Oyster Consumption
Table 3 shows the daily and weekly uptake of metals and the maximum consumption of the studied metals for adults and children in the present study. According to the results, in both daily and weekly absorption in adults and children, the copper metal rate was the highest and the lowest uptake was recorded for cadmium metal.
|
EWI children |
EWI adults |
EDI children |
EDI adults |
RFD (µg/kg/day) |
|
|
0.20 |
0.047 |
0.029 |
0.0067 |
1 |
Cd |
|
20.70 |
4.73 |
2.95 |
0.67 |
20 |
Ni |
|
393.3 |
89.9 |
56.19 |
12.84 |
40 |
Cu |
Table 3: EDI and EWI Heavy Metals for Adults and Children in S. Cucullata
THQ Assessment
Consumption risk for Cd, Ni and Cu estimated through the use of S. cuculata has been shown in Table 4. According to USEPA, the acceptable value for THQ is 1. In the present study, THQ wascalculated to be less than one for both adults and children (Figure Consumption risk for Cd, Ni and Cu estimated through the use of S. cuculata has been shown in Table 4. According to USEPA, the acceptable value for THQ is 1. In the present study, THQ was 4). Therefore, eating S. cucullata in the study area does not pose a cancerous health risk to adults and children.
|
THQ (children) |
THQ (adults) |
|
|
0.0011 |
0.0067 |
Cd |
|
0.057 |
0.033 |
Ni |
|
0.54 |
0.32 |
Cu |
|
0.60 |
0.036 |
TTHQ |
Table 4: Daily and Weekly Uptake of Heavy Metals in S. Cucullata
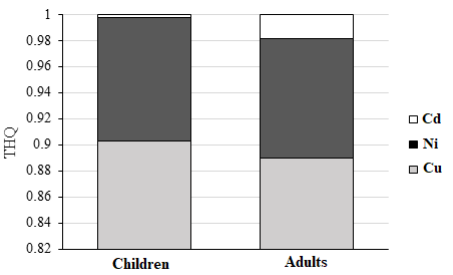
Figure 4: THQ Changes of Heavy Metals
Discussion
A notable feature of S. cucullata on the shores of the Oman Sea is its ability to accumulate and disperse widely. Given its importance as an important oyster in human nutrition, it is one of the suitable species to study the concentration of heavy metals in the environment. Feeding oysters with a high intake capacity of metals is a concern. Edible oysters such as S. cucullata have high nutritional and economic value, so the analysis of heavy metal accumulation in them in terms of biomarkers of aquatic environments in close periods is very important to maintain the health of oyster’s consumers and study areas exposed to pollutants. Heavy metals in these oysters accumulate and biomagnate through the food chain and are consumed by humans and will have many health risks over time [16]. Anbuselvan and Sridharan stated that changes in science and industry, increasing world population, especially in coastal areas, developing industrial activities along the coast and discharging their wastewater into the aquatic environment has led to the spread of persistent pollutants in this environment [17]. Although pollution has existed since the beginning of human existence, it has not accumulated to the extent that it causes harm to human health. Over time, due to the increase and development of societies and human powers, there has been a significant increase in pollutants, especially stable and non-degradable pollutants that are occurring at a worrying level of non-stop [17].
As shown in the results, the concentrations of heavy metals fluctuated in all stations and in both hot and cold seasons. According to researchers, different levels of heavy metals in different bivalves may be due to differences in the accumulation properties of heavy metals [18]. Also, it has been stated that the concentration of heavy metals in the edible tissue of bivalves is mainly influenced by environmental factors [19].
Various studies have shown that organisms need small contents of essential metals for their biological and biochemical functions, Cu and Ni in the present study. In contrast non-essential metals (including Cd in this study) have no biological function in living organisms. Therefore, the concentration of Cu metal in the present study was higher. Cu is an important component of several enzymes that play a key role in the metabolism of organisms [20]. The deficiency of copper in organisms can lead to immune disorders, loss of appetite and stunted growth. However, studies have shown that excessive consumption can lead to poor health problems and even disease [19,21]. Cadmium binds to low molecular weight proteins called metallothionein, which reduces its toxicity. Therefore, using the detoxification mechanism, the soft tissue of the oyster can collect more contents of this element. The researchers stated that mollusca use copper metal to make homocyanins for the respiratory process, which is the main reason for the high concentration of copper [8]. High levels of copper in soft tissue may be due to the tendency of oysters to accumulate this element essential for cell growth and metabolism. Therefore, the high concentrations of copper compared to other metals in the present study indicate the need for oyster’s bodies. Because of this, high content is absorbed from the environment. In addition, Liang et al., stated that oysters use copper as an oxygen delivery system in the blood [22]. The highest source of copper metal in coastal waters is the anti-sediment paints used in most ships. This metal enters the aquatic environment through industrial effluents containing CuSO4 used in metal plating operations and fishing equipment [23]. Also, high levels of copper accumulate in aquatic animals and environments due to increased transportation activity, industrial waste, untreated wastewater and leached sediments [24]. One of the factors of nickel metal origin in the Oman Sea is due to the presence of Ophiolites in the seabed, which has a large amount of nickel sulfide [25]. Also, study results by Talebimatin et al., showed that S. cuculata has a high ability to absorb nickel and cadmium metals. For this reason, S. cuculata is the most suitable species for measuring the pollution of the environment by nickel and cadmium metals [26].
In recent years, many factors such as increasing ship traffic, industrialization and increasing population of Chabahar Bay and the Sea of Oman have polluted this important commercial area. In Chabahar Bay, which does not have much advanced industry, most pollutants have entered the aquatic ecosystem due to increased traffic of ships, boats and fishing boats, ship repairs in coastal areas, ship water balance discharge, dredging of municipal sewage (Keshavarz et al., 2013) [27]. Continuous monitoring of Chabahar Bay is necessary due to its valuable marine resources, including commercial fish and molluscs, and due to the increase in pollution, so as not to endanger the health of the natives and important resources of this region. One of the sources of nickel in the Oman Sea is the presence of Ophiolites in the seabed, which has a large amount of nickel sulfide. Therefore, the origin of nickel metal in the sediments of the Chabahar coasts is due mainly to the presence of these rocks [25].
Qin et al. investigated the concentrations of heavy metals (copper, zinc, arsenic, cadmium, lead and mercury) in the five marine bivalves (Ruditapes philippinarum, Paphia undulata, Meretrix meretrix, Sinonovacula constricta and Meretrix lyrata) in the three major coastal cities of Guangxi in China [19]. A comparison of this study with the present study showed that the concentration of copper metal was much lower and cadmium was approximately consistent with the present study. The research of Jana et al., on the accumulation of heavy metals in the rocky oyster (S. cucullata) showed that the concentration of copper metal was much higher than the present study [28]. Copper metal concentrations in the study of Rodrigues et al., of S. cucullata in Zuari estuary (India) were the same as in the present study [29]. Comparison of metal concentrations obtained by Jahromi et al., in five bivalve species of Hormozgan with the present study showed that the copper metal concentration is much higher and nickel and cadmium metals are approximately consistent with the present study [30]. Liu et al. investigated the bioaccumulation of heavy metals in three bivalve species of Mactra veneriformis, Ruditapes philippinarum and Scapharca subcrenata of Laizhou waters of China and comparing their study with the present study showed that cadmium and copper were higher than the present study [31]. Shakouri and Gheytasi investigated the accumulation of heavy metals of S. cucullata in Chabahar Bay [32]. Comparison of the results showed that the concentrations of all three metals nickel, copper and cadmium are much higher than the present study. Shaari et al. have examined the bioaccumulation of heavy metals in Crassostrea sp. oyster in Setiu Wetland (Malaysia) [33]. The results, showed that the concentration of copper metal was much lower and cadmium metal was approximately consistent with the present study. The difference in heavy metal concentrations of the present study with other studies is usually different environmental and geographical conditions, different ways of municipal and industrial wastewater disposal, different species, differences in feeding habits, gender, age, size, and different conditions in measurement and laboratory activities [34].
It is well accepted that aquatic organisms accumulate different levels of heavy metals depending on several factors including species differences, physiological mechanisms in the body, metabolic activity, diet, growth rate, habitat and lifestyle, nutritional levels, longevity as well as chemical characteristics and heavy metal surfaces [21,30,35]. According to Huanxin et al., the accumulation of heavy metals in oyster tissue is affected by their metabolism and the availability of various metals in the environment [36].
Table 5 compares the concentrations of the studied metals in different mollusks of other places with the present study. Numerous studies have presented the differences in the accumulation of metals to various factors such as geographical differences in latitude and longitude, different species, and different effects of human activities (Yilmaz et al., 2018). Cobbett and Goldsbrough (2002) stated that the neutralization of some metals potentially occurs when they are bound to proteins and may be removed from living organisms by various metabolic methods (Sandrin and Maier 2002, Flora et al. 2012) [37]. A comparison of the concentrations of heavy metals in Table 2 showed the safety of Cu and Cd concentrations in the soft texture of S. cucullata. But the concentration of Ni metal was higher than WHO, FAO and FDA standards.
According to the results obtained for daily and weekly intake in adults and children showed that the highest uptake rate is associated with copper and the lowest uptake rate is related to cadmium. According to USEPA, the acceptable value for THQ is less than 1 [13]. In the present study, less than one THQ was obtained for both adults and children. Therefore, feeding on S. cucullata in the study area does not pose a non-cancerous health risk to adults and children.
Potential health risks from eating contaminated seafood can be carcinogenic and non-carcinogenic. Non-cancerous risk is based on the risk factor (THQ) derived from the estimated daily intake (EDI) [38]. The risk assessment of seafood consumption is based on the fact that the rate of ingestion is equal to the rate of absorption and cooking does not affect on the total content of metals. Seafood is consumed as the cheapest source of protein available. Consumption risk (THQ) of the three metals Cu, Ni and Cd obtained by Yap et al. in S. cucullata of the present study was consistent and less than one was calculated [39]. The study by Jahromi et al., as in the present study, showed that THQ and TTHQ for all heavy metals studied in binocular samples were below the safe level (THQ=1) for children and adults, indicating no potential non-cancerous risk of consuming six bivalve species (Saccostrea cucullata, Circenita callipyga, Barbatia helblingii, Solen brevis, Amiantis umbonella, Telescopium telescopium and Saccostrea cucullate) on the coast of Hormozgan [30]. Consumption risk in the study of Wang et al. on oysters (Crassostrea angulate and Crassostrea hongkongensis) of the Jilong estuary in China was very high [15]. Therefore, they had great concerns for human consumption and were not consistent with the present study.
TTHQ <1 indicates that the exposure level is less than RfD , indicating that daily exposure at this level is unlikely to have any negative impact on the lifespan of a human population (Bogdanovic et al., 2014). However, in most studies, TTHQ values vary across all bivalves.
The health risk assessment showed that although there is no non-cancerous health risk to oyster users in the study area, continuous and lifelong use for more than 70 years may increase cancer risk. Therefore, in the long run, monitoring and controlling bivalve consumption will be necessary.
The results obtained in both daily and weekly intake in adults and children showed that the highest uptake rate was related to copper metal and the lowest was recorded for Cd.
EDI is an estimate for the safe range of daily food intake without adverse health effects. Estimated daily intake (EDI) can be a guide for daily food consumption for humans (Puspitasari and Suratno, 2020). Estimation of daily intake (EDI) in the study of Zhao et al., on 17 bivalve species was less than the RfD guidelines for all metals studied in their study, which did not match the results of daily intake of copper metal in the present study [21,40]. Therefore, they showed that consumers of the studied bivalves would not experience significant health risks. Zhao et al., also stated that people are much more exposed to seafood contaminants than people who live in remote or landless urban areas [21].
|
Area |
species |
Cd |
Ni |
Cu |
Source |
|
Tropical Island of Thailand |
Strombus canarium |
--- |
0.46 |
8.4 |
Kobkeatthawin et al., 202142 |
|
Malaysian peninsular beaches |
Saccostrea cucullata |
0.9 |
0.4 |
11.5 |
Yap et al., 202140 |
|
Qeshm Island |
Saccostrea cucullata |
51.09 |
12.94 |
206.03 |
Nourozifard et al., 202010 |
|
Estuary potengi (Brazil) |
Crassostrea rhizophorae |
0.82 |
0.93 |
4.02 |
Senez-Mello et al., 202043 |
|
El-Mex Bay, Egypt (summer) |
Patella caerulea |
21.75 |
21.93 |
39.77 |
Abdel-Halim, 201944 |
|
Gulf of California |
Atrina Maura |
26.33 |
9.86 |
11.96 |
Góngora-Gómez et al., 201845 |
|
Marudu Bay (Malaysia) |
Crassosstrea gigas |
1.38 |
1.4 |
24.8 |
Denil et al., 201746 |
|
Assaluyeh beach |
Saccostrea cucullata |
1.54 |
0.26 |
283.78 |
Delshab et al., 201725 |
|
North shores of the Oman Sea |
Saccostrea cucullata |
0.1 |
7.67 |
158.37 |
The present study of autumn |
|
North shores of the Oman Sea |
Saccostrea cucullata |
0.051 |
7.52 |
132.12 |
The present study of spring |
Table 5: Comparison of Metal Concentrations of the Present Study with Other Studies in Domestic and Foreign Waters in the Soft Tissue of Bivalve Molluscs
Conclusion
The heavy metal accumulation sequence of the present study in both seasons and all stations was obtained as Cu> Ni> Cd. A comparison of heavy metal concentrations with world standards showed that Ni was higher than some standards. The results for daily and weekly intake in adults and children showed that the highest rate was associated with copper metal and the lowest value was related to Cd. In the present study, THQ was calculated to be less than one for both adults and children. Therefore, eating S. cucullata in the study area does not pose a non-cancerous health risk to adults and children.
Acknowledgements
The authors are grateful to the biology laboratory experts of the Chabahar Maritime University.
Conflict of Interest
Authors have been declared that there is no conflict of interest.
Author Contributions
YF: Data collection, analysis; ML: analysis and manuscript writing.
References
- Sobihah, N. N., Zaharin, A. A., Nizam, M. K., Juen, L. L., & Kyoung-Woong, K. (2018). Bioaccumulation of heavy metals in maricultured fish, Lates calcarifer (Barramudi), Lutjanus campechanus (red snapper) and Lutjanus griseus (grey snapper). Chemosphere, 197, 318-324.
- Rajeshkumar, S., & Li, X. (2018). Bioaccumulation of heavy metals in fish species from the Meiliang Bay, Taihu Lake, China. Toxicology reports, 5, 288-295.
- Khodami, S., Surif, M., WO, W. M., & Daryanabard, R. (2017). Assessment of heavy metal pollution in surface sediments of the Bayan Lepas area, Penang, Malaysia. Marine Pollution Bulletin, 114(1), 615-622.
- Hellberg, R. S., DeWitt, C. A. M., & Morrissey, M. T. (2012). Riskâ?benefit analysis of seafood consumption: A review. Comprehensive reviews in food science and food safety, 11(5), 490-517.
- Barchiesi, F., Branciari, R., Latini, M., Roila, R., Lediani, G., Filippini, G., ... & Ranucci, D. (2020). Heavy metals contamination in shellfish: benefit-risk evaluation in Central Italy. Foods, 9(11), 1720.
- Lehel, J., Bartha, A., Dankó, D., Lányi, K., & Laczay, P. (2018). Heavy metals in seafood purchased from a fishery market in Hungary. Food Additives & Contaminants: Part B, 11(4), 302-308.
- Han, B. C., Jeng, W. L., Hung, T. C., Ling, Y. C., Shieh, M. J.,& Chien, L. C. (2000). Estimation of metal and organochlorine pesticide exposures and potential health threat by consumption of oysters in Taiwan. Environmental Pollution, 109(1), 147-156.
- Nourozifard, P., Mortazavi, S., Asad, S., & Hassanzadeh, N. (2020). Using Saccostrea cucullata as a biomonitor of heavy metals (Cu, Pb, Zn, Cd, Ni, and Cr) in water and sediments of Qeshm Island, Persian Gulf. Ecopersia, 8(3), 181-190.
- Agah, H., Bastami, K. D., & Fumani, N. S. (2016). Ecological risk, source and preliminary assessment of metals in the surface sediments of Chabahar Bay, Oman Sea. Marine pollution bulletin, 107(1), 383-388.
- Hosseini, R., Sayadi, M. H., Aazami, J., & Savabieasfehani,M. (2020). Accumulation and distribution of microplastics in the sediment and coastal water samples of Chabahar Bay in the Oman Sea, Iran. Marine pollution bulletin, 160, 111682.
- Staniskiene, B., Matusevicius, P., Budreckiene, R., & Skibniewska, K. A. (2006). Distribution of heavy metals in tissues of freshwater fish in Lithuania. Polish Journal of Environmental Studies, 15(4).
- Ruilian, Y. U., Xing, Y., Yuanhui, Z. H. A. O., Gongren,H. U., & Xianglin, T. U. (2008). Heavy metal pollution in intertidal sediments from Quanzhou Bay, China. Journal of Environmental Sciences, 20(6), 664-669.
- EPA, U. (2000). Estuarine and coastal marine waters:Bioassessment and biocriteria technical guidance. Technology.
- Chien, L. C., Hung, T. C., Choang, K. Y., Yeh, C. Y., Meng, P. J., Shieh, M. J., & Han, B. C. (2002). Daily intake of TBT, Cu, Zn, Cd and As for fishermen in Taiwan. Science of the total environment, 285(1-3), 177-185.
- Wang, X. N., Gu, Y. G., Wang, Z. H., Ke, C. L., & Mo, M.S. (2018). Biological risk assessment of heavy metals in sediments and health risk assessment in bivalve mollusks from Kaozhouyang Bay, South China. Marine pollution bulletin, 133, 312-319.
- Agah, H., Leermakers, M., Elskens, M., Fatemi, S. M. R., & Baeyens, W. (2009). Accumulation of trace metals in the muscle and liver tissues of five fish species from the Persian Gulf. Environmental monitoring and assessment, 157, 499-514.
- Anbuselvan, N. D. S. N., & Sridharan, M. (2018). Heavy metal assessment in surface sediments off Coromandel Coast of India: Implication on marine pollution. Marine pollution bulletin, 131, 712-726.
- Rajaram, R., Ganeshkumar, A., & Vinothkannan, A. (2020). Health risk assessment and bioaccumulation of toxic metals in commercially important finfish and shellfish resources collected from Tuticorin coast of Gulf of Mannar, Southeastern India. Marine Pollution Bulletin, 159, 111469.
- Qin, L. Y., Zhang, R. C., Liang, Y. D., Wu, L. C., Zhang, Y. J., Mu, Z. L., ... & Yu, Z. P. (2021). Concentrations and health risks of heavy metals in five major marketed marine bivalves from three coastal cities in Guangxi, China. Ecotoxicology and Environmental Safety, 223, 112562.
- Mendil, D., Demirci, Z., Tuzen, M., & Soylak, M. (2010). Seasonal investigation of trace element contents in commercially valuable fish species from the Black sea, Turkey. Food and Chemical Toxicology, 48(3), 865-870.
- Zhao, L., Yang, F., & Yan, X. (2013). Concentrations of selected trace elements in marine bivalves and the dietary risk to residents of Dalian City, Northern China. Human and Ecological Risk Assessment: An International Journal, 19(1), 145-150.
- Junrong, L., Jun, W., Yongquan, S., Dexiang, W., & Jiugang,Y. (2001). Effects of metals on embryodevelopment of Tachypleus tridentatus. Acta Ecologica Sinica, 21(6), 1009-1012.
- Mitra, A., Barua, P., Zaman, S., & Banerjee, K. (2012). Analysis of trace metals in commercially important crustaceans collected from UNESCO protected world heritage site of Indian Sundarbans. Turkish journal of fisheries and aquatic sciences, 12(1), 53-66.
- Delshab, H., Farshchi, P., Mohammadi, M., & Moattar, F. (2017). Assessment of heavy metals contamination and its effects on oyster (Saccostrea cucullata) biometry parameters in the Asaluyeh port coasts, Persian Gulf, Iran. International Journal of Environmental Studies, 74(6), 1031-1043.
- De Mora, S., Fowler, S. W., Wyse, E., & Azemard, S. (2004). Distribution of heavy metals in marine bivalves, fish and coastal sediments in the Gulf and Gulf of Oman. Marine pollution bulletin, 49(5-6), 410-424.
- Talebimatin, M., Shamsaei-Mahdajan, M., Haqbayan, S. (2013). A comparative study of the concentration of heavy metals nickel and cadmium in the oyster (Saccostrea cucullata) and bed sediments in a part of the beach of Bandar Dayer, Bushehr province, J Mar Sci, 5(1), 25-34.
- Sadeghi, P., & Darbazi, M. (2016). Assessment of heavy metals concentration Cu, Ni and Zn in Acanthopleura vaillantii in the coast of Chabahar Bay. Journal of Aquatic Ecology, 6(3), 115-123.
- Jana, H., Malik, B. K., Shit, P., Chakraborty, P. P., & Mondal,K. C. (2021). Effect of some heavy metal (zn, cu, pb) pollutants and microbial load on the edible oyster (saccostrea cucullata) in sundarban, West Bengal.
- Rodrigues, R. J., Nasnodkar, M. R., Nayak, G. N., & Tiwari,A. K. (2021). Bioaccumulation of metals by edible bivalve Saccostrea cucullata and its application as a bioindicator of metal pollution, tropical (Zuari) estuary, Goa, India. Arabian Journal of Geosciences, 14(12), 1192.
- Jahromi, F. A., Keshavarzi, B., Moore, F., Abbasi, S., Busquets, R., Hooda, P. S., & Jaafarzadeh, N. (2021). Source and risk assessment of heavy metals and microplastics in bivalves and coastal sediments of the Northern Persian Gulf, Hormogzan Province. Environmental Research, 196, 110963.
- Liu, J., Cao, L., & Dou, S. (2017). Bioaccumulation of heavy metals and health risk assessment in three benthic bivalvesalong the coast of Laizhou Bay, China. Marine pollutionbulletin, 117(1-2), 98-110.
- Shakouri, A., & Gheytasi, H. (2018). Bioaccumulation of heavy metals in oyster (Saccostrea cucullata) from Chabahar bay coast in Oman Sea: Regional, seasonal and size-dependent variations. Marine pollution bulletin, 126, 323-329.
- Shaari, H. B., Raven, B., Sultan, K., Mohammad, Y., & Yunus,K. (2016). Status of heavy metals concentrations in oysters (Crassostrea sp.) from Setiu Wetlands, Terengganu, Malaysia.
- Rainbow, P. S. (2002). Trace metal concentrations in aquatic invertebrates: why and so what?. Environmental pollution, 120(3), 497-507.
- Soltani, N., Moore, F., Keshavarzi, B., Sorooshian, A., & Javid, R. (2019). Potentially toxic elements (PTEs) and polycyclic aromatic hydrocarbons (PAHs) in fish and prawn in the Persian Gulf, Iran. Ecotoxicology and environmental safety, 173, 251-265.
- Huanxin, W., Lejun, Z., & Presley, B. J. (2000). Bioaccumulation of heavy metals in oyster (Crassostrea virginica) tissue and shell. Environmental Geology, 39, 1216-1226.
- Cobbett, C., & Goldsbrough, P. (2002). Phytochelatins and metallothioneins: roles in heavy metal detoxification and homeostasis. (1), 159-182.
- Satapathy, S., Panda, C. R., & Jena, B. S. (2019). Risk-based prediction of metal toxicity in sediment and impact on human health due to consumption of seafood (Saccostrea cucullata) found in two highly industrialised coastal estuarine regions of Eastern India: a food safety issue. Environmental geochemistry and health, 41, 1967-1985.
- Yap, C. K., Azmizan, A. R., & Hanif, M. S. (2011). Biomonitoring of trace metals (Fe, Cu, and Ni) in the mangrove area of Peninsular Malaysia using different soft tissues of flat tree oyster Isognomon alatus. Water, Air, & Soil Pollution, 218, 19-36.
- Kobkeatthawin, T., Sirivithayapakorn, S., Nitiratsuwan, T., Muenhor, D., Loh, P. S., & Pradit, S. (2021). Accumulation of trace metal in sediment and soft tissue of Strombus canarium in a tropical remote island of Thailand. Journal of Marine Science and Engineering, 9(9), 991.
- Gautam, P. K., Gautam, R. K., Banerjee, S., Chattopadhyaya,M. C., & Pandey, J. D. (2016). Heavy metals in the environment: fate, transport, toxicity and remediation technologies. Nova Sci Publishers, 60, 101-130.
- Ji, Y., Wu, J., Wang, Y., Elumalai, V., & Subramani, T. (2020). Seasonal variation of drinking water quality and human health risk assessment in Hancheng City of Guanzhong Plain, China. Exposure and health, 12, 469-485.



