Research Article - (2026) Volume 5, Issue 1
Green Fabrication of Silver Nanoparticles Using Symphyotrichum Subulatum Stem Extract, their Characterization and Biological Properties
2Lecturer of Botany, Govt Degree College, Gulabad, Kazakhstan
3Department of Chemistry, Nazarbayev University, Astana, Kazakhstan
4Department of Chemistry, University of Malakand, Kpk, Pakistan
5Department of Chemistry, Govt Degree College Gulabad, Kpk, Pakistan
Received Date: Dec 16, 2025 / Accepted Date: Jan 09, 2026 / Published Date: Jan 22, 2026
Copyright: ©2026 The authors. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Ali, W., Alam, A., Ahmad, W., Wahab, A., Khan, A., et al. (2026). Proximate Composition of Symphyotrichum subulatum Stem: A Potential Source of Nutritional Value. J Traditional Medicine & Applications, 5(1), 01-06.
Abstract
Silver nanoparticles (AgNPs) were synthesized via a green method using the aqueous stem extract of Symphyotrichum subulatum Michx. The extract effectively reduced Ag+ ions from AgNO3 without additional chemicals, confirmed by a visible color change. Characterization through XRD, FTIR, and SEM revealed crystalline FCC-structured, spherical AgNPs (~50 nm) stabilized by plant phytochemicals. The AgNPs exhibited significant antibacterial activity against Staphylococcus aureus (93%) and E. coli (80%), along with broad antifungal activity, notably against Trichophyton rubrum (87%). This study highlights a simple, eco- friendly synthesis route and the biomedical potential of plant-mediated AgNPs.
Keywords
Green Synthesis, Characterization, AgNPs, Antimicrobial Activity, Antifungal Activity
Introduction
Nanotechnology is a developing field that deals with materials at the 1 to 100 nm scale with different structures and scales. through silver ion release, Silver nanoparticles (AgNPs) show strong antibacterial activity and are widely used in agriculture, optics, pharmaceutics, catalysis, cosmetics, sensors, health, food, and textiles green synthesis using plant extracts is cost- effective, eco-friendly, and stable, fabricating nanoparticles with strong antimicrobial potential [1-8]. Symphyotrichum subulatum Michx., a medicinal plant rich in flavonoids and alkaloids, is used for asthma, cold and cough, and its extracts minimize toxicity in nanoparticle synthesis [9]. Nanotechnology was first hypothesized by Feynman (1959), is now an important 21st-century expertise [10,11]. AgNPs show high biomedical, catalytic stability and conductivity applications, while S. subulatum is locally used in Asia for various disorders [12,13]. AgNPs cause ROS-mediated DNA and protein destruction, and green synthesis improves antimicrobial effectiveness against MDR pathogens [13,14].
This plant, known as annual saltmarsh aster, is applied for piles, bleeding, and respiratory complaints, with bioactive composites like vasicinone and vaccine [15,16]. Locally, it treats excessive menses, bone fractures, jaundice, diabetes, and fever [17-19]. The present study focuses on AgNPs synthesis from S. subulatum stem extract, their characterization (UV–Vis, SEM), and antimicrobial evaluation [18,19].
Materials and Methods
Preparation of the Plant Extracts
The plant symphyotrichum subulatum Michx. Was collected from Galabad, Tehsil Adenzai, District Dir Lower, Khyber Pakhtunkhwa Pakistan Wajid Ali and was identified and authenticated from the Flora of Pakistan. The plant stems were cleaned with distilled water, dried in a shaded area and then grinded to powder form using a mortar and pistil. About 10g plant materials were weighed and mixed with 200mL of distilled water, heated for 30 min while being constantly stirred in a clean, sterilized conical flask. The mixture solution was filtered using Whatman filter paper No. 1 to get the plant extract. The filtered extract obtained was refrigerated in closed vessel till further use.
Chemicals and Other Materials
In the present study chemicals used were silver nitrate and distilled water. These chemicals were of analytical grade. These chemicals and reagents were of analytical grade and of high purity.
Biosynthesis of Agnps Mediated by Symphyotrichum Subulatum Michx. Stem Extract
Ag-NPs were prepared via a standard method Symphyotrichum subulatum Michx [20]. extract was used as bio reductant for the synthesis of silver nanoparticles. 0.95g of silver nitrate was dissolved in 200ml of distilled water to get AgNO3 solution (2mM solution of silver nitrate) Silver nitrate solution was first stirred for one minute at room temperature and then Symphyotrichum subulatum Michx.stem extract was added. Reaction mixture was kept on shaker for one hour. After the addition of stem extract, the color of the solution immediately changed from colorless to brownish black color. This change in color indicated the formation of AgNPs.
Characterization of the Synthesized AgNPs
SEM Analysis
The morphological characterization of the samples was done using JEOL Jsm- 6480 LV for SEM analysis. The samples were dispersed on a slide and then coated with platinum in an auto fine coater. After that the material was subjected to analysis.
XRD Analysis
The sample was ground into a fine powder and placed in the XRD machine. The machine was set up with specific settings and the sample was scanned. The resulting data showed the sample’s crystalline structure. The scan settings were adjusted for high- quality results. The data was compared to known patterns to identify the sample’s phases. Calculations were done to determine the crystallite size and lattice parameters. The test was done at room temperature. The results gave useful information about the sample’s properties.
FTIR Analysis
The characterization of functional groups on the surface of AgNPs by plant extracts were investigated by FTIR analysis (Shimadzu) and the spectra was scanned in the range of 4000–400 cm−1range at a resolution of 4 cm−1. The sample was prepared by dispersing the AgNPs uniformly in a matrix of dry KBr, compressed to form an almost transparent disc. KBr was used as a standard in analysis of the samples.
Antibacterial Activities of Extract and AgNPs
The zone of inhibition (ZI), minimum inhibitory concentration (MIC), and minimum bactericidal concentration (MBC) approaches were used to assess the antibacterial activities of the fruit extract and NPs against the selected bacterial strains. The strain of selected bacteria was inoculated onto sterilized agar culture dishes, and 2mm holes were made in the medium at a distance of 4cm from each other and from the reference standard antibiotic (amoxicillin). Using a micropipette, the solution of the extract and NPs was inserted into the holes. The bacterial culture plates were incubated at 37°C for 24h. Each inhibition around each hole was measured as a zone of inhibition. The entire process was completed in laminar flow to avoid any contamination during the experiment.
Antifungal Activities of Extract and AgNPs
The antifungal activity of the stem extract of Symphyotrichum subulatum Michx. And nanoparticles (NPs) were evaluated using the zone of inhibition (ZI) method. Fungal spores were inoculated onto sterilized agar plates, and 2mm wells were created in the medium. The extract and NP solutions were added to the wells, and the plates were incubated at 25°C for 48-72 hours. The zone of inhibition around each well was measured to assess the antifungal activity. The entire process was conducted under sterile conditions to prevent contamination. The results provided insight into the efficacy of the extract and NPs against fungal growth.
Results
Due to the use of expensive equipment and toxic components like solvents, reducing agents, and precursors, most physical, chemical, and hybrid techniques are generally expensive and dangerous in nature [21]. Using green ways to prepare NPs is more affordable, environ mentally friendly, and simple to implement. As a result, these methods are more widely used for the synthesis of metallic of NPs. A range of natural resources, including microbes, biomolecules, plant extracts, etc., have been used in the attempts at green synthesis of NPs so far [22].Plant extracts are preferred over other natural resources, despite the fact that a sizable number of research works have been published in the literature on the green synthesis of NPs employing biomolecules, microbes, and plant extracts, as plant extract-based methods are affordable, effective, and do not require a laborious procedure for isolating and keeping microbiological cultures [23]. The green synthesis of NPs is effectively carried out using extracts from several plant species, their organs, and isolated chemicals. In addition to being environmentally beneficial, the synthesis of nanomaterials may be made more sustainable by utilizing industrial and agricultural waste [24].
SEM Confirmation of AgNPs Synthesis
SEM analysis of AgNPs synthesized using S. subulatum stem extract revealed predominantly spherical nanoparticles with occasional irregularities and an average size of 50 nm. Some aggregation was observed, likely due to natural clustering, while phytochemicals in the extract acted as reducing and capping agents. These results confirm successful nanoparticle formation and provide key morphological insights for potential biological applications.
Figure 1: SEM Micrograph of AgNPs Synthesized using Symphyotrichum Subulatum Stem Extract
Structural Characterization of AgNPs by XRD
XRD analysis of AgNPs synthesized using S. subulatum stem extract showed sharp peaks at 2θ = 24.25°, 28.4°, 30.05°, 32.4°, 34.1°, 37.85°, 40.55°, and 50.2°, confirming a face-centered cubic (FCC) crystalline structure. The most intense peak at 30.05° indicates preferential crystallographic growth, while minor peaks suggest capping by plant biomolecules. The sharpness of the peaks confirms the nanoscale and polycrystalline nature of the AgNPs.
Figure 2: XRD Spectrum of AgNPs
FTIR Analysis
The FTIR spectrum of the synthesized AgNPs exhibited peaks corresponding to O–H, C–H, C≡C/C≡N, C=C/amide, C–H bending, and C–O stretching vibrations, along with Ag–O/Ag–N bands, confirming nanoparticle formation. Shifts and reduced intensities compared to the GT extract indicate that hydroxyl, carboxyl, carbonyl, and methyl groups actively participated in the reduction, nucleation, and stabilization of AgNPs.
Figure 3: FTIR Spectra of AgNPs
Antibacterial Activity of Synthesized AgNPs
The synthesized compound was evaluated against five bacterial strains using the Microplate Alamar Blue Assay (MABA) at 300 µg/µL. The compound showed strong activity against S. aureus (93%), E. coli (80%), and B. subtilis (60%), while no inhibition was observed for P. aeruginosa and S. typhi, indicating selective effectiveness toward Gram-positive bacteria.
|
Name of Bacteria |
% Inhibition of Compound |
% Inhibition of Drug |
|
Escherichia coli |
80 |
90 |
|
Bacillus subtilis |
60 |
80 |
|
Staphylococcus aureus |
93 |
95.5 |
|
Pseudomonas aeruginosa |
0 |
90 |
|
Salmonella typhi |
0 |
89.3 |
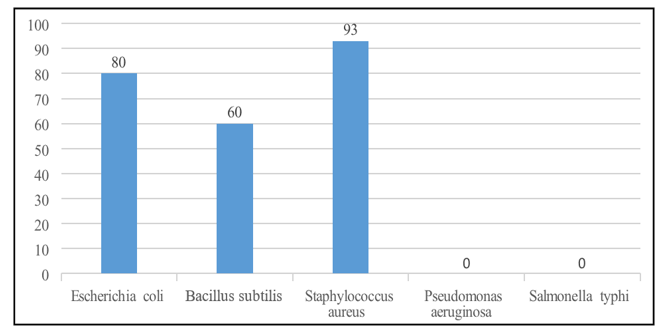
Figure 4: Antibacterial Activity of AgNPs
Antifungal Activity of Synthesized AgNPs
The synthesized compound was evaluated via the agar tube dilution method at 3000 µg/mL in DMSO. Tubes inoculated with fungal strains were incubated at 28 ± 1°C for 7–10 days. The compound showed broad-spectrum antifungal activity: Trichophyton rubrum (87%), Candida albicans (56%), Aspergillus niger (74%), Microsporum canis (82%), Fusarium lini (86%), Candida glabrata (77%), and Aspergillus flavus (84%) inhibition, indicating potential as a lead antifungal agent.
|
Name of Fungus |
Linear Growth (mm) |
% Inhibition |
Std. Drug |
MIC (µg/mL) |
|
Sample |
Control |
|||
|
Trichophyton rubrum |
13 |
100 |
87% |
Miconazole |
|
Candida albicans |
44 |
100 |
56% |
Miconazole |
|
Aspergillus niger |
26 |
100 |
74% |
Amphotericin B |
|
Microsporum canis |
18 |
100 |
82% |
Miconazole |
|
Fusarium lini |
14 |
100 |
86% |
Miconazole |
|
Candida glabrata |
23 |
100 |
77% |
Miconazole |
|
Aspergillus fumigatus |
16 |
100 |
84.50% |
Miconazole |
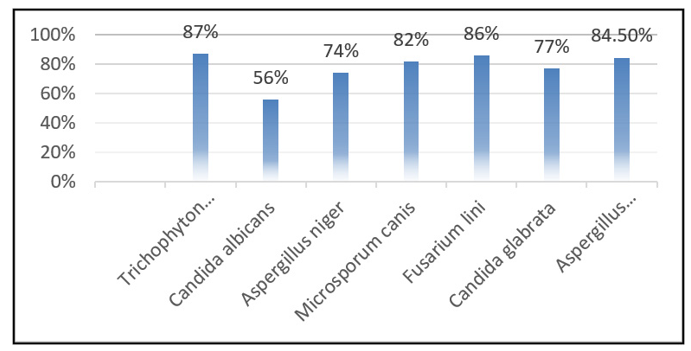
Figure 5: Antifungal Activity of AgNPs
Conclusion
This study demonstrates the green synthesis of silver nanoparticles (AgNPs) using Symphyotrichum subulatum Michx. stem extract as a natural reducing and stabilizing agent. XRD confirmed their crystalline face-centered cubic structure, FTIR indicated hydroxyl, carbonyl, and alkyl groups in reduction and capping, and SEM showed mostly spherical nanoparticles (~50 nm) with minor aggregation. Biologically, AgNPs exhibited strong antibacterial activity against S. aureus (93%), E. coli (80%), B. subtilis (60%) and antifungal activity against T. rubrum (87%), F. lini (86%), and A. flavus (84.5%). The method is simple, eco-friendly, and cost-effective, with potential applications in medicine, agriculture, and environmental science. Future work may explore scale-up, cytotoxicity, and stability [25].
References
- Greenwood, N. N., Earnshaw, A. (1997). Chemistry of the Elements 2nd Edition, Butterworth-Heinemann.
- Prasad, N., Swamy, V. (2015). Green synthesis of silver nanoparticles using Terminalia chebula extract and their antimicrobial properties. Int J Nanomedicine 10, 431-438.
- Roy, A., Bulut, O., Some, S., Mandal, A. K., & Yilmaz, M. D. (2019). Green synthesis of silver nanoparticles: biomolecule- nanoparticle organizations targeting antimicrobial activity.RSC advances, 9(5), 2673-2702.
- Iravani, S. (2011). Green synthesis of metal nanoparticles using plants. Green chemistry, 13(10), 2638-2650.
- Ahmed, S., & Ikram, S. (2016). Biosynthesis of gold nanoparticles: a green approach. Journal of Photochemistry and Photobiology B: Biology, 161, 141-153.
- Zambare, P., Survase, A., & Kanase, S. (2023). Green Synthesis of Copper Nanoparticles Using Leaf Extract of Ocimum sanctum and its Antimicrobial Activity. International Journal of Pharmaceutical Investigation, 13(1).
- Ahmad, S., Deen, R. A. (2020). Antibacterial activity of biogenic silver nanoparticles synthesized from Azadirachta indica leaf extract. Arab J Chem, 13(1), 824-834.
- Li, N., Zhao, W., Wang, J. (2016). Synthesis and characterization of silver nanoparticles using green methods, J Chem Pharm Res, 8(3), 151-158.
- Khalil, M. M., Ismail, E. H., El-Baghdady, K. Z., & Mohamed,D. (2014). Green synthesis of silver nanoparticles using olive leaf extract and its antibacterial activity. Arabian Journal of chemistry, 7(6), 1131-1139.
- Arya, V., Komal, R., Kaur, M., & Goyal, A. (2011). Silver nanoparticles as a potent antimicrobial agent: a review. Pharmacologyonline, 3, 118-124.
- Mulvaney, P. (1996). Surface plasmon spectroscopy of nanosized metal particles. Langmuir, 12(3), 788-800.
- Singh, A., Jain, D., Upadhyay, M. K., Khandelwal, N., & Verma, H. N. (2010). Green synthesis of silver nanoparticles using Argemone mexicana leaf extract and evaluation of their antimicrobial activities. Dig J Nanomater Bios, 5(2), 483-489.
- Pal, S., Tak, Y. K., & Song, J. M. (2007). Does the antibacterial activity of silver nanoparticles depend on the shape of the nanoparticle? A study of the gram-negative bacterium Escherichia coli. Applied and environmental microbiology, 73(6), 1712-1720.
- Shrivastava, J., Patel, R., Verma, A. (2010). Antimicrobial activity of Ocimum sanctum (Tulsi) extract. Int J Green Pharm, 4(4), 210-213.
- Shah, M. A., Khan, M. F. (2015). Green synthesis of silver nanoparticles using Ficus carica extract and their antibacterial activity. J Nanomater Mol Nanotechnol, 4(2), 1-6.
- Naqvi, H. S. A., Majeed, A. K. (2012). Microwave-assistedgreen synthesis of AgNPs using plant extract. Mater Sci EngC. 32(3), 940–946.
- Waghmare, R., Salunkhe, J. V., Pokharkar, S. K., Pujari, V.B. (2014). Green synthesis of silver nanoparticles using Ficus racemosa leaf extract and their antibacterial activity. Int JAdv Res, 2(6), 141-147.
- Huang, J., Li, Q., Sun, D., Lu, Y., Su, Y., Yang, X., ... & Chen,C. (2007). Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphoraleaf. Nanotechnology, 18(10), 105104.
- Mohanpuria, P., Rana, N. K., & Yadav, S. K. (2008).Biosynthesis of nanoparticles: technological concepts and future applications. Journal of nanoparticle research, 10(3), 507-517.
- Sharma, V. K., Yngard, R. A., & Lin, Y. (2009). Silver nanoparticles: green synthesis and their antimicrobial activities. Advances in colloid and interface science, 145(1- 2), 83-96.
- Shaik, M. R., Albalawi, G. H., Khan, S. T., Khan, M., Adil, S. F., Kuniyil, M., ... & Khan, M. (2016). “Miswak” based green synthesis of silver nanoparticles: evaluation and comparison of their microbicidal activities with the chemical synthesis. Molecules, 21(11), 1478.
- Khan, F. A., Zahoor, M., Jalal, A., & Rahman, A. U. (2016). Green synthesis of silver nanoparticles by using Ziziphus nummularia leaves aqueous extract and their biological activities. Journal of Nanomaterials, 2016(1), 8026843.
- Khan, M., Karuppiah, P., Alkhathlan, H. Z., Kuniyil, M., Khan, M., Adil, S. F., & Shaik, M. R. (2022). Green synthesis of silver nanoparticles using Juniperus procera extract: their characterization, and biological activity. Crystals, 12(3), 420.
- Iftikhar, M., Zahoor, M., Naz, S., Nazir, N., Batiha, G. E. S., Ullah, R., ... & Mahmood, H. M. (2020). Green synthesis of silver nanoparticles using Grewia optiva leaf aqueous extract and isolated compounds as reducing agent and their biological activities. Journal of Nanomaterials, 2020(1), 8949674.
- Sadiq, M., Saeed, K. (2012). Biological applications of ABC.Am J Chem, 45(2), 1230–1232.