Research Article - (2026) Volume 4, Issue 1
Fossil Stories
2Center for Veterinary Health Sciences (CVHS), Department of Veterinary Pathobiology, Oklahoma State University (OSU), Stillwater, OK 74078, USA
Received Date: Dec 19, 2025 / Accepted Date: Jan 27, 2026 / Published Date: Feb 03, 2026
Copyright: ©2026 José de la Fuente. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Fuente, J. D. L. (2026). Fossil Stories. Env Sci Climate Res, 4(1), 01-21.
Abstract
Organisms well preserved in fossil inclusions in amber, limestones and bone beds provide information to explore the ancient world with stories that can illustrate their origin, evolution and interactions between different organisms. This information is relevant for the study factors associated with the environment and climate. In this article, 16 stories were selected to address different findings and challenges in the study of the fossils. These stories provide information and questions on fossil inclusions and body remains inspiring and guiding research and education for planetary exploration and evolution, and material sciences.
Keywords
Amber, Climate, Copal, Environment, Evolution, Fossil, Paleontology
Introduction
Fossils are evidence of the evolution of life preserved in a geologic context such as amber, limestones and bone beds e.g., [1]. The study of fossils provides information of extinct organisms and the evolution of living species throughout geologic time. The fossils are also a challenge for the identification of preserved organisms associated with the quality of the inclusions and possible interactions shaping the ancient environment. In this context, the present study provides 12 stories addressing the role and behavior of fossil organisms in (1) The origin of the forest, (2) The Devonian Sea, (3) The Carboniferous Sea bottom, (4) Fish predation by a ravenous crocodile, (5) Bivalve predation in the Pliocene Sea, (6) Association of species in the Pliocene Sea, (7) Ant syninclusions, (8) Fossil organisms preserved in both amber and calcium carbonate, (9) Fossil corals, (10) Fish fossil remains, (11) Fossil ambiguity, and (12) Arthropod syninclusions in copal.
Story . 1 The Origin and Evolution of the Forest
Tree-like plants appeared during the Devonian period around 400 Mya. During the Late Devonian (ca. 385-359 Mya) is considered the epoch when tree-like plants formed forest-like stands with growing populations [2]. Earliest tree-like plants included Lycophytes and extinct Archaeopteris (Dawson 1871) as ancestor of gymnosperms. Seed plants appeared at the end of Devonian with currently existing groups of lycophytes, sphenopsids, ferns and gymnosperms and extinct pteridosperms or seed ferns expanding in the late Palaeozoic. The differentiation of forest was probably associated with geographical location and climate through Carboniferous with development of coal swamp forest (Figure 1A), extinction of most arborescent Lycophytes and dominance of tree ferns (Figure 1B), and until the Permian with first occurrence of Ginkgophytes and Cycadophytes and increasing conifers before the mass extinction around 372 Mya. Fossil inclusions in amber provide additional evidence on the evolution of plants. These inclusions highlight a period of "floral experimentation," with many lineages not surviving to the present day [3,4]. All Tropidogyne species lack petals but have five sepals of approximately similar length and are extinct angiosperms that flourished in the Burmese amber forest in the Cretaceous aproximately 99 Mya (Figure 1C). Also representative of Cretaceous flora are grass Programinis burmitis with spikelets (Figure 1D) and gymnosperm leaf (Figure 1E) found in ancient conifers and extinct Cordaitales with diversity representing evolutionary changes and adaptations to ancient climates [3]. Then moving to the more recent Eocene (38-54 Mya), moss (Figure 1F) and Eograminis sp. (proposed Eograminis balticus) represent the Baltic Sea region.
Taken together, these fossils represent the origin and evolution of the forest from Carboniferous to Eocene periods with 359 to 38 Ma history.

Figure 1: The Origin of the Forest
Story 1. (A) Extinct fossil tree-sized horsetail plant, Asterophyllites equisetiformis (Schlotheim; Brongniart 1828). The A. equisetiformis has a cylindrical stem, with a variable number of whorls that appear increasingly closer together as they approach the apex of the stem. Each whorl has lanceolate-shaped leaves, with a conspicuous central nerve, measuring 10-25 mm, slightly wider than the stem, and inserted slightly obliquely, with the bases fused, forming an elliptical nodal area. (B) Extinct fossil seed fern, seed-producing pteridosperm, Neuropteris ovata, reference in paleobotanics early evolution of seed plants. (C) Five sepals flower, Tropidogyne pentaptera, (D) Programinis burmitis spikelet, and (E) Gymnosperms leaf in Burmese amber (Cretaceous, ca. 99 Mya). (F) Moss and (G) Eograminis sp. (proposed Eograminis balticus) in Baltic Sea amber (Eocene, 38-54 Mya).
Story 2.The Devonian Sea
During the Devonian period in the Paleozoic Era (ca. 345-385 Mya) of Oklahoma (Haragan Formation, Coal County, OK, USA), three different trilobites and a brachiopod represented the Devonian Sea on a large fossil plate (Figure 2A). A large cephalon of Huntonia, a trilobite with a strange cephalic process, is at the top of the plate. An unidentified brachiopod, with excellent detail, is below the Huntonia cephalon. A complete, enrolled Paciphacops campbelli is positioned to the right of the brachiopod. Above, and to the right, of this 3-dimensional Paciphacops are the "horns" of a Dicranurus trilobite. There is also another cephalon of Paciphacops and several other different brachiopods exposed on the large plate.
Story 3. The Carboniferous Sea Bottom
During the Carboniferous (Mississippian) period in the Paleozoic Era (ca. 320-345 Mya) from the Mississippian of Missouri (Upper Burlington Formation, Ralls County, Missouri, USA), multiple crinoid fossils represented the Carboniferous Sea bottom (Figure 2B). The crinoid on the left is Uperocrinus pyriformis with a long, smooth-plated cup and a multi-plated tegmen. The crinoid in the center of the slab is an unusual Macrocrinus konincki camerate crinoid with very nodose plates. At the bottom of the plate is a very ornate example of Platycrinites sp. with very three-dimensional fine details. The plate structure of the crinoids is very nicely defined. There is also a partial Steganocrinus and many crinoid stem fragments (putative Actinocrinites sp.) on the matrix block.
Story 4. Fish Predation by a Ravenous Crocodile
During the Cretaceous period in the Mesozoic Era (ca. 65-146 Mya) from the Tegana Formation (Taouz, Morocco), a well- preserved fish scale recreates predation by a ravenous crocodile (Figure 2C). This large, multi-colored fish scale exhibits several puncture marks made by the sharp teeth of a hungry crocodile, Crocodylus sp. In fact, a three-dimensional crocodile tooth is still present in one of the holes. This tooth has a reddish-brown coloration. The thdisplays the corrugated enamel and exhibits only a slight amount of wear to the very tip.
Figure 2: fossil stories inspired by well-preserved organisms. (a-e) stories 2-6
Story 5. Bivalve Predation in the Pliocene Sea
A fossil bivalve, Macrocallista nimbosa, from the Pliocene period in the Cenozoic Era (ca. 1.6-5 Mya) of the Pinecrest Formation (Sarasota, Florida, USA) displays in one of the valves a small, perfectly round hole, presumably caused by some boring organism (Figure 2D). The lustrous surface of the shell is intact, and the only matrix is the sandstone-like filling of one of the valves. This is a very intriguing fossil clam that has been bored by a mysterious gastropod.
Story 6. Association of Species in the Pliocene Sea
A fossil bivalve, Pecten sp., from the Pliocene period in the Cenozoic Era (ca. 1.6-5 Mya) of the Pinecrest Formation (Sarasota, Florida, USA) represents an association between species in the Pliocene Sea (Figure 2E). This fossil bivalve is totally free of matrix and exhibits good detail. The coarse, radial ornamentation of the Pecten shell, contains several three-dimensional Barnacles attached to the clam at the time of preservation. These Barnacles became fossilized along with their resting place. Accordingly, this specimen gives a unique picture of different organisms living together and interacting during the Pliocene. The commensal association between Pecten scallops and Barnacles do not affect the bivalve while provides to the attached Barnacles a stable place to live without competition for space in the seafloor and free access to flow of water and suspended food particles such as diatoms and zooplankton as the scallop moves or feeds. Additionally, the movement of the host maintains the Barnacle free of suffocating bottom sediments. However, if the barnacles become too large or numerous it may affect the scallop with an antagonistic and non- commensal relationship affecting host movement and increasing predation risk and food competition.
Story 7. Ant syninclusions
As approached with bibliometric analysis of ant syninclusions, ants are common in amber inclusions, but the presence of other organisms represent coexistence and raise questions about possible interactions [5]. In accordance with previous analyses, ant syninclusions are a scientific challenge [5]. For example, ants use moss for nesting, foraging, and shelter due to its humid and protective capacity those providing evidence on ancient environments with the co-existence of these different species (Figure 3A). In the second example (Figure 3B), syninclusions of ants, flies and a salamander amphibian tail may be a random process but raising the question about the evolution of predator- prey relationships with predation by salamanders eating both ants and flies, and flies sometimes acting as predators or parasites of ants. However, syninclusions of extinct grass flower (Programinis sp.) and bee in Cretaceous burmite only reflect the coexistence on these organisms without interactions as grasses are typically wind- pollinated and do not attract bees (Figure 3C).
Additionally, classification is a key component to approach the analysis of amber syninclusions. For example, as illustrated in Figure 3D, a syninclusion of a wingless insect with a Crown ant in an amber piece from Baltic Sea (Eocene, 38-54 Mya) is a challenge. Based on the characteristic elongated, cylindrical body with three long terminal filaments (two cerci and a median filament) and very long segmented antennae and the head with a large, prominent compound eyes and a slightly arched the thorax, the wingless insect can be morphologically classified as a Silverfish or a Jumping bristletail. Silverfish (Zygentoma:Insecta) are rare scarce inclusions in amber and those syninclusions with ants may not be random but reflecting silverfish-ant interactions [6]. Silverfish and ants have multiple interactions with symbiotic relationships from commensalism to kleptoparasitism. Myrmecophiles silverfish have evolved specialized chemical and behavioral strategies to live within ant colonies by hiding in plain sight [7]. These ant-associated silverfish are kleptoparasites by stealing nutrient drops or ants collected prey or seeds without benefit to the ants [8,9]. However, if classified as Jumping bristletail (Archaeognatha, e.g., extinct families, also represented with a cross (†), Dasydorythidae or Praesentiidae among others), one of the most primitive of all living insects, the interactions with ants are different. Jumping bristletails generally have a limited, largely unspecialized interaction with ants, primarily relating to shared habitat use and the potential for predation avoidance without complex mutualistic or parasitic relationships like with Silverfish [10,11]. Finally, and based on detailed head morphology, the wingless insect was classified as a Jumping bristletail (large, often contiguous compound eyes meeting at the midline and a monocondylic mandible hinge) and not a Silverfish (smaller or separated eyes and dicondylic mandibles). Therefore, these syninclusions reflect ancient environment but without interactions between them.
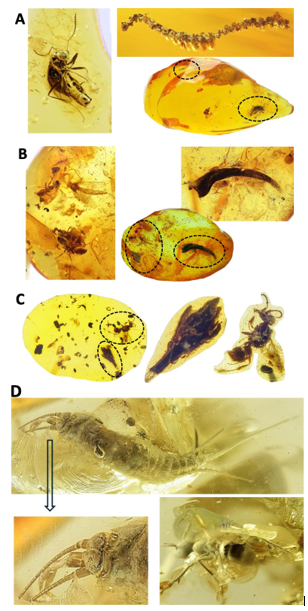
Figure 3: Ant syninclusions in amber
Story 7. (A) Ant and moss. Baltic Sea (Eocene, 38-54 Mya, size amber 50 x 14 x 26 mm and ant 5.5 mm). (B) Ants, flies and salamander amphibian tail. Baltic Sea (Eocene, 38-54 Mya, size amber 41 x 14 x 4 mm and ants more than 3 mm). (C) Bee and extinct grass flower (Programinis sp.) with lizard remains to consider. Hukawng Valley, Myanmar (Cretaceous Cenomanian, ca. 93.9-100.5 Mya, size amber 21.5 x 15.6 x 5.5 mm). (D) Crown ant and Jumping bristletail. Baltic Sea (Eocene, 38-54 Mya, size amber 22 x 10 x 17 mm).
Story 8. Fossil Organisms Preserved in Both Amber and Calcium Carbonate
Fossils are preserved in amber with resin polymerization into copal and then into amber after millions of years with inclusions. Fossil organisms preserved in calcium carbonate such as calcite, aragonite, or dolomite often occur as replacements, molds or encrustations, particularly in aquatic environments or stalagmitic deposits. These fossils can preserve three-dimensional details of the exoskeleton and in occasions also internal soft tissues. In this way, it is possible to find similar organisms in both amber and calcium carbonate. For example, beetles (Insecta: Coleoptera) are preserved with details in amber (Figure 4A) and fossil stones (Figure 4B). The extinct group of marine mollusks, ammonites (Cephalopoda) and Land Snails (Gastropoda) are also found in both amber inclusions (Figure 4C) and fossil stones (Figs. 4D, 20E). In this way, it is possible to study the evolution of these organisms from the analysis of different fossil samples (e.g., for Land Snails, )[12].
Figure 4: Fossils in amber and calcium carbonate
Story 8. (A) Spider beetle (Coleoptera: Ptinidae) in Baltic amber from Eocene (38-54 Mya). (B) Beetle (Coleoptera, Cybister sp.) with beetle elytron and the surrounding material showing the texture and sheen of hardened asphalt or roofing tar from Rancho La Brea Formation, Kern County, California, USA, Pleistocene (ca. 1 Mya) (C) Land Snail (Gastropoda, likely Cyclophoridae/ Assimineidae) with defined spiral shell, visible whorls and good structural detail with insect remains in Burmese amber from Cretaceous (ca. 99 Mya). (D) Ammonite (Eubranoceras sp.) from Ammonite Beds Formation in Huanzala, Peru, from Cretaceous (66-145 Mya). (E) Ammonite designed for decoration from North America, Cretaceous (66-145 Mya).
Story 9. Fossil Corals
Sphere of fossil coral displays an exceptionally clear and regular pattern of well-preserved polyp cross-sections (Figure 5). The light, creamy-beige matrix is enlivened by the radial structure of the individual coral polyps, which are distributed like small suns or flowers across the entire surface. The fine details of the fossil structure can be seen with the naked eye and attest to the excellent quality and polish of the piece. These corals formed millions of years ago in warm, shallow seas, where their calcareous skeletons fossilized into rock. Today, this material is highly valued, not only for its fossil origin but also for its energetic properties. Fossil coral is regarded as a stone of grounding and natural balance. It is said to strengthen emotional well-being, gently dissolve old patterns, and have a harmonizing effect on the body and mind, a symbolic connection between ancient life and the present. Shaped as a sphere, this balancing character is further enhanced by its perfect symmetry.
Figure 5: Natural fossil coral sphere
Story 9. Corals without treatment and well-preserved polyp cross- sections from Western Australia, Devonian period (ca. 359-419 Mya; weight 360.79 g, size 64 mm diameter).
Story 10. Fish Fossil Remains
Fishes are the earliest vertebrates that have been identified, and their study is known as paleoichthyology. Fossil fishes are a key component of ancient ecosystems and climates from different epochs and with different species remains (Fig. 6). The study of fossil fishes reveals paleobiodiversity and developmental patterns from the Devonian "Age of Fishes" to modern ecosystems (Fig. 6). As revised by Cloutier (2010), more than 90 fossil fishes have been described with developmental patterns in the evolution of fishes from the Silurian to the Quaternary and a good representation during the Devonian.
Figure 6: Fossil Fishes and Remains
Story 10. Green River fish, Knightia sp. (Eocene, Wyoming, USA, ca. 9.1 cm long). Chinese fish, Lycoptera sp. (Jurassic, Lycoptera Layers Laioning Province, China, ca. 8.9 cm long). Santana Formation fish, extinct Rhacolepis sp. (Cretaceous, Brazil, ca. 16.5 cm long). Shark teeth (a) extinct Carcharocles megalodon (Miocene-Neogene, River Deposits, Cuba - South Carolina, ca. 6.4 cm long), (b) Serratolamna gafsana White, 1926 (Eocene, Morocco, ca. 6 cm long), (c) Sand tiger shark, Carcharias taurus (Cretaceous, Morocco, ca. 1.2 cm long. Ray teeth, dental plate (Jurassic, Morocco, ca. 1.2 cm long). Predator fish Cockerellites Jordan & Hanibal1923, Cockerellites liops Cope 1877 (Cenozoic Eocene ca. 47-56 Mya, Green River Formation, Kemmerer, Wyoming, USA, fish 15 x 7 cm).
The Cockerellites fossil predator fish (Family Priscacaridae, Order Perciformes) from the Green River Formation shows high quality and conservation of complete skeleton in a plate (Figure 7). Priscacara is an extinct genus of perch from the Eocene commonly found in this area where the habitat were probably lakes and freshwater streams and fed on small organisms such as tadpoles, shrimp, crabs and snails that tore with their powerful jaws (Figure 7).
Figure 7: High quality and conservation of Cockerellites fossil predator fish complete skeleton
Story 10. Plate size of 20 x 29.5 x 1.8 cm and weight of 2.7 kg.
Story 11. Fossil Ambiguity
Fossil ambiguity in samples with trusted origin is mainly associated with taphonomic biases based on the parts and quality of preserved organisms. For example, a certified fossil inclusion in Burmese amber (Cretaceous, ca. 99 Mya; certificates in Appendix 1) was initially classified as an animal bone. Then, expert colleagues suggested other possibilities that were supported by the analysis of different parts (Figure 8A-E) with the Fossil Identifier (https:// www.identifyrock.net/tools/fossil-identifier). The results identified with the analysis of individual parts: Centipede (Figure 8A,B,E). The inclusion shows a clearly segmented, elongated trunk with repeating segmental nodes and paired lateral appendages projecting from each segment, which matches the basic body plan of a centipede. The body is dorsoventrally flattened, and the appendages appear single and leglike rather than bundled, consistent with Chilopoda rather than Diplopoda. Centipedes and millipedes are known from amber deposits and often preserve their legs and segment outlines in this way. Springtail (Figure 8C). The specimen shows a distinctive forked, taillike structure made of two long, thin, parallel rami matching the furcula (springing organ) of Collembola. Its small size, simple body outline and preservation in Burmese amber are also consistent with abundant springtail fossils from that deposit. Plant trichome (Figure 8D). The object is a single, thin, tapering filament with a slight basal swelling and no obvious joints or repeated ringed segments; that shape and attachment to a nearby green-brown mass are typical of a plant hair (trichome) embedded in amber. Trichomes are commonly preserved in Burmese amber as isolated hairs or hairs still attached to fragments of leaf/plant tissue, and the smooth surface and simple taper match known fossil trichomes. An arthropod appendage such as an antenna or seta is not discarded. Then, by using all parts together, the analysis identified a Caterpillar (Figure 8A-E). The inclusion shows a soft, elongated, clearly segmented arthropod body with multiple short, fleshy lateral appendages on the abdominal segments that look like prolegs rather than long jointed legs. The overall body shape and the presence of a possible head/ anterior end with a thin paired-looking appendage are consistent with an insect larva (a lepidopteran/caterpillar) preserved in amber. The stubby proleg-like structures sometimes show small hook-like projections in the photos that resemble crochets typical of caterpillar prolegs. Burmese amber commonly preserves insect larvae, including caterpillars.
To improve resolution quality, the best conserved part was used for analysis (Figure 8F), and the results confirmed the Caterpillar identification in amber. To provide an analysis with reduced biases, the inclusion image was extracted and combined in a single body for analysis (Figure 8G). Considering fossil amber origin, the specimen was classified as sawfly larva. The specimen shows a soft, elongated, segmented body with repeated lateral bulges and a series of regularly spaced openings that line up with abdominal segments (consistent with abdominal spiracles). The overall shape and size (ca. 12 mm) and the multiple repeated fleshy segmental lobes/protrusions are characteristic of sawfly Symphyta larvae, which resemble caterpillars but retain many similar looking prolegs on most abdominal segments. Cretaceous Burmese amber commonly preserves hymenopteran larvae, and the preserved external segmentation and chewing-head region toward the right of the image match known sawfly larval morphologies. However, when both images (Figure 8F,G) were used together the analysis classified a mayfly nymph based on gill-like structures along many abdominal segments and the terminal filament.
Using the Insect Identifier application (https://insect-identifier. netlify.app) with the integrated image (Figure 8G), inclusion morphology was associated with a Myriapod (Millipede or Centipede relative) and a caterpillar larva, likely a Slug Caterpillar family (Limacodidae), respectively. Therefore, the conclusions considering this evidence is that the specimen is likely a Cretaceous arthropod. However, to confirm the results, the specimen needs to be examined under higher magnification or with microCT to resolve diagnostic details at genus or species level and discard the animal bone. The results confirmed the identification of a lepidopteran caterpillar larva with stout spines dorsally and other structures (Figure 8H) as previously reported in Myanmar amber [13,14]. Nevertheless, the holes and fragmented structure in the inclusion suggested a section of a small vertebral column or fish gill-arch elements, those supporting a fragment of a bone from an animal such as marine reptile, Keichousaurus hui (Figure 8I,J).
Figure 8: Difficulties in classification of fossil amber inclusion
Story 11. Burmese amber (Cretaceous, ca. 99 Mya; certificates in Appendix 1) inclusion was initially classified as an animal bone. The analysis with Fossil Identifier resulted in (A, B, E) centipede, (C) springtail, (D) plant trichome, (A-F) caterpillar, and (G) sawfly Symphyta larva. (H) High magnification confirmed a lepidopteran caterpillar larva with (1) dorsal spines, (2) appendage pair of abdomen segment, (3) trunk/thorax appendage and (4) anterior part of head capsule. (I, J) Fragmented structure with holes supports a fragment of a bone from an animal such as marine reptile, Keichousaurus hui.
Story 12. Arthropod Syninclusions in Copal
Arthropod inclusions are also rare in copal (Figure 9). Copal is an intermediate stage of polymerization between fresh tree resin and true amber, which forms over millions of years with sufficient heat and pressure. The copal from Madagascar is produced by Hymenaea trees and is estimated to be around 300 years old in Malagasy soils before deterioration). In this context, arthropods in copal provide information on the Madagascar´s environment hundreds of years ago. Supported by Fossil Identifier (https:// www.identifyrock.net/tools/fossil-identifier), the arthropod syninclusions include:
Beetle (Coleoptera). The specimen shows a hardened pair of elytra covering the dorsal surface, a distinctly segmented body with a separate head and pronotum, and jointed legs and antennae visible at the front. The overall oval, elongate shape and the clearly delineated elytral suture are typical of Coleoptera preserved in amber. Spider (Araneae). The specimen shows a clear two-part body plan (cephalothorax and rounded abdomen) with multiple long, jointed legs radiating from the front body region. The leg posture with legs folded around the body and overall silhouette are typical of small spiders trapped in resin.
Chalcid wasp (Hymenoptera). The specimen is very small with a slender petiole, a compact mesosoma and a narrow metasoma, and shows the reduced, simple wing outline typical of chalcidid wasps. The antenna appears geniculate (elbowed) with a distinct funicle and terminal club, and the overall body proportions and size match abundant tiny parasitoid chalcids commonly trapped in Malagasy copal Pseudoscorpion (Chelicerata). The specimen shows a compact, arachnidlike body with a pair of conspicuous, forwardprojecting raptorial appendages that resemble pedipalps ending in hooked tips rather than long segmented antennae. Its overall proportions and the presence of stout grasping limbs are typical of pseudoscorpions commonly found preserved in resins and copal.
True fly (Diptera). The specimen shows the overall body plan of a true fly with a compact head–thorax–abdomen layout, long legs attached to a thorax, and a single visible wing plane rather than two clearly separate pairs. The proportions with a relatively large thorax compared with abdomen and long slender legs and the wing outline that can be seen through the copal are typical of small flies preserved in amber/copal.
Ant (Formicidae). The specimen shows the classic three-part ant body plan with a distinct head, mesosoma, and a narrow waist region with at least one petiole node visible. The head appears to have a constricted neck and what look like elbowed antennae projecting forward, and the overall proportions and leg placement match worker ants commonly preserved in resin.
Cockroach nymph (Dictyoptera). The specimen shows a dorsoventrally flattened, oval body with a distinct head bearing chewing mouthparts and what appear to be long, multisegmented antennae, which are classic Blattodea cockroach features. The thoracic region looks shielded (pronotum-like) and the legs are positioned laterally in a way consistent with a roach nymph rather than a true flying insect. Similar small roach nymphs are commonly trapped in Malagasy copal.

Figure 9: Madagascar copal with arthropod syninclusions
Story 12. Copal (Holocene – Pleistocene, ca. 300 ya) from Diego-Suarez, Madagascar includes beetle, spider, chalcid wasp, pseudoscorpion, true fly, ant and cockroach nymph syninclusions.
Story 13. Fossil Marine Fauna: Unique Fossil Horseshoe Crab and Fishes
Fishes and crustaceans are key fauna components of the marine ecosystem. Horseshoe crabs are characterized by a horsehoe- shaped shield or carapace that covers the entire body except for the tail with the exoskeleton divided into a semicircular cephalothorax followed posteriorly by a relatively short abdomen and a long spine extends. Fossil and living horseshoe crabs show similar morphological characteristics supporting and evolutionary stasis for at least 445 millions of years and the presence of "living fossils" in the current marine environment [15].
Although fossil horseshoe crab extant species of the Limulus arthropod genus (O. F. Müller 1785) have been described ,its presence surrounded by two fishes of different species of extinct genera, Diplomystus of marine clupeomorph fish and Armaginatus of marine teleost fish, is rare and provide information about the fossil marine ecosystem (Figure 10) [16,17]. Fishes and horseshoe crabs and have a significant ecological relationship because crab eggs are an important food source for multiple fish species, while adult crabs are prey for sharks and turtles. Additionally, horseshoe crabs are currently key marine species in coastal ecosystem and a resource used in baits for commercial fisheries of eels and conchs. In biomedicine, their blue copper-based blood, Limulus Amebocyte Lysate (LAL) is used in safety test of medical products and drugs for bacterial contamination [18]. On the bad side of the coin, the abuse of these applications and habitat loss have resulted in the population decline leading the development of management and conservation interventions in the Atlantic coast [18].


Figure 10: Cretaceous fossil marine fauna
Story 13. Fossil limestone from Lebanon (Upper Cretaceous, 100.5-66 Mya) with Limulus horseshoe crab, Diplomystus extinct genus of marine clupeomorph fish, and Armaginatus extinct genus of marine teleost fish. Plate size of 28 x 21.5 x 1.4 cm and weight of 1.9 Kg.
Story 14. Cretaceous Pseudoscorpion-Chalcid Wasp Interactions.
Pseudoscorpions are considered an example of living fossils with significant morphological stasis preserved in Cretaceous Burmese amber from ca. 99 Mya (Figure 11) [19-21]. The pseudoscorpion and chalcid wasp syninclusions may represent ancient interactions between these organisms that are considered antagonistic by pseudoscorpion predation of the wasp with complex phoretic- parasitic relationships.

Figure 11: Cretaceous Pseudoscorpion and Chalcid Wasp Syninclusions
Story 14. Burmese amber (Cretaceous, ca. 99 Mya) with pseudoscorpion and chalcid wasp syninclusions. Amber piece diameter 19 mm.
Story 15. Fossil Rocks
Fossil rocks are sedimentary layer rocks representing the Earth geological history and preserving fossil remains or traces of ancient living organisms (e.g., Figure 1, 7, 10). These rocks include agate rock pieces, fossil slates and mudstone (Figure 12A-12D). The origin of agate lies in silica-rich fluids filling gas bubbles or vesicles in volcanic rocks structured in concentric layers over millions of years (Figure 12A). Mudstones were deposited in the Jurassic Sea and are commonly found in the UK (Figure 12B). Fossil slate forms from low-grade metamorphism of shale or mudstones form when heat and pressure align clay minerals into parallel layers to create slaty cleavage favoring it to split into thin flat sheets (Figure 12C). The Simbirzit, also known as Simbirskite,is considered a unique type of a Russian rock composed of fossils such as clam shells or ammonites filled with calcite and pyrite (Figure 12D). In cultural and metaphysical implications, Simbirzit is an important component of Ulyanovsk folk art and holistic practices rather than traditional Russian culture associated with prosperity, family well- being, happiness, and inner strength and stability. It is claimed to help recognize, release, and realign emotional patterns.
Figure 12: Fossil rocks
Story 15. (A) Agate rock pieces from North America (Eocene, 38-54 Mya). (B) Mudstone from UK (Jurassic, 145-201 Mya). (C) Fossil slate from North America (Devonian, 358-419 Mya). (D) Simbirzit, also known as Simbirskite, from Volga, Ulyanovsk (former Simbirsk), Russia (Cretaceous, 66-145 Mya).
Story 16. Evolution of Host-Host Interactions
The possible interactions between different organisms are also suggested in fossil amber inclusions (Figure 13A-13D). Although less common, it is possible to find fossil amber pieces with multiple organisms including snail, spider web, insects, acari, seeds, ant, fly, mosquito and multiple species of unclassified arthropods (Figure 13A-13D). These arthropods show different feeding behaviors such as herbivores, detritivores, arthropod predators, opportunistic feeders, and predators/scavengers with implication of multiple host species (Figure 13A-13D). The study of these fossil syninclusions provides insights into the evolution of host- host interactions with possible impact on arthropod holobiont, defined as the ecobiological assemplage of the host and the other organisms living in or close to it.
Figure 13: Insights into the evolution of host-host interactions with possible impact on arthropod holobiont
(A) Amber with snail, spider web and insects. Burmite or Kachin amber piece. Dimensions: 18 x 10 x 6 mm. Age: Cretaceous, ca. 99 mya. Expert/Provider: Manuel Quiring. (B) Amber with acari and insects. Baltic (Lithuania) amber piece. Dimensions: 17 x 12 x 3 mm. Age: Paleogene, Eocene (56 - 34 mya). Expert/ Provider: Manuel Quiring. (C) Amber with seeds of Fabaceae or Leguminosae and many arthropods (Collembola, Acari, Beetle, Diptera and others). Burmite or Kachin amber piece. Dimensions: 14 x 12 x 5 mm. Age: Cretaceous, ca. 99 mya. Expert/Provider: David Leggett (Holding Historic Inc.). (D) Amber with ant, fly and mosquito. Baltic (Poland) amber piece. Dimensions: 16 x 24 x 6 mm. Expert/Provider: Frédéric Diebold and Lambert van Es (WorldAmber). Most common feeding behaviors are illustrated in green square images.
Story 17. Fossil Collection and Conclusions
It is not possible to reach a final fossil story because the number of inclusions is very big and always growing with new findings worldwide. Nevertheless, as disclosed with stories here, the analysis of fossil collections provides information to inspire investigations in this area.
As an example, the KJG Colección (Ciudad Real, Spain) with fossils included in this study, contains 332 pieces including decorative art (210 amber and 122 limestone/remains) with documented 504 fossil inclusions (338 in amber and 166 in limestone/remains). Despite collector´s biases in collecting fossils that may be considered, the results illustrate the variety and composition of fossils in collections (Table 1, Figure 14-16).
|
Amber |
||||
|
Organism classification |
No. pieces |
Origin |
Epoch |
No. inclusions |
|
Arthropods - Chelicerata |
39 |
AF, 1 CI, 4 |
Holocene-Pleistocene |
77* |
|
|
|
EU, 6 |
Miocene-Oligocene |
|
|
|
|
AS, 27 |
Eocene |
|
|
|
|
|
Cretaceous |
|
|
Arthropods - Insects |
111 |
AF, 1 |
Holocene-Pleistocene |
208* |
|
|
|
AF, 1 |
Pliocene |
|
|
|
|
CI, 20 |
Miocene-Oligocene |
|
|
|
|
MX, 1 |
Miocene-Oligocene |
|
|
|
|
EU, 49 |
Eocene |
|
|
|
|
AS, 38 |
Cretaceous |
|
|
Arthropods - Myriapoda |
4 |
EU, 1 |
Eocene |
6* |
|
|
|
AS, 3 |
Cretaceous |
|
|
Plants |
9 |
CI, 3 |
Miocene-Oligocene |
15* |
|
|
|
EU, 3 |
Eocene |
|
|
|
|
AS, 3 |
Cretaceous |
|
|
Nematodes |
3 |
AS, 3 |
Cretaceous |
8* |
|
Mollusca |
3 |
AS, 3 |
Cretaceous |
9* |
|
Amphibians |
1 |
EU, 1 |
Eocene |
5* |
|
Reptiles |
2 |
AS, 1 |
Cretaceous |
2 |
|
Birds |
2 |
AS, 4 |
Cretaceous |
4* |
|
Decorative art |
36 |
BA, 4 |
Miocene-Oligocene |
4 |
|
|
|
MX, 1 |
Miocene-Oligocene |
|
|
|
|
EU, 26 |
Eocene |
|
|
|
|
AS, 5 |
Cretaceous |
|
|
Limestones and remains |
||||
|
Organism classification |
No. pieces |
Origin |
Epoch |
No. inclusions |
|
Arthropods - Chelicerata |
2 |
AS, 1 |
Cretaceous |
4* |
|
|
|
AF, 1 |
Ordovician |
|
|
Arthropods - Insects |
12 |
AU, 1 |
Pleistocene |
12 |
|
|
|
EU, 1 |
Miocene |
|
|
|
|
AS, 1 |
Miocene |
|
|
|
|
NA, 5 |
Eocene |
|
|
|
|
SA, 2 |
Cretaceous |
|
|
|
|
AS, 1 |
Jurassic |
|
|
|
|
UNK, 1 |
UNK |
|
|
Arthropods - Crustacea |
4 |
AS, 1 |
Pleistocene |
4 |
|
|
|
FL, 1 |
Pliocene |
|
|
|
|
AS, 1 |
Cretaceous |
|
|
|
|
SA, 1 |
Cretaceous |
|
|
Arthropods - Trilobites |
11 |
NA, 4 |
Devonian |
16 |
|
|
|
SA, 1 |
Devonian |
|
|
|
|
AF, 1 |
Devonian |
|
|
|
|
NA, 3 |
Ordovician |
|
|
|
|
NA, 2 |
Cambrian |
|
|
Sponges and Bryozoans |
1 |
NA, 1 |
Carboniferous |
1 |
|
Corals |
3 |
NA, 1 |
Carboniferous |
4* |
|
|
|
AU, 1 |
Devonian |
|
|
|
|
AF, 1 |
Ordovician |
|
|
Anthomedusae |
1 |
NA, 1 |
Carboniferous PA |
1 |
|
Echinoderms |
6 |
FL, 1 |
Miocene |
10* |
|
|
|
NA, 1 |
Carboniferous |
|
|
|
|
NA, 1 |
Carboniferous MS |
|
|
|
|
NA, 2 |
Ordovician |
|
|
|
|
NA, 1 |
Cambrian |
|
|
Brachiopods |
4 |
NA, 1 |
Carboniferous |
11* |
|
|
|
NA, 1 |
Carboniferous MS |
|
|
|
|
NA, 2 |
Devonian |
|
|
Mollusca |
27 |
FL, 1 |
Pleistocene |
46 |
|
|
|
FL, 3 |
Pliocene |
|
|
|
|
CA, 1 |
Eocene |
|
|
|
|
NA, 2 |
Eocene |
|
|
|
|
NA, 7 |
Cretaceous |
|
|
|
|
SA, 1 |
Cretaceous |
|
|
|
|
AF, 1 |
Cretaceous |
|
|
|
|
EU, 5 |
Jurassic |
|
|
|
|
AF, 1 |
Jurassic |
|
|
|
|
NA, 2 |
Carboniferous |
|
|
|
|
EU, 2 |
Devonian |
|
|
|
|
AF, 1 |
Silurian |
|
|
Amphibians |
1 |
NA, 1 |
Permian |
4 |
|
Reptiles |
10 |
FL, 1 |
Pleistocene |
10 |
|
|
|
NA, 2 |
Cretaceous |
|
|
|
|
AF, 5 |
Cretaceous |
|
|
|
|
AS, 1 |
Triassic |
|
|
|
|
EU, 1 |
Triassic |
|
|
Mammals |
7 |
EU, 1 |
18th century |
7 |
|
|
|
EU, 1 |
2nd to 5th century AD |
|
|
|
|
NA, 2 |
Pleistocene |
|
|
|
|
FL, 3 |
Pleistocene |
|
|
Birds |
1 |
NA, 1 |
Eocene |
1 |
|
Plants |
14 |
AS, 1 |
Neogene PlioMiocene |
15 |
|
|
|
NA, 1 |
Pliocene |
|
|
|
|
NA, 1 |
Miocene |
|
|
|
|
NA, 2 |
Eocene |
|
|
|
|
EU, 1 |
Eocene |
|
|
|
|
EU, 1 |
Jurassic |
|
|
|
|
NA, 1 |
Jurassic |
|
|
|
|
AS, 1 |
Jurassic |
|
|
|
|
NA, 1 |
Permian |
|
|
|
|
EU, 2 |
Carboniferous |
|
|
|
|
NA, 2 |
Carboniferous PA |
|
|
Fishes |
14 |
FL, 1 |
Pleistocene |
16* |
|
|
|
FL, 1 |
Pliocene |
|
|
|
|
NA, 1 |
Miocene |
|
|
|
|
NA, 2 |
Eocene |
|
|
|
|
AF, 1 |
Eocene |
|
|
|
|
NA, 1 |
Eocene-Paleocene |
|
|
|
|
SA, 1 |
Cretaceous |
|
|
|
|
AF, 2 |
Cretaceous |
|
|
|
|
AS, 2 |
Cretaceous |
|
|
|
|
AS, 1 |
Jurassic |
|
|
|
|
AF, 1 |
Jurassic |
|
|
Decorative art |
4 |
NA, 2 |
Pleistocene |
4 |
|
|
|
CI, 1 |
Pleistocene |
|
|
|
|
AU, 1 |
Cretaceous |
|
Table 1: Fossils in the KJG Colección (Ciudad Real, Spain)
*With syninclusions of other organisms. Abbreviations: CI - Dominican amber, EU - Baltic amber & Europe (including Russia), AS - Burmese amber & Asia, MX - Mexican amber, BA - Sumatra amber, AF - Africa, AU - Australia, Oceania & Pacific Islands, NA - North America (USA & Canada), FL - Florida, CI - Caribbean Islands, CA - Central America (Panama to Mexico), SA - South America, UNK - unknown origin and epoch. Data obtained from the collection on January 14, 2026. The fossil inclusions are distributed across the phylogenetic tree with some organisms such as Mollusca, amphibians, reptiles, birds, Chelicerata, insects and plants identified in both amber and limestone/remains (Figures 14, 15). Other organisms are present only in amber (nematodes) and limestone/remains (echinoderms, bryozoans, brachiopods, crustacea, fishes and mammals) inclusions (Figure 14, 15). Even for fossil organisms preserved in both amber and limestone/remains, insects and Chelicerata are more represented in amber while Mollusca and reptiles are more commonly found in limestone/remains (Figure 15). Some organisms such as birds and amphibians are rarely found in both amber and limestone/ remains (Figure 15). These differences between fossil inclusions in amber and limestone/remains are associated with the specific environments represented by these materials that provide unique specialized conditions for the preservation of certain life organisms that under other conditions would decompose.
Figure 14: Fossil hylogeny
Data was obtained from Table 1 and analyzed with same classifications for inclusions in amber and in limestones and remains. Legend: I (amber), II (limestones and remains).
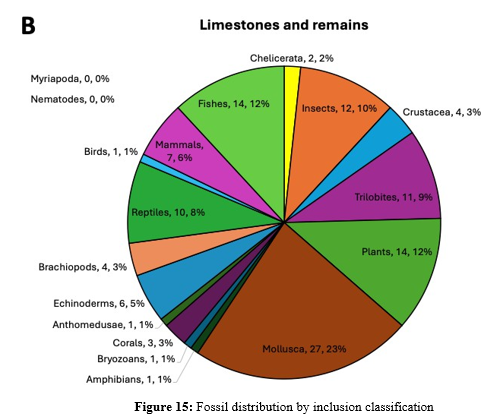
Figure 15: Fossil distribution by inclusion classification
Data was obtained from Table 1 and analyzed with same classifications for inclusions in amber and in limestones and remains. The epochs covered by fossil inlusions in amber and limestone/remains provide information on the evolution of the ecosystem and life on Earth for more than 500 millions of years since the Cambrian period (Figure 16). In the fossilization process, limestone/remains fossils provide information on the Paleozoic and Mesozoic era while amber is mostly found in the Cenozoic era (Figure 16). Nevertheless, Burmese amber comes from Mesozoic Cretaceous period and limestone/remains are also found across the Cenozoic era (Figure 16).
Figure 16: Epochs covered by fossils in amber and in limestones and remains
Taken together, fossil inclusions in both amber and limestone/ remains provide a better representation of the life evolution on Earth with implications in the study of environmental and climatic factors.
Acknowledgements
The author thanks Margarita Villar, Agustín Estrada-Peña and all other collaborators worldwide in this research topic. Art, images and fossil pieces are from author´s collection, KGJ Colección (Ciudad Real, Spain). The study was partially supported by University of Castilla La Mancha 2025-AYUDA-38326- Vaccines for the control of tick infestations in sub-Saharan Africa (ZENDAL)-01110DO064.
References
1. de la Fuente, J., Villar, M., Estrada-Peña, A. (2026). Paleontological approaches for the study of fossils. AmericanJ. Sci. Edu. Res., in press. https://www.cmjpublishers.com/american-journal-of-science-education-research/
2. Çolak, A. H., Kirca, S., & Rotherham, I. D. (Eds.). (2023). Ancient woods, trees and forests: Ecology, history and management. Pelagic Publishing Ltd.
3. Poinar, G. (2022). Burmese amber flowers. In Flowers in Amber (pp. 1-67). Cham: Springer International Publishing.
4. Poinar, G. (2025). The fossil record of grasses preserved in amber.
5. De La Fuente, J., & Estrada-Peña, A. Description of fossil amber with ant syninclusions. Frontiers in Ecology and Evolution, 14, 1724595.
6. Mendes, L. F., & Poinar Jr, G. O. (2008). A new fossil silverfish (Zygentoma: Insecta) in Mesozoic Burmese amber. European Journal of Soil Biology, 44(5-6), 491-494.
7. Molero-Baltanás, R., Degallier, N., Gaju-Ricart, M., & Parmentier, T. (2024). Finding a nest: Ant-associated silverfish (Zygentoma: Lepismatidae) moving outside granivorous Messor nests (Hymenoptera: Formicidae). Entomological Science, 27(4), e12592.
8. von Beeren, C., Schulz, S., Hashim, R., & Witte, V. (2011). Acquisition of chemical recognition cues facilitates integration into ant societies. BMC ecology, 11(1), 30.
9. Parmentier, T., Gaju-Ricart, M., Wenseleers, T., & Molero- Baltanás, R. (2022). Chemical and behavioural strategies along the spectrum of host specificity in ant-associated silverfish. BMC zoology, 7(1), 23.
10. Sánchez-García, A., & Engel, M. S. (2016). Springtails from the Early Cretaceous amber of Spain (Collembola: Entomobryomorpha), with an annotated checklist of fossil Collembola. American Museum Novitates, 2016(3862), 1-47.
11. Sánchez-García, A., Peñalver, E., Delclòs, X., & Engel, M.S. (2019). Jumping bristletails (Insecta, Archaeognatha) from the Lower Cretaceous amber of Lebanon. Papers in Palaeontology, 5(4), 679-697.
12. Neubauer, T. A., Xing, L., & Jochum, A. (2019). Land snail with periostracal hairs preserved in Burmese amber. IScience, 20, 567-574.
13. Haug, J. T., & Haug, C. (2021). A 100 million-year-old armoured caterpillar supports the early diversification of moths and butterflies. Gondwana Research, 93, 101-105.
14. Haug, C., Haug, J. T., Haug, G. T., Müller, P., Zippel, A., Kiesmüller, C., ... & Hörnig, M. K. (2024). Fossils in Myanmar amber demonstrate the diversity of anti-predator strategies of Cretaceous holometabolan insect larvae. Iscience, 27(1).
15. NZCBI. (2022). 10 Incredible Horseshoe Crab Facts. Smithsonian's National Zoo & Conservation Biology Institute (Connecticut Ave., NW Washington, DC, USA. June 3, 2022. https://nationalzoo. si.edu/animals/news/10-incredible-horseshoe-crab- facts#:~:text=1.,often%20called%20
16. Stoermer, L. (1952). Phylogeny and taxonomy of fossil horseshoe crabs. Journal of Paleontology, 630-640.
17. Bicknell, R. D., Bazzi, M., Mehling, C., Rashkova, A., Pankowski, M. V., Botton, M., & Cuomo, C. (2024). A unique example of the Late Cretaceous horseshoe crab Tachypleus syriacus preserves transitional bromalites. Alcheringa: An Australasian Journal of Palaeontology, 48(3), 495-500.
18. Krisfalusi-Gannon, J., Ali, W., Dellinger, K., Robertson, L., Brady, T. E., Goddard, M. K., ... & Dellinger, A. L. (2018). The role of horseshoe crabs in the biomedical industry and recent trends impacting species sustainability. Frontiers in Marine Science, 5, 185.
19. Harms, D., & Dunlop, J. A. (2017). The fossil history of pseudoscorpions (Arachnida: Pseudoscorpiones). Fossil Record, 20(2), 215-238.
20. Geißler, C., Kotthoff, U., Hammel, J. U., Harvey, M. S., & Harms, D. (2022). The first fossil of the pseudoscorpion family Ideoroncidae (Arachnida: Pseudoscorpiones): a new taxon from the mid-Cretaceous of northern Myanmar. Cretaceous Research, 130, 105030.
21. Feng, C., Guo, X., Zhuang, Y., Zhang, Q., Jouault, C., Jarzembowski, E. A., & Liu, Y. (2025). The first Cheliferidae (Pseudoscorpiones: Cheliferoidea) from mid-Cretaceous Kachin amber. Journal of Systematics and Evolution.



