Research Article - (2020) Volume 1, Issue 1
Foot and Mouth Disease Virus Infection Seroprevalence Study in Dairy Cattle Reared by Smallholder Farmers in Welmera District, Central, Oromiya Ethiopia
Received Date: Sep 28, 2020 / Accepted Date: Oct 03, 2020 / Published Date: Oct 12, 2020
Copyright: ©Copyright: ©2020 Beksisa Urge. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Foot and mouth disease (FMD) is a highly contagious viral disease of cloven-hoofed animals and one of the endemic diseases in Ethiopia. A study was conducted to determine foot and mouth disease virus seroprevalence and potential risk factors in welmera district, central Oromiya, Ethiopia. Purposive sampling was performed in the respective district and kebeles where there was recent history of outbreaks. A total of 126 sera samples were collected from randomly selected cattle and tested using ELISA for antibodies against nonstructural proteins of foot and mouth disease virus. The seroprevalence of foot and mouth disease virus infection in Welmera district was 49.2%. The result indicated that seropositivity of FMD infection FMD was higher in females (54%) than in males (46.1%) and this variation was statistically significant (χ2= 12.93, p = 0.001). The associations of seropositivity of FMD in relation to body condition score and management system were statistically significant (χ2= 5.34, p = 0.023, χ2= 6.7, p = 0.003) respectively. Age related seropositivity was not statistically significant (P>5%). Cross bred cattle were 2.17 (OR = 2.17, 95% CI: 2.10 – 5.74) times more likely to be infected by FMD than local breeds. This showed that FMD infection is widely prevalent in the district. Therefore, foot and mouth disease control will depend on identifying virus serotypes circulating in the area and regular vaccination program.
Keywords
FMDV, Risk factor, Seroprevalence, Welmera,
Introduction
Foot and mouth disease (FMD) is a highly contagious and economically significant viral disease of cloven hoofed animals caused by an Aphthovirus of the family Picornaviridae. Seven distinct serotypes have been identified (A, O, C, SAT1, SAT2, SAT 3, and Asia 1) and there is no cross-immunity between these strains. The disease is characterized by fever, loss of appetite, salivation, and vesicular eruptions in the mouth, on the feet, and teats [1]. The disease primarily affects cloven hoofed animals such as cattle, pigs, sheep and goats. The infection typically courses with high morbidity rate, close to 100% in cloven hoofed animals and the mortality rate is 1–5% in adult animals and may increase up to 20% in young stock due to acute myocarditis [2,3]. Foot and mouth disease is also characterized by loss of production, restriction of local and international export of animal and animal products, and socio-economic burden in the country [4]. The Egyptian ban of 2003 on Ethiopia’s livestock market alone resulted in market loss of 14.36 million USD [5]. Foot and Mouth Disease (FMD) is one of the major endemic and widely prevalent disease in Ethiopia, However, there is remarkable variation in the different production system and agro-ecologies of the country [6]. FMD outbreaks have been reported in Ethiopia with a sero-prevalence of 9-26% at animal and 28% at herd level [7]. Other studies conducted by also showed that the herd and individual prevalence of the disease was 57.6 % and 11.9% respectively [8]. Beside this differences, there is genetic heterogenecity because of error-prone replication which improves viral fitness by changing antigenically important sites of the virus that enabled them to escape from protection by the developed vaccine [9]. In spite of the annual FMD burden in Ethiopia, seroepidemiology and serotyping studies for FMD infections are inadequate. The current trend of FMD occurrence in Ethiopia showed that there are regular outbreaks, poor control measures, and reporting system. In majorities of the current study areas or villages, there was outbreak of foot and mouth disease and the presence of antibodies to fight against FMDv cattle kept by smallholder farmer is missing. Knowing the status of FMD through serological surveillance and field based outbreak monitoring could aid in generating baseline information about foot and mouth disease and institute better mitigation measures in the area. Therefore, the objectives of this study were to determine FMDv seroprevalence and potential risk factors in cattle of smallholder farmers
Materials and Methods
Study are
Welmera district is 40 km away from the capital city, Addis Ababa and the area is situated at 9o04’- 9o13’ N latitude and 38o29’-38o39’ E longitude. It is bordered on the south by the Sebeta Hawas, on the west by West shewa zone, on the North by Mulo district, on the Northeast by the Sululta. Menagesha Kolobo and Holeta Genet are the towns located in the district. The average altitude of the area ranges from 2200-2500 meter above sea level. The district is known by crop-livestock mixed farming system is a common practice. The rainfall pattern of the district is bimodal, with a short rainy period from February to April and a long rainy season from mid-June to September. The annual temperature and rainfall ranges from 18°C to 24°C and 1000 to 1100 mm, respectively [10].
Study Population
The study population were cattle that were kept under smallholder farmers and experienced outbreaks of disease and manifested clinical signs of the disease. FMD in the outbreaks and those in close contact with outbreaks. The animal level factors such as Age, Sex, BCS, and Breed as well as management systems were collected and recorded.
Study Design
A cross-sectional study design was conducted to investigate seroprevalence of FMD infection and its associated risk factors.
Sampling technique and Sample size determination
The district was purposively selected based on its accessibility, geographical location, proximity to livestock market, population density and recent outbreak. A systematic random sampling was applied to select study animals. The required sample size in this district was 96 by using 6.7% previous report by at precision of 5% within 95% Confidence interval consideration [11,12]. However, to increase the representativeness and improve precision, a total of 126 bovine sera were collected for serological test using Chekit FMDV NSP ELISA to determine the seropositivity of FMD in the district.
Sera sample collection
About 10 ml of blood sample was collected from the jugular veins of each animal using plain vacutainer tube, and the tube containing the blood sample was kept and protected from direct sunlight and put in slant position at room temperature until the blood clotted for serum separation. The separated serum was transferred into sterile cryovials bearing the names of the owner and herd code, species, village, age, sex, date of collection and transported in ice box to the laboratory and then stored at -80°C for analysis [13].
Serological Diagnostic Tests
All sera were tested for the presence of antibodies produced against nonstructural proteins 3 ABC of FMD virus infection regardless of serotypes involved using a commercially available Chekit FMD 3 ABC bo-ov ELISA kit. Antibody to The assay was performed according to manufacturer’s instruction and results were analyzed and interpreted using: X 100 The test was conducted as per the manufacturer’s instructions (IDEXX Laboratories Inc). The optical density (OD) reading was recorded using a spectrophotometer at a wavelength of 450 nm. According to the ELISA test kit manual, the samples were categorized based on their optical density (OD values as negative if OD value < 20%, ambiguous if OD value is between 20-30 % positive if OD value is > 30%).
Data Management and Analysis
Data generated from laboratory investigations were recorded and coded using Microsoft Excel spreadsheet and analyzed using STATA version 13 for Windows (Stata Corp. College Station, TX, USA) and Statistical Analysis System (SAS version 9). Odd ratio (OR) was used to determine the degree of association of risk factors with disease occurrence as indicated by 95% confidence intervals. Stepwise logistic regression model was used to analyze and regress those factors having significant putative effect on the occurrence of disease based up on p value < 0.05 as significance threshold for entries and removals. The association of potential risk factors (age, sex, breed type,) with FMD infection was computed by Pearson’s chi square test. In all the cases, 95% Confidence limit and P Values <0.05 was set for significance level.
Results
Clinical characteristics of cattle diagnosed with FMD infection
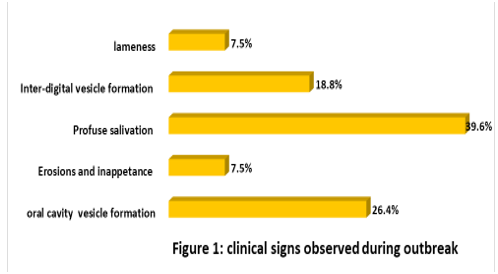
The result of the field study showed that profuse salivation was the most frequently observed clinical sign (39.6%) followed by oral cavity vesicle formation (26.4%) and inter-digital vesicle formation ( 18.8%) in sick cattle ( Figure 1).
A total of 126 bovine sera were tested by CHEKIT FMD 3ABC Bo-Ov ELISA test for detection of antibody to 3ABC Non-structure of FMD virus of which , 49.2% (n=62) cattle were positive and the remaining were negative for the test. The result indicated that seropositivity of FMD infection FMD was higher in females (54%) than in males (46.1%) and this variation was statistically significant (χ2= 12.93, p = 0.001). The associations of seropositivity of FMD in relation to body condition score and management system were statistically significant (χ2= 5.34, p = 0.023, χ2= 6.7, p = 0.003) respectively. Age related seropositivity was not statistically significant (P>5% (Table 1).
Table 1: FMD seropositivity and risk factors by using chekit 3ABC Bov-Ov ELISA test
|
Risk factors |
Category |
No. of bovine sera tested |
Seropositivity of FMD (%) |
Pearson chi square |
P value |
|
Sex |
Male |
76 |
35 (46.1) |
12.93 |
0.001 |
|
|
Female |
50 |
27(54) |
|
|
|
Age |
Young |
33 |
16(48.4) |
0.63 |
0.203 |
|
|
Adult |
93 |
46(49.5) |
|
|
|
BCS |
Good |
22 |
8(36.3) |
5.34 |
0.023 |
|
|
Moderate |
61 |
29(47.5) |
|
|
|
|
poor |
43 |
25(58.1) |
|
|
|
Mgt St |
Intensive |
20 |
13(65) |
6.7 |
0.003 |
|
|
Semi intensive |
106 |
49 (46.2) |
|
|
|
Over all (%) |
|
126 |
62 (49.2) |
|
|
**Mgt St-Management System
The result showed that crossbred animals were more seropositive (62.5%) than Local bred animals (31.4%) (Figure 1).
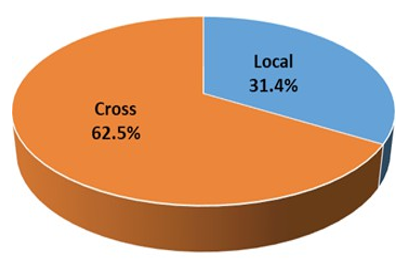
Figure 2:Breed Wise Serprevalance
The logistic regression analysis showed that breed, sex, body condition and management systems were statistically significant (P<5%). Cross bred cattle were 2.17 more likely to be infected by FMD than local breeds (OR = 2.17, 95% CI: 2.10 – 5.74). Antibodies to FMDv infection were significantly higher in female cattle than males (P<0.05) and the risk of FMD occurrence was increased by 3.1 times (OR = 3.1, 95% CI: 1.84 – 5.98). The results also showed that animals with poor body condition were 2.6 times more FMD seropositive than animals with good body condition. Cattle that were kept under intensive management system were 3.25 times more prone to FMD infection than those managed semi intensively (OR= 3.25, 95% CI: 2.21-6.02) (Table 2).
Table 2: Logistic regression analysis of FMD infection seropositivity with various risk factors
|
Variables |
Category |
OR |
95% CI for OR |
P value |
|
Breed |
Cross Local* |
2.17 |
2.10-5.74 |
0.001 |
|
Sex |
Female Male* |
3.1 |
1.84-5.98 |
0.001 |
|
Management |
Intensive semi intensive* |
3.25 |
2.21-6.05 |
0.002 |
|
BCS |
Poor Good* |
2.6 |
1.27-4.70 |
0.04 |
OR-Odd ratio, BCS-Body condition score, CI= confidence interval
Foot and mouth disease infection is higher in adult animals than young animals and vaccination of cattle reduce infection (Table 3).
Table 3: Linear regression analysis of FMD infection
|
Infection Coefficient standard error z P>[t] [95% Conf. Interval] |
|
Age Adult* 0.7982656 0.1929834 3.06 0.002 0.3280288 1.472503 Vaccine 4.39 1.49 2.9 0.03 1.47 7.31 Yes * -2.533172 .894455 -6.58 0.000 -2.810293 -1.65605 |
|
sex Male * 0.3621 0 .1952 0.0641 0.003 0.473 1.025 Cons 0.6542657 0 .223926 2.48 0.013 0.1153789 0.99315 |
Discussion
The results of the present study indicated that the overall seropositivity of FMD was 49.2 % in the study area and this finding was lower than the findings of Ayelet et al. (2012) who reported 32.7% and 30 % in Guji zone of Oromia region and Yeka district of the city of Addis Ababa, respectively [14]. The result was also lower than the findings reported in Western Ethiopia (9%) by and closed to reports in the eastern zone of Tigray with 41.5% [14,15]. On the other hand, the result was lower than the seroprevalence of seroprevalence of 53.6% FMD reported by Mekonnen et al. (2005) in Ethiopia [16]. A report from neighboring Sudan indicated that, after an active outbreak of the disease, seroprevalence of FMD was 79% in cattle which indicated a significant variation in seropositivity among cattle [7]. Previous studies conducted by Hafez et al. (2014), in Saudi Arabia and Namatovu et al. (2015) in Uganda also showed FMD seropositivity of 53% and 77% in infected cattle [17,18]. A report from neighboring Sudan also indicated that, after an active outbreak of the disease, the seroprevalence of FMD was 79% in cattle [19]. This seropositivity variation might be resulted from differences in individual animals breed, immune status, interaction of cattle with other animals and production system as well as differences in geographical area or the way sampling was conducted based on the existence of recent outbreak and this could also contribute to higher seropositivity recorded in this study area.
The study also revealed that age-specific seropositivity in adult was 49.5% and 48.4% in young cattle. This was in accordance with earlier studies of Murphy et al. (1999) who reported that seropositivity increment was due to the cumulative experience of the population with the agent. The reaserch works conducted by Bayissa (2009) indicated relative low seropositivity in age group less than two years that might be indicative of the existence of passive maternal immunity and low frequency of exposure [20].
Age association with FMD seropositivity was also consistent with the previous study of [21]. The probable reason might be, aged animals might have acquired the infection from multiple serotypes, and could produce antibodies against serotypes of FMD. In the study areas, young animals were often managed separately at around homestead and have less exposure to the disease.
The results also showed that seropositivity of FMD was observed to be higher in females (54%) than in males (46.1%) and this variation was statistically significant (p<5%). This finding was higher than the previous findings in Ethiopia, 8.27% in male and 15.07% in female cattle and lower than the previous works in Kenya, 67% in female and higher 33% in male as reported by Chepkwony [22,23]. The significant seroprevalence variation observed between the sex of cattle may be related to the effect of the higher number of females included in the study than males. This might also be related with the personal observation where dairy farm owners are usually keeping few males only for breeding purpose. The greater percentage of seropositivity in females might be due to physiological stresses which include oestrus, pregnancy and lactation which are known to affect their resistance to infection [24]. Cross breed cattle was more affected (62.5%) than local breed (31.4%). This could be related to differences in management practices. Those genetically improved cattle are more prone to FMD infection than local breeds since they may suffer from deficient housing and malnutrition. The management factors which influenced the seroprevalence of FMD in the study areas of cattle were intensive (65%) and semi intensive (46.2%) management system. In line with this result, Vosloo et al. (2002) reported that intensive livestock production is highly vulnerable to the effect of FMD [25]. This could be attributed to crowding of animals that can facilitate frequency of direct contact and hence enhances chances of transmission. The present study also identified those cattle with poor body condition scores had more FMD infection (58.1%) than moderate (47.5%) and good (36.3%) body condition which was also reported by Fraser [26]. The probable reason could be related to the weak protective immune response in poor body conditioned cattle as animals with good body condition have relatively good immunological response to infection (Radostats et al., 2007). In the current study, Cross bred cattle had higher odds (2.17 time (OR = 2.17, 95% CI: 2.10 – 5.74) of infection than local breeds. The relative higher seropositivity in crossbred cattle might be attributed to the genetic variation of animals [1].
Conclusion
The findings of the present study showed that foot and mouth disease is highly prevalent in welmera district central Oromiya. The regression analysis showed that breed, sex, body condition and management systems were significantly associated (P<5%) with the occurrence of infection. Cross bred cattle were 2.17 more likely to be infected by FMD than local breeds (OR = 2.17, 95% CI: 2.10 – 5.74). Antibodies to FMDv infection were significantly higher in female cattle than males (P<0.05) and the risk of FMD occurrence was increased by 3.1 times (OR = 3.1, 95% CI: 1.84 –5.98). A comprehensive active assessment and serotyping of foot and mouth disease outbreak field strains are required to detect the emergence and circulation of new serotypes and variants involved in the outbreaks and regular vaccination of cattle should be conducted in the areas.
References
- OIE (2019) in Foot and Mouth Disease (FMD), OIE, Paris, France.
- Radostits OM, Gay CC, Hinchcliff KW, Constable PD (2007) Veterinary Medicine: A text book of the disease of cattle, sheep, goat, pigs and horses. 10th Ed, London, Bailliere Tindall 1223-1230.
- Kandeil A, ElShesheny R, Kayali G, Moatasim Y, Bagato O, Darwish M (2013) Characterization of the recent outbreak of FMD virus serotype SAT2 in Egypt. Arch Virol 158: 619-627.
- James A, Rushton J (2002) The economics of Foot and mouth disease. OIE scientific and Techinical Rev 3: 637-644.
- MoARD (2007) Livestock Development Master Plan Study. Phase-I, Report, Data Collection and Analysis. Volume B Meat Production. Ministry of agriculture and rural development, Government of Ethiopia, Addis Ababa, Ethiopia.
- Wubshet K, Dai J, Li Q, Zhang J (2019) Review on outbreak dynamics, the endemic serotypes, and diversified topotypic profiles of foot and mouth disease virus isolates in Ethiopia from 2008 to 2018, Viruses 11: 1076.
- Mohamoud A, Tessema E, Degefu H (2011) Sero-prevalence of bovine Foot and mouth disease (FMD) in Awbere and Babille districts of Jijiga zone, Somalia Regional State, Eastern Ethiopia. African J Mic Res 5: 3559-3563.
- Gulima D (2011) Disease reporting, presentation on VACNADA Project Close out workshop, 5th to 7th December, Bishoftu, Ethiopia.
- Grazioli S, Fallacara F, Brocchi E (2013) Mapping of antigenic sites of foot-and-mouth disease virus serotype Asia 1 and relationships with sites described in other serotypes. J Gen Virol 94: 559-569.
- WoWAHA (2015) Wolmera Wereda Animal Health Agency. StatisticalAbstract. Wolmera, FinfineLiyuZuria Zone, Oromia, Ethiopia.
- Shanko K, Rufael T, Kasaye E (2015) A study on sero prevalence of foot and mouth diseases in West and South West Shoa zones of Oromia regional state, central Ethiopia,Academic Journal 7: 1-21.
- Thrusfield M (1995) Veterinary Epidemiology.2nd ed, Blackwell Science Ltd, Uk 233-250.
- OIE (2004) Foot and mouth disease. In: Manual of Standards for Diagnostic Tests and Vaccines. 5th ed, Office int. des Epizooties (OIE), Paris, France 111-128.
- Ayelet G, Gelaye E, Negussie H, Asmare K (2012) Study on the epidemiology of foot and mouth disease in Ethiopia. Rev Sci off Int Epiz 31: 789-798.
- Beyene B, Tolosa T, Rufael T, Hailu B, Teklue T (2015) Seroprevalence and associated risk factors of Foot and mouth disease in selected districts of western Ethiopia. Rev Sci Tech Off Int 34.
- Mekonnen S, Workineh M, Bayleyegn (2005) Antimicrobial susceptibility profiles of mastitis isolates from cows in three major Ethiopian dairies 156: 391.
- Hafez SM, Farag MA, Mazloum KS, Albokmy AM (2014) Serological survey of FMD virus in Saudi Arabia. Rev Sci Tech Off Int Epiz 13: 711-719.
- Namatou A, Sebenzia N, Tjornehoj K, Muwanika, Siegismund H, Ayebazibwe C (2015) Characterization of FMD virus from Ugandan cattle outbreaks during 2012-2013: Evidence for circulation of multiple seirotypes. J Resea 10: 44-116.
- OIE (2012) Foot and Mouth disease, Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, Version adopted by the World Assembly of Delegates of the OIE in May 2012, OIE Terrestrial 1-29.
- Bayissa B (2009) Foot and mouth Disease sero-prevalence and implications in Borana pastoral and agro pastoral systems, southern Ethiopia. MSc Thesis, Addis Ababa University, Faculty of Veterinary Medicine, Bishoftu, Ethiopia.
- Olabode OH, Kazeem HM, Raji, Ibrahim M (2013) Seroprevalence of foot and mouth disease virus antibodies in trade cattle (Bosindicus) in Kwara state of Nigeria. Vet World 6: 828-832.
- Gelaye E, Ayelet G, G/Egziaber B, Zeleke A (2009) A study on FMD virus serotypes circulating in Ethiopia. National Veterinary Institute, Bishoftu, Ethiopia 116-125.
- Chepkwony C, Gitao G, Muchemi M (2012) Seroprevalence of foot and mouth disease in the Somali eco-system in Kenya. Int J Anim Veter Adv 4: 198-203.
- Susan EA (1998) The Merck Veterinary Manual, 8th Ed. Whitehouse Stat NJ Merck and Co. Inc 1879.
- Vosloo W, Bastos A, Sangare S, Hargreaves OS, Thomson R (2002) Review of the status and control of foot and mouth disease in sub-Saharan Africa. Rev Sci Tech Off Int Epizt 21: 437-449.
- Fraser CM (1991) The Merck veterinary manual, 7th edition, Merck Co 338-340.


